Abstract
There are numerous herbal products on the Ghanaian market that are purported to cure various ailments, including cancer. However, scientific investigations on efficacy and toxicity of most of these products are not done. The aim of the study was to assess the anticancer potentials of herbal products on the Ghanaian market. Antiproliferative effects of Kantinka BA (K-BA), Kantinka Herbaltics (K-HER), Centre of Awareness (COA), a stomach (STO) and multicancer (MUT) product were evaluated in vitro using liver (Hep G2), breast (MCF-7), prostate (PC-3 and LNCaP), and blood (Jurkat) cancer cell lines. Cytotoxicity of the medicinal products was assessed using tetrazolium-based colorimetric assay, and total phenolic content and antioxidant activity of the products were determined using Folin-Ciocalteau and 1,1-diphenyl-2-picrylhydrazyl (DPPH) assays, respectively. Phytochemical screening resulted in the detection of terpenoids and flavonoids in most of the products, and alkaloids were detected in only MUT. Tannins were absent from all the products. The highest and lowest concentrations of phenolics were recorded for MUT and K-BA, respectively. The highest and lowest antioxidant activities were measured for MUT and K-HER, respectively. Only 2 products (STO and MUT) were cytotoxic to Hep G2 cells; with MUT being the only product that was cytotoxic to MCF-7 cells. All but K-BA were cytotoxic to PC-3 cells, while all products except K-HER were cytotoxic to LNCaP and Jurkat cells. The study thus confirms that the herbal products have selective cytotoxic activities against the tested cancer cell lines. However, comprehensive toxicity studies must be conducted to establish their safety.
Cancer is the second leading cause of mortality after heart diseases worldwide, and it is anticipated to be the primary cause of death in the next few years. 1 About 8.2 million cancer-related deaths and 14 million new cancer cases were recorded in the year 2012, with the new cases are projected to rise to 22 million annually in the next 2 decades. 2 By the year 2020, it is anticipated that 15 million new cancer cases will be recorded annually; 70% of which will be in developing countries. 3 Cancers are a major public health burden in both developed and developing countries, and they pose a great challenge despite numerous advances in cancer therapy research. 4 Worldwide, the most common cancers include lung, stomach, colorectal, liver, prostate, and breast. Lung and breast cancers are commonly diagnosed in men and women, respectively.
Various treatment regimens are available for the management of cancers, and the choice of regimen depends on the type and stage of the cancer. Although surgery and radiotherapy are effective in the treatment of cancer at its early stages, many cancers are diagnosed at later stages when the tumor has metastasized, especially in developing countries like Ghana, thus making chemotherapy the main form of treatment. 5 However, high cost of chemotherapy drugs, coupled with the side effects associated with their usage has accounted for the patronage of herbal products for the management of various cancer types in developing countries.
Natural products have received a lot of attention over the past decades due to their potential as novel preventive and therapeutic agents. 6 The medicinal prospects of plants are supported by the fact that an estimated 25% to 28% of all orthodox medicines are either directly or indirectly derived from higher plants. 7,8 About 60% of all orthodox anticancer drugs, which are actively in use, were isolated from natural products 9 ; the plant kingdom being the most significant contributor. This implies that medicinal plants have the potential of providing leads to the discovery of novel drugs. Numerous medicinal plant products are preferred in the treatment of various ailments in developing countries, however little or no scientific evidence is available to support the safety and efficacy of most of these herbal products. 10 The efficacy and safety of anticancer herbal products used in developing countries thus need to be scientifically assessed, with the possibility of isolating potent anticancer compounds.
In order to survive in their natural habitat, plants produce phytochemicals against herbivorous insects, and these phytochemicals end up being toxic to humans. 11 Also the contamination of herbs by fungal toxins, pesticides, and heavy metals absorbed from the soil contributes to the toxicity of herbal products. 12 This study was conducted to determine the efficacy of 5 herbal formulations that are marketed and commonly used for the management of cancer in Ghana.
Methods
Herbal Products
The herbal products used in this study were obtained from drug outlets and herbal centers in the Greater Accra and Eastern Regions of Ghana. All samples were bottled as aqueous solutions.
Kantinka BA (K-BA)
Plants used in preparing K-BA are Piper umbellatum, Vernonia conferta, and Sporobolus pyramidalis. The product is used for the treatment of sexually transmitted diseases (STDs), boils, skin rashes, and cancer.
Kantinka Herbaltics (K-HER)
Plants used in preparing K-HER are Spathodea campanulata, Mangifera indica, and Alstonia boonei. This product is used for the treatment of gastritis, pelvic and chest pains, skin infections, and cancers.
Centre of Awareness (COA) Drug
The active components of the COA drug are triterpenes extracted from different plant sources. The product is used for the treatment of viral infection, hepatitis, blood-related diseases, and cancers.
Stomach Cancer Product (STO) and Multicancer Product (MUT)
Plants used in the preparation of STO and MUT were not disclosed by the traditional medicine practitioners due to issues about confidentiality. However, the STO product is used for the treatment of stomach cancer, while the MUT product is used to treat multiple types of cancer.
Freeze Drying
The herbal products were vigorously shaken to obtain a uniform mixture of the concoction and poured into round bottom flasks. The samples were frozen at −20°C and freeze-dried (FreeZone 2.5 Liter Benchtop Freeze Dry System, USA) to obtain dried powdery forms of the herbal extracts. All the extracts were stored at −20°C until use.
Phytochemical Analysis
Phytochemical tests for the presence of saponins, tannins, terpenoids, flavonoids, and alkaloids were carried out as previously described, 13 with slight modifications. An aliquot of 2 mL of distilled water was added to 0.1 g of each dried herbal product and shaken vigorously. Observation of a stable and persistent (≥10 minutes) froth suggests the presence of saponins. For terpenoids, 400 µL of chloroform was added to 0.1 g of herbal product, followed by the gentle addition of 600 µL of concentrated H2SO4. The formation of an interface with a reddish-brown color suggests the existence of terpenoids. Tannins were tested for by adding 2 mL of distilled water to 0.1 g of dried herbal product and boiled, followed by the addition of 3 drops of 0.1% FeCl3. A brownish-green or blue-black coloration indicates the presence of tannins. An aliquot of 1 mL dilute ammonia solution was added to 100 mg/mL of each herbal product, followed by the addition of 200 µL of concentrated H2SO4 to test for the presence of flavonoids. A yellow coloration that disappears on standing at room temperature indicates the presence of flavonoids. To test for the presence of alkaloids, 0.1 g of dried herbal product was mixed with 2 mL of acid alcohol (1% v/v) and boiled; 400 μL of dilute ammonia (10% v/v) was added, followed by the addition of 1 mL of chloroform and shaken. The chloroform layer was extracted by adding 1 mL of acetic acid and Mayer’s test was conducted by taking 1 mL of the chloroform layer and adding 1 mL of Mayer’s reagent (potassium mercuric iodide solution). Appearance of a whitish or cream-colored precipitate indicates the presence of alkaloids.
Determination of Total Phenolic Content
Total phenolic content of the herbal products was determined using the Folin-Ciocalteau method as previously described, 14 with some modifications. Two-fold dilutions were prepared from a stock solution of 10 mg/mL of each herbal product. In brief, 790 µL of distilled water was added to 10 µL of each sample dilution followed 50 µL of Folin-Ciocalteau reagent. The mixture was thoroughly mixed and incubated for 8 minutes in the dark. Subsequently, 150 µL of 7% Na2CO3 was added and the mixture incubated in the dark for 2 hours at room temperature. The absorbance of the mixture was measured using a microplate reader (Tecan Infinite M200 PRO, Switzerland) at a wavelength of 750 nm. A standard gallic acid calibration curve was plotted and used to estimate the concentration of total phenolic content of each herbal product. Total phenolic content of each sample was expressed in grams of gallic acid equivalents (GAE).
Determination of Antioxidant Activity
The free radical scavenging ability of each herbal product was determined as previously described. 15 An aliquot of 0.5 mM methanolic DPPH (Sigma-Aldrich, Urbana, IL, USA) solution was added to equal volumes of different concentrations of each herbal product (concentration range 0-10 mg/mL) and incubated for 20 minutes in the dark. Absorbance readings were taken using a microplate reader (Tecan Infinite M200 PRO, Switzerland) at a wavelength of 517 nm. Butylated hydroxytoluene was used as a standard, and the diluents (water or methanol) were used as blanks. The percentage antioxidant activity of the extracts was determined using the following formula:
Cell Culture
Five human cancer cell lines—liver (HepG2), breast (MCF-7), prostate (PC-3 and LNCap), and blood (Jurkat), obtained from RIKEN BioResource Centre Cell Bank (Japan) were used for this study. The cell lines were cultured as previously described, 16 in RPMI 1640 (PC-3, LNCap, and Jurkat) and Dulbecco’s modified Eagle medium (DMEM) (MCF-7, HepG2) medium supplemented with 10% fetal bovine serum (FBS) (Sigma-Aldrich, Urbana, IL, USA) and 1% of penicillin streptomycin L-glutamine (Sigma-Aldrich, Urbana, IL, USA). Cells were grown in a humidified incubator (Panasonic Healthcare Company Limited, Japan) at 37 °C, supplied with 5% CO2 and passaged on reaching about 90% confluency.
Determination of Anticancer Activity via Cytotoxicity
The ability of the herbal products to inhibit proliferation of cancer cells was evaluated using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) method previously described. 17 Briefly, cells were seeded in 96-well plates at a cell density of 1 × 105 cells per well and supplemented with 10% FBS and 1% penicillin streptomycin L-glutamine. The cells were treated with different concentrations of reconstituted herbal extract (10-0.625 mg/mL), curcumin as positive control, or media alone (negative control) and incubated at 37 °C, 5% CO2 for 72 hours. After incubation, 20 µL of MTT solution (2.5 mg/mL) was added to each well, and the plates were incubated at room temperature for 4 hours. A volume of 150 µL of acidified isopropanol was added to each well to dissolve any formazan crystals formed and absorbance of each well determined at a wavelength of 570 nm using a microplate reader (Tecan Infinite M200 PRO, Switzerland). All experiments were performed in triplicates, and percentage viability of cells at each concentration of extract was calculated as follows:
Data Analysis
The data were analyzed using Microsoft Office Excel 2013 and GraphPad Prism 6.01 (GraphPad Software Inc, San Diego, CA) software. Data were presented as mean ± standard deviation (SD) of 3 replicates.
Results
Phytochemical Composition of Herbal Products
Table 1 shows that terpenoids were detected in most of the products (n = 4), followed by flavonoids (n = 3). However, tannins were not detected in any of the herbal products, and alkaloids were detected in only the MUT product. None of the phytochemical compounds tested were detected in the STO product. On the other hand, we confirmed the presence of terpenoids in the COA product, the sole active component present in the product as indicated by the producers (Table 1).
Phytochemical Composition of Herbal Products.a

Abbreviations: K-BA, Kantinka BA; K-HER, Kantinka Herbaltics; COA, Centre of Awareness; STO, a stomach cancer product; MUT, multicancer product; +, present; −, absent.
aAsterisk (*) indicates that tests were not performed because the product label suggested that the herbal preparation contain is composed of only triterpenes.
Total Phenolic Content of Herbal Products
Phenolics were found to be present in varied amounts in all the 4 herbal products that were assayed (K-BA, K-HER, STO, and MUT) (Figure 1). The COA product was not taken through the assay since its active components were not phenolic compounds (triterpenes). Stock concentration of 10 mg/mL of each herbal product was prepared and used for the determination of total phenolics. Among the 4 herbal products, MUT had the highest phenolic content (4.516 g GAE/100 g) followed by STO (1.855 g GAE/100 g), K-HER (1.230 g GAE/100 g), and K-BA (1.024 g GAE/100 g).
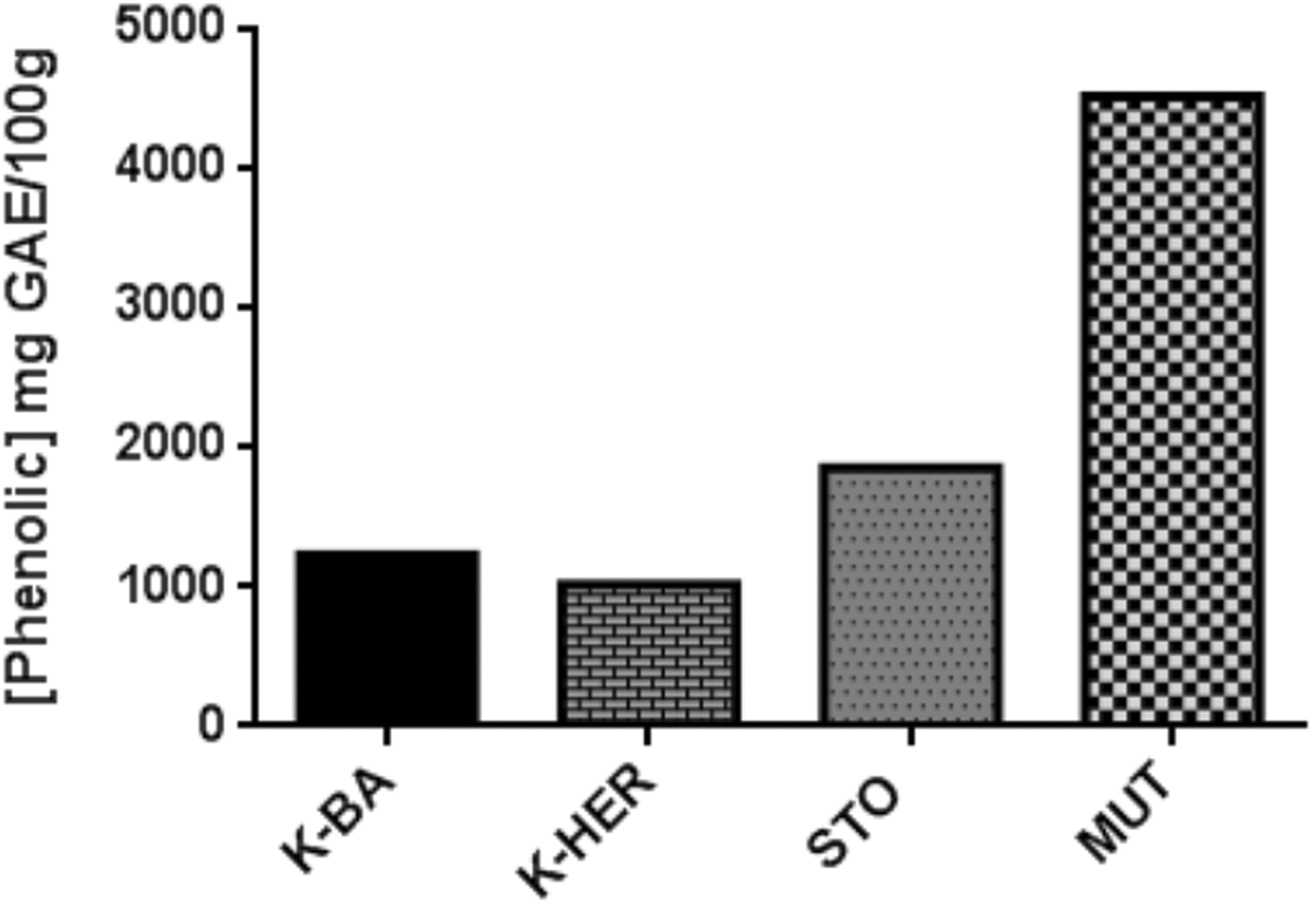
Total phenolic content of selected herbal products. Plotted values were calculated using a standard gallic acid curve.
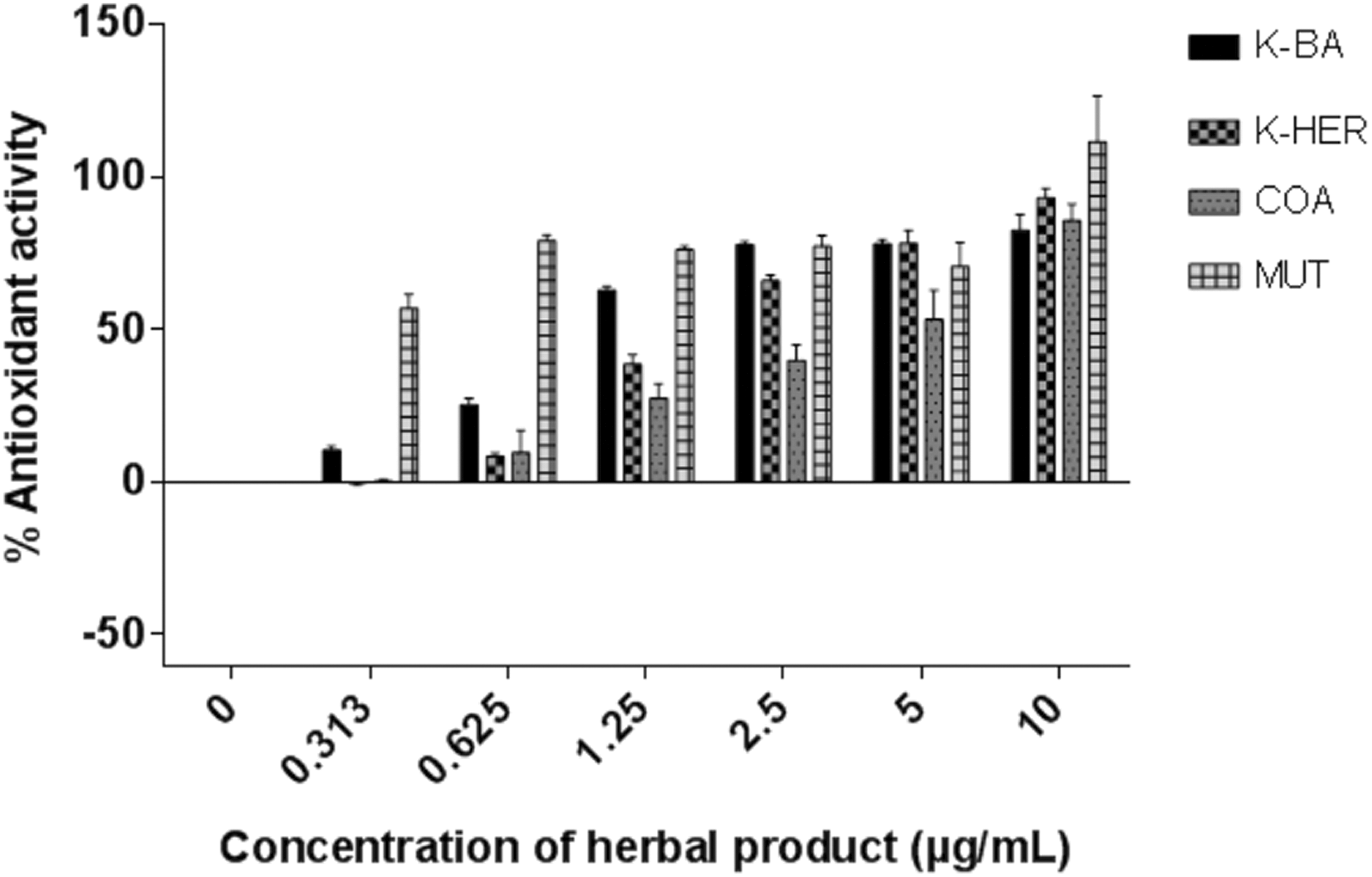
Antioxidant activity of selected herbal products.
Antioxidant Activity of Herbal Products
The antioxidant assay conducted on the herbal products showed a concentration dependent increase in antioxidant activity of all the products; with the STO product exhibiting significant activity only at the highest concentration of 10 mg/mL. The calculated EC50 (half maximal effective concentration) values were 4.38 mg/mL, 1.77 mg/mL, 1.04 mg/mL, and 0.27 mg/mL for COA, K-HER, K-BA, and MUT, respectively.
Cytotoxic Activity of Herbal Products on Cancer Cells
The cytotoxic activity of the herbal products was evaluated on 5 different cell lines (MCF-7, Hep G2, PC-3, LNCap, and Jurkat) using the MTT assay. In general, the herbal products had weak cytotoxic activity on MCF-7 and Hep G2 cell lines, but relatively stronger toxicities against PC-3, LNCap, and Jurkat cell lines.
At a maximum concentration of 1000 µg/mL, 3 (K-BA, K-HER and COA) out of the 5 herbal products still had low toxicity on Hep G2 cells. However, STO and MUT reduced the viability of Hep G2 cells with IC50 (Half maximal inhibitory concentration) values of 965.7 µg/mL and 879.1 µg/mL, respectively (Figure 3).

Growth inhibitory effect of herbal products on Hep G2 cells.
With regard to MCF-7 cell line, only one of the herbal products (MUT) decreased the cell viability by more than 50% with an IC50 value of 705.8 µg/mL (Figure 4). The other 4 herbal products (K-BA, K-HER, COA, and STO) reduced the viability of MCF-7 cells by different amounts, with percentage reduction of <20% even at maximum concentrations of 1000 µg/mL (Figure 4).

Growth inhibitory effect of herbal products on MCF-7 cell line.
All the herbal products decreased the viability of PC-3 cells by more than 50%, except for K-BA, and this occurred at different concentrations of the products (Figure 5). The IC50 values for K-HER, COA, MUT, and STO were 322.0 µg/mL, 162.50 µg/mL, 185.0 µg/mL, and 122.2 µg/mL, respectively.

Growth inhibitory effect of herbal products on PC-3 cell line.
The products STO and MUT showed moderate toxicities against LNCap cells, inhibiting more than 50% of the cells at a concentration of 62.5 µg/mL (Figure 6), while K-BA and K-HER had weak toxicities against growth of the same cells. K-HER gave less than 50% growth inhibitory activity at the highest concentration tested. The IC50 values for K-BA, COA, STO, and MUT were 583.0 µg/mL, 479.2 µg/mL, 54.5 µg/mL, and 49.2 µg/mL, respectively.

Growth inhibitory effect of herbal products on LNCap cell line.
The K-BA, MUT, and COA products had moderate toxicities against the viability of Jurkat cells, with COA inhibiting about 90% of the cells at a concentration of 500 µg/mL (Figure 7). K-HER and STO however exhibited less than 50% growth inhibitory activity at the highest concentration tested. The IC50 values for K-BA, MUT, and COA were 489.2 µg/mL, 431.0 µg/mL, and 301.3 µg/mL, respectively.

Growth inhibitory effect of herbal products on Jurkat cell line.
Discussion
Phytochemical Analysis
Phytochemical screening resulted in detection of terpenoids in most of the products (4), followed by flavonoids (3). Tannins were, however, not detected in any of the herbal products, and alkaloids were detected in only the MUT product. The different phytochemicals found in the herbal products is as expected, since the products are prepared using more than one plant/plant part; except the COA drug. The metabolite profile of a plant (nature of metabolites and concentration in specific tissues) is determined by the genetic composition and developmental stage of the plant, coupled with dominant environmental influences. 18 Phytochemical profiles of medicinal plants in different geographical locations and seasons vary distinctively 19 –22 and might account for the differences in the phytochemical profiles of the herbal products used in this study.
Alkaloids and tannins were detected in Piper umbelletum in Cameroon, with only flavonoids detected in Vernonia conferta. 23 However, we did not detect alkaloids in the K-BA product. Ethanolic extract of the stem bark of Magnifera indica has been found to contain saponins, terponoids, and flavonoids. 13 Tannins and flavonoids were detected in the aqueous and methanolic fractions of Spathodea campulantum, with alkaloids detected in the methanolic but not the aqueous fraction; and saponins were absent in both fractions. 24 Flavonoids, terpenoids and alkaloids have been detected in the aqueous and methanolic root extracts of Alstonia boonei, with the detection of saponins in only the methanolic fraction. 25 Nonetheless, we were unable to detect saponins and tannins in the K-HER product. The inability to detect some of these phytoconstituents could be due to the colored nature of the herbal products. The variation in the phytochemical profile of the herbal products may also be due to alteration of plant material during harvesting, processing, and storage. 26 –28
Total Phenolic Acid Content
Plants produce secondary metabolites such as phenolics and alkaloids, which play numerous roles in their growth and development, including protection from predators and environmental stress. The differences in the total phenolic content of the herbal products used in this study may be due to differences in the plants and the part(s) of the plants used for the preparation of each herbal product (Figure 1). In the extraction of polyphenols from plant materials, their solubility in the extraction solvent, the kind of solvent, the interaction of phenols with other plant components, the level of polymerization of phenols and the formation of insoluble complexes are some of the factors that influences the process. 29 Thus, the use of different plants or plant parts, and water in the preparation of the herbal products may account for differences in total phenolic content of the products.
Antioxidant Activity
It has been reported that oxidative stress caused by reactive oxygen species is one of the characteristics of cancer cells 30 due to genetic and metabolic alterations; hence supplementation with antioxidants has been recommended to reduce the risk of cancer in humans. However, results from a study conducted by Klein et al 31 were inconsistent with what was hypothesized. Nonetheless, with the mitochondrion as the major source of reactive oxygen species, mitochondrion-targeted antioxidants have been shown to strongly inhibit the growth of cancer cells. 32,33
The antioxidant activity of plant extracts or herbal products is usually attributed to the presence of phenolic and nitrogen compounds, as well as other potent metabolites. For phenolic compounds, the number and position of hydroxyl (–OH) groups and methoxy (–OCH3) substituents, as well as galloyl group and ortho-hydroxyl structure plays a vital role in their activity. 34 The free radical scavenging activities of plant extracts depends on the category of components, including secondary metabolites such as phenolics that are present in the extract, and these could account for differences in antioxidant activities of the herbal products. However, a concentration-dependent increase in free radical scavenging activity was observed in all the products (Figure 2), thus highlighting the fact that the protective effects of the herbal products is dose dependent. There was a positive correlation between total phenolic content (Figure 1) and antioxidant activity (Figure 2) of the herbal products, as observed by Cai et al. 35 A positive correlation was also observed between the total phenolic content and the cytotoxicities of the herbal products.
Anticancer Activity
Previous studies have shown that the phenolic content of plant extracts have an impact on both the antioxidant and antitumor activities of the extracts. 35,36 Thus it is possible that the cytotoxic activities of K-BA, K-HER, STO, and MUT are partly due to the synergistic or additive action of polyphenols that are present in these products.
Compared to K-BA and K-HER, STO and MUT contained higher amounts of phenolics (Figure 1). This may account for the relatively significant toxicities of these products on Hep G2, PC-3, and LNCap cells (Figures 3, 5, and 6) compared with the other products. The MUT, which had the highest phenolic content, exhibited much stronger cytotoxic activity against MCF-7 and LNCap cells, compared with the other herbal products (Figures 4 and 6).
Triterpenes have been demonstrated to exert some level of cytotoxic activity on HepG2, MCF-7, and Caco-2 cancer cells. 37,38 Triterpenes present in the COA product could account for the observed cytotoxic activity against Hep G2 and MCF-7, PC-3, LNCap, and Jurkat cells (Figures 3 –7). Several studies have been conducted on the anticancer potential of the individual plants used to prepare the K-BA and K-HER products as well. Ethanolic extract of Piper umbelletum leaves has been demonstrated to inhibit the growth of Hela cells, 39 with other species of the Piper genus reported to exhibit activity against MCF-7, HepG2, and HL-60 cells. 40 –42 Ayim et al 43 reported that methanolic root extract of Vernonia conferta has activity on breast (MCF-7) and colon (DLD-1) cancer and melanoma (M14) cells. The aqueous leaf extract of a specie of the Vernonia genus (Vernonia condensata) has been shown to inhibit the growth of leukemic cell lines, breast (MCF-7) and kidney (HEK 293T) cancer cell lines in a dose-dependent manner. 44 It also inhibits tumor progression in allograft-bearing mice. Moustafa et al 45 demonstrated that ethanolic extract of a species of the Sporobolus genus (Sporobolus pungens) has activity against colon (HCT-116) and lung (A-549) cancer cells. Methanolic extract of the bark and roots of Spathodea campunulata has also been demonstrated to have cytotoxic effect on CCRF-CEM leukemia cells. 46 Abdullah et al 47 reported that ethanolic extract of the kernel of Magnifera indica L has activity on breast cancer cell lines (MDA-MB-231 and MCF-7), with low toxicity on normal breast cell line (MCF-10A). Furthermore, Jagetia and Baliga 48 have demonstrated that ethanolic fractions of the stem bark of Alstonia scholaris—a species of the genus Alstonia, harvested in monsoon, winter, and summer—inhibits the growth of Hela cells in a dose-dependent manner.
Unlike the aforementioned studies that investigated the anticancer activity of a single plant, and used methanolic, ethanolic, and aqueous extracts, water was used to prepare the K-BA and K-HER products, and these products consisted of 3 different plant extracts. The low toxicities measured may be due to the products acting as prodrugs; therefore in vivo systems may be better in establishing their true activities. Some studies have shown that organic solvent extracts of plants usually possess significant anticancer activity, compared to aqueous extract. 49,50 Although the individual plant material used in the preparation of these herbal products may have some potent anticancer agents, the solvent used in the extraction procedure might have an effect on the availability of anticancer agents in the extracts.
Conclusions
For the first time, the efficacy and toxicity of some anticancer herbal products on the Ghanaian market has been evaluated. The results show that the herbal products have varied phytochemical profiles, phenolic content, antioxidant activities, and cytotoxic activities. From the IC50 values, the herbal products exerted weak toxicity against the breast, liver, prostate, and blood cancer cell lines tested. Two of the herbal products (MUT and COA) were comparatively potent against the blood cancer cell line. In Ghana, patients who patronize herbal products consume the products chronically. Chronic intake of the herbal products evaluated in this study could lead to the maintenance of an effective concentration of the active agents, thus immensely contributing to the anticancer activity of the herbal products in vivo. This could, however, have an effect on the toxicity of the herbal products due to continuous availability of the product in the body. Although the findings confirm the claims that the selected herbal products are cytotoxic to cancer cells, in vivo studies should be carried out to affirm the potency and toxicities of these herbal products. It is prudent to carry out in vivo studies, since liver enzyme activity has an effect on the efficacy and toxicity of consumed herbal products/drugs.
Limitations
The major limitations of the study were the inability to determine some of the plants or plant parts that were used in the preparation of the products due to issues of confidentiality, and there was no normal cell line to compare with the cancer cell lines.
Footnotes
Authors’ Note
The funding agency played no role in the design of the study, analysis and interpretation of data, and the writing of the manuscript.
Acknowledgments
The authors wish to thank the West African Center for Cell Biology of Infectious Pathogens (WACCBIP) for awarding a research grant to Sylvester Languon to conduct this research at the Noguchi Memorial Institute for Medical Research (NMIMR). The authors also thank the staff of the Clinical Pathology Department of NMIMR.
Author Contributions
SL was involved with the conception and study design; data collection and analysis; interpretation of data and manuscript writing, and gave final approval. EEQ performed data collection and analysis, interpretation of data and manuscript writing, and gave final approval. IT was involved with data collection and analysis, interpretation of data and manuscript writing, and gave final approval. RA-O conceived and designed the study, performed data analysis, interpreted the data, wrote the manuscript, and gave final approval. OQ was involved with conception and study design, data analysis, interpretation of data and manuscript writing, and gave final approval.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a WACCBIP-World Bank ACE Masters/PhD fellowship (ACE02-WACCBIP: Awandare).
Ethical Approval
Not applicable.


