Abstract
A herbal preparation, Shemamruthaa (SM), was formulated to investigate the molecular mechanism by which it exhibits anticancer effects in mammary carcinoma bearing rats. Female Sprague-Dawley rats were used for the study, and mammary carcinoma was induced by administration of 7,12-dimethylbenz(a)anthracene, intragastrically. After 3 months of induction period, the rats were treated with SM (400 mg/kg body weight) for 14 days. Our study shows that SM-treated mammary carcinoma rats showed regression in tumor volume with concomitant increase in p53, Bax, caspase-3, and caspase-9 mRNA and protein levels compared with mammary carcinoma–induced rats. Proliferating cell nuclear antigen and anti-apoptotic Bcl-2 were markedly increased in mammary carcinoma–induced rats, whereas the SM treatment significantly decreased the expression of these proteins. The expression pattern of apoptotic signaling molecules analyzed in the present study signifies the therapeutic efficacy of SM against breast cancer.
Breast cancer is the most common malignant disease affecting women worldwide, with 1 290 000 new cancer cases and 519 000 deaths annually. 1 It accounts for about one fourth of all cancers in Indian women and about half of all cancer-related deaths. It is the second leading cause of cancer death among women in India. 2 Despite the major scientific and technological advancement in the treatment and management of cancer, no reliable and definitive cure has been found. 3 Therefore, the identification of new and efficient anticancer drugs has always been a focal point in cancer research. 4,5
In countries such as India and China, use of herbal medicines is a very common practice from ancient time, and herbal medicines are considered to be much safer and less expensive therapeutic strategies for the treatment of various diseases. 6 Increasing side effects caused by chemotherapeutic agents and drugs have brought about a renaissance in the use of natural products–based drugs to minimize hazards caused by synthetic drugs. 7 More than 60% of the currently used anticancer agents are derived in one way or other from natural sources including plants and marine organisms. 8 The use of herbal drugs is once again becoming attractive in the form of food enhancements, nutraceuticals, and complementary and supplementary medicine. It has been shown that the phytochemicals present in fruits and vegetables or formulations are more effective than their individual constituents in preventing cancer through both additive and synergetic effects. 9,10 In this perspective, Shemamruthaa (SM), a herbal formulation, was evaluated for the first time with a view to study its anticancer property.
Hibiscus rosa-sinensis Linn (wild-type, red flower variety), commonly known as China rose, is a potent herb in the traditional system of medicine. 11 It is being used against cough, fever, dysentery, venereal diseases, and cancerous swellings. 12 There is an important evidence of the anticancer action of flowers against the effect of benzoyl peroxide and ultraviolet radiation–induced 2-stage skin carcinogenesis in mouse. 13 Hibiscus flowers are reported to contain antioxidants and other bioactive metabolites such as flavonoids, flavonoid glycosides, hibiscetin, cyanidine, and anthocyanin pigments. 14 Emblica officinalis Gaertn (Phyllanthus emblica Linn) has been used in Ayurveda, the ancient Indian system of medicine; Chinese herbal medicine; and Tibetan medicine. 15 Previous studies suggest that consumption of Emblica fruits can have beneficial effects against diseases such as cancer. Emblica fruits contains tannins, alkaloids, phenolic compounds, amino acids, carbohydrates, and the highest vitamin C content. 16 Honey was found to be rich in compounds known to possess anticancer properties, such as polyphenols and phenolic acids, and exhibits significant anticancer effects in human cancer cells. 17 –19 In order to promote intellect and prevent senility and for longevity, 20 honey was also added in SM.
Earlier reports from our laboratory have demonstrated that SM exerts profound anticancer activity through its role in reinstating the normal levels of glycoprotein components, revitalizing the membrane stability, restoring antioxidant status, and regulating lipid profile and lipid-metabolizing enzymes in dimethylbenz(a)anthracene (DMBA)–induced mammary cancer rats. 21 –25 The present study investigates the molecular mechanism underlying the anticancer effects of SM in DMBA-induced mammary carcinoma bearing rats.
Materials and Methods
Animal Care and Handling
The experimental design was performed in accordance with the current ethical norms approved by the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Environment & Forests (Animal Welfare Division), Government of India, and Institutional Animal Ethics Committee (IEAC) Guidelines (IAEC No. 01/030/2011). Animals were kept under a 12-hour light/dark cycle in a temperature-controlled environment, with free access to food and water.
Chemicals and Suppliers
All chemicals and reagents used in the present study were of molecular and analytical grade and were purchased from Sigma Chemical Company, St Louis, MO, USA; Amersham Biosciences, Buckinghamshire, UK; and Sisco Research Laboratories, Mumbai, India. Total RNA isolation reagent from Invitrogen (Grand Island, NY), reverse transcriptase enzyme from New England Biolabs (Ipswich, MA), and polymerase chain reaction (PCR) mastermix from Promega (Madison, WI) were used for the present study. The primers for PCNA, p53, Bax, Bcl-2, caspase-3, and caspase-9 and the β-actin monoclonal antibody were purchased from Sigma Chemical Company.
Cancer Induction
7,12-Dimethylbenz(a)anthracene was used as a carcinogen for the present investigation. Breast cancer was induced in overnight-fasted animals using a single dose of DMBA in olive oil (25 mg/kg body weight) by means of gastric intubation. 26,27
Experimental Design
Adult female Sprague-Dawley rats (8 weeks old) were divided into 4 groups having 8 animals in each group and given the following dose regimens: Group I, normal control animals received a single oral dose of olive oil (1 mL) at the age of 8 weeks (Control). Group II, animals induced for mammary carcinoma with a single oral dose of 25 mg of DMBA dissolved in 1 mL of olive oil after overnight fasting and received no treatment. After 2 months, the rats were palpated regularly to find out the appearance of mammary tumor(s). After 3 months, mammary carcinoma was confirmed by histological examinations (DMBA-induced). In group III, mammary carcinoma was induced as in group II; in addition, after 3 months, animals were treated with SM at the dose of 400 mg/kg body weight and continued for 14 days by gastric intubation (DMBA + SM treated). Group IV, animals received 400 mg/kg body weight of SM alone and served as SM control.
Evaluation of Antitumor Activity of SM
After the experimental period (ie, 3 months after DMBA induction and 14 days SM treatment), 6 animals from each group were sacrificed by cervical dislocation, tissues were collected, and tissues were stored under appropriate conditions. Blood was also collected for further analysis. Mammary tissues were excised and used for the assay of various parameters.
Size of the developing tumor was measured in both group II and III animals by using Vernier calipers. Tumor volume was calculated using the formula V = 0.5ab 2, where a and b indicate the major and minor diameters, respectively. 28,29
Isolation of Total RNA
Total RNA was isolated from control and experimental mammary tissues using total RNA isolation reagent from Invitrogen. The concentration and purity of RNA were determined spectrophotometrically at A260/280 nm. A ratio of absorbance of >1.8 was considered as good-quality RNA. 30 The purity of RNA obtained was 1.85. The yield of RNA is expressed in micrograms.
Reverse Transcriptase Polymerase Chain Reaction (RT-PCR)
Total RNA was used for the synthesis of complementary DNA (cDNA). The following specific oligonucleotide primers were used for the generation of complementary DNAs.
PCNA (amplicon size 354 bp)
Sense primer: 5′-AGTTTTCTGCGAGTGGGGAG-3′
Antisense primer: 5′-AAGACCTCAGAACACGCTGG-3′
P53 (amplicon size 372 bp)
Sense primer: 5′-TACTTCCCAGCAGGGTGTCA-3′
Antisense primer: 5′-GGTGGAAGCCATAGTTGCCT-3′
Bax (amplicon size 310 bp)
Sense primer: 5′-CAAGCCGGGAGAACAGGGTA-3′
Antisense primer: 5′-GAGGAAGTCCAGTGTCCAGC-3′
Bcl-2 (amplicon size 460 bp)
Sense primer: 5′-AGTTTTCTGCGAGTGGGGAG-3′
Antisense primer: 5′-CCCACCGAACTCAAAGAAGGC-3′
Caspase-3 (amplicon size 434 bp)
Sense primer: 5′-ACTGGAATGTCAGCTCGCAA-3′
Antisense primer: 5′-TAACCGGGTGCGGTAGAGTA-3′
Caspase-9 (amplicon size 293 bp)
Sense primer: 5′-CCCCAGTGACATCCTTGTGT-3′
Antisense primer: 5′-CCAGGCCATGAAGGCTTTTG-3′
β-Actin (amplicon size 195 bp)
Sense primer: 5′-GAG ATT ACT GCC CTG GCT CC-3′
Antisense primer: 5′-CAG CTC AGT AAC AGT CCG CC-3′
PCR products were separated by 2% agarose gel electrophoresis along with 100 bp marker DNA. Gels were densitometrically (Bio-Rad, Hercules, CA) scanned, and the cDNAs were normalized to that of the house keeping gene or internal control (β-actin), which was co-amplified along with the cDNA of interest.
Western Blot Analysis
The mammary tissue was homogenized in 20 mM Tris–HCl (pH 7.5), containing 150 mM NaCl, 1% Nonidet P40, 1 mM phenylmethylsulphonyl fluoride, and 1 μg/mL aprotinin. After incubation on ice for 30 minutes, the homogenate was centrifuged at 14 000 rpm for 10 minutes at 4°C. The protein concentration of the homogenate was estimated by the method of Lowry et al. 31 The samples were boiled with sample solubilizing buffer for 5 minutes and separated on 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis by the method of Lamelli. 32 The gel was transferred onto a nitrocellulose membrane (Hybond C+, Amersham Life Sciences) at 30 V for 3 hours. Membrane was then washed thrice with phosphate-buffered saline and blocking was done with Tris-buffered saline with Tween-20 (TBST buffer: 20 mM Tris, 500 mM NaCl, and 0.1% Tween-20, pH 7.5) containing 5% nonfat dried milk. Then, the membrane was incubated with primary antibody (rabbit monoclonal antibody, Bax, and Bcl-2 [Cell Signaling Technology]) and rabbit monoclonal antibody p53, PCNA, caspase-3, and caspase-9 (Sigma) in TBST buffer containing 1% nonfat dried milk and agitated gently at room temperature for 3 hours. After incubation with the primary antibody, the blots were washed thrice for 5 minutes with TBST buffer and incubated for 75 minutes at room temperature with goat rabbit anti-goat horseradish peroxidase conjugated secondary antibody in phosphate-free TBST buffer containing 5% nonfat dried milk. The bands were detected using chemiluminescence (Pierce).
Data Analysis
The data were subjected to statistical analysis using one-way analysis of variance and Duncan multiple range test to assess the significance of individual variations between the control and treatment groups using computer-based software (SPSS 16). In the Duncan test, the significance was considered at the level of P < .05.
Results
Effect of SM on Tumor Volume
Table 1 shows the mean body weight and tumor volume in different groups. Rats in the DMBA-induced group showed a tendency to be lower in body weight gain during the experiment and the mean final body weights were decreased significantly (P < .05) compared to DMBA + SM treated rats. No significant differences in the body weights were observed in groups I and IV. Administration of SM effectively decreased the tumor volume as well as the pathological changes. Furthermore, in groups I and IV, no tumors were observed, and the lobules and ductules were normal (Figure 1).
Effect of SM on changes in body weight, organs weight and tumor volume in control and experimental animals.

Abbreviations: DMBA, 7,12-dimethylbenz(a)anthracene; SM, Shemamruthaa. Values are expressed as mean ± SD of six animals in each group.
Comparison is made as:
aDMBA-induced vs. Control
bDMBA-induced vs. DMBA + SM treated
cControl vs. SM control
Statistical significance: *p < 0.05. NSNon Significant.

The gross appearance of the mammary adenocarcinoma noticed in DMBA alone (A) and tumor regression in DMBA + SM treated (B) rats. Normal mammary gland in control (C) and SM control animals (D).
Effect of SM on PCNA
Figure 2 depicts the level of PCNA in mammary tissues of control and experimental rats. DMBA-induced rats showed a marked increase in PCNA, whereas SM treatment significantly restored it to near normal level.
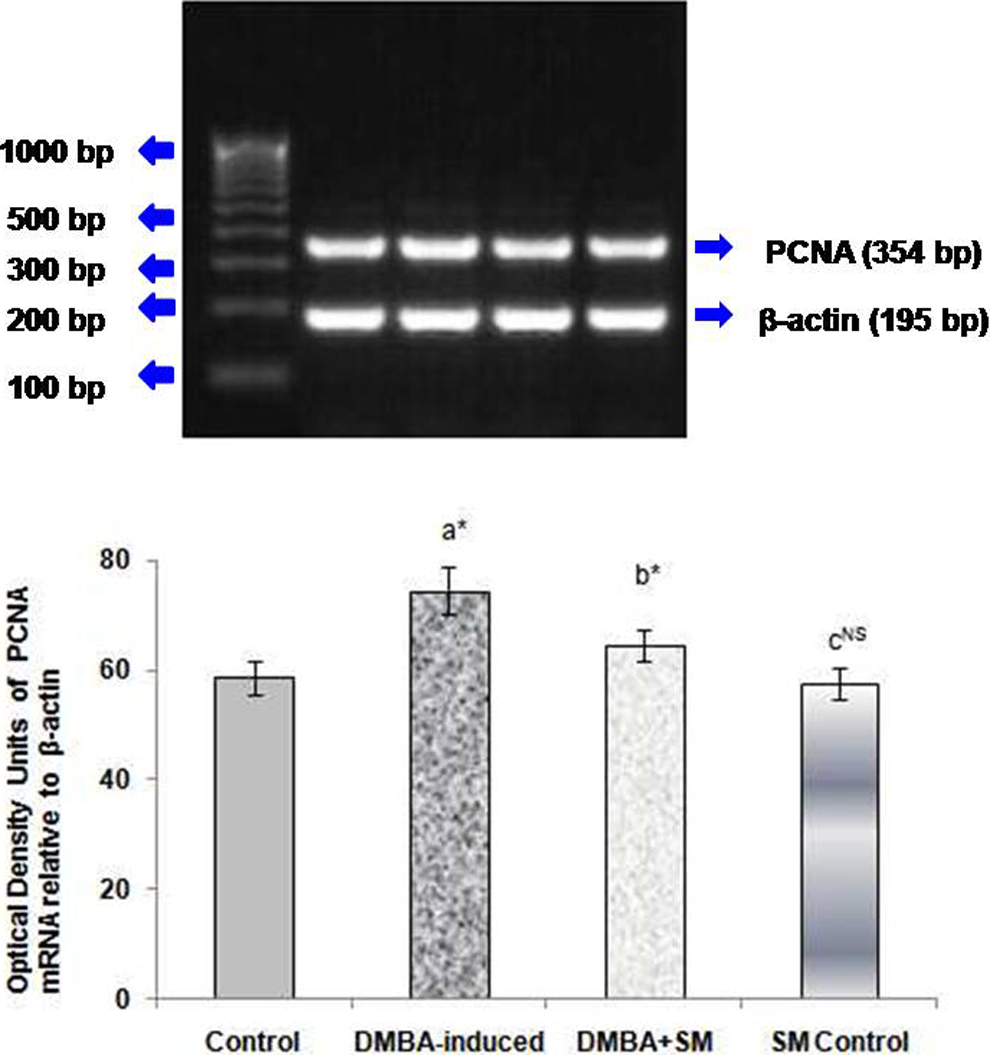
Effect of SM on PCNA mRNA level in the mammary tissues of control and experimental rats. Each bar represents mean ± standard deviation of six animals. Lane 1: 100 bp DNA marker, Lane 2: Control, Lane 3: DMBA-induced, Lane 4: DMBA + SM treated, Lane 5: SM Control.
Effect of SM on p53
The effect of SM on p53 expression in the mammary tissues of control and experimental rats is depicted in Figure 3. Downregulation of p53 was noticed in DMBA-induced rats (Group II). SM treatment at a dose of 400 mg/kg body weight to DMBA-induced cancer rats significantly restored the expression of the above marker.
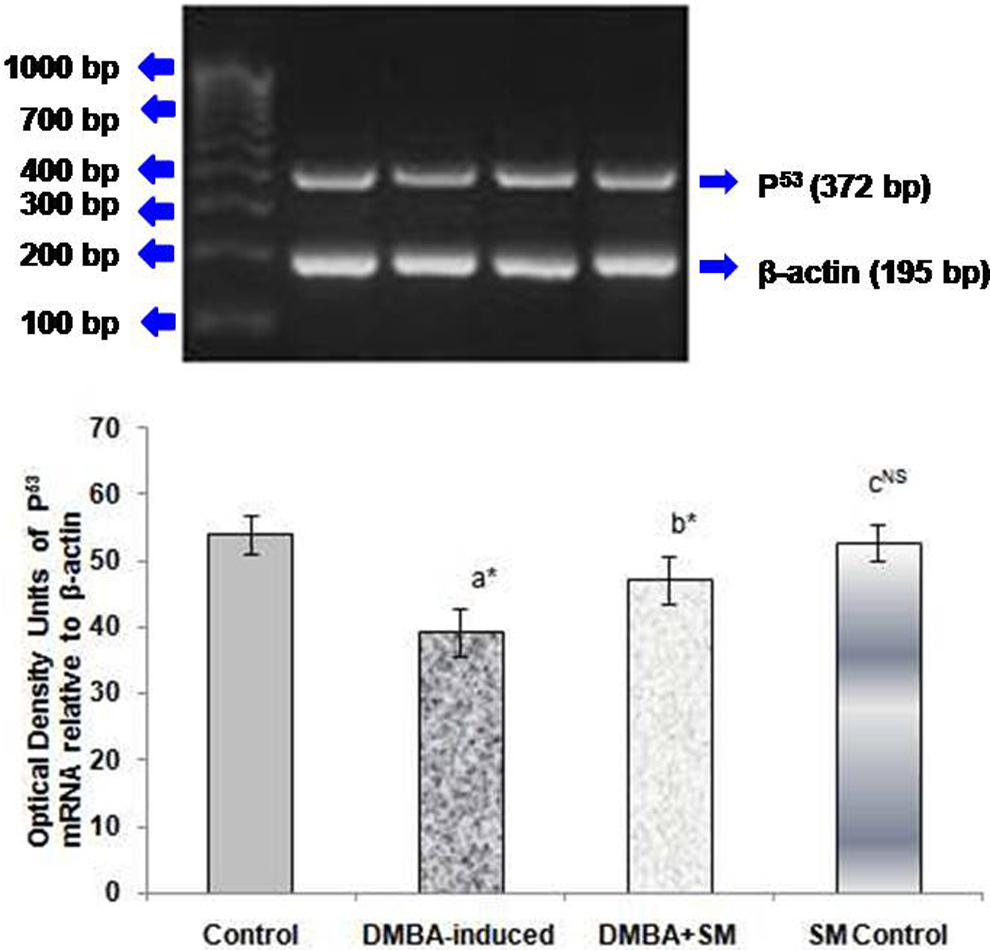
Effect of SM on p53 mRNA level in the mammary tissues of control and experimental rats. Each bar represents mean ± standard deviation of six animals. Lane 1: 100 bp DNA marker, Lane 2: Control, Lane 3: DMBA-induced, Lane 4: DMBA+ SM treated, Lane 5: SM Control.
Effect of SM on Bax, Bcl-2, and Caspases
In order to point out the pro-apoptotic effects of SM in our study, the specimens of mammary tissues from all experimental groups were evaluated for mRNA and protein expression of anti-apoptotic Bcl-2 and pro-apoptotic Bax genes. In DMBA-induced animals (group II), the expression of Bcl-2 was significantly higher, and that of Bax, caspase-3, and caspase-9 were significantly lower compared to control animals (group I). SM treatment (400 mg/kg body weight) significantly decreased expression of Bcl-2 (Figure 4) and increased the Bax (Figure 5), caspase-3 (Figure 6), and caspase-9 (Figure 7) expressions compared to the DMBA-induced group. Rats treated with SM alone (group IV) revealed expression similar to that of normal control rats (group I).

Effect of SM on Bcl-2 mRNA level in the mammary tissues of control and experimental rats. Each bar represents mean ± standard deviation of six animals. Lane 1: 100 bp DNA marker, Lane 2: Control, Lane 3: DMBA-induced, Lane 4: DMBA + SM treated, Lane 5: SM Control.

Effect of SM on Bax mRNA level in the mammary tissues of control and experimental rats. Each bar represents mean ± standard deviation of six animals. Lane 1: 100 bp DNA marker, Lane 2: Control, Lane 3: DMBA-induced, Lane 4: DMBA + SM treated, Lane 5: SM Control.

Effect of SM on caspase-3 mRNA level in the mammary tissues of control and experimental rats. Each bar represents mean ± standard deviation of six animals. Lane 1: 100 bp DNA marker, Lane 2: Control, Lane 3: DMBA-induced, Lane 4: DMBA + SM treated, Lane 5: SM Control.

Effect of SM on caspase-9 mRNA level in the mammary tissues of control and experimental rats. Each bar represents mean ± standard deviation of six animals. Lane 1: 100 bp DNA marker, Lane 2: Control, Lane 3: DMBA-induced, Lane 4: DMBA + SM treated, Lane 5: SM Control.
Figure 8 shows the effect of SM on p53, PCNA, Bcl-2, Bax, caspase-3, and caspase-9 protein levels in the mammary tissues of control and experimental rats. PCNA and Bcl-2 proteins were significantly decreased, while p53, Bax, caspase-3, and caspase-9 proteins were significantly increased in DMBA-induced mammary carcinoma rats. SM treatment reestablishes the expression of the above-mentioned proteins. Overexpression of PCNA and Bcl-2 observed in the mammary tumor tissues of DMBA-induced rats indicates abnormal cell proliferation and evasion of apoptosis.

Effect of SM on p53, PCNA, Bcl-2, Bax, caspase-3 and caspase-9 proteins levels in the mammary tissues of control and experimental rats using Western blot analysis.
Discussion
In the current study, we have evaluated the anticancer property of a herbal preparation Shemamruthaa (SM) using a DMBA-induced mammary carcinoma rat model. The chemotherapeutic effects of SM observed in the present study may be related to the constituent phytochemicals. Gas chromatography–mass spectrometry analysis revealed the presence of pyrogallol, 5-hydrxoymethylfurfural, dipalmitin, and trilinolein as the major bioactive constituents, and gallic acid, quercetin, caffeic acid, rutin, and ferulic acid in the SM validating the high-performance liquid chromatography data reported by us earlier. 25 Pyrogallol, a catechin compound, is an active component of Emblica officinalis and has been documented to exert an antiproliferative effect on some human cancer cell lines, H441 (lung adenocarcinoma) and H520 (lung squamous cell carcinoma). 33 Trilinolein, bioactive constituent of SM, has also been reported to induce the upregulation of pro-apoptotic Bax and downregulation of anti-apoptotic Bcl-2 expression, which was associated with the proteolytic activation of caspases and the concomitant degradation of poly(ADP-ribose) polymerase (PARP) protein. 34 Quercetin, a bioflavonoid present in SM, has also been reported to downregulate signal transduction pathways in human breast carcinoma cells. Furthermore, quercetin inhibits tumor cell growth by downregulating the expression of PCNA, GST-P, NF-jB, and Bcl-2 and upregulating caspase-3 expression. 35,36 Thus, SM offers promise as a potential anticancer drug due to its ability to modulate key hallmark capabilities of cancer cells such as cell proliferation and apoptosis.
There was a sharp fall in the body weight and weights of organs (liver and kidneys) in mammary carcinoma–induced animals. This may be due to tumor cachexia, characterized by weakness, lethargy, anorexia, depletion of host components, tissue wasting, and a progressive waning of vital functions. 37 The SM-treated animals showed a gradual increase in their body weights indicating the counteractive property of SM. Many studies in different cell lines, animal models, and human epidemiological trials have shown the potential of dietary polyphenols as anticarcinogenic agents. 38,39 The polyphenolic compounds, flavonoids, ascorbic acid, β-carotene, and other bioactive components present in the SM might influence the growth due to their antioxidant, immunopotentiating, and anti-inflammatory properties.
The SM-treated rats showed a significant reduction in tumor volume when compared with DMBA-induced mammary carcinoma rats. This might be due to the protective effect of flavonoids present in the SM, since flavonoids are known to display a vast array of cellular events, they can control the overall process of carcinogenesis by several mechanisms including modulation of survival/proliferation pathways, 40,41 activation of caspases, 40 downregulation of Bcl-2 and Bcl-xL expression and enhanced expression of Bax and Bak, 42,43 and modulation of nuclear factor κB, 44 which might be responsible for tumor regression in SM-treated animals.
Proliferating cell nuclear antigen (PCNA), a 36 kDa protein that functions as a cofactor of DNA polymerase δ, 45 plays a pivotal role in the cell cycle and is an important proliferative marker for evaluating several cancers including breast cancer. Its expression level is high during S-phase of cell cycle where active multiplication of cells takes place. Elevated expression of PCNA in the mammary tissues of DMBA-induced rats indicates the massive proliferation of tumor cells and is consistent with similar findings by other researchers. 46,47 SM-treated rats resulted in decreased expression of this proliferative marker, which clearly revealed its antiproliferative efficacy.
The most common anti-apoptotic lesion that is detected in cancers is the inactivation of the p53 tumor-suppressor pathway. Several responses can be provoked by p53, including DNA repair, cell-cycle arrest, senescence, differentiation, and apoptosis, with the option chosen being dependent on many factors that are both intrinsic and extrinsic to the cell. 48 Hence, the effect of SM on p53 expression in mammary tissues was analyzed using RT-PCR and western blot analyses. In DMBA-induced mammary carcinoma bearing rats, a significant decrease was observed in p53. In contrast to frequent p53 mutations in human breast cancer, DMBA-induced breast cancer is rarely associated with p53 alterations, although such alterations have been well documented. 49,50 The reduced activity of p53 observed in DMBA-induced rats (group II) may be due to overexpression of mouse double minute 2 homolog (Mdm2) and other cellular factors that downregulate its level. 51 SM treatment showed an elevation in the expression of p53 in parallel with Bax, caspase-3, and caspase-9 when compared with DMBA-induced rats. In most cases, induction of p53 leads to an irreversible inhibition of cell growth, most decisively by activating apoptosis. 52 Earlier studies by Srivastava et al 53 and Kim et al 54 have reported the increased expression of p53 during treatment with herbal extracts. Therefore, it can be inferred that the inhibition of mammary cancer by SM may be mediated through the elevated expression of the p53 gene.
In order to point out the pro-apoptotic effects of SM in our study, the specimens of mammary tissues from all experimental groups were evaluated for mRNA and protein expression of anti-apoptotic Bcl-2 and pro-apoptotic Bax genes along with caspase-3 and caspase-9. The increase in Bcl-2 expression with downregulation of Bax (increase in Bcl-2/Bax ratio) provides an evidence for apoptosis evasion in DMBA-induced rats. 55 Dysregulation of apoptosis due to an increase in Bcl-2/Bax ratio has been suggested to contribute to the pathogenesis of breast cancer. 56 On SM treatment (400 mg/kg body weight), pro-apoptotic shift in Bax/Bcl-2 mRNA expression ratio was confirmed in our study. The decrease in Bcl-2 expression, a reliable indicator of the overall propensity of a cell to undergo apoptosis, coupled with activation of caspase-3 and caspase-9 underscore the apoptosis-inducing potential of SM.
The antiproliferative and apoptotic effects of SM observed in the present study may be related to the constituent phytochemicals. Quantitative analysis of SM showed the presence of 62.67 mg GAE of total phenolics, 21.35 mg QE/g of flavonoids, and 9.74 mg QE of proanthocyanidins per gram of extract. Gas chromatography–mass spectrometry analysis revealed the presence of 5-hydrxoymethylfurfural(2-furancarboxaldehyde,5-(hydroxymethyl)-), dipalmitin and trilinolein, and gallic acid (992.25 μg/g), quercetin (335.75 μg/g), caffeic acid (156.6 μg/g), rutin (6.0 μg/g), and ferulic acid (0.3 μg/g) in the SM validating the high-performance liquid chromatography data reported by us earlier. 25 Trilinolein has been documented to exert antiproliferative and apoptosis inducing effects on cancer cell lines. 34,57 Hydrxoymethylfurfural at a concentration of (300 μg/mL) was tested to study the antiproliferative activity associated with this compound. At this concentration, the HMF inhibited the proliferation of colorectal adenocarcinoma cell lines HT-29 and SW-480. 58 In view of recent data, it can be inferred that hydroxymethylfurfural might have antitumor potential. 59 Thus, the abundance of HMF in the SM implicates its possible role in the antiproliferative activities observed in the present study. Gallic acid, ferulic acid, and caffeic acid, the flavonoid constituents of SM, have been reported to exhibit antiproliferative, pro-apoptotic, and of antitumor effects in preclinical models of human breast cancer animal models. 60 –64 Therefore, the anticancer and apoptotic effect is possibly due to the synergistic effect of these compounds present in the formulation.
Conclusions
Based on the results, it might be concluded that the formulation, Shemamruthaa, constituted of dried flowers of Hibiscus rosa-sinensis, fruits of Emblica officinalis, and honey has been found to exhibit pronounced antiproliferative and apoptotic effect. Furthermore, our studies revealed that it can modulate the expression of p53 and can activate the mitochondrial pathway of apoptosis to abrogate cancer cell proliferation. Thus, our study clearly shows that the herbal preparation SM can offer an effective, holistic approach for the management of breast cancer.
Footnotes
Acknowledgments
The authors would like to thank Mr S. Kumaravel, Senior Scientist, Food Testing Laboratory, Indian Institute of Crop Processing Technology (Ministry of Food Processing Industries, Government of India), Tanjavur 613005, Tamil Nadu, India, for providing gas chromatography–mass spectrometry and high-performance liquid chromatography facilities.
Author Contributions
AP was responsible for conducting experiments, analyzing data, and writing the manuscript. EN supported in conducting experiments and the preparation of the manuscript. PS helped in histopathological analysis and offered valuable suggestions for the study. TPS provided commentary and analysis of the biochemical and molecular aspects and significance of the findings. He also participated in the preparation of the manuscript and had the overall editing responsibilities.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The experimental design was performed in accordance with the current ethical norms approved by the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Environment & Forests (Animal Welfare Division), Government of India, and Institutional Animal Ethics Committee (IEAC) Guidelines (IAEC No. 01/030/2011).
