Abstract
Objectives:
Mammary analog secretory carcinoma (MASC) is a classification of salivary gland tumors, recently included within the term secretory carcinoma. Previous descriptions of this diagnosis have largely consisted of case reports and case series with few studies investigating its clinical characteristics as compared to non-MASC tumors. Our objective was to use a large patient database to compare the clinical characteristics of mammary analog secretory carcinoma vs. non-mammary analog secretory carcinoma salivary gland tumors.
Methods:
The National Cancer Database was queried between September and October 2022 for histological diagnosis of mammary analog secretory carcinoma and non-MASC salivary tumors. Patients diagnosed with mammary analog secretory carcinoma and non-mammary analog secretory carcinoma salivary tumors between the period of 2004 through 2019 were included in this analysis. Various demographic and clinical variables were abstracted from the database and compared using Wilcoxon rank sum and chi-square tests. Survival was compared between cohorts using Cox proportional hazards regression.
Results:
Overall, compared to non-mammary analog secretory carcinoma diagnoses (
Conclusions:
Clinically, mammary analog secretory carcinoma salivary tumors have a more indolent course compared to other salivary cancers. Additional studies are needed to determine the natural history of this tumor type.
Keywords
Introduction
The 2017 World Health Organization (WHO) classification of head and neck tumors characterized the diverse group of salivary gland carcinomas by demographic, clinical, and histologic features.
1
This 4th edition included mammary analog secretory carcinoma (MASC) as an entity distinct from acinic cell carcinoma.
2
In the recently updated 5th edition of WHO classification of head and neck salivary gland tumors, secretory carcinomas harboring an
Current literature describing MASC of salivary glands predominantly features case reports and case series, with a focus on cytopathologic features. MASC of salivary glands was first described as a distinct tumor syndrome in 2010 by Skalova
Clinically, the literature describes cases of MASC invading various body tissues, including the maxillary and ethmoid sinuses, buccal mucosa, the palate, thyroid gland, lung, liver, and vulva, and affects children and adults alike.9–16 Meaningful synthesis of large-scale clinical data describing MASC, however, is lacking. Although Anderson
Materials and methods
This study is a retrospective cohort study. National Cancer Database (NCDB) data were accessed between September and October 2022, and histological diagnoses of MASC and non-MASC salivary malignancies were collected over the period of 2004 through 2019. 18 Subsequent analyses compared MASC diagnoses to a select cohort of non-MASC diagnoses, namely squamous cell carcinoma, adenocarcinoma, adenoid cystic carcinoma, mucoepidermoid carcinoma, lobular carcinoma, and acinar cell carcinoma. Finally, individual comparisons between MASC and each of the six individual non-MASC diagnoses included in the cohort were also performed. Data abstracted for both diagnosis categories include patient demographics (age, sex, race, income, and education), year of diagnosis, primary tumor site, grade, stage, size, nodal status, tumor extension, lymphovascular invasion, surgical treatment and approach, margin status, radiation therapy, chemotherapy, and patient mortality. Inclusion criteria included all adults (aged ⩾18) with a listed diagnosis of MASC and non-MASC salivary cancer, and exclusion criteria included patients with diagnoses of non-salivary gland cancer.
Statistical analysis
Demographic and clinical characteristics were compared between the MASC and non-MASC tumor cohorts using the Wilcoxon rank sum test for continuous measures and the chi-square test for categorical measures.
19
A Kaplan–Meier curve was generated to compare survival between cohorts, and Cox proportional hazards regression models were fit both with and without adjustment for important baseline characteristics. All statistical analyses were performed using SAS version 9.4 (SAS Institute Inc, Cary, NC), and statistical significance was defined as
Results
During the study period, a total of 384 cases of MASC and 47,668 cases of non-MASC tumors were collected from the NCDB, with only one MASC case (identified in 2006) identified prior to 2010. Table 1 demonstrates the demographic characteristics of MASC and non-MASC patients. On average, patients with MASC salivary tumors were younger at the time of initial diagnosis compared to patients with non-MASC salivary tumors (53.4 years vs. 63.6 years,
Demographic characteristics of MASC vs. non-MASC tumors.
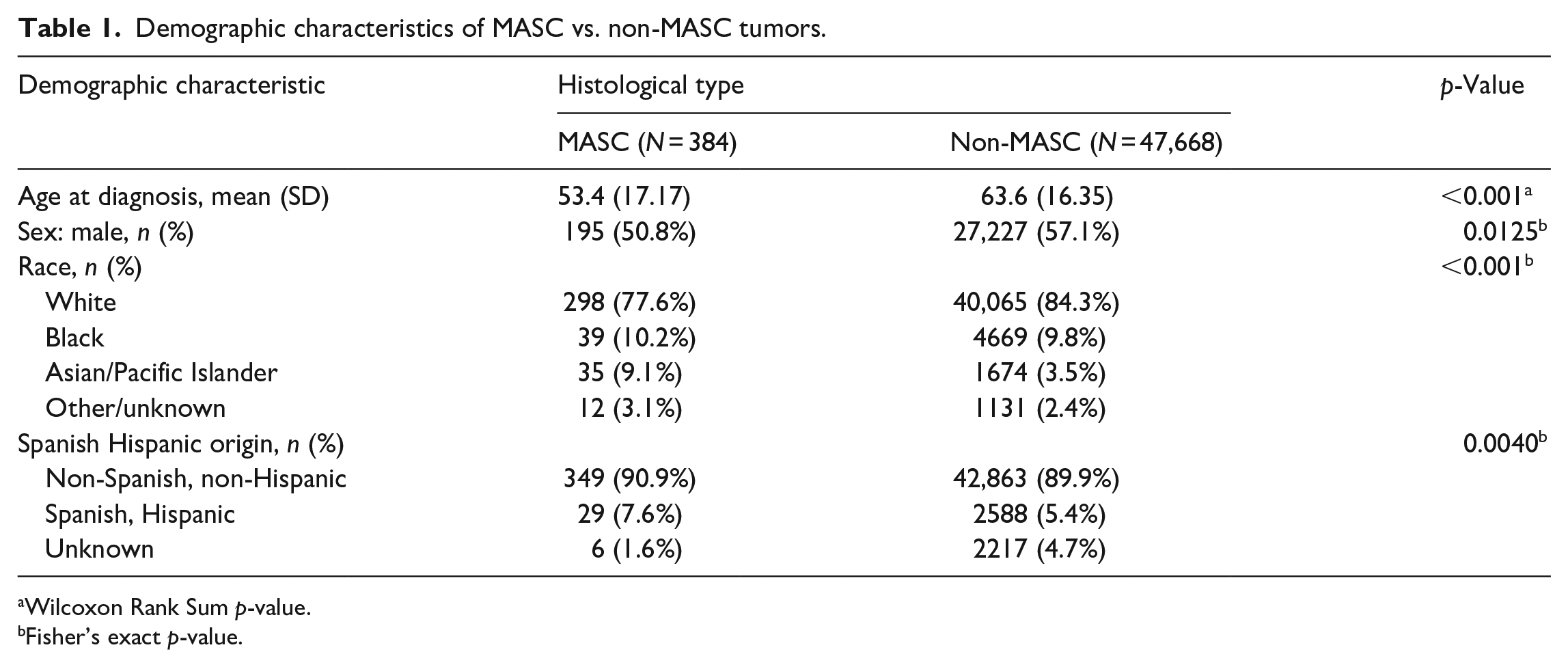
Wilcoxon Rank Sum
Fisher’s exact
Table 2 describes the clinical characteristics of MASC and non-MASC tumors. There was a significant difference between cohorts with respect to the primary tumor site (
Clinical characteristics of MASC vs. non-MASC tumors.

Fisher’s exact
Table 3 presents the treatment characteristics of patients in the MASC and non-MASC tumor cohorts. Overall, MASC patients were able to receive treatment in fewer days after diagnosis than non-MASC patients (18.0 days vs. 21.4 days,
Treatment demographics of MASC vs. non-MASC tumors.
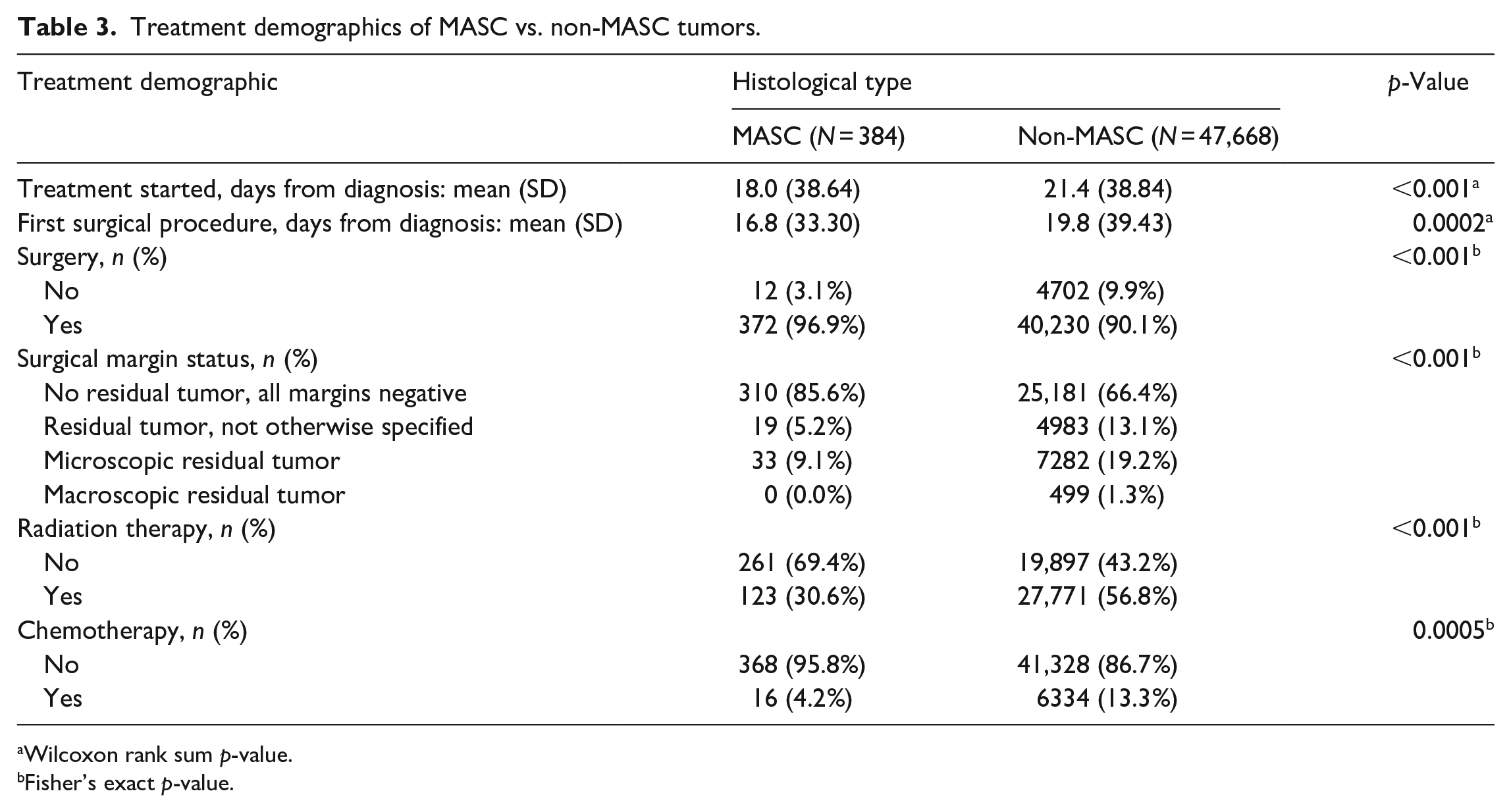
Wilcoxon rank sum
Fisher’s exact
Table 4 shows the difference in clinical grades of patients diagnosed with MASC and non-MASC tumors (
Clinical grades and AJCC pathologic staging of MASC vs. non-MASC tumors.

Fisher’s exact
Overall, there was a significant difference in the survival of MASC vs. non-MASC patients according to the log-rank test (unadjusted
The relationship between MASC cases and a select group of non-MASC cases (squamous cell carcinoma, adenocarcinoma, adenoid cystic carcinoma, mucoepidermoid carcinoma, lobular carcinoma, and acinar cell carcinoma) was also investigated (Supplemental Tables 1–7). Overall, these additional comparisons recapitulate the findings of MASC tumors affecting younger, male patients, leading to a less severe clinical course and better treatment-based outcomes. With adjustment for age, insurance type, clinical stage of tumor, pathological stage of tumor, and regional lymph node involvement, the risk of death was 4.1 times greater for the select group of non-MASC vs. MASC patients (HR = 4.1, 95% CI [2.30–7.48],
Discussion
MASC is a recently recognized histological type of salivary gland tumor, and despite previous attempts to characterize its clinical properties, a comprehensive analysis comparing MASC to non-MASC salivary gland tumors has not yet been reported in the literature. In comparison to non-MASC salivary tumors, our analysis demonstrates MASC tumors are more likely to affect younger patients, be well- to moderately-differentiated, have decreased rates of lymphovascular invasion, and have less nodal disease. In comparison to the findings published by Anderson
The literature contains previous reports differentiating MASC from other salivary gland tumors. In a retrospective analysis by Alves
A systematic review of salivary gland SCs reported 403 cases of MASC tumors in the current literature after histopathological reclassification of cases previously diagnosed as other salivary gland tumors.
26
Their findings recapitulate those described in the present study, namely, MASC diagnoses more commonly affected males, affected the parotid gland, and were commonly treated surgically, with isolated cases of tumor recurrence, metastasis, and death.
26
Furthermore, in a recent study by Silver
The present study highlights clinical distinctions among patients with both MASC and non-MASC salivary tumors. The
Our study is not without limitations in both data collection and review. As a retrospective analysis of a preexisting patient database, we are inherently limited by the data that are both reported and lacking within the database, in addition to inconsistent reporting across all patients. Therefore, we are unable to analyze individual patients’ genetic or histological records to confirm the accuracy of their diagnosis and must rely on the good faith of accurate reporting by clinicians to the National Cancer Database. In addition, our analysis includes data ranging from 2004 through 2019, despite MASC being classified as a distinct malignancy in 2010. Therefore, MASC tumors may have been misclassified as non-MASC diagnoses prior to 2010 and included in our analysis as non-MASC cancers. Our motivation for including data ranging from 2004 to 2019 was to maximize the number of patient diagnoses (of both MASC and non-MASC tumors) included in the analysis. Given its status as a rare disease, mortality data are available only for a limited number of MASC patients. Finally, power analysis for sample size calculation was not performed for this study; all MASC and non-MASC cases available within the 2004–2019 timeline were included in the study.
Due to its rare incidence and recent disease classification, the clinical features and natural history of MASC are still under investigation. Although our analysis of a comprehensive cancer database reveals demographic, clinical, and treatment differences in MASC vs. non-MASC salivary tumors, additional studies are needed to fully characterize this new diagnosis. We hope that as more data continue to become available, more effective therapeutic guidelines can be established for patients with this diagnosis.
Conclusions
Overall, the findings suggest that MASC salivary tumors are less aggressive than non-MASC malignant salivary tumors. Our data are consistent with prior reports and add additional measures not previously studied. MASC tumors were found to affect younger, male patients with the majority of cases isolated to the parotid gland, representing low AJCC staging, and demonstrating favorable histology. Furthermore, patients with MASC tumor diagnoses received treatment quicker than patients with non-MASC tumors, in addition to experiencing decreased mortality compared to patients with a non-MASC salivary tumor diagnosis. Clinicians must become aware of the unique characteristics of MASC tumors in how they can be differentiated from non-MASC tumors, thus helping guide patient care and management.
Supplemental Material
sj-docx-1-smo-10.1177_20503121231200103 – Supplemental material for Defining the clinical characteristics of mammary analog secretory carcinoma of the salivary gland: Analysis of the National Cancer Database
Supplemental material, sj-docx-1-smo-10.1177_20503121231200103 for Defining the clinical characteristics of mammary analog secretory carcinoma of the salivary gland: Analysis of the National Cancer Database by Akshilkumar Patel, Brandon LaBarge, Tonya S King, Sandeep Pradhan, Joshua Warrick and Neerav Goyal in SAGE Open Medicine
Footnotes
Acknowledgements
We thank Dr. Vonn Walter and Dr. Joseph Miccio for their expertise and assistance in gaining access to the updated National Cancer Database data used in this study. The findings in this article were shared through a poster presentation at the 2022 American Head & Neck Society Annual Meeting.
Authors’ Contribution
AP and BL conceptualized and designed the study; analyzed and interpreted the data; collected the data; wrote and revised the manuscript; and approved the manuscript. TSK and SP collected the data; wrote and revised the manuscript; approved the manuscript; and performed statistical analysis. JW wrote and revised the manuscript and approved the manuscript. NG conceptualized and designed the study; analyzed and interpreted the data; wrote and revised the manuscript; and approved the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
This study was exempt from requiring ethics approval by the IRB because it is a retrospective analysis of a de-identified, preexisting national oncologic data set.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was not supported by any grant or other source of funding.
Informed consent
This study was exempt from requiring written informed consent by the IRB because this study is a retrospective study analyzing a national oncologic database sourced from over 1500 cancer facilities and containing de-identified patient data. As such, informed consent is not possible to obtain.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
