Abstract
Vitamin D deficiency is prevalent across all geographic areas, age groups, and seasons and is increasing in incidence. Vitamin D receptors have been found to be present in almost every body cell and tissue. Clinical research has alluded to a preventive effect of vitamin D supplementation on cardiovascular disorders such as myocardial infarction, hypertension, and dyslipidemia. Furthermore, a low serum vitamin D level has been associated with an increased risk for cardiovascular disease. This article examines the association between serum vitamin D levels and cardiovascular disease mortality and risk.
Vitamin D is a secosteroid derived from diet or ultraviolet (UV)-B radiation in the epidermis. 1,2 Dietary sources rich in vitamin D include egg yolks and animal fat (cholecalciferol or vitamin 25(OH)D3) and green vegetables (ergocalciferol or vitamin 25(OH)D2). 1,2 Vitamin D receptors have been found to be present in almost every body cell and tissue. 1 –3 Plasma or serum 25(OH)D is the most stable and abundant vitamin D metabolite in the bloodstream and is, therefore, the best measure of vitamin D status, achieved through either sunlight or diet. 1,4
Vitamin D deficiency is prevalent across all geographic areas, age groups, and seasons and is increasing in incidence. 1,5 Risk factors for vitamin D deficiency include obesity, decreased exposure to sunlight, decreased cutaneous synthesis related to age, low dietary intake, and high degree of skin pigmentation. 1 Table 1 reports other behaviors and conditions affecting risk for vitamin D deficiency.
Behaviors and Conditions Affecting Risk for Vitamin D Deficiency a

There is no consensus among experts on the optimum serum 25(OH)D level. Thus, recommendations regarding serum 25(OH)D are varied, depending on the reference. For most populations, the optimal range for serum 25(OH)D levels is at least 30 to 32 ng/mL (75-80 nmol/mL). 1,3 However, Bischoff-Ferrari et al 6,7 report that serum 25(OH)D levels of 30 to 40 ng/mL (75-100 nmol/L) have been demonstrated to provide optimal benefits without an associated increase in health risk. An even higher range, 40 to 50 ng/mL (100-125 nmol/L), has also been reported as the ideal goal for serum 25(OH)D levels. 3
It has been speculated that more than 75% of Americans have abnormally low levels of serum 25(OH)D. 5 Data from the National Health and Nutrition Examination Survey showed that 75% to 80% of subjects had serum 25(OH)D levels of <30 ng/mL (<75 nmol/L), numbers that experts unanimously agree are too low. 1,5 The United States Institute of Medicine, however, believes a 75% approximation of vitamin D deficiency in Americans to be an overestimation. The Institute of Medicine reports that 25% of Americans have inadequate levels of serum 25(OH)D and that only 8% of Americans are at risk for deficiency. 3 The Institute of Medicine also estimates that 1% of Americans have serum 25(OH)D levels that are too high and, consequently, possibly causing harm. 3 Serum 25(OH)D levels ranging from deficient to intoxicated are classified in Table 2.
Serum 25 (OH)D Levels Ranging From Deficiency to Intoxication a

b To convert from ng/mL to nmol/L, multiply by 2.496.
Some reports of vitamin D supplementation note no adverse or toxic effects with dosages as high as 10 000 IU/day. 8,9 However, when dosages are equal to or greater than 50 000 IU/day, over weeks to months, toxic adverse events such as hypercalcemia leading to hypercalciuria and eventual renal and cardiovascular failure result in death from vitamin D toxicity. 8 Despite acknowledging no adverse events with dosages as high as 10 000 IU/day, 8 the Institute of Medicine currently recommends more conservative daily allowances (see Table 3). 10
Institute of Medicine Dietary Reference Intakes for Vitamin D a
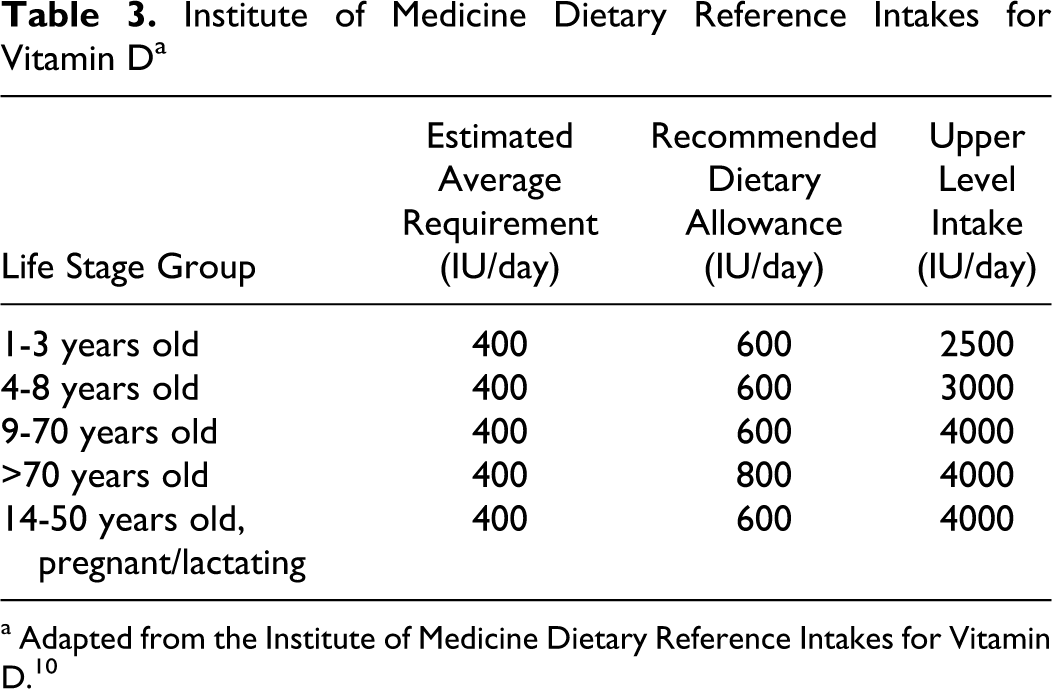
a Adapted from the Institute of Medicine Dietary Reference Intakes for Vitamin D. 10
Cardiovascular Effects
The effect of vitamin D supplementation on bone health is well documented. 11,12 Heart disease is currently the leading cause of death for both men and women, with a $316.4 billion estimated annual cost in the United States alone. 13 Emerging evidence suggests that optimal serum 25(OH)D levels could also improve cardiovascular outcomes, such as reducing hypertension, improving lipid management, and reducing incidence of myocardial infarction and cardiovascular mortality. 14 –19 Several observational studies have linked vitamin D deficiency with a higher incidence of cardiovascular disease in the general population. 14,15,20 Epidemiological studies note a proportional increase in the rate of both coronary heart disease and hypertension in those living farther away from the equator. 3 This increased rate of cardiovascular disease in locations farther away from the equator could potentially suggest an association with lower serum vitamin D levels. 3
Vitamin D regulates many genes either directly or indirectly, including those involved in the proliferation of cardiac muscle cells, vascular smooth muscle, macrophages, and lymphocytes. 3 Vitamin D is also postulated to improve cardiovascular health through multiple pathways, including inhibition of vascular smooth muscle proliferation, increased regulation of inflammatory mediators, suppression of vascular calcification, and improved modulation of the renin–angiotensin system. 14,21 This article examines the association between serum 25(OH)D levels and cardiovascular disease mortality and cardiovascular disease risk.
Methods
An electronic search was conducted to identify studies from 2006 to 2012 in the following databases: CINAHL, Medline, PsycInfo, and the Cochrane library. Search terms included vitamin D, cardiovascular, cardiac, myocardial infarction, hypertension, and lipids. The search was limited to English, peer-reviewed studies. Using these criteria, 54 articles were initially identified. Studies were further limited to clinical trials, meta-analyses, and epidemiological studies. Studies were excluded if animals were used as subjects. References from articles were reviewed for additional relevant studies and reviews; multiple resources were identified, bringing the total number of articles for inclusion to 36.
Results
Cardiovascular Mortality
Without supplementation
A prospective cohort study of 3258 patients referred for elective coronary angiography and followed for 7 years demonstrated an independent association between low serum 25(OH)D and cardiovascular mortality (hazard ratio [HR] = 2.22, 95% confidence interval [CI] = 1.57-3.13 for the lowest serum 25(OH)D quartile [median = 7.3 ng/mL] when compared with patients in the highest serum 25(OH)D quartile [median = 28.4 ng/mL]). 22 However, the researchers noted that it had yet to be proven by interventional trial that a causal relationship exists between vitamin D and mortality. 22
In another prospective study, Ginde and colleagues 15 demonstrated an independent, inverse association between baseline serum 25(OH)D levels and cardiovascular disease mortality and all-cause mortality in the Third National Health and Nutrition Examination Survey. Compared with subjects with serum 25(OH)D > 40 ng/mL, the adjusted hazard ratio HR of all-cause mortality for subjects with serum 25(OH)D < 10 ng/mL was 1.83 (95% CI = 1.14-2.94). 15 There was an even stronger association in subjects with lower serum 25(OH)D levels (<10 ng/mL) when compared with subjects with higher serum 25(OH)D levels (>40 ng/mL) for cardiovascular disease mortality (adjusted HR = 2.36; 95% CI = 1.17-4.75). 15
Researchers examining 2312 healthy subjects from the Cardiovascular Health Study noted at the 14-year follow-up a greater incidence of sudden cardiac death in subjects with lower serum vitamin D levels. 23 Researchers documented 1 sudden cardiac death events per 1000 in subjects with serum vitamin D levels ≥20 ng/mL and 4 events per 1000 in subjects with serum 25 (OH)D levels <20 ng/mL. 23 While these researchers also examined parathyroid hormone levels, they concluded that the combination of lower serum 25(OH)D levels and higher parathyroid hormone levels were associated with an increased risk of sudden cardiac death in adults presenting without cardiovascular disease as baseline. 23
With vitamin D supplementation
In a meta-analysis of randomized controlled trials, Autier and Gandini 24 examined the risk of dying from any cause in subjects who participated in randomized controlled trials testing the effect of vitamin D supplementation on any health condition. Subjects were followed for a mean of 5.7 years. Daily doses of vitamin D supplementation ranged from 300 to 2000 IU, with a trial size-adjusted mean dose of 528 IU/day. They identified a modest, but statistically significant reduction in the relative risk (RR) of all-cause mortality (RR = 0.92; 95% CI = 0.87-0.99) with vitamin D supplementation. 24 However, they noted their meta-analysis was not powered sufficiently to identify cause of mortality or variation in specific populations; nor could it account for baseline vitamin D status.
In an investigation to determine whether vitamin D or calcium supplementation affected vascular disease mortality or cancer in the elderly, 5292 subjects aged 70 years and older were studied. 25 Participants were randomly assigned to 1 of 4 groups: vitamin D3 800 IU, calcium 1000 mg, both, or placebo. These groups were supplemented for 24 to 62 months and then received follow-up 3 years postintervention. There was no difference in vascular disease mortality (HR = 1.07; 95% CI = 0.92-1.24). 25 However, in post hoc analysis, adjusting for compliance with fewer participants, trends for reduced mortality with vitamin D supplementation were accentuated but still remained nonsignificant. Vitamin D supplementation did not appear to affect mortality or vascular disease in this long-term follow-up study. The researchers noted that they chose a 3-year postintervention follow-up because the effects of calcium supplementation tend to diminish 2 years after stopping supplementation. 25
In the Cardiovascular Health Study, 1018 subjects were studied over a median of 11 years. 26 Researchers discovered that serum 25(OH)D levels of 20 ng/mL or less were associated with an increased risk for clinical disease events. They also determined season-specific serum 25(OH)D concentrations for winter, spring, summer, and fall, which were, respectively, 17, 20, 24, and 22 ng/mL. 26 Because of these results the authors postulate that using season-specific targets for serum 25(OH)D levels can be better than using standard normal ranges when evaluating risks to health. 26
Myocardial Infarction
The modest decrease in all-cause mortality reported by Autier and Gandini 24 could have been due to vitamin D’s effects on vasculature. Vitamin D has been purported to affect vascular smooth muscle proliferation, inflammation, vascular calcification, and blood pressure, all of which affect the risk of cardiovascular disease and myocardial infarction. 21
However, the only randomized controlled trial identified examining the effect of vitamin D supplementation on myocardial infarction failed to find a significant effect of supplementing calcium carbonate 1000 mg and vitamin D3 400 IU daily over 7 years. 27 This randomized controlled trial was associated with the Women’s Health Initiative, in which the dose of vitamin D was likely too low to have an effect, adherence was poor, and concurrent calcium and vitamin D supplementation was allowed, thus impairing the ability of researchers to identify any treatment effect. 27
Despite inconclusive results from the Women’s Health Initiative, observational data seem to be compelling for an effect of serum vitamin D and myocardial infarction risk. In a prospective study of 18 225 men with no known coronary disease, Giovannucci et al 14 observed that subjects with serum 25(OH)D levels < 15 ng/mL are at an increased risk for myocardial infarction compared with those with sufficient serum 25(OH)D levels (>30 ng/mL; RR = 2.09; 95% CI = 1.53-3.84). This 2-fold increase in risk of myocardial infarction was present even after controlling for factors known to be associated with coronary artery disease, including family history of myocardial infarction, history of diabetes mellitus or hypertension, dyslipidemia, body mass index, and physical activity. 14
In another observational study, Lee et al 16 found that among a multicenter myocardial infarction registry, 75% of patients were vitamin D deficient (serum 25(OH)D ≤ 20 ng/mL); the researchers reported 96% of myocardial infarction patients presented with abnormally low serum 25(OH)D levels (<30 ng/mL). Interestingly, this report far exceeds the estimated 25% of the American population with serum 25(OH)D levels < 30 ng/mL reported by the Institute of Medicine. 3
Hypertension
Epidemiological data from the National Health and Nutrition Examination Survey links vitamin D deficiency with hypertension. 18,19 Several interventional studies have included blood pressure as a secondary end point with mixed results. 27 –30 No significant change in blood pressure was reported by Maki et al, 28 who primarily examined the effect of 1200 IU vitamin D/day on serum lipids, or by Zittermann et al, 29 who studied the effect of 3332 IU vitamin D/day on several cardiovascular risk factors. Although Jorde et al 30 documented a slight but statistically significant increase in diastolic blood pressure with vitamin D supplementation when compared with the placebo group, Hsia et al 27 reported a slight but significant decrease in diastolic blood pressure and increase in systolic blood pressure in the treatment group during the first 2 years of follow-up in the Women’s Health Initiative randomized controlled trial on bone fracture; these differences in groups became insignificant, however, through later years of follow-up.
In a meta-analysis examining the effect of vitamin D supplementation on blood pressure, Witham et al 31 found weak evidence for a positive effect. No reduction in blood pressure was seen in those who were normotensive at baseline. Among those hypertensive at baseline, researchers reported no significant reduction in systolic blood pressure (−3.6 mm Hg; 95% CI = −8 to 0.7) and a small but statistically significant reduction in diastolic blood pressure (−3.1 mm Hg; 95% CI = −5.5 to −0.6). 31 Significant heterogeneity was present between studies included in the meta-analysis by Witham et al, 31 in addition to multiple self-reported methodological weaknesses; the researchers disclosed that results should be treated with considerable caution.
Even if no true effect of vitamin D supplementation on blood pressure exists, there could still be beneficial outcomes from supplementation among hypertensive patients. Wang et al 20 examined baseline serum 25(OH)D levels and the associated risk of cardiovascular events in 1739 Framingham Offspring Study participants without prior know cardiovascular disease. Cardiovascular disease events included myocardial infarction, coronary insufficiency, angina, stroke, transient ischemic attack, peripheral claudication, or heart failure. Individuals with serum 25(OH)D < 15 ng/mL had a multivariable-adjusted hazard ratio of 1.62 (95% CI = 1.11-2.36; P = .01) for incident cardiovascular events compared with serum 25(OH)D > 15 ng/mL. 20 The effect was evident in those with hypertension (HR = 2.13; 95% CI = 1.30-3.48; P = .003) but not in those without hypertension (HR = 1.04; 95% CI = 0.55-1.96; P = .90), suggesting that vitamin D deficiency somehow exacerbates the harmful effects of hypertension. 20
Serum Lipids
The increased rate of cardiovascular mortality among vitamin D–deficient subjects demonstrated in prospective studies may be explained, partially, by the theorized beneficial effects of vitamin D on serum lipids. 15,22,24 In a cross-sectional analysis, Maki et al 17 observed that each additional 10 ng/mL of serum 25(OH)D corresponded to a 4.2 mg/dL increase in high-density lipoprotein cholesterol (P < .01). However, in a randomized controlled trial, supplementation with vitamin D3 at a dose of 1200 IU/day over 8 weeks failed to affect cardiovascular disease markers such as total cholesterol, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, non-high-density lipoprotein cholesterol, and triglycerides in subjects with elevated waist circumference. 28 These results came while documenting a mean increase of 4.9 ± 1.2 ng/mL in serum 25(OH)D levels in the treatment group versus 0.5 ± 1.0 in the placebo group (P = .003). 28 In a study of 1529 postmenopausal women, no significant difference was noted on serum lipids from supplementation with 1 g calcium carbonate and 400 IU vitamin D3/day over 5 years. 32 A Norwegian study raised serum 25(OH)D levels 31.8 ± 12.5 ng/mL from a baseline 23.2 ± 8.5 ng/mL with 40 000 IU vitamin D3 weekly over 12 months (P < .001 vs the placebo group), yet failed to demonstrate an effect on serum lipids. 30 However, serum lipids were secondary endpoints in this study; the researchers disclosed the study was not designed or powered to detect such effects. 30
In contrast to these equivocal results of vitamin D supplementation, Zittermann et al 29 demonstrated a reduction in triglycerides (−13.5% vs +3% with placebo, P < .001) while noting an unexpected increase in low-density lipoprotein cholesterol (+5.4% vs −2.5%, P < .001); high-density lipoprotein cholesterol appeared unaffected. All subjects in this study were overweight and significantly vitamin D deficient (mean baseline serum 25(OH)D = 12 ng/mL); they were supplemented at a dose of 3332 IU vitamin D3 daily or with placebo for 12 months while participating in a weight reduction program. 29 Reporting similar results, Salehpour et al 33 also demonstrated in a randomized controlled trial an increased low-density lipoprotein cholesterol and total cholesterol while documenting an improved high-density lipoprotein cholesterol by supplementing 77 healthy premenopausal overweight and obese women with 1000 UI vitamin D3 daily for 12 weeks.
In a retrospective chart review, significant reductions were reported in total cholesterol, low-density lipoprotein cholesterol, and triglycerides (−48.6 mg/dL, −45.5 mg/dL, −44.7 mg/dL, respectively; P < .001), by use of statin therapy, niacin and/or omega-3 fatty acid supplementation, and vitamin D supplementation titrated to achieve serum 25(OH)D levels 50 to 60 ng/mL. 34 It should be noted that this study had several reported methodological weaknesses, including an all-white, highly motivated population and the lack of a control group. Given the design of the study, it is not possible to determine which variables were responsible for observed outcomes, and results should be interpreted with caution. 34
Discussion
While evidence for epidemiological and clinical studies tend to support an association between vitamin D deficiency and the incidence of cardiovascular disease, caution must be taken due to the number of potential confounding variables that are not possible to control. 35 Observational data are compelling, but not adequate for establishing causation. Because interactions between vitamin D, calcium, and parathyroid hormone are complex, separating the effects distinct to vitamin D in clinical trial can be difficult. 35 Furthermore, Ullah et al 35 note statistical significance in a study does not prove that one factor has caused another; these 2 factors may be closely related to a third. 35 Since there is increasing evidence linking vitamin D deficiency and cardiovascular disease, treating hypovitaminosis D in patients at risk could be prudent. 35 Furthermore, a recent Endocrine Society’s scientific statement notes that research is inconsistent on the associated benefits of supplemental vitamin D outside of bone health effects. 36
Recommendations for Future Research
Few randomized controlled trials to date have used vitamin D doses that would raise serum 25(OH)D levels above 30 ng/mL. 28,30 As such, appropriate hope remains about potential cardiovascular benefits of vitamin D supplementation. Randomized controlled trials that primarily evaluate the effects vitamin D supplementation doses in the range of 2000 to 5000 IU/day on cardiovascular health outcomes are needed. 12 Large randomized controlled trials are needed along with dose–response data to determine the effects of vitamin D on heart disease and hypertension. 36
Current Recommendations
The Institute of Medicine currently recommends a dietary intake of 600 IU/day of vitamin D for all adults aged 18 to 70 years and 800 IU/day for adults aged 70 years and older with an upper level intake of 4000 IU/day for all adults. 10 Holick et al 12 propose supplementation with at least 1000 IU of vitamin D per day to guarantee vitamin D sufficiency in normal circumstances. Screening for vitamin D deficiency should only be performed for those individuals at risk for deficiency (see Table 1). 12 At present, the Endocrine Society recommends that all adults who are vitamin D deficient be treated with 50 000 IU of vitamin D2 or vitamin D3 once a week (or the equivalent dose of 6000 IU/day) for 8 weeks to achieve rapid repletion, followed by a maintenance dose of 1500 to 2000 IU/day. 12 Supplementation of vitamin D doses at least 2 to 3 times larger may be needed for obese individuals (body mass index > 30 kg/m2; body fat sequesters the fat-soluble vitamin) and individuals on anticonvulsant medications, glucocorticoids, antifungals (such as ketoconazole), or medications for AIDS (these medications increase the catabolism of 25(OH)D). 12
Conclusion
In conclusion, there are a large number of observational studies demonstrating an association between vitamin D deficiency and multiple adverse cardiovascular outcomes, including dyslipidemia, hypertension, myocardial infarction, and cardiovascular disease mortality. Yet observational data do not establish causation. Though lacking sufficient evidence for the benefits of vitamin D supplementation in reducing risk of cardiovascular disease mortality, the literature does give reassurance that long-term supplementation with ordinary doses of vitamin D does not seem to be associated with an adverse effect. However, it is premature to prescribe vitamin D supplementation beyond recommended daily needs for preventing cardiovascular disease or death.
Footnotes
Authors’ Note
Brigham Young University College of Nursing was the location where this work was completed.
Author Contributions
The first rough draft of this article was written by Aaron Bennion. Dr Beckstrand was the senior investigator who acted as mentor and also contributed to this work. Dr Luthy and Dr Freeborn contributed to the editing and clarification in writing for the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
