Abstract
Background
Frailty among surgical patients correlates with the risks of adverse outcomes, including postoperative complications, extended recovery, and elevated mortality rates. The ERAS protocol is a comprehensive, team-based care pathway that focuses on refining perioperative management, lowering the risk of complications, and promoting faster recovery. This study investigates the effects of implementing ERAS on hospitalization duration, clinical outcomes, and perioperative complications in frail individuals undergoing short-segment lumbar fusion.
Methods
We included frail patients ≥65 years who underwent one- or two-level posterior lumbar fusion for degenerative spinal disease from January 2018 to October 2020 (non-ERAS cohort) and from October 2020 to June 2023 (ERAS cohort). Demographic information, comorbidities, and surgical details were collected. The primary outcome was to compare the hospitalization duration. Additionally, other clinical outcomes, such as postoperative recovery parameters, pain scores, complications, and the Oswestry Disability Index (ODI), were recorded.
Results
The study enrolled 114 frail patients, equally distributed into ERAS and non-ERAS cohorts. ERAS intervention significantly reduced the length of hospital stay (6.95 ± 2.74 days vs. 8.72 ± 4.54 days, p = 0.024) and blood transfusion rates (19.3% vs. 49.1%, p = 0.001). Time to remove the drain and indwelling catheter showed no statistically significant difference. Postoperative VAS scores and Oswestry Disability Index (ODI) scores at 6-week, 6-month, and 12-month follow-up intervals were comparable between groups. There was no significant difference in the incidence of major or minor complications between the two groups.
Conclusions
Our study suggests that applying an ERAS pathway to frail individuals undergoing short-segment lumbar fusion can shorten hospital stay and lower transfusion rates, while not increasing complications, reoperations, or readmissions.
Introduction
As the population ages, the prevalence of lumbar degenerative disease requiring surgical management has increased.1,2 Lumbar fusion represents the primary surgical option for addressing these conditions. With the growing number of elderly patients undergoing such procedures, it is essential to recognize the influence of age-related physiological changes on recovery and postoperative outcomes. Reduced physiological reserve and the presence of multiple comorbidities place older adults at higher risk of perioperative complications and extended hospital stays, underscoring the importance of proactive perioperative management. 3
Frailty is a clinical syndrome marked by reduced physiological reserves, impaired strength, and limitations in daily functioning.4,5 In the context of lumbar fusion surgery, frailty has been recognized as an important determinant of postoperative outcomes. Recent evidence indicates that frailty is a more reliable predictor of postoperative complications than chronological age.6,7 Specifically in spine surgery, frailty increased risks in correlation with postoperative complications, prolonged hospital stays, and higher mortality rates.8-10 Frail patients exhibit delayed wound healing, an increased susceptibility to infections, and a prolonged recovery trajectory. 11 These complications emphasize the critical need for a comprehensive preoperative assessment, in which the identification of frailty directly informs surgical decision-making. Frailty in older adults was assessed using the Fried frailty phenotype proposed by Fried and colleagues in 2001, which is based on five criteria: unintentional weight loss, self-reported exhaustion, reduced grip strength, slow gait speed, and low levels of physical activity. 12 Each criterion is objectively measured or self-reported, allowing for a standardized assessment of frailty status. Individuals who meet three or more criteria are categorized as frail, whereas those who meet one or two criteria are classified as pre-frail. This phenotype serves as a valuable clinical instrument for identifying at-risk individuals and implementing targeted interventions to mitigate the adverse outcomes associated with frailty in aging populations. Furthermore, the FRAIL scale, developed by Morley, designed as a rapid screening tool using five self-reported components: Fatigue (feeling tired), Resistance (difficulty climbing one flight of stairs), Ambulation (difficulty walking one block), Illnesses (presence of multiple chronic diseases, typically ≥5), and Loss of weight (unintentional weight loss). Each component is scored dichotomously (yes/no), allowing quick classification of individuals as robust, pre-frail, or frail without the need for equipment or trained assessors, making the FRAIL scale especially suitable for primary care and community-based settings. 13 The T-FRAIL scale is a Thai-adapted version of the FRAIL scale, culturally and linguistically validated for Thai older adults, retaining the simplicity of the original FRAIL scale while demonstrating acceptable validity and good inter-rater reliability, supporting its use in routine clinical practice and population-based screening. 14
Effective management of frailty in patients receiving lumbar fusion requires a coordinated, multidisciplinary strategy. Enhanced Recovery After Surgery (ERAS) represents a structured perioperative care pathway that incorporates multimodal, evidence-based interventions tailored to specific surgical specialties and procedures.15,16 Although originally designed for colorectal surgery, ERAS has since been adapted to spine operations, where it has demonstrated benefits in reducing perioperative morbidity, accelerating postoperative recovery, and improving resource utilization. 17 ERAS protocols integrate evidence-based interventions across the preoperative, intraoperative, and postoperative phases to facilitate early recovery and minimize the overall healthcare burden.15,16 Several studies have reported favorable outcomes with ERAS in spine surgery, including shorter hospital stays and fewer complications.18-20 Nevertheless, limited evidence exists regarding its effectiveness in frail patients undergoing lumbar fusion. This study, therefore, aimed to evaluate clinical outcomes in frail individuals who underwent lumbar fusion surgery with and without ERAS application.
Methods
Subjects
This retrospective cohort study was approved by the Institutional Review Board of Siriraj Hospital, Mahidol University (COA No. Si 842/2023). Informed consent was waived due to the retrospective design of the study. This study was conducted and reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Frail patients diagnosed with degenerative lumbar spine disease and treated with one- or two-level posterior lumbar spinal fusion, either before or after the application of the Enhanced Recovery After Surgery (ERAS) protocol, were included in the analysis. Frailty status was determined according to the T-FRAIL criteria. Frailty status was assessed preoperatively during the outpatient clinic evaluation using the T-FRAIL scale. For this retrospective cohort, frailty scores were obtained from standardized preoperative assessment records documented in the electronic medical record system. All frailty assessments were completed prior to hospital admission and surgical intervention. The retrospective nature of frailty assessment may introduce inherent limitations related to documentation accuracy; however, the use of standardized assessment forms minimized variability across the study period. Inclusion criteria comprised patients aged over 65 years with a T-frail score of ≥3, who were treated with posterior lumbar spinal fusion for one to two levels due to degenerative conditions such as spinal stenosis, spondylolisthesis, or degenerative scoliosis. The study excluded individuals with a history of lumbar spine operations, those undergoing concurrent cervical spine procedures, and those lacking complete one-year follow-up data.
The sample size calculation was the study by Ken Porche et al. 21 The mean length of stay (LOS) in the Pre-ERAS group was 4.8 days, while that in the ERAS group was 3.8 days, with a standard deviation (SD) of 1.9. These values were used to detect a statistically significant difference between the ERAS and non-ERAS (Traditional) groups. Assuming a Type I error (α) of 0.05 and a Type II error (β) of 0.20 (corresponding to a power of 80%). The estimated sample size was 57 participants per group. Therefore, the total sample size required for the study is 114 participants.
A total of 509 consecutive patients aged ≥65 years who underwent one- or two-level posterior lumbar fusion for degenerative lumbar disease between January 2018 and June 2023 were retrospectively reviewed. During the non-ERAS period (January 2018–October 2020), 273 patients underwent surgery, including 67 frail patients (T-FRAIL ≥3). During the ERAS period (October 2020–June 2023), 236 patients underwent surgery, of whom 62 were frail. Patients with revision surgery, concomitant spinal procedures, or incomplete clinical data were excluded. After exclusions, 57 frail patients remained in each cohort, yielding a final sample of 114 patients. All consecutive eligible frail patients were included without matching or additional sampling. The patient selection process is illustrated in Figure 1. Flow diagram of patient selection and cohort allocation
Bias and Confounding
Given the retrospective design and non-contemporaneous comparison between pre-ERAS and ERAS periods, potential selection and temporal biases may exist. To minimize selection bias, all consecutive eligible frail patients were included without matching or additional sampling. Surgical indications, operative techniques, and the surgical team remained consistent throughout the study period to reduce performance-related confounding.
Surgical Technique
After general anesthesia was induced, patients were placed in the prone position on a radiolucent surgical table. Standard sterile preparation and draping techniques were applied. A midline posterior approach was used to expose the relevant spinal elements. Instrumentation was achieved with a pedicle screw–rod construct. Decompression by laminectomy was performed according to preoperative clinical symptoms (lumbar and radicular) and MRI findings. Fusion procedures included standard open posterolateral fusion, posterior lumbar interbody fusion, or transforaminal lumbar interbody fusion, using the CD HORIZON® LEGACY Spinal System and CAPSTONE® PEEK implants (Medtronic Inc., Memphis, TN, USA). Surgical drains were routinely placed in all patients postoperatively.
ERAS Protocol
The orthopedic surgery department-initiated application of the Enhanced Recovery After Surgery (ERAS) protocol in October 2020, coinciding with the establishment of a multidisciplinary perioperative care team. The protocol incorporated interventions across the preoperative, intraoperative, and postoperative phases.
Preoperative measures included: (1) patient education and counseling to explain surgical risks and familiarize patients with the ERAS pathway; (2) nutritional assessment with individualized dietary guidance and supplementation for malnourished patients; (3) encouragement of smoking and alcohol cessation for at least two months before surgery; (4) prescribed preoperative conditioning exercises, including back, leg, and respiratory training; (5) avoidance of prolonged fasting, permitting solid food up to six hours and carbohydrate-rich fluids up to two hours before surgery; and (6) preemptive analgesia with nonsteroidal anti-inflammatory drugs, acetaminophen, pregabalin, and proton pump inhibitors.
Intraoperative interventions included: (1)administration of prophylactic antibiotics within 30 minutes prior to incision; (2) administration of tranexamic acid within 30 minutes before incision; (3) maintenance of normothermia using warming blankets; (4) mechanical prophylaxis for deep vein thrombosis with intermittent pneumatic compression devices; (5) adherence to standardized anesthetic protocols; and (6) local wound infiltration analgesia with 0.5% bupivacaine, with or without ketorolac.
Postoperative interventions comprised: (1) Multimodal analgesia regimens; (2) early removal of urinary catheters and consideration of drain removal within 48 hours; (3) promotion of early mobilization, with patients encouraged to ambulate using assistive devices as needed.
The Non-ERAS conventional care regimen lacked standardized guidelines and did not include the structured preoperative, intraoperative, and postoperative interventions outlined in the ERAS protocol. All patients in both cohorts were managed by same spine surgery team, using uniform surgical indications, operative approaches, and implant systems throughout the study period. No major changes in surgical staffing, operative techniques occurred during the study timeframe.
Data Collection
Data collected for analysis included demographic characteristics (age, sex, weight, height, and BMI), baseline clinical variables (surgical level, comorbidities, ASA classification, and preoperative ODI), and intraoperative parameters (operative duration, estimated blood loss, and transfusion requirements). Postoperative assessments comprised visual analog scale (VAS) scores, recovery milestones (drain and catheter removal), complications, 30-day reoperations, 90-day readmissions, and postoperative ODI scores. The primary outcome was hospital length of stay, compared between the ERAS and non-ERAS groups. Secondary outcomes consisted of additional clinical and perioperative measures. A subgroup analysis was further conducted within the ERAS cohort, stratified by the mean length of stay, to identify factors associated with prolonged hospitalization.
Statistics
The statistical analysis was conducted utilizing SPSS version 18 (SPSS, Inc., Chicago, IL, USA). Continuous variables were tested for normality using the Shapiro-Wilk test. Variables with a normal distribution are expressed as mean ± SD, and between-group differences were assessed with the independent t-test. For data that did not follow a normal distribution, nonparametric methods, including the Mann-Whitney U test and the Wilcoxon signed-rank test, were applied. Categorical variables were analyzed using the Chi-square test. A P-value< 0.05 was considered indicative of statistical significance.
Results
Baseline Demographics and Surgical Diagnosis of Patients
Baseline Demographics and Surgical Diagnosis of Patients in ERAS and Non-ERAS Groups

†Values are presented as mean ± SD or number (%). Exact p-values are reported. Comparisons were performed using the independent t-test or chi-square/Fisher’s exact test, as appropriate. A two-sided p value < 0.05 was considered statistically significant.
Abbreviation: ERAS: Enhanced Recovery After Surgery, BMI: Body Mass Index, ASA: American Society of Anesthesiologists physical status classification, ODI: Oswestry Disability Index, SD: standard deviation.
Fusion techniques, including posterolateral fusion (PL fusion), posterior lumbar interbody fusion (PLIF), and transforaminal lumbar interbody fusion (TLIF), were similarly distributed between groups, with no statistically significant differences (PL fusion, p = 0.260; PLIF, p = 0.130; TLIF, p > 0.99).
Clinical and Surgical Outcome
Perioperative Parameters and Surgical Outcomes Between ERAS and Non-ERAS Groups

†Values are presented as mean ± SD or number (%). Exact p-values are reported. Comparisons were performed using the independent t-test or chi-square/Fisher’s exact test, as appropriate. A two-sided p value < 0.05 was considered statistically significant.
Abbreviation: ERAS: Enhanced Recovery After Surgery, VAS: Visual Analog Scale, mL: milliliters, min: minutes, SD: standard deviation.
Comparison of Postoperative Complications Between ERAS and Non-ERAS Groups

Values are presented as number (%). Exact p-values are reported. Comparisons were performed using the chi-square or Fisher’s exact test. A two-sided p value < 0.05 was considered statistically significant.
Abbreviation: ERAS: Enhanced Recovery After Surgery, UTI: Urinary Tract Infection, DVT: Deep Vein Thrombosis, SD: standard deviation
Postoperative Pain and Functional Outcomes (VAS and ODI Scores) Between Groups

†Values are presented as mean ± SD. Exact p-values are reported. Comparisons were performed using the independent t-test. A two-sided p value < 0.05 was considered statistically significant.
Abbreviation: ERAS: Enhanced Recovery After Surgery, VAS: Visual Analog Scale, ASA: American Society of Anesthesiologists physical status classification, BMI: Body Mass Index, CKD: Chronic Kidney Disease, SD: standard deviation.
Comparison of Clinical and Perioperative Factors Associated With Prolonged Length of Stay (≥7 days) Within the ERAS Group
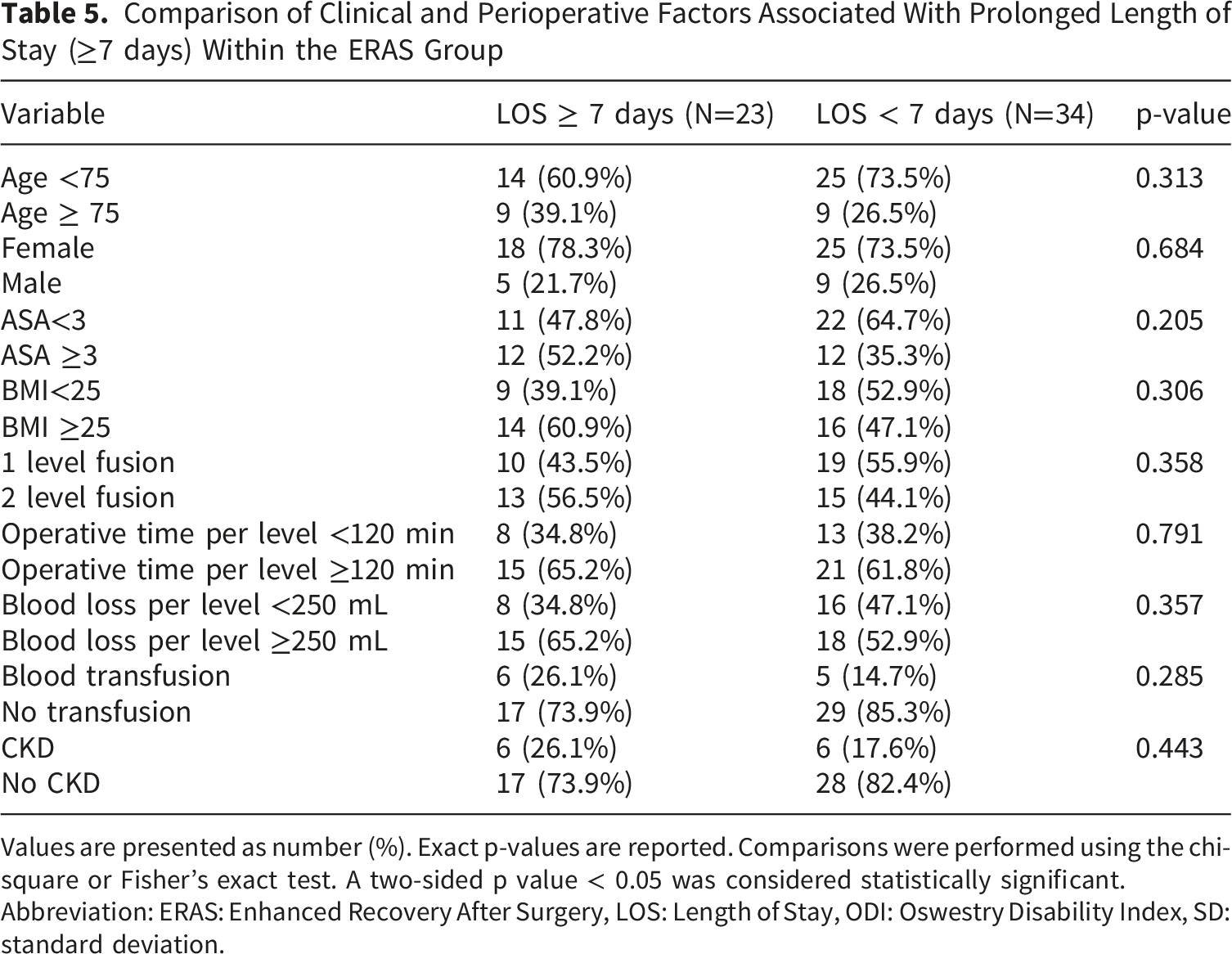
Values are presented as number (%). Exact p-values are reported. Comparisons were performed using the chi-square or Fisher’s exact test. A two-sided p value < 0.05 was considered statistically significant.
Abbreviation: ERAS: Enhanced Recovery After Surgery, LOS: Length of Stay, ODI: Oswestry Disability Index, SD: standard deviation.
Discussion
Frailty presents a significant challenge in the management of patients undergoing lumbar fusion surgery due to its association with a higher incidence of postoperative complications and prolonged recovery times.4,7 This study was conducted to examine the influence of the Enhanced Recovery After Surgery (ERAS) protocol on the clinical outcomes of frail patients undergoing 1-2 level lumbar fusion surgery. For the purposes of this study, frailty was assessed using the T-FRAIL scale, a straightforward, five-question tool that has demonstrated high sensitivity in identifying individuals at risk of developing disabilities, health function decline, and increased mortality. Moreover, the T-FRAIL scale has shown satisfactory validity and reliability in detecting frailty among elderly patients.13,14
Our findings demonstrate that ERAS implementation is associated with a significant reduction in hospital length of stay for frail patients. This result is consistent with prior studies highlighting the benefits of ERAS in accelerating recovery and decreasing hospitalization duration. Wang et al. 18 reported a retrospective analysis of elderly patients (≥65 years) undergoing short-level lumbar fusion, showing a significant decrease in LOS in the ERAS group compared with the non-ERAS cohort (12.30 ± 3.03 vs. 15.50 ± 1.88 days). Li et al. 19 similarly examined patients over 70 years undergoing lumbar arthrodesis and found shorter LOS in the ERAS group compared with traditional care (13.6 ± 4.0 vs. 15.6 ± 3.9 days). In a study focusing specifically on frail patients undergoing one-to two-level transforaminal lumbar interbody fusion, Porche et al. 21 observed a one-day reduction in LOS (pre-ERAS: 4.8 ± 1.6 days vs. post-ERAS: 3.8 ± 1.9 days) together with a 3.2-day improvement in the return of physiological function. Likewise, Cui et al. 22 demonstrated that ERAS implementation in multilevel lumbar fusion significantly shortened LOS (12.18 ± 4.69 vs. 10.44 ± 4.60 days), highlighting the protocol’s role in improving perioperative efficiency through strategies such as optimized management and early mobilization.
The ERAS group demonstrated a significantly longer operative duration per level compared with the non-ERAS group (86.28 ± 35.21 vs. 71.46 ± 25.38 minutes, p = 0.027); however, this increase was not accompanied by a corresponding rise in estimated blood loss or complication rates. Several factors may account for this difference, including additional time required for ERAS-adherent intraoperative measures such as local wound infiltration, maintenance of normothermia, and implementation of multimodal prophylactic strategies. Although these interventions may modestly prolong operative time, they likely contribute to improved physiologic stability and reduced perioperative blood loss, thereby decreasing transfusion requirements without increasing complications. Furthermore, the prolonged operative duration may partially reflect a learning curve associated with the initial adoption of the ERAS pathway, which necessitates enhanced coordination among surgical, anesthetic, and nursing teams. Importantly, the absence of increased blood loss or complications suggests that the additional operative time did not adversely affect patient safety. From a clinical standpoint, a modest increase in operative duration appears to be a sustainable and acceptable trade-off when balanced against the observed benefits of shorter length of stay and reduced transfusion rates, particularly in frail patients who are more susceptible to postoperative morbidity. As familiarity with the ERAS protocol improves, operative efficiency may increase, potentially attenuating this difference in operative duration over time.
Although the intraoperative blood loss was similar between the ERAS and non-ERAS groups, patients in the ERAS cohort required fewer blood transfusions. This is consistent with previous studies that have reported reduced blood transfusion rates with the implementation of ERAS protocols in various surgical procedures. 23 The reduced transfusion rate observed may be attributed to core ERAS strategies, such as optimizing preoperative hemoglobin, administering tranexamic acid intraoperatively, and following restrictive transfusion protocols. In particular, numerous studies have confirmed the effectiveness of tranexamic acid in minimizing intraoperative blood loss during spine surgery.19,24,25
Additionally, while the differences were not statistically significant, frail patients in the ERAS group experienced shorter durations of both drain and indwelling catheter use. This could indicate that the ERAS protocol contributes to faster resolution of postoperative drainage and urinary catheterization, thereby facilitating earlier ambulation and potentially decreasing complication rates such as urinary tract infections. Despite these positive findings, no significant differences were found in VAS pain scores or ODI functional measures between the ERAS and non-ERAS cohorts. These results contrast with some prior studies that have reported improvements in pain management and functional recovery following the implementation of ERAS protocols. 19 The absence of significant differences in pain and functional outcomes in our study may be attributed to various factors, including heterogeneity in patient populations, variability in pain management strategies, and limitations in sample size.
Previous studies have demonstrated the potential of ERAS to reduce postoperative complications in various surgical populations. 19 In our study, however, this benefit was not statistically evident among frail patients receiving lumbar fusion surgery. In the non-ERAS group, three patients required reoperation within 30 days postoperatively: one due to implant malposition and two due to surgical site infection. Additionally, two patients in this group were readmitted within 90 days postoperation: one for Polyetheretherketone (PEEK) cage migration and the other for severe radicular pain. In contrast, no reoperations or readmissions were observed in the ERAS group. Several considerations may help explain why complication rates did not differ significantly between the ERAS and non-ERAS cohorts. First, frail patients receiving lumbar fusion surgery are a particularly vulnerable population, with inherently elevated risks of complications, which suggests that larger sample sizes may be required to identify subtle differences attributable to the ERAS protocol. Furthermore, the heterogeneity of frailty among participants in our study may have influenced their responses to the ERAS protocol and its potential impact on complication rates. Frail patients constitute a heterogeneous population with varying degrees of physiological vulnerability and functional impairment across multiple domains, including physical endurance, mobility, comorbidity burden, and functional reserve, which may influence both postoperative risk profiles and responsiveness to perioperative interventions such as ERAS. Additionally, variability in the implementation and adherence to the ERAS protocol across different healthcare settings and surgical teams may have influenced its effectiveness in reducing complications. Moreover, variations in the implementation and adherence to the ERAS protocol across surgical teams and healthcare settings may have influenced its effectiveness. The success of ERAS depends on consistent, multidisciplinary collaboration and strict adherence to evidence-based practices.
Despite these positive outcomes, our subgroup analysis within the ERAS cohort did not identify any significant preoperative or intraoperative factors associated with prolonged LOS. We defined prolonged LOS as a hospital stay equal to or exceeding the mean LOS, yet variables such as age, gender, ASA score, fusion level, operative time, blood loss, or transfusions showed no significant correlation with hospitalization duration. These findings contrast with prior studies, which have identified multiple risk factors for extended LOS. For instance, Cui et al. 26 reported age ≥75 years, female sex, ASA ≥3, operative time, fusion level, and allogenic transfusion as potential predictors of prolonged LOS. Similarly, Gruskay et al. 27 found that advanced age and the presence of systemic comorbidities were associated with longer hospital stays, although intraoperative factors and fusion levels were not significantly associated. Kobayashi et al. 28 also identified age ≥70 years, ASA ≥3, open procedures, fused levels ≥3, operative time ≥300 minutes, and blood loss ≥500 mL as significant predictors. The absence of significant associations between prolonged LOS and traditional risk factors in the present ERAS subgroup may be explained by the relatively homogeneous frail population, in which baseline characteristics such as age, ASA grade, and fusion level may have limited discriminatory value. Additionally, the standardized perioperative care pathways inherent to ERAS implementation may mitigate the influence of these variables on postoperative recovery. The small subgroup sample size (n = 23) may have further limited statistical power, while unmeasured confounding factors, including social support, discharge planning efficiency, rehabilitation availability, and institutional practices during ERAS adoption, may have played a more substantial role in determining length of stay.
An important consideration in interpreting our results is the non-contemporaneous comparison between the ERAS and non-ERAS cohorts. The non-ERAS group was treated prior to the COVID-19 pandemic, whereas the ERAS group was managed during or after the pandemic. This temporal separation introduces potential confounding, as the pandemic prompted substantial restructuring of perioperative workflows. Modifications implemented to reduce exposure risk, preserve hospital bed capacity, and enhance discharge efficiency became widely adopted in spine surgery during this period. Reports have described accelerated discharge practices, constrained rehabilitation access, and reallocation of perioperative staffing, any of which may have independently influenced LOS outcomes.29,30 These system-level changes may be particularly relevant to frail patients, for whom altered discharge planning and heightened infection control protocols could modify recovery trajectories independent of the ERAS pathway. Although no major changes occurred in surgical indications, operative techniques, or implant systems at our institution beyond ERAS adoption, the possibility remains that pandemic-related policy shifts interacted synergistically with ERAS principles and amplified the observed benefits. As such, caution is warranted when attributing outcome improvements solely to ERAS implementation.
Ultimately, our findings emphasize the necessity of a comprehensive, team-based strategy for the care of frail patients in spinal surgery. While ERAS shows promise in improving postoperative recovery and reducing transfusion rates, future research with larger patient populations, frailty stratification, and multivariate analysis is warranted to better elucidate the factors influencing outcomes in this vulnerable population.
Strengths and Limitations
Our study has some limitations; the first is the retrospective design. Additionally, the use of various lumbar spine fusion techniques across the study cohort could introduce variability in clinical outcomes. Another limitation is the limited sample size, which may limit the secondary outcome of our analysis. Furthermore, collecting cohorts across different time frames could introduce confounding variables, including changes in surgeon proficiency. These potential confounders might influence the outcomes independently of the ERAS protocol implementation. Although functional outcomes were assessed using the Oswestry Disability Index at 12 months, data on long-term complications—such as pseudarthrosis or implant failure—and sustained functional improvement beyond one year were not available. Given that frail patients may experience prolonged or delayed recovery, longer-term follow-up (e.g., 2–5 years) would be valuable to further validate the durability of ERAS benefits. In light of these limitations, future prospective studies with larger sample sizes and extended follow-up periods are warranted to more comprehensively evaluate the impact of ERAS protocols in a broader spine surgery population and to assess their implications for long-term outcomes and healthcare resource utilization.
However, our study also had several strengths. First, it was one of the few studies to specifically evaluate the impact of the ERAS protocol on frail patients receiving 1–2 level posterior lumbar fusion surgery—a population often underrepresented in spine surgery literature. Second, our study employed the T-FRAIL scale, a simple and validated screening tool, which allowed consistent identification of frail individuals in clinical settings. Third, we comprehensively assessed clinical outcomes, including LOS, transfusion rate, pain score, functional outcome, complications, reoperations, and readmissions, along with a subgroup analysis of prolonged hospitalization. Finally, all clinical data were collected from a single institution with a standardized surgical and postoperative protocol, which enhanced internal validity and reduced variability in perioperative care. The findings provide practical implications for the implementation of ERAS protocols in frail surgical populations within similar healthcare contexts. Unlike prior ERAS studies that primarily focused on general elderly populations, the present study exclusively evaluated objectively defined frail patients using a validated T-FRAIL scale and demonstrated that ERAS implementation was associated with reduced transfusion requirements and shorter length of stay without an increase in perioperative complications in this high-risk subgroup.
Conclusion
Our study suggests that applying an ERAS pathway to frail individuals undergoing short-segment lumbar fusion can shorten hospital stay and lower transfusion rates, while not increasing complications, reoperations, or readmissions. These findings support the role of ERAS as a valuable perioperative strategy for this high-risk population.
Footnotes
Acknowledgements
The authors thank Miss Pinprapha Boonhyad, research officer, for statistical analysis. This research project was supported by the Faculty of Medicine, Siriraj Hospital, Mahidol University.
ORCID iDs
Ethical Considerations
Ethical approval for this study was granted by the Siriraj Institutional Review Board, Faculty of Medicine, Siriraj Hospital, Mahidol University, Bangkok, Thailand (COA No.Si 842/2023).
Author Contributions
Conceptualization and Study Design: Ekkapoj Korwutthikulrangsri (EK), Monchai Ruangchainikom (MR), Werasak Sutipornpalangkul (WS).
Data Collection: Tinh Laoharojanaphand (TL).
Data Analysis and Interpretation: Tinh Laoharojanaphand (TL), Panlop Tirawanish (PT).
Methodology and Validation: Ekkapoj Korwutthikulrangsri (EK), Monchai Ruangchainikom (MR), Werasak Sutipornpalangkul (WS).
Writing – Original Draft: Tinh Laoharojanaphand (TL).
Writing – Review & Editing: Panlop Tirawanish (PT), Ekkapoj Korwutthikulrangsri (EK), Monchai Ruangchainikom (MR), Werasak Sutipornpalangkul (WS).
Supervision: Ekkapoj Korwutthikulrangsri (EK), Monchai Ruangchainikom (MR), Werasak Sutipornpalangkul (WS).
Final Approval of Manuscript: All authors Accountability: All authors agree to be accountable for all aspects of the work, ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data analyzed in this study are accessible from the corresponding author upon reasonable request.
