Abstract
Background
To estimate 5-year mortality after major upper-extremity fractures in adults aged ≥65 years and to identify clinical and fracture-related factors associated with mortality.
Methods
We conducted a retrospective single-center study of adults aged ≥65 years who presented between 2014 and 2020 with a suspected major upper-extremity fracture. Fracture diagnoses were confirmed on archived radiographs and analyzed by index diagnosis and by anatomic region. Candidate factors included age, sex, comorbidity burden (age-adjusted Charlson Comorbidity Index [aCCI] categories), presence of concomitant fractures outside the upper extremity, injury mechanism (low- vs high-energy), upper-extremity fracture multiplicity, and treatment type (operative vs nonoperative). Five-year mortality was the primary outcome.
Results
A total of 1,240 patients were included (median age 73 years; 67.3% female) with a median follow-up of 6.0 years. Five-year mortality was 28.2% (95% CI 25.7–30.8). Mortality differed by fracture location, lowest after wrist-region fractures (especially distal radius) and highest after arm-region fractures (especially humeral shaft). Mortality increased with age and was higher in men. Concomitant fractures outside the upper extremity and greater comorbidity burden were associated with higher mortality, with a stepwise gradient across aCCI categories (2–3, 4–5, and ≥6). Injury mechanism, upper-extremity fracture multiplicity, and treatment type were not significantly associated with 5-year mortality.
Conclusions
In older adults with major upper-extremity fractures, long-term mortality is substantial and is most strongly associated with baseline vulnerability—especially comorbidity burden—the presence of concomitant fractures outside the upper extremity, and fracture location. Larger multicenter studies incorporating standardized frailty and functional measures are needed to confirm these associations and to inform care pathways for high-risk patients.
Keywords
Introduction
Upper-extremity fractures represent a heterogeneous injury spectrum with a bimodal age distribution, showing higher incidence in younger males and older postmenopausal females, and mechanisms ranging from high-energy trauma in younger individuals to low-energy/fragility mechanisms in older adults with compromised bone quality.1-4 Across different healthcare settings, incidence estimates for upper-extremity fractures vary but are commonly in the range of a few hundred per 100,000 person-years.2,3 Epidemiologic studies and registry-based analyses consistently identify the distal radius as one of the most frequent upper-extremity fracture sites, while more proximal injuries—including proximal humerus, diaphyseal humerus, and proximal forearm (e.g., olecranon) fractures—are disproportionately represented among older adults.2,5-9
Beyond functional impairment, an increasing body of evidence suggests that certain upper-extremity fractures in older adults are associated with excess mortality, supporting the concept that a clinically significant upper-extremity fracture may act as a sentinel marker of frailty and medical vulnerability. 10 Proximal humerus fractures, in particular, have been linked to higher short- and medium-term mortality compared with matched population controls, with the greatest relative excess risk observed early after injury and a persistently elevated risk thereafter.11,12 In contrast, mortality findings after distal radius fractures appear more heterogeneous across cohorts, with some studies reporting little or no excess mortality after adjustment for baseline risk, while other fracture locations such as humeral shaft and proximal forearm/olecranon fractures have been associated with substantial comorbidity burden and poorer survival in older populations.13-15 Collectively, these observations imply that fracture location alone may be insufficient to explain long-term outcomes, and that patient-level vulnerability (e.g., comorbidity burden) and injury pattern (e.g., concomitant fractures) likely play a major role in determining survival after upper-extremity fractures.
Although post-fracture mortality has been characterized most extensively after hip fractures, 16 this evidence base has also supported the development of guideline-directed and multidisciplinary care pathways. 17 However, the literature remains more fragmented for major upper-extremity fractures, as prior studies often focus on a single anatomic site, selected treatment cohorts, or shorter follow-up. Moreover, long-term outcomes after upper-extremity fractures are likely determined by an interaction between baseline vulnerability (e.g., comorbidity burden) and injury pattern (fracture location and concomitant fractures), yet these factors are not consistently examined within the same analytic framework.
Therefore, the primary aim of this study was to quantify five-year mortality following major upper-extremity fractures in adults aged sixty-five years and older. The secondary aim was to identify factors associated with mortality, focusing on clinical and fracture-related characteristics.
Materials and Methods
Study Design, Patient Identification, and Eligibility Criteria
This retrospective single-center cohort study included adults who presented between 2014 and 2020 with a suspected upper-extremity fracture. Ethical approval was obtained from the Clinical Research Ethics Committee (decision no. 2023/68). Potentially eligible cases were identified through the hospital information management system by screening predefined ICD codes for upper-extremity fractures.
Inclusion criteria were age sixty-five years or older, presentation during the study period with a suspected major upper-extremity fracture, and radiographic confirmation of the fracture diagnosis on archived radiographs. For patients with more than one eligible presentation during the study period, only the first (index) fracture episode was included.
Exclusion criteria were pathological fractures (e.g., malignancy-related), isolated hand/finger fractures, severe multisystem trauma requiring critical care-level management at presentation, and insufficient imaging or medical records to verify the diagnosis, ascertain comorbidity burden, or determine mortality status. Patients with a history of prior surgery on the same extremity (including fracture fixation or arthroplasty) were also excluded. To restrict the cohort to first-fracture presentations, patients with any documented history of a prior upper- or lower-extremity fracture were not included. High-energy trauma alone was not considered an exclusion criterion.
Radiographic Confirmation
All candidate diagnoses identified through ICD codes were radiographically verified. Two independent observers reviewed archived radiographs to confirm the presence and anatomical location of the fracture. Discrepancies were resolved by consensus review and, when necessary, adjudicated by a senior reviewer.
Definitions and Variable Classification
Fracture diagnoses were categorized into 12 predefined categories based on anatomical location: scapula fracture, clavicle fracture, proximal humerus fracture, humeral shaft fracture, distal humerus fracture, proximal ulna fracture, proximal radius fracture, ulnar shaft fracture, radial shaft fracture, both-bone forearm shaft fracture, distal radius fracture, and both distal radius and distal ulna fracture. Isolated ulnar styloid fractures accompanying distal radius fractures were classified as distal radius fractures rather than as a separate distal ulna fracture category. Fractures were additionally grouped by anatomical region into five categories (shoulder, arm, elbow, forearm, and wrist): scapula, clavicle, and proximal humerus fractures were classified as shoulder; humeral shaft fractures as arm; distal humerus, proximal ulna, and proximal radius fractures as elbow; ulnar shaft, radial shaft, and both-bone forearm shaft fractures as forearm; and distal radius fractures and combined distal radius–distal ulna fractures as wrist. Fracture multiplicity was categorized as a single fracture, multiple fractures within the same region, or multiple fractures involving different regions. Trauma mechanism was categorized as low-energy (simple fall) or high-energy trauma. Treatment approach was categorized as nonoperative or operative management.
At the time of diagnosis, the presence of concomitant fractures outside the upper extremity was recorded. Comorbidity status was assessed from electronic medical records, and the age-adjusted Charlson Comorbidity Index 18 (aCCI) was calculated for each patient using standard definitions. For categorical analyses, aCCI was categorized as low (2–3), moderate (4–5), and high (6 or higher). During follow-up, subsequent fractures were defined as any new fracture episode documented in the hospital digital records after the index fracture. Mortality status and the date of death were obtained from institutional electronic records and verified through linkage using each patient’s unique national identification number. Because all patients had a minimum follow-up of 5 years in the institutional records, five-year all-cause mortality was calculated as the proportion of patients who died within 5 years of the date of fracture diagnosis; patients without a recorded death within this interval were classified as alive at 5 years. Follow-up time was calculated from the index date, defined as the date of fracture diagnosis.
Statistical Analysis
Baseline characteristics were summarized as mean ± standard deviation or median (interquartile range, IQR), as appropriate, for continuous variables and as counts (percentages) for categorical variables. Five-year all-cause mortality was the primary outcome and was defined as death within 5 years of the index fracture diagnosis. Associations between categorical variables and 5-year mortality were assessed using the chi-square test (or Fisher’s exact test when appropriate). Univariable logistic regression was used to estimate odds ratios (ORs) with 95% confidence intervals (CIs) for candidate predictors of 5-year mortality; age was modeled as a continuous variable, and clinically relevant reference categories were selected for categorical predictors (e.g., distal radius fracture for diagnosis and the wrist region for anatomic grouping). For comorbidity burden, analyses used predefined age-adjusted Charlson Comorbidity Index (aCCI) categories (low, moderate, high), and a p value for trend was obtained by modeling the ordered categories in logistic regression. A two-sided p value <0.05 was considered statistically significant.
Results
Patient Characteristics and Injury Profile
Five-Year Mortality by Upper-Extremity Fracture Diagnosis

*Rare categories (<5% frequency) were combined under ‘Other’. OR=odds ratio; CI=confidence interval. ORs were derived from univariable logistic regression with distal radius fracture as the reference; p<0.05 was considered statistically significant.

Distribution of upper-extremity fracture diagnoses (fracture instances, n=1303). Values are shown as n (%)

Distribution of fracture regions (patients, n=1240). Values are shown as n (%)

Distribution of comorbidities (patients, n=1240). Values are shown as n (%)
Factors Associated With 5-Year Mortality
Patients with 5-year mortality were older than those without (77.8 ± 7.7 vs 72.5 ± 5.9 years; p<0.0001). Five-year mortality increased across age groups (65–74: 18.4% [136/740]; 75–84: 36.1% [137/380]; ≥85: 64.2% [77/120]; p for trend<0.0001). Each 10-year increase in age was associated with higher odds of 5-year mortality (OR 3.14, 95% CI 2.59–3.81; p<0.0001).
Among female patients, 5-year all-cause mortality was 25.9% (216/834) compared with 33% among male patients (134/406) (p=0.005). Male sex was associated with higher odds of mortality (OR 1.41, 95% CI 1.09–1.83) compared with female sex.
Fracture diagnosis was significantly associated with 5-year mortality (p<0.001; Table 1). Mortality was highest in patients with humeral shaft fractures, and in univariable logistic regression (reference: distal radius fracture), humeral shaft fractures showed markedly increased odds of mortality (OR 4.67, 95% CI 2.8–7.77; p<0.001). Compared with distal radius fractures, mortality odds were also higher for proximal humerus fractures (OR 2.17, 95% CI 1.59–2.98; p<0.001), clavicle fractures (OR 1.76, 95% CI 1.08–2.87; p=0.029), and the “other” diagnosis group (OR 2.27, 95% CI 1.47–3.51; p<0.001), whereas the multiple-fracture group did not differ from the reference category (OR 1.04, 95% CI 0.63–1.73; p=0.879).
Five-Year Mortality by Upper-Extremity Fracture Region
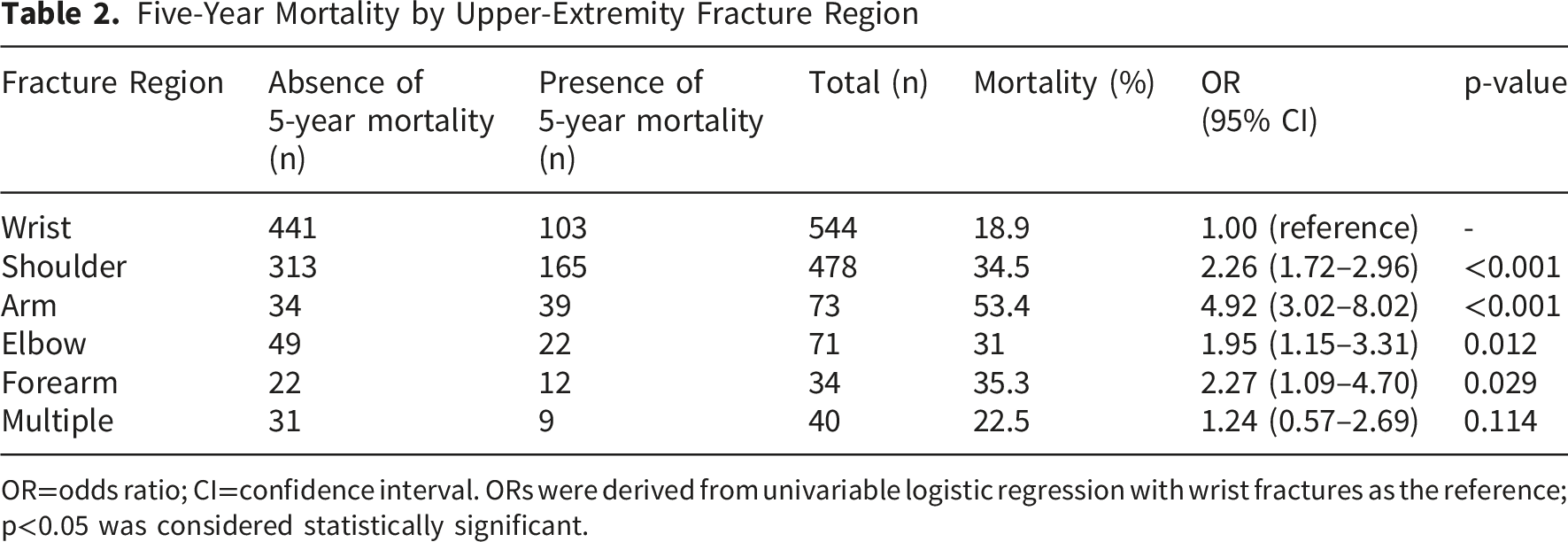
OR=odds ratio; CI=confidence interval. ORs were derived from univariable logistic regression with wrist fractures as the reference; p<0.05 was considered statistically significant.
The number of upper-extremity fractures was not significantly associated with 5-year mortality (p=0.084). Five-year mortality was 29% (327/1127) in patients with a single fracture, compared with 19.2% (14/73) in those with multiple fractures in the same region and 22.5% (9/40) in those with multiple fractures in different regions; neither multiple-fracture category differed significantly from the single-fracture reference (same region: OR 0.61, 95% CI 0.34–1.09; p=0.094; different regions: OR 0.71, 95% CI 0.33–1.51; p=0.477).
Mechanism of injury was not significantly associated with 5-year mortality (p=0.387). Mortality was 29.1% (55/189) in the high-energy trauma group and 28.1% (295/1051) after simple falls. High-energy trauma was not associated with the odds of mortality (OR 1.05, 95% CI 0.75–1.48).
Concomitant fractures outside the upper extremity were significantly associated with 5-year mortality (p=0.0027). Mortality was 38% (63/166) among those with a concomitant fracture outside the upper extremity, compared with 26.7% (287/1074) among those without. The presence of a concomitant fracture outside the upper extremity was associated with higher odds of mortality (OR 1.68, 95% CI 1.19–2.36).
The presence of comorbidity was significantly associated with 5-year mortality (p<0.001). Mortality was 35.3% (253/717) among those with at least one comorbidity, compared with 18.5% (97/523) among those without. Comorbidity was associated with higher odds of mortality (OR 2.39, 95% CI 1.83–3.13).
Five-Year Mortality by Age-Adjusted Charlson Comorbidity Index (aCCI) Category

OR=odds ratio; CI=confidence interval. ORs were derived from univariable logistic regression with the low aCCI category as the reference; p<0.05 was considered statistically significant.
Treatment type was not significantly associated with 5-year mortality (p=0.160). Mortality was 31.6% (79/250) after surgery and 27.4% (271/990) with nonoperative treatment (OR 1.23, 95% CI 0.91–1.66).
Discussion
In this retrospective study of older adults with major upper-extremity fractures and a minimum of five years of follow-up, long-term mortality was substantial and was largely explained by baseline vulnerability and fracture location. Mortality differed across diagnoses and anatomic regions, with the lowest risk after distal radius and wrist-region fractures and the highest risk after humeral shaft fractures. Comorbidity burden emerged as a central driver, with both the presence of comorbidity and increasing age-adjusted Charlson Comorbidity Index categories strongly associated with higher mortality.
In our study, distal radius fractures had the lowest five-year mortality (19.7%), whereas humeral shaft fractures had the highest (53.4%), and proximal humerus fractures were intermediate (34.8%). After grouping diagnoses by anatomic region, five-year mortality was lowest after wrist fractures (18.9%) and highest after arm fractures (53.4%), with intermediate mortality after shoulder (34.5%), elbow (31%), and forearm (35.3%) fractures. In a Swedish register-based cohort of patients aged ≥80 years with distal radius fractures, Arvidsson et al 19 reported a low 1-year mortality (5%) and a mortality ratio of 0.44 compared with the background population. Ruffner et al, 20 using U.S. inpatient data on surgically treated humeral shaft fractures, reported higher odds of in-hospital mortality in patients aged ≥80 years than in younger geriatric groups. In a nationwide Swedish register study, Sumrein et al 11 reported 1-year and 4-year mortality rates of 8.5% and 24% after proximal humerus fractures, rising to 12% and 33% among patients aged ≥65 years. Although elbow-region fractures have been less frequently studied, available studies report 1-year mortality ranging roughly from 4% to 16% after olecranon fractures in older adults and around 14% after distal humerus fractures in inpatient cohorts.10,15,21,22 Overall, this site-specific gradient is broadly consistent with the limited literature and supports interpreting fracture location as a practical marker of vulnerability in older adults. We suspect the higher risk after humeral shaft fractures reflects greater functional burden and deconditioning with immobilization, whereas the relatively low risk after distal radius fractures may partly reflect better baseline health.
Older age and male sex were associated with higher mortality in our study. This pattern is consistent with prior studies examining mortality after upper-extremity fractures in older adults.10-13 Comorbidity burden was common, and cardiometabolic conditions such as hypertension and diabetes were among the most frequently recorded. This comorbidity profile is typical for older upper-extremity fracture populations, where cardiometabolic conditions are commonly reported and frequently included in risk-adjustment models.13,23 Accordingly, the presence of at least one comorbidity and increasing aCCI category were both associated with higher mortality, indicating a stepwise risk gradient with greater comorbidity burden. Similar relationships have been reported after proximal humerus fractures, where higher Charlson scores predict mortality and patients with substantial comorbidity (e.g., CCI >5) consistently represent a higher-risk group.24,25 Large registry analyses further show that specific comorbid conditions commonly captured within these indices (e.g., COPD, cerebrovascular disease, chronic kidney disease, and congestive heart failure) are independently associated with mortality risk, supporting the clinical relevance of comorbidity-based risk stratification.22,23 Beyond proximal humerus fractures, comorbidity burden has also been incorporated into distal radius fracture studies using the Charlson Comorbidity Index, reinforcing the relevance of comorbidity-based risk stratification across upper-extremity fracture sites. 19 Taken together, these findings emphasize that baseline comorbidity burden should be considered a key driver of mortality risk after upper-extremity fractures, alongside injury-related factors.
Although we could not identify a directly comparable upper-extremity fracture study that examines “non–upper-extremity concomitant fractures” in the same way as our variable, the geriatric hip-fracture literature provides a useful framework for interpretation. In older adults with hip fractures, concomitant fractures or injuries have repeatedly been linked to worse short-term outcomes, including longer hospital stay, higher in-hospital mortality, delayed surgery, and reduced likelihood of discharge home, plausibly reflecting greater overall trauma burden, impaired early mobilization, and higher complication risk.26-28 Recent hip-fracture studies suggest that the prognostic impact depends on the type of the concomitant upper-extremity injury, with proximal humerus or humeral fractures generally identifying higher-risk subgroups than concomitant wrist fractures.29,30 In our study, such concomitant fractures were associated with higher mortality. Framed in this way, our finding supports treating concomitant fractures outside the upper extremity as a practical clinical “red flag” that may warrant closer monitoring and more proactive multidisciplinary care in older patients presenting with major upper-extremity fractures.
In our study population, 5-year mortality did not differ meaningfully by the number of upper-extremity fractures, injury mechanism, or treatment type. For fracture multiplicity, the absence of an association may reflect heterogeneity in what “multiple fractures” captures and limited power in the smaller multi-fracture strata. Mechanism also showed little separation in long-term risk, which may be expected in older adults because even low-energy falls can lead to clinically consequential injury patterns; national stair-fall data similarly suggest that downstream severity varies substantially by fracture location, despite a shared low-energy mechanism. 31 With respect to treatment, our dichotomous operative/nonoperative classification may have been too coarse to capture differences in indications and physiologic reserve over a five-year horizon. Prior studies report mixed findings on treatment, with some suggesting higher early mortality after nonoperative management, while others show little difference once baseline vulnerability and comorbidity burden are considered.14,23,32 Taken together, variation in case-mix, endpoint timing, and treatment-definition granularity likely contributes to these discrepancies; in our cohort, broad categories for mechanism, multiplicity, and treatment may have pooled clinically distinct subgroups, diluting any association over five years.
The results of the present study have practical implications for early risk stratification in older adults presenting with major upper-extremity fractures. Patients with more proximal fracture patterns, higher comorbidity burden, and concomitant fractures outside the upper extremity appear to represent a subgroup with greater vulnerability and may benefit from closer follow-up and proactive multidisciplinary care. Practical steps may include early geriatric input where available, prompt optimization of comorbid conditions, and an emphasis on early mobilization and complication prevention during the acute phase. Although evidence is limited, care pathways conceptually similar to those used for hip fractures may be worth considering for selected high-risk upper-extremity fracture patients, while acknowledging that prospective studies are needed to define the most effective approach.
Our findings should also be interpreted in the context of the existing literature, much of which relies on nationwide registries or administrative datasets that provide very large sample sizes but typically limit analyses to a relatively narrow set of coded variables.11-14 In contrast, our study was not based on a national database and therefore included a more modest sample size; however, it allowed a more detailed clinical review. Patients were identified through institutional records and fracture diagnoses were confirmed on archived radiographs, enabling classification by both diagnosis and anatomic region. Key covariates, including comorbidity burden and concomitant fractures, were obtained from medical records rather than inferred solely from administrative coding. In addition, the distribution of fracture diagnoses and regions in our sample was consistent with commonly reported patterns in older adults with wrist- and shoulder-region injuries accounting for the majority of cases.2,3,5 Finally, because all included patients had at least five years of follow-up, mortality ascertainment was straightforward and consistent across the study population.
This study has several limitations. Its retrospective, single-institution design may limit generalizability, and no a priori sample size calculation was performed because the cohort comprised all eligible patients identified during the study period. Residual confounding is possible because several geriatric vulnerability markers were not available beyond comorbidity burden (e.g., formal frailty or functional status measures, ASA class, BMI, socioeconomic factors, medication burden, osteoporosis status, detailed endocrine conditions, and hormone therapy use). Detailed injury-level factors, such as fracture complexity and soft tissue injury severity, were also not uniformly available. Cause-specific mortality could not be examined. In addition, some diagnosis and region subgroups were relatively small, which may have reduced power for detecting differences in less common fracture patterns and in multi-fracture strata. Our analyses also relied on pragmatic binary classifications for injury mechanism and treatment, which likely pooled heterogeneous clinical scenarios and may have attenuated associations over a five-year horizon. Moreover, our eligibility criteria (including exclusions related to prior upper-extremity surgery/fracture history and incomplete records) may have introduced selection bias and could limit applicability to more complex or recurrent-injury populations. Nevertheless, the study has important strengths, including radiographic confirmation of fracture diagnosis, clinically detailed covariate capture from medical records (including comorbidity burden and concomitant fractures), and complete long-term outcome ascertainment supported by national identification number linkage with a minimum of five years of follow-up for all included patients. Comorbidities were ascertained through review of electronic medical records rather than administrative coding alone. Taken together, these features support the robustness of our main observation that long-term mortality after major upper-extremity fractures in older adults is driven primarily by baseline vulnerability and fracture location, and they provide a practical basis for bedside risk stratification.
Conclusion
In conclusion, major upper-extremity fractures in older adults were associated with substantial long-term mortality. Our results suggest that baseline vulnerability, particularly comorbidity burden, and fracture location are important markers of risk, but these observations should be interpreted in light of the study’s retrospective, single-center design. Larger multicenter studies with standardized frailty and functional measures are needed to confirm these associations and to inform the most effective care pathways for high-risk patients.
Footnotes
Ethical Considerations
Ethical approval was obtained from the Mersin University Clinical Research Ethics Committee (decision no. 2023/68).
Consent to Participate
Not applicable/Waived due to retrospective study design.
Author Contributions
Conceptualization: EÖ, EE, MYH, EG, ZMA
Methodology: EÖ, EE, MYH
Data curation: EÖ, EE, MYH
Formal analysis: EG, ZMA
Investigation: EÖ, EE, MYH, EG, ZMA
Writing – original draft: EÖ, EE, MYH
Writing – review & editing: EG, ZMA
Supervision: ZMA
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
