Abstract
Phytochemicals present in plants are more effective than their individual constituents in preventing cancer through synergetic effects. From this perspective, Shemamruthaa, a herbal formulation was evaluated with a view to potentiate more intense anticancer property. This study investigates the anticancer activity of Shemamruthaa in breast cancer (MDA-MB 231) cell lines and its cancer therapeutic potential in 7,12-dimethylbenz[a]anthracene induced breast cancer rats. Results of MTT, trypan blue, and apoptotic marker assays suggested that Shemamruthaa can induce cytotoxicity in cancer cells, in a concentration- and time-dependent manner. Oral administration of Shemamruthaa effectively suppressed the tumor progression as evidenced by decrease in tumor volume and modulation of oxidant-antioxidant status and resulted in extended life span. Gas chromatography–mass spectrometry and high-performance liquid chromatography analysis of Shemamruthaa revealed the presence of pyrogallol, 5-hydrxoymethylfurfural, trilinolein, and flavonoids. Finally, we show that Shemamruthaa contains potential anticancer agents acting either singly or in combination against breast cancer cell proliferation.
Breast cancer is the most common malignant disease affecting women worldwide and the second leading cause of death due to cancer among women. 1,2 A diet rich in fruits and vegetables has been associated with a reduced risk of diseases such as cardiovascular disorders and cancer. 3 It has been shown that the phytochemicals present in fruits and vegetables or formulations are more effective than their individual constituents in preventing cancer through both additive and synergetic effects. 4,5 Hence, it is important to study the potential activity of fruits and vegetables using whole formulation containing various phytochemicals, instead of using purified molecules or fractions enriched with certain classes of molecules. From this perspective, Shemamruthaa, a herbal formulation was evaluated for the first time with a view to potentiate more intense anticancer property.
Hibiscus rosa-sinensis Linn commonly known as China rose is a potent herb in traditional system of medicine. 6 It is being used against cough, fever, dysentery, venereal diseases, and cancerous swellings. 7 In South Asian traditional medicine, various parts of the plant are used in the preparation of a variety of foods. There is very important evidence of the anticancer action of flowers against the effect of benzoyl peroxide and ultraviolet radiation in mouse skin by means of antioxidant protection, decreasing the ornithine decarboxylase activity and thymidine incorporation in DNA. 8 It has also been reported that the plant’s flower possesses androgenic, antitumor, and anticonvulsant properties. 9,10 Hibiscus flowers are reported to contain antioxidants and other bioactive metabolites such as flavonoids, flavonoid glycosides, hibiscetin, cyanidine, cyanidin glucosides, taraxeryl acetate, β-sitosterol, campesterol, stigmasterol, ergosterol, citric acid, tartaric acid, oxalic acid, cyclopropenoids, and anthocyanin pigments. 11,12
Emblica officinalis Gaertn (Phyllanthus emblica Linn.) enjoys a hallowed position in Ayurveda—an Indian indigenous system of medicine—Chinese herbal medicine, and Tibetan medicine. According to ancient Indian mythology, it is the first tree to be created in the universe. 13,14 Siddha literature is based on the three humors of subtle energy such as Vatham (Wind), Pitham (Fire), and Kapham (Water), which mutually coordinate to perform the normal functions of the body. Yugi Vaidhya Chinthamani, a book on Siddha pathology addresses in detail about the Vippurudhi Roga Nidhanam (diagnosis of the disease) according to which 7 types are established on functional disorders and organ involvement. Based on the functional disorders, they are classified as Vadha vippurudhi, Pitha vippurudhi, and Kapha vippurudhi. Based on the organs involved, they are classified as Kuvalai (lung) vippurudhi, Karpa (uterine) vippurudhi, Sandu (Bone and joints) vippurudhi, Oodu (metastatic) vippurudhi. According to Siddha, E officinalis possesses the properties of Vatham, Pitham, Kapham. 13,14
Emblica officinalis is certainly a medicinal plant of interest. 15 It has played an important medicinal role for centuries in the Indian system of medicine. Fruits of E officinalis are used for the treatment of a number of diseases, such as dyslipidemia 16 and atherosclerosis, 15 as hepatoprotective 17 and anti-inflammatory agent. 18 In many cases, E officinalis has been shown to be a potent free radical scavenging agent thereby preventing carcinogenesis and mutagenesis. 19
Emblica officinalis fruits have been reported to contain polyphenols, flavones, tannins, and the highest vitamin C content (∼478.56 mg/100 mL). It has been reported to have antimetastatic activity and can have beneficial effects against diseases such as cancer. 20,21
Honey was found to be rich in compounds known to possess anticancer properties, such as polyphenols and phenolic acids and exhibit significant biological effects in human cancer cells. 22 –24 Therefore; honey was also incorporated in the formulation with the aim to potentiate anticancer effect.
Earlier reports from our laboratory have demonstrated that Shemamruthaa exerts profound anticancer activity through its role in reinstating the normal levels of glycoprotein components, revitalizing the membrane stability, restoring antioxidant status, regulating lipid profile and lipid-metabolizing enzymes and inducing apoptosis in 7,12-dimethylbenz[a]anthracene-induced cancer-suffering animals. 25 –29
In the present study, we show that Shemamruthaa induces cytotoxicity and activates intrinsic pathway of apoptosis, through a p53 independent mechanism in MDA-MB-231 breast cancer cells. Shemamruthaa also interferes with progression of tumours in breast cancer rat models and results in the extended life span without affecting other cellular functions and body weight.
Materials and Methods
Chemicals and Cell Line
7,12-Dimethylbenz[a]anthracene (DMBA) was obtained from Sigma Chemical Co (St Louis, MO, USA). Annexin-V staining kit was purchased from Bio Vision Inc (Milpitas, CA, USA). The reverse transcriptase–polymerase chain reaction was performed using a reverse transcriptase kit (New England Biolabs, Hitchin, UK). DNA markers, protein markers, normal melting agarose, acrylamide, nitrocellulose membrane and N,N′-methylene bisacrylamide were purchased from Amersham Pharmacia (Munich, Germany). All other chemicals and reagents used were of analytical grade and purchased from Glaxo Laboratories, BDH Division (Mumbai, India), SISCO Research Laboratories, SD Fine Chemicals (Mumbai, India), and Sarabhai Chemicals (Vadodara, India).
Human breast adenocarcinoma (MDA-MB-231) cell line was obtained from the National Center for Cell Science, Pune, India.
Cell Culture
The cancer cell line MDA-MB-231 was maintained in Dulbecco’s modified Eagle’s medium supplemented with 10% fetal bovine serum, 0.1 mM minimum essential medium, nonessential amino acids, 2 mM glutamine, 10 mM HEPES (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid), 1 unit/mL penicillin, and 1 mg/mL of streptomycin by feeding every 3 to 4 days. Cells were incubated at 37°C in a humidified atmosphere of 5% CO2 and 95% air. Cells were passaged every 3 to 4 days, using a trypsin/ethylenediamine tetraacetic acid solution for detachment.
Preparation of Shemamruthaa
The fruits of E officinalis Gaertn. (Family: Euphorbiaceae; Synonym: Phyllanthus emblica Linn.), at the mature stage, were purchased commercially from the local market and the rinds were carefully removed from the seeds. The flowers of H rosa-sinensis and Emblica rinds were air dried under shade, pulverized to fine powder using a laboratory scale cutting mill, and mixed with pure honey in a definite ratio.
MTT Assay
The cytotoxic effect of H rosa-sinensis, E officinalis, and Shemamruthaa was evaluated by MTT assay using MDA-MB-231 cell line. MTT assay was performed according to a slight modification of the procedure reported by Mosmann. 30 Cells were cultured in minimum essential medium supplemented with glutamine (0.6 g/L), gentamycin (25 mg/mL), and 10% foetal calf serum at 37°C and in humidified 5% CO2. For experiments, cells were plated in 96-well plate. After 24 hours, various concentrations (0.1-1.0 mg/mL) of H rosa-sinensis, E officinalis, and Shemamruthaa dissolved in dimethyl sulfoxide were added to each well and incubated for 48 hours. The final dimethyl sulfoxide concentration did not exceed 0.1% throughout the study, all the control groups treated with 0.1% dimethyl sulfoxide. Triplicate wells were analysed at each concentration. Growth of cell lines was quantified by ability of living cells to reduce the yellow dye MTT to a blue formazan product. At the end of 48-hour incubation, the medium in each well was replaced by fresh medium containing 0.5 mg/mL of MTT. Four hour later, the formazan product of MTT reduction was dissolved in dimethyl sulfoxide and absorbance of the plate was measured in an enzyme-linked immunosorbent assay reader at 550 nm (Sirios, Seac Radim Group, Firenze, Italy). Drug effect was quantified as the percentage of control absorbance of reduced dye at 550 nm. The growth inhibition (%) was determined by the formula: Control OD − Sample OD/Control OD × 100, where OD represents optical density. Experiments were repeated 3 independent times and the data were presented as bar diagram with standard deviation.
Trypan Blue Cell Viability Assay
The effect of Shemamruthaa on cell viability was determined by trypan blue dye exclusion assay. Trypan blue (0.4%) was dissolved in phosphate buffered saline to be used for cell counting. Cells were cultured (0.75 × 105 cells/mL) and increasing concentrations (0.1-1.0 mg/mL) of H rosa-sinensis, E officinalis, and Shemamruthaa dissolved in dimethyl sulfoxide were added to each well and incubated for 48 hours. The final dimethyl sulfoxide concentration did not exceed 0.1% throughout the study, all the control groups composed of 0.1% dimethyl sulfoxide. Cells were collected after 48 hours of incubation. Triplicate wells were analyzed at each concentration. Number of viable cells was determined by trypan blue staining. Trypsinized cell solution (0.2 mL) was placed in a tube and 0.3 mL medium was added and then mixed following addition of 0.5 mL 0.4% trypan blue. After 5 minutes, cells were counted using hemocytometer. Number of cells not stained with trypan blue was counted as viable cells. Cell viability (%) is calculated using the following formula: Total viable cells (unstained)/Total cells (stained and unstained) × 100. Experiments were repeated three independent times and the data were presented as bar diagram with standard deviation.
DNA Gel Electrophoresis
DNA fragmentation was determined by electrophoresis of extracted genomic DNA from MDA-MB-231cells. Cells (0.75 × 105 cells/mL) in 6-well plates were incubated with and without Shemamruthaa for 24 hours. Cells were lysed in lysis buffer (10 mM ethylenediamine tetraacetic acid, 50 mM Tris buffer, pH 8.0, 0.5% (w/v) sodium dodecyl sulfate and proteinase K 0.5 mg/mL) and incubated at 50°C for 1 hour. Lysate was further incubated with RNase A (0.5 mg/mL) at 50°C for 1 hour. Finally, DNA loading dye was added to the DNA obtained and immediately resolved on to 1.5% agarose gel at 50 V for 2.5 hours.
Flow Cytometric Analysis of Apoptosis
The cells (1 × 106) were plated in 60-mm dish and allowed to settle down and adhere to the surface. After 12 hours, Shemamruthaa dissolved in dimethyl sulfoxide (0.125 mg/mL) was added to each well (final dimethyl sulfoxide concentration did not exceed 0.1%) and incubated for 48 hours to induce apoptosis. The control groups received the same amount of dimethyl sulfoxide. After incubation, the cells were trypsinized and collected in a 15-mL Falcon tubes. The cells were counted and 1 × 105 cells were resuspended in 500 µL of 1X binding buffer. Annexin V–fluorescein isothiocyanate (Annexin V-FITC) (5 µL) and 5 µL of propidium iodide was added and incubated in the dark at room temperature for 5 minutes. Annexin V-FITC binding was analyzed by flow cytometry. FITC-stained cells at Ex (excitation) = 488 nm; Em (emission) = 530 nm, using FITC signal detector (usually FL1) and propidium iodide staining by the phycoerythrin emission signal detector (usually FL2) were followed up.
Western Blot Analyses
After Shemamruthaa treatment, cells were harvested and cell lysates were made in a cell lysis buffer (50 mM Tris-HCl, 150 mM NaCl, pH 8.0, with 0.1% Triton X-100, 0.01 mg/mL aprotinin, and 0.05 mg/mL phenylmethylsulfonyl fluoride. The protein content was quantified using the method of Lowry et al. 31 An equal amount of the protein (40 mg/sample) was electrophoretically separated by 15% sodium dodecyl sulfate–polyacrylamide gel electrophoresis. Proteins were transferred onto Hybond C-Super nitrocellulose (Amersham International, Amersham, UK) in a Bio-Rad Trans blot apparatus. Nitrocellulose matrices were preblocked with 5% nonfat milk powder in phosphate buffered saline and 0.05% Tween-20 for 1 hour at room temperature. Following phosphate buffered saline–Tween washes, preblocked matrices were incubated with the following primary antibodies: p53 (sc-126, 1:250), PCNA (sc-7907, 1:300), Bcl-2 (sc-783, 1:200), Bax (sc-7480, 1:100), cytochrome c (sc-13156, 1:500), caspase-3 (H-277, 1: 100) caspase-9 (H-83, 1: 100), and β-actin (Internal control, 1:200). Appropriate horseradish peroxidase–conjugated secondary antibodies were then added and colour developed with H2O2-diaminobenzidine.
Animals and Experimental Design
Adult female Sprague-Dawley rats (8 weeks old) were divided into 4 groups having 8 animals in each group and given the following dose regimen: group I—normal control animals received a single oral dose of olive oil (1 mL) at the age of 8 weeks (Control); group II—animals induced for mammary carcinoma with a single oral dose of 25 mg of DMBA dissolved in 1 mL of olive oil after overnight fasting (DMBA). The rats were palpated regularly to find out the appearance of mammary tumor(s); group III—mammary carcinoma was induced as in group II rats, in addition, after 3 months; animals were treated with Shemamruthaa at the dose of 400 mg/kg body weight per day and continued for 14 days by gastric intubation (DMBA + Shemamruthaa); group IV—animals received 400 mg/kg body weight per day of Shemamruthaa alone and served as Shemamruthaa control.
The experimental design was performed in accordance with the current ethical norms approved by the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Environment & Forests (Animal Welfare Division), Government of India and Institutional Animal Ethics Committee (IEAC) Guidelines (IAEC. No. 01/030/2011).
Gross Observations and Tumor volume
The tumor incidence and changes in body weights of animals were measured and the weights were recorded in grams. The tumor volume (v) was measured as described by Escrich et al.
32
Estimation of Oxidative Stress Markers
The levels of lipid and protein oxidation in the mammary and liver tissues of control and experimental animals were analyzed. The levels of lipid peroxides were measured as thiobarbituric acid reactive substances by the method described by Okhawa et al. 33 Lipid hydroperoxides were measured as described by Jiang et al. 34 The protein carbonyl content was quantified by the method of Levine et al. 35
Statistical Analysis
The results are expressed as mean ± standard deviation. Differences between groups were assessed by analysis of variance using the SPSS software package for Windows. Post hoc testing was performed for intergroup comparisons using the least significance difference. Differences were considered significant at P < .05.
Results and Discussion
Active Principles of Shemamruthaa
Shemamruthaa, a phytochemical combination constituting dried flowers of Hibiscus rosa-sinensis, fruits of Phyllanthus emblica, and honey was formulated and evaluated for the first time with a view to potentiate more intense anticancer property.
Phytochemical screening of the formulation revealed that the Shemamruthaa is rich in flavonoids, alkaloids, glycosides, saponins, steroids, terpenoids, tannins, and phenolic compounds. Shemamruthaa was found to have 62.67 ± 2.38 mg GAE/g of total phenolics, 21.35 ± 1.20 mg QE/g of flavonoids, and 9.74±0.86 mg CE/g of proanthocyanidins. Gas chromatography–mass spectrometry analysis of herbal constituents of Shemamruthaa showed the presence of bioactive constituents such as pyrogallol, 5-hydrxoymethylfurfural, levoglucosan, dipalmitin, and trilinolein. High-performance liquid chromatography analysis of flavonoids indicated the presence of gallic acid (992.25 μg/g), quercetin (335.75 μg/g), caffeic acid (156.6 μg/g), followed by rutin (6.0 μg/g) and ferulic acid (0.3 μg/g). 26 Thus the anticancer effect of Shemamruthaa might be attributed to the synergistic action of the compounds present in the Shemamruthaa formulation.
Cytotoxic Effect of Shemamruthaa on MDA-MB-231 Cell Line
We have evaluated the cytotoxic effect of Shemamruthaa on human breast cancer (MDA-MB-231) cells using MTT assay and trypan blue dye exclusion test. Results showed a dose-dependent effect on cell viability of MDA-MB-231 cell lines on treatment with increasing concentrations of Shemamruthaa (Figure 1). Comparable results were also obtained when MTT assay was used to determine the effect of Shemamruthaa on proliferation of MDA-MB-231 cell lines (Figure 2). Thus, our results suggest that Shemamruthaa was capable of inducing cytotoxicity in breast cancer.

Assessment of Shemamruthaa-induced cytotoxicity in MDA-MB-231 cell line (cell viability by trypan blue assay).

Assessment of Shemamruthaa-induced cytotoxicity in in MDA-MB-231 cell line (MTT assay).
Previously, it has been reported that sole Emblica extracts induce cytotoxicity in microgram range 36 whereas, in the present study it was in milligram range. This difference could be attributed to the enrichment of polyphenolic compounds by the removal of free sugars, organic acids, vitamin C, and so on. Moreover, when enriched polyphenolic compounds were used, normal cells also showed equal sensitivity as cancer cells. 37
DNA Fragmentation Analysis
In the present study, we demonstrated that Shemamruthaa induced cytotoxicity and DNA fragmentation with DNA ladder formation (Figure 3) characteristic of apoptosis. The pro-oxidant action of plant-derived phenolics rather than their antioxidant action may be an important mechanism for their anticancer and apoptosis-inducing properties as reactive oxygen species and can mediate apoptotic DNA fragmentation. 38 –40
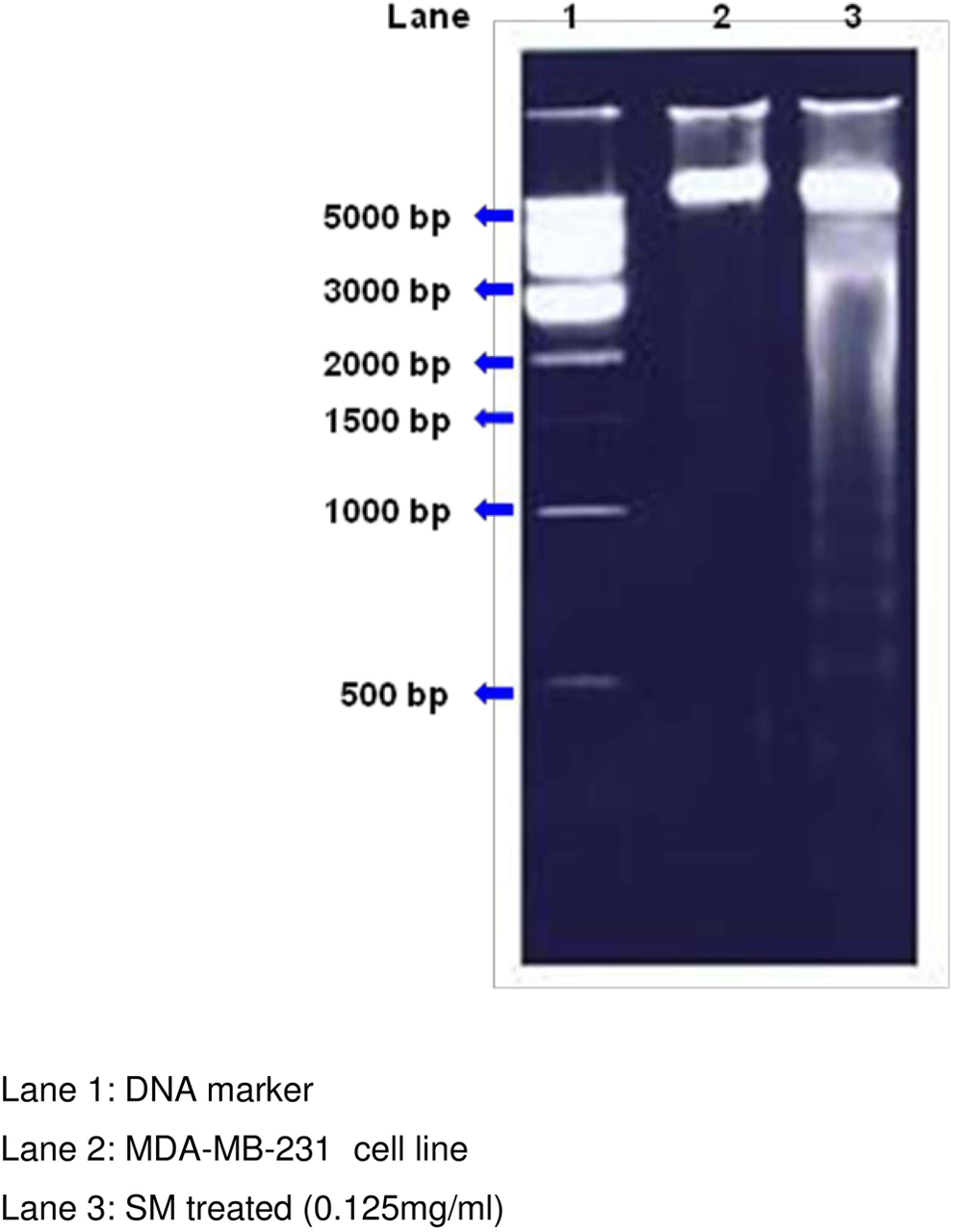
Effect of Shemamruthaa on DNA fragmentation in control and Shemamruthaa-treated MDA-MB-231 cells.
Flow Cytometric Analysis of Apoptosis
After performing cell viability assay, to determine the effect of Shemamruthaa on the cell proliferation and to confirm apoptosis, analysis of the cells treated with vehicle (0.1% dimethyl sulfoxide) and 0.125 mg/mL of Shemamruthaa was done by flow cytometry. The results, shown in Figure 4, indicate that Shemamruthaa-induced apoptosis and the proportion of early-stage apoptotic cells increased significantly from 2.43% to 4.31%, while the proportion of late-stage apoptotic/necrotic cells increased from 4.27% to 26.48%. To estimate the degree of apoptosis induced by Shemamruthaa, we utilized Annexin V-FITC (fluorescein isothiocyanate) and propidium iodide to distinguish between apoptotic and necrotic cells. Annexin V is a binding protein with high affinity and selectivity for phosphatidylserine. The appearance of phosphatidylserine on the cell surface is a general indicator of apoptosis; however, the translocation of phosphatidylserine to the cell surface also occurs during necrosis. Therefore, the measurement of Annexin V binding to the cell surface was performed in conjunction with propidium iodide. Flow cytometric analysis showed that Shemamruthaa induced death of breast cancer cells by apoptosis. High-performance liquid chromatography analysis of flavonoids constituents of Shemamruthaa revealed the presence of anticancer compounds such as gallic acid, caffeic acid, rutin, quercetin, and ferulic acid. Further gas chromatography–mass spectrometry investigation has demonstrated the presence of hydroxymethyl furfural, pyrogallol, dipalmitin, and trilinolein. Hence, the synergistic action of these compounds might be attributed to the observed apoptotic effect.

Flow cytometric analysis of MDA-MB-231 cell line incubated with Shemamruthaa for 48 hours.
Western Blot Analysis of Apoptotic Markers
Once we confirmed that Shemamruthaa induced apoptosis, we set out to establish the primary targets that were affected by Shemamruthaa. The expression of p53 in MDA-MB-231 cells with and without Shemamruthaa treatment was tested by Western blot analysis. It is important to note that MDA-MB-231 cells possess a mutation in the p53 gene (codon 280, AGA to AAA, arginine to lysine). 41
As shown in Figure 5, on treatment with Shemamruthaa, the levels of mutant p53 protein in MDA-MB-231 cells were decreased by about one-third. This result suggests that Shemamruthaa significantly suppressed the expression of a mutant gene (p53) and put forward new possibilities for regulation of mutated gene products. MDA-MB-231 cells express the mutant form of p53 protein that is no longer proapoptotic and is believed to impair the function of any remaining wild-type p53, thereby inhibiting apoptosis. It was also apparent that in these estrogen-negative cells, Shemamruthaa induction of apoptosis was through a p53-independent mechanism.
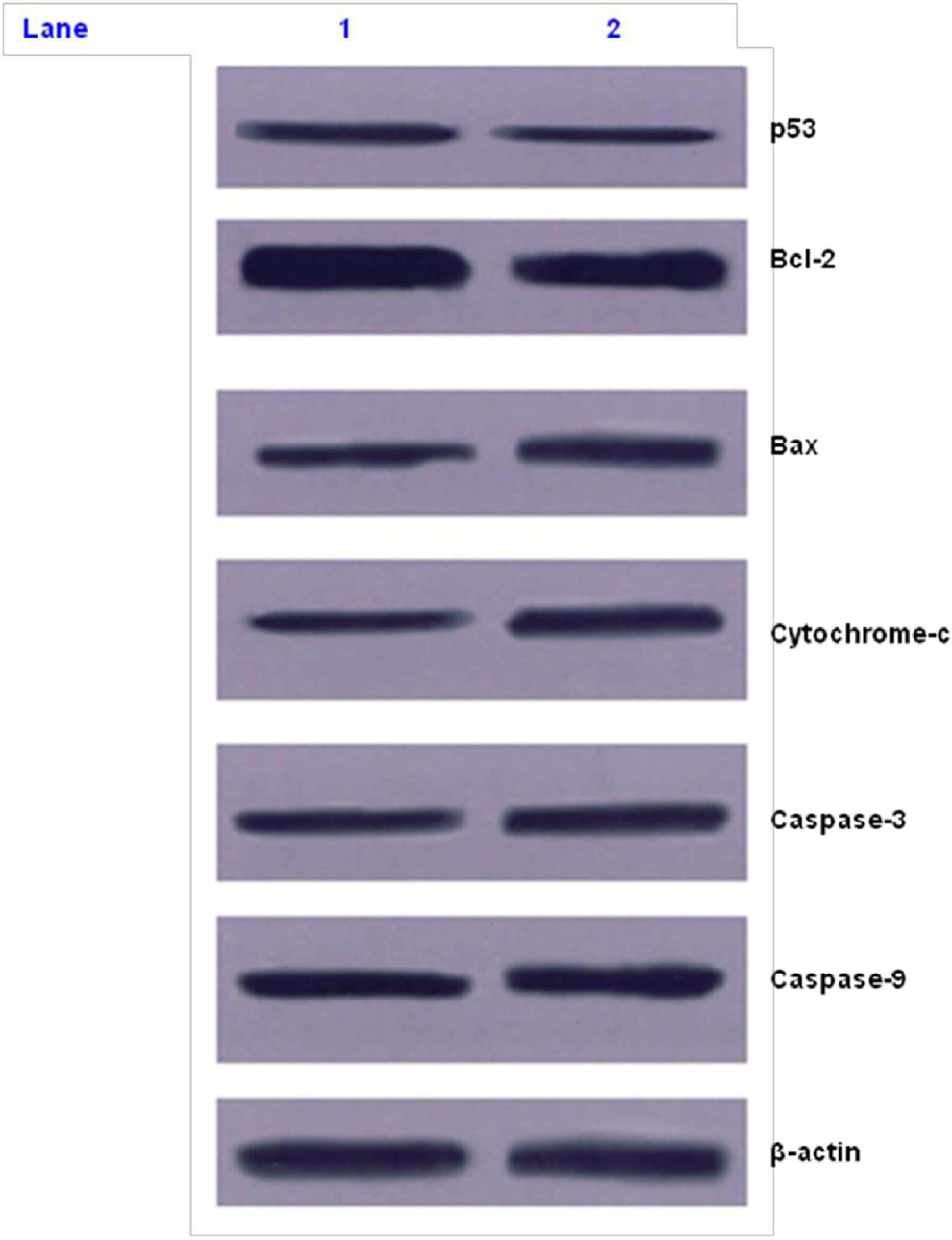
Western blot analysis of p53, BCl-2, Bax, cytochrome c, caspase-9, and caspase-3 protein expression in control and Shemamruthaa-treated MDA-MB-231 cells. Lane 1: MDA-MB-231 cells (dimethyl sulfoxide, 0.1%); lane 2: Shemamruthaa-treated MDA-MB-231 cells (0.125 mg/mL).
The present study also investigated the paradoxical increase in Bcl-2 protein expression in MDA-MB-231 cells by assessing the Shemamruthaa-induced expression of Bax, one of the proapoptotic members of the bcl-2 family of proto-oncogenes. These proteins can oppose the effects of Bcl-2 and the ratio of Bax to Bcl-2 is regarded as a more important regulator of apoptosis than the absolute amounts of the individual proteins. 42 Incubation of these estrogen receptor negative cells with Shemamruthaa elicited a significant increase in the expression of Bax assessed by Western blot. The concomitant induction of expression of these analogs of Bcl-2 by Shemamruthaa observed in the MDA-MB-231 cells resulted in a higher ratio of Bax to Bcl-2, which could explain the apoptosis elicited by Shemamruthaa in these cells.
Since cytochrome c is reported to be involved in the activation of the caspase that executes apoptosis 43 we examined the level of cytochrome c by Western blot analysis. Results showed that the amount of cytosolic cytochrome c increased in the Shemamruthaa-treated MDA-MB-231 cells. Once cytochrome c is released into the cytosol, cytochrome c together with Apaf-1 and procaspase-9 forms a wheel-like structure known as an apoptosome, which finally activates caspase-9 and then caspase-3. Earlier studies have shown that the release of cytochrome c contributes to activation of caspase-9, which then leads to activation of caspase-3. Activation of caspase-3 is an important step in the execution phase of apoptosis. 44 Activation of caspases is directly responsible for many of the molecular and structural changes in the cells undergoing apoptosis. 45 Thus, we conclude that Shemamruthaa primarily targets the mitochondria, inducing loss of membrane potential and loss of mitochondrial integrity, leading to apoptosis.
Animal Studies
Gross Observations
Table 1 presents the changes in the body weight and tumor volume in control and experimental animals. The body weight was significantly decreased (P < .05) in DMBA-induced mammary carcinoma–bearing rats when compared with control animals. Oral administration of Shemamruthaa (400 mg of Shemamruthaa/kg body weight per day) significantly recouped the body weight. The Shemamruthaa-treated rats showed a significant reduction in tumor volume when compared with DMBA-induced mammary carcinoma–bearing rats (Figure 6).

Photograph showing (A) tumor in situ in 7,12-dimethylbenz[a]anthracene (DMBA)-induced rat and (B) tumor excised from DMBA-induced and DMBA + Shemamruthaa–treated rats.
Effect of Shemamruthaa on Changes in Body Weight, Organ Weight, and Tumor Volume in Control and Experimental Animals.

Abbreviations: DMBA, 7,12-dimethylbenz[a]anthracene; NS, nonsignificant; SM, Shemamruthaa.
Comparison is made as follows: aDMBA-induced versus control, bDMBA-induced versus DMBA+ SM–treated, ccontrol versus SM control.
*P < .05.
The Shemamruthaa-treated animals showed a gradual increase in their body weights and significant reduction in tumour volume when compared to DMBA-induced mammary carcinoma rats. This might be due to the protective effect of flavonoids present in the Shemamruthaa. Since flavonoids are known to display a vast array of cellular events, they can control the overall process of carcinogenesis by several mechanisms including modulation of survival/proliferation pathways, activation of caspases, downregulation of Bcl-2 and Bcl-xL expression and enhanced expression of Bax and Bak and modulation of nuclear factor κB might be responsible for tumour regression in Shemamruthaa-treated animals. 46 –50
Effect of Shemamruthaa on Oxidative Stress Markers
Figure 7a-c shows the extent of thiobarbituric acid reactive substances, lipid hydroperoxides and protein oxidation measured as protein carbonyl content, respectively, in the mammary and liver tissues of control and experimental animals. The extent of lipid and protein oxidation was significantly higher in the mammary and liver tissues of DMBA-induced rats (group II) when compared to control rats. Shemamruthaa treatment resulted in free radical scavenging and there by significantly (P < 0.05) decreasing lipid peroxidation, protein oxidation, and restoring the enzymatic antioxidant activities to near normal levels in Shemamruthaa-treated rats.

(A) The levels of thiobarbituric acid reactive substances (TBARS) in the mammary tissue and liver of control and experimental animals. (B) The levels of hydroperoxides (LOOH) in the mammary tissue and liver of control and experimental animals. (C) The levels of protein carbonyl content (PC) in the mammary tissue and liver of control and experimental animals.
Reactive oxygen species generated during DMBA metabolism may be responsible for the increased lipid and protein oxidation and compromised antioxidant defences observed in group II (DMBA-induced) cancer rats. High-performance liquid chromatography analysis of Shemamruthaa showed the presence of bioactive compounds such as gallic acid, quercetin, caffeic acid, rutin, and ferulic acid.
26
An in vitro oxidation model showed that quercetin, myricetin and rutin are more powerful antioxidants than the traditional vitamins. The gas chromatography–mass spectrometry analysis of Shemamruthaa showed the presence of 5-hydroxymethylfurfural, 1,2,3-benzenetriol (pyrogallol), 1,6-anhydro-α-
Conclusion
Based on the results of in vitro studies, it might be concluded that the formulation, Shemamruthaa, constituted of dried flowers of H rosa-sinensis, fruits of E officinalis, and honey has been found to exhibit pronounced antiproliferative and apoptotic effect. This enhanced anticancer effect of Shemamruthaa might be attributed to the synergistic action of polyphenols such as flavonoids, tannins, alkaloids, glycosides, saponins, steroids, terpenoids, vitamin C, niacin, pyrogallol, hydroxymethylfurfural, trilinolein, and other compounds present in the Shemamruthaa formulation. Collectively, these results demonstrate that Shemamruthaa holds potential for the development of potent apoptosis-inducing chemotherapeutics for breast carcinoma.
Footnotes
Acknowledgments
The authors would like to thank Mr S. Kumaravel, Senior Scientist, Food Testing Laboratory, Indian Institute of Crop Processing Technology (Ministry of Food Processing Industries, Government of India), Tanjavur 613 005, Tamil Nadu, India for providing gas chromatography–mass spectrometry and high performance liquid chromatography facilities.
Authors Contributions
AP was responsible for conducting experiments, analyzing data, and writing the manuscript.
EN supported in conducting experiments and the preparation of the manuscript.
PS devoted her time and service for data analysis and offered valuable suggestions that related to the topic of our project.
TPS provided commentary and analysis of the biochemical and molecular aspects and significance of the findings. He also participated in the preparation of the manuscript and had the overall editing responsibilities.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The experimental design was performed in accordance with the current ethical norms approved by the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Environment and Forests (Animal Welfare Division), Government of India and Institutional Animal Ethics Committee (IEAC) Guidelines (IAEC No. 01/030/2011).

