Abstract
Background
Psoriasis is nowadays regarded as a systemic inflammatory disorder. Among the topicals, vitamin D derivates are often applied on the skin for their anti-inflammatory and immune-modulatory properties. Vitamin D serum levels in psoriasis (PsO) patients are still debated and an eventual depletion may offer the rational to integrate anti-psoriatic therapies with oral vitamin D. Then, we aimed to perform a systematic review and meta-analysis on the current evidence towards serum vitamin D level in PsO.
Methods
We searched in PubMed, Scopus, Web of Sciences, ScienceDirect and Science Information Database (SID) using the terms “Vitamin D” and “Psoriasis” including manuscripts in English, Italian and Persian. Duplications were excluded using EndNote software and records were screened by title, abstract and full-text. Quality assessment of studies was assessed using Newcastle Ottawa Checklist (NOS). Psoriasis odds ratio (OR) and mean serum vitamin D levels were calculated and displayed in Forest-plots. Heterogeneity indexes were evaluated using I2 and Q. Sensitivity analysis and publication biases were also considered.
Results
From 3006 records extracted, after removing duplicates and analyzing full texts we finally included 19 manuscripts involving a total of 1387 PsO cases and 6939 controls. PsO patients exhibited a substantial odds ratio (3.07, 95% CI: 1.56-6.04) for lower serum vitamin D levels compared to the control group. Standardized Mean Difference (SMD) of vitamin D in PsO versus controls was −0.92 (−1.33 to −0.51).
Conclusion
Psoriatic patients displayed higher risk to have a vitamin D deficiency. Interventional studies to verify the preventive value are mandatory.
Introduction
Psoriasis (PsO) is currently regarded as a systemic inflammatory disorder1‐3 with a great comorbidity burden, ranging from cardiovascular to respiratory diseases.4‐6 Prevalence of psoriasis in general population has been estimated 2–3% with significant variability among ethnicities.7,8 Although PsO pathogenesis is still debated, the evidence of susceptible genes, 9 the identified triggers 10 and nutritional data11,12 seem to suggest a multifactorial pathogenesis between a susceptible genetic asset is triggered by a predisposing environment. 2
Topicals containing vitamin D derivates are widely used as single or combined therapy in mild PsO, and sometimes also in moderate to severe patients during systemic anti-psoriatic therapies. 1
Vitamin D deficiency is a common finding worldwide, especially in patients affected by autoimmune disorders.13,14 Beside race and congenital vitamin D metabolic problems, the other factors conditioning serum vitamin D levels are modifiable including insufficient sunlight exposure, intestinal malabsorption and insufficient intake.13‐15 Since vitamin D also modulates immunity, both innate and adaptive, its deficiency may trigger or inefficiently counteracts systemic inflammation. 16 Furthermore, low vitamin D levels were described cardiovascular and metabolic disorders, malignancies and several autoimmune (ie systemic lupus erythematous and rheumatoid arthritis) and inflammatory (ie inflammatory bowel disease and psoriasis) diseases. 17
Vitamin D supplementation was studied in several dermatological diseases, such as psoriasis, characterized by low vitamin D serum levels, without any objective improvement. 7 However, low levels of vitamin D are commonly regarded as a detrimental parameter to correct with supplementation. Despite the association between vitamin D low level and PsO-related aspects is still debated, low levels of vitamin D is notoriously related with osteoporosis and at the same time also psoriasis is related to osteoporosis. 18
In 2019 a meta-analysis have been done, studying association between vitamin D level and psoriasis combining results of 10 primary study (pooled mean difference: −6.13 ng/ml). 19 Further investigation in databases showed there were more records and primary studies investigating this association, so a new meta-analysis is essential in order to update the results. In the study by Pitukweerakul et al, 19 Mean Difference (MD) have been reported instead of Standardized Mean Difference (SMD) although they have mentioned that SMD have been calculated in methods section.
Since several studies assessed vitamin D serum levels in PsO patients with discordant results,3,15,20‐31 we aimed to analyze the current evidence in order to legitimate dermatologists to methodically test every PsO patient.
Materials and Methods
This study was designed based on Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) and the protocol has been registered in PROSPERO registration number “CRD42018100192”. 32
Inclusion and Exclusion Criteria
Inclusion and exclusion criteria were structured with PICO. We included every observational study that report serum vitamin D levels in psoriatic patients and controls. Vitamin D levels below 20 UI were considered as deficiency. 33 Every study present in the assessed databases from the beginning until January 19th 2020 in English and Persian were included.
Search Strategy
Databases including PubMed, Scopus, ScienceDirect, Web of Science and Google Scholar search engine were searched with designated MeSH terms and OR, AND, NOT operators. Persian database SID was searched using Persian keywords for “Vitamin D” and “Psoriasis”. Search strategy was as follows:
(“Vitamin D” OR “Cholecalciferol” OR “25-Hydroxyvitamin D” OR “25-OH vitamin D” OR “25-(OH) D” OR “25-hydroxy Vitamin D”) AND (“Psoriasis” OR “Pustulosis of Palms and Soles” OR “Pustulosis Palmaris et Plantaris” OR “Palmoplantar Pustulosis” OR “Pustular Psoriasis of Palms and Soles”)
We carefully also evaluated references in the included studies to improve the sensitivity of our searching phase. EndNote X9 was used to manage the studies.
Study Selection
We preliminary deleted duplicates, then we have screened studies by title and abstract to exclude unrelated records. Selected full-texts were carefully evaluated to verify inclusion and exclusion criteria. This process was performed by two authors and in case of disagreement the study was discussed with the third author that made the final decision.
Data Extraction
Data extraction was performed independently by two authors. From each included study we extracted: first author name, study title, journal title, publication year, study country, article language, numerosity of PsO patients and controls, mean and standard deviation of vitamin D level, number of individuals with vitamin D level <20 UI and ≥ 20 UI, serum vitamin D level measurement unit, pool of patients and selection methods, cases-controls matching criteria, patients’ age and gender.
Risk of Bias Assessment
Risk of bias was assessed using Newcastle-Ottawa Scale (NOS) checklist. 34 This checklist includes three sections, namely Selection, Comparability and Exposure, and globally score studies from 0 to 9 points. Studies that obtained a score < 5 points were excluded. Selection criteria maximum score is 4, Comparability is 2 and Exposure is 3. Assessment was done independently by two people.
Statistical Analysis
Data were analyzed using STATA, version11. Heterogeneity index was performed with Cochran (Q) and I2 tests. Odds ratios and 95% confidence intervals were performed using Metan command, random effect model and reverse variance. To evaluate standardized difference in vitamin D mean serum levels in cases and controls, Metan command, random effect model and reverse variance method and Cohen's d were used. Forest plot was used to graphically represent the estimate standardized difference in the mean serum vitamin D with 95% confidence interval. Sensitivity analysis was performed to evaluate the weight of each study in the overall meta-analysis. Funnel diagram and Egger test were used to investigate publication bias. The potential presence of publication bias was evaluated with Trim and fill analysis.
Results
The preliminary search results in 3006 records that after removing duplicates 1719 remained. Then, after title and abstract screening phase 1698 were excluded and only 21 articles were further screened with the full text. Finally, only 19 papers2,3,15,16,20‐26,28,30,31,35‐39 met the inclusion criteria (Figure 1).

Study selection using PRISMA flow diagram.
Table 1 displays the characteristics of the included primary studies published between 1990 to 2018. Twenty six percent of the studied were published in 2018. Interestingly two studies included only male subjects.28,38 In the great majority of the included studies the matching criteria were age and gender. Characteristics of included studies are summarized in details in Table 1.
Characteristics of Primary Studies Included to the Present Meta-Analysis.

*Quality of studies were assessed using Newcastle-Ottawa Scale Checklist
In our meta-analysis 1387 PsO and 6939 controls from the 19 studies were included. Remarkably, from 13 studies reported vitamin D serum levels, we extracted the odd ratios in psoriatic patients versus controls, maintaining the vitamin D cut-off level of 20 UI (Figure 2).
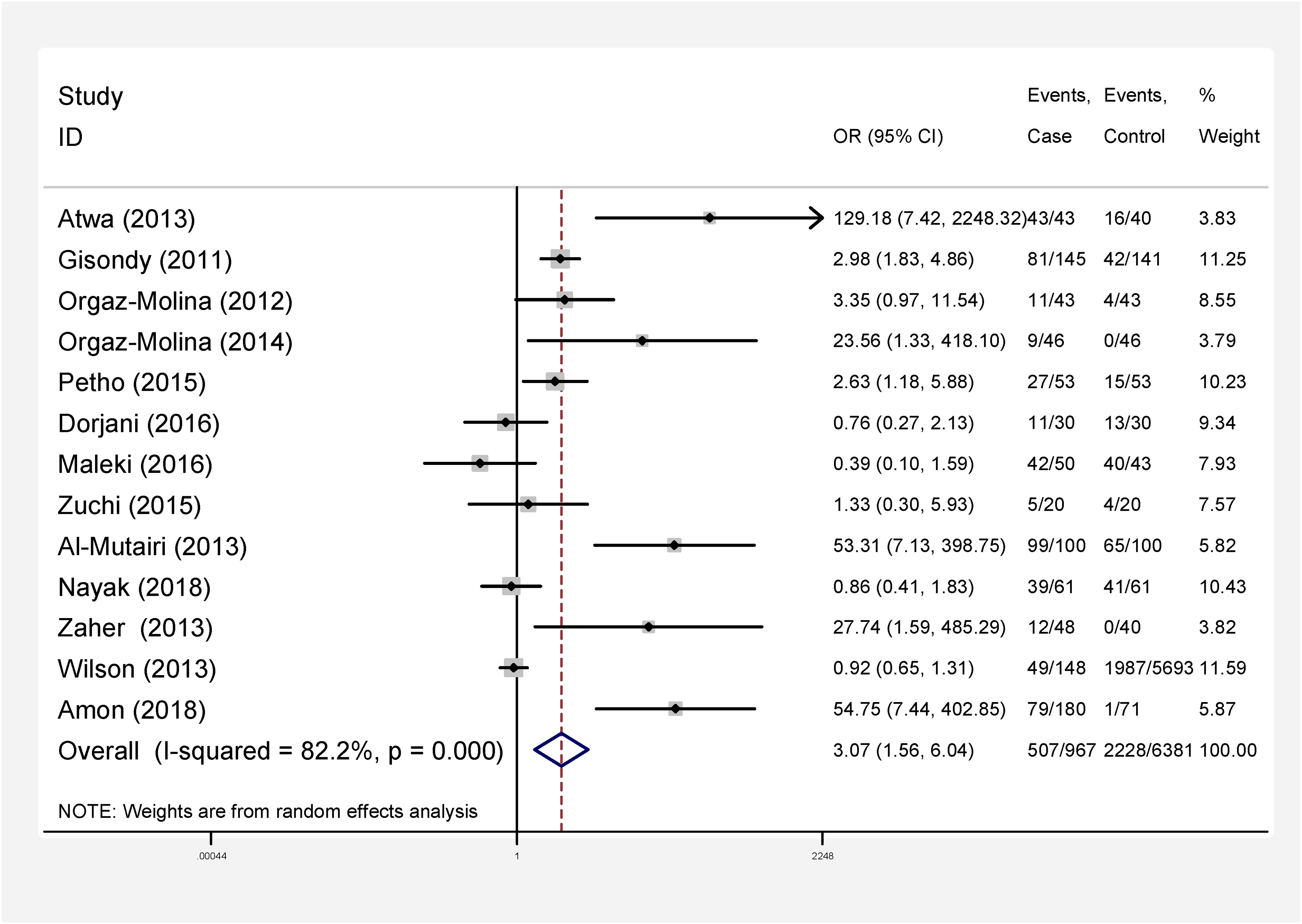
Point odds ratio and 95% confidence interval for each primary study included and overall estimation.
Studies heterogeneity were high in the 19 considered studies (I-squared = 82.2, Q = 67.25, P < 0.001). In 13 studies comparing PsO patients Vs controls, pooled odd ratio was 3.07 (95% CI, 1.56-6.04, p < 0.001) (Figure 2).
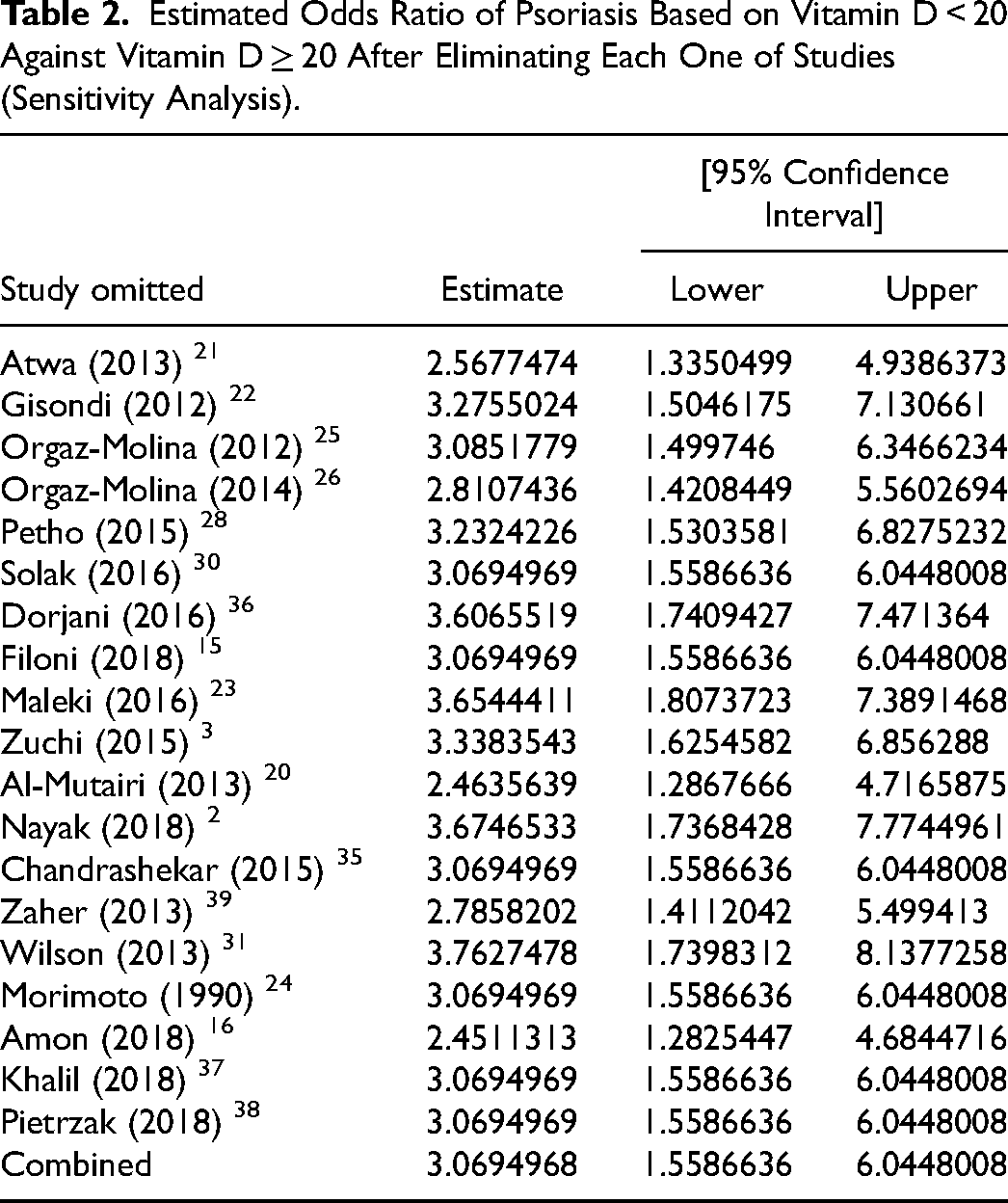
Sensitivity analysis is displayed in Figure 3 and Table 2. Funnel plot (Figure 4) showed the presence of publication biases, as well as Egger test (β=2.32, P = 0.028).
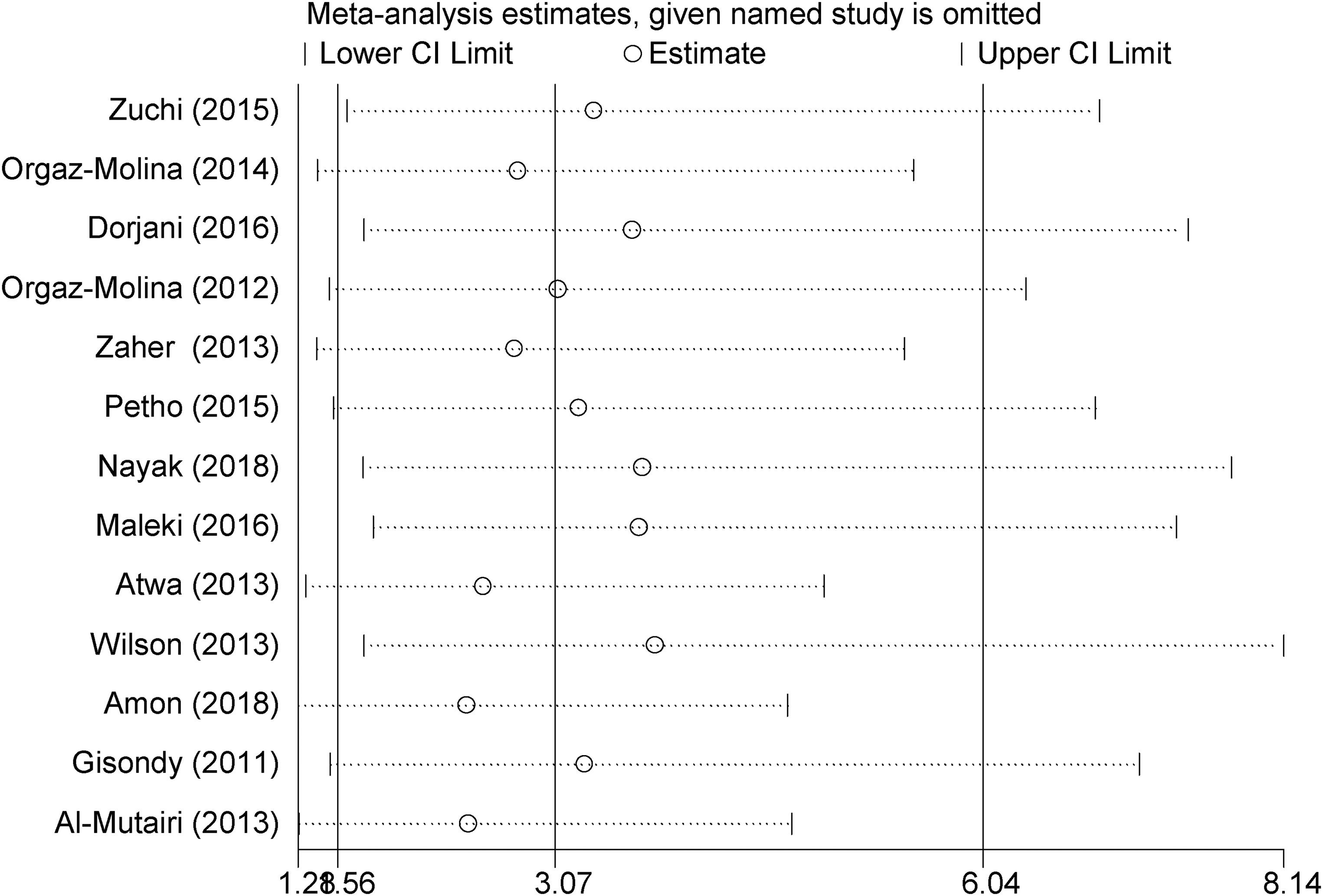
Sensitivity analysis in order to investigate effect of each primary study on overall odds ratio estimate.

Funnel plot describing publication bias.
Estimated Odds Ratio of Psoriasis Based on Vitamin D < 20 Against Vitamin D ≥ 20 After Eliminating Each One of Studies (Sensitivity Analysis).
Trim and fill test estimated 3 possible missing studies (Figure 5). Considering this fact, odds ratio became 1.40 (CI 95% 1.12-1.76) using fixed effect model and 1.76 (CI 95% 0.86-3.59) using random effect model.

Filled funnel plot of psoriasis odds ratio.
In 19 studies mean and standard deviation of serum vitamin D level in case and control group were reported. Combining the results of 19 primary studies, pooled SMD of serum vitamin D level estimated −0.92 (−1.33, −0.51) (Figure 6). Heterogeneity was high (I-squared = 96%, Q = 421.46, P < 0.001) but based on Egger test results publication bias for estimating SMD was not statistically significant (β=1.25, P = 0.197).

Standardized mean difference (SMD) of serum vitamin D level in patients with psoriasis in compare with control group.
Discussion
In this meta-analysis the association between psoriasis and vitamin D have been investigated. Low vitamin D (<20 UI) was significantly associated with psoriasis. Standardized mean vitamin D level was significantly lower in psoriatic patients than healthy controls.
Vitamin D is a crucial nutrient synthesized in the skin through exposure to ultraviolet B (UVB) sunlight, leading to the conversion of 7-dehydrocholesterol to pre-vitamin D3 and further to vitamin D3. 40 Measurement of vitamin D status involves assessing various markers and metabolites. One of the most widely used markers is the circulating concentration of 25-hydroxyvitamin D (25(OH)D). This marker is favored due to its relatively long half-life, stability, and reflection of both endogenous synthesis and dietary intake of vitamin D. Serum 25(OH)D concentration provides a reliable indicator of an individual's overall vitamin D status, allowing healthcare professionals to classify individuals as deficient, insufficient, or sufficient. 33 This approach aids in determining appropriate supplementation and intervention strategies to maintain optimal vitamin D levels and prevent associated health risks.33,40,41 Vitamin D also plays a pivotal role is skin homeostasis, in fact it is involved in antimicrobial peptides synthesis and regulation. 42 Vitamin D also showed immunomodulatory effects exerted on Langerhans cells, preventing activated T-cell proliferation.43,44 Its pleiotropic effect affects both plasmacytoid dendritic cells (pDC) 45 Th-17 ones, two abundant cytotypes in psoriatic active lesions. 46
Interventional trials that assessed vitamin D oral integration in psoriatic patients are controversial. Disphanurat et al administered 60,000 IU of vitamin D2 twice weeks for 6 months in a cohort of psoriatic patients obtaining a significant PASI reduction without adverse events. 47 Conversely, Ingram et al did not appreciate a severity improvement after 100,000 IU vitamin D3 supplementation for 12 month. 48
Previous meta-analysis on vitamin D and psoriasis severity by Pitukweerakul et al 19 have estimated pooled MD of vitamin D, −6.13 (CI 95% −10.93, −1.32). Since we have used SMD instead of MD the pooled estimate is smaller. However, Pitukweerakul et al 19 have missed number of studies. They have also did not report pooled OR for psoriasis and vitamin D deficiency.
Despite our meta-analysis shed light on the association existing between vitamin D deficiency and psoriasis, we are far to plenty understand the temporal association. Furthermore, future studies should focus on the potential relationship between vitamin D levels and PsO severity. One approach could involve longitudinal cohort studies that track individuals over time to better establish whether vitamin D deficiency precedes the onset of psoriasis or if psoriasis contributes to lower vitamin D levels. Additionally, examining the molecular mechanisms underlying this relationship could provide insights into the causal direction.
Our meta-analysis focused on the difference in vitamin D levels in psoriatic patients compared to controls, but did not account for the constellation of well-known factors that influence vitamin D serum levels. In literature, vitamin D serum levels recognized as modulators several factors, included air pollution, drugs, vaccination, infection, lifestyle, obesity, sun exposure, dyslipidemia, mental stress, hypertension, diabetes and psoriasis family history. 49 Ultraviolet radiation has been shown to be linked with improvement in psoriasis by increasing Vitamin D and immunomodulatory effects. 50 Vitamin D also plays a crucial role in modulating the interaction between the innate and adaptive immune systems, contributing to a reduction in autoimmune disease activity and reducing the psoriasis flares in these patients. 51
Thus, we suggest that PsO patients with such a risk factor should be mandatory tested in order to potentially be supplemented with vitamin D. Despite the numerous uncertainties on the effect of oral vitamin D supplementation in psoriasis, an interventional approach should be encouraged to prevent osteoporosis in these patients.
Publication biases and language inclusion criterium may have limited the studies evaluated, however we statistically account for these aspects. The main limitation of any meta-analysis remains the primary studies quality. In several included studies extracting data based on gender and age group was not possible.
Conclusion
This meta-analysis showed that odds of vitamin D deficiency is higher in psoriatic patients than control. Critically evaluating the current literature remains unclear if the vitamin D deficiency is present before psoriasis development or the psoriasis causes itself vitamin D deficiency. Hence, we recommend checking vitamin D serum levels in psoriatic patients and supplement them in case of low levels.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Author Contributions
M.M has contributed in study design, analysis and data interpretation. G.D has contributed in data interpretation, writing and editing manuscript. M.K, M.Kh and F.N have contributed to data collection, analysis and interpretation. A.H has contributed in writing manuscript, data interpretation, writing and editing manuscript.
