Abstract
Introduction
Total knee replacement (TKA) is a treatment that removes articular surfaces that cannot be repaired by themselves and replaces them with artificial joints to eliminate pain and stabilize the knee joint. It can be unilateral for one-sided lesions or bilateral for two-sided lesions.1,2 According to a survey in 18 countries in 2012, about 1.1 million people received TKA every year, and the number increased by 11% yearly. 3 In China, the overall prevalence of symptomatic knee osteoarthritis was 8.1%, 4 and the total number of TKA has now reached nearly 300 000 each year. 5 However, the procedure is associated with substantial perioperative blood loss, often leading to acute anemia and blood transfusion, which can lead to serious morbidities such as hemolytic reactions, acute lung injuries, and transfusion-related infection. 6 Therefore, reducing blood loss and minimizing the risk of transfusions in TKA have drawn the great attention of orthopedic surgeons. 7
Tranexamic acid (TXA) is a synthetic agent that exerts its antifibrinolytic effects by inhibiting plasminogen. Tranexamic acid inhibits plasminogen activation by binding plasmin to fibrin, which leads to clot stabilization and reduces blood loss. 8 TXA can be delivered topically, intravenously, or by combined application. Though the effectiveness of TXA is well established in the literature, the best practices regarding the route or dosage of TXA in TKA remain controversial.
This study aimed to compare the efficacy and safety of intravenous, topical, or combined routes of TXA for patients with TKA.
Materials and Methods
Patients
From Jan 2019 to June 2021, medical records of patients aged 65 years or older who underwent primary unilateral TKA for primary osteoarthritis were retrospectively reviewed. The exclusion criteria were secondary osteoarthritis, allergy to TXA, a history of coexisting diseases that cannot tolerate surgery or general anesthesia, and active cancer.
The study protocol was reviewed and approved by the Ethics Committee of our hospital. As it is a retrospective study, consent to participate is waived by the Ethic Committee of our hospital.
Study Design
The included patients were divided into 3 groups according to the methods of TXA application: intravenous (IV) group, topical group, or combined group. In the IV group, the patients received .5 g of IV TXA prior to tourniquet inflation and a second .5 g of IV TXA 3 hours after the first administration; In the topical group, patients received 1.0 g of TXA in 10 mL of saline solution 5 minutes prior to final tourniquet release; In the combination group, patients received .5 g of IV TXA prior to tourniquet inflation, 1.0 g of TXA in 10 mL of saline solution 5 minutes prior to final tourniquet release and a second .5 g of IV TXA 3 hours after the first administration. All patients received combined spinal-epidural anesthesia, an adductor canal block with .25% bupivacaine, and an arterial line.
Propensity-score match (PSM) was used to reduce the bias and imbalance of confounding variables. In this study, patient demographic characteristics including age, gender, American Society of Anesthesiologists (ASA) physical status classification, and body mass index (BMI), and pre-operative laboratory values including prothrombin time, international normalized ratio (INR), platelet count, hematocrit, and hemoglobin (Hb) were used to estimate the propensity score. A 1:1:1 greedy match was performed based on a caliper width of .2 for the propensity score. Initially, 367 patients (159 in the IV group, 143 in the topical group, and 65 in the combination group) met the inclusion and exclusion criteria, and after PSM, 65 patients in each group were included in this analysis.
Surgical Technique and Postoperative Care
All surgeries were performed by the same orthopedic surgeon using an anterior midline incision and a medial parapatellar approach with a tourniquet (pressure of 260 mmHg). The Scorpio NRG Knee System implants (Stryker, Kalamazoo, Michigan, USA) were inserted, balanced, adjusted, and cemented. After surgery, a drainage tube was placed to connect to an autologous blood transfusion device (Stryker, Kalamazoo, USA) without clipping and a pressure dressing was applied with an elastic bandage before the tourniquet was slowly released. Half a dose of low-molecular-weight heparin (LMWH) (.2 mL 2000 IU) was started 6 h postoperatively and repeated at 24-hour intervals with a full dose (.4 mL 4000 IU) in subsequent days. In addition, an intermittent foot slope pump system was used as a routine practice to prevent deep-vein thrombosis (DVT). After discharge, 10 mg of rivaroxaban was administered orally to patients for 10 days. Postoperative rehabilitation included mobilization with a walker, muscle strengthening, and range of motion exercises as tolerated on the first postoperative day. The patients were discharged from the hospital when they were able to walk independently with a walker.
Outcome Measures
The primary outcome was total blood loss. The Nadler formula was used to calculate blood volume and the Gross formula was used to calculate and report total blood loss.9,10 Nadler’s formula for men: Patients’ blood volume (PBV) = .3669 × H3 + .03219 × W + .6041; Nadler’s formula for women: PBV = .3561 × H3 + .03308 × W + .1833, where H is the patient’s height in m, and W is the body weight in kg. 9 Gross’ formula: Blood loss = PBV × (H0-HF)/((H0 + HF)/2), where PBV is the patient’s blood volume, H0 is the patient’s initial hematocrit value, and HF is the patient’s minimum hematocrit value. 10
Secondary outcomes included hidden blood loss, the reduction of Hb concentration, transfusion rate, length of hospital stays, and incidence of DVT. Hidden blood loss was defined as total blood loss minus intra-operative blood loss. The reduction of Hb concentration was defined as pre-operative Hb levels minus Hb levels measured on the day after the operation. Intra- and post-operatively, blood transfusion was performed on all patients with a Hb below 8 g/dL or Hb below10 g/dL in patients with significant symptoms of anemia despite volume repletion. Doppler ultrasound examination was routinely used to detect DVT at the time of discharge and 6- month follow-up assessments or at any time there was clinically suspected DVT.
Statistical Analysis
Statistical power was determined on the primary outcome of total blood loss. It was determined that 65 patients in each group could provide a power of >90% with an alpha of 5% to determine a difference of 100 mL in the total blood loss among the 3 groups.
Continuous data are presented as means ± standard deviations (SDs) and compared using the one-way analysis of variance (ANOVA), followed by post-hoc analysis. Categorical data are presented as numbers and percentages and compared using the chi-square test. A P value <.05 was considered significant. The analyses were performed with the use of SPSS software v. 20.0 (IBM Corp, Armonk, NY).
Results
Patients’ Baseline Demographics and Clinical Characteristics.

Comparison of Efficacy Outcomes Among Combined, IV, and Topical TXA Groups.

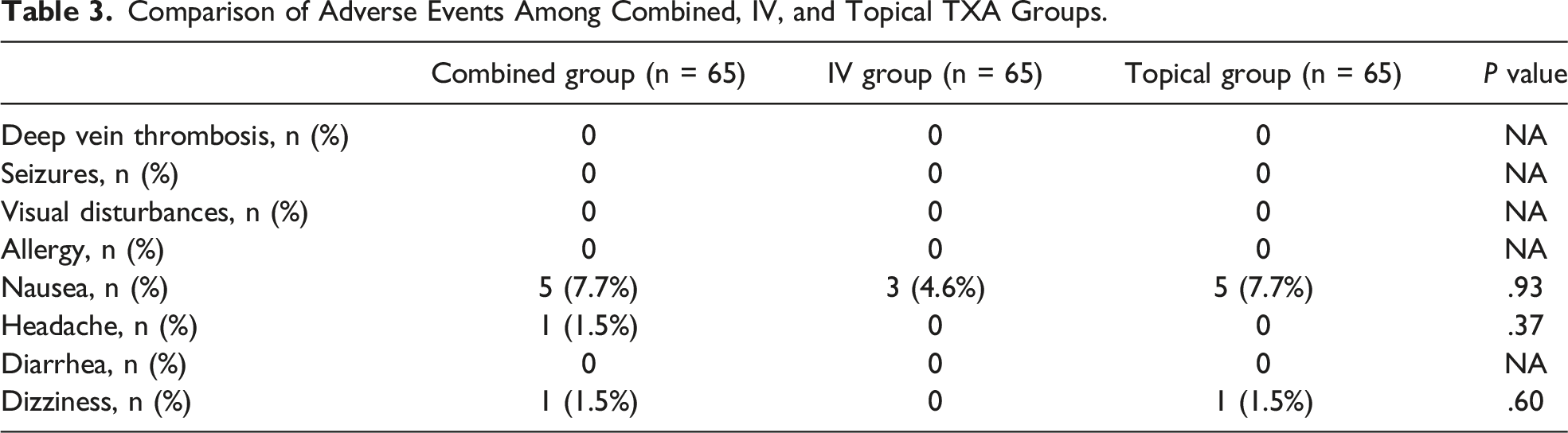
Comparison of Adverse Events Among Combined, IV, and Topical TXA Groups.
Discussion
With the growing data on the efficacy and safety of TXA, various routines of TXA in primary and revision procedures have been widely adopted for reducing blood loss during total joint replacement and ensuring fast postoperative recovery. 11 IV administration achieves a rapid TXA level in the synovial fluid of the target joint 12 but there are contraindications such as a previous history of pulmonary thromboembolism, deep vein thrombosis or ischemic events. 13 For its part, topical administration provides maximum concentration of TXA in the surgical site associated with a minimal systemic effect, thus reducing possible systemic adverse effects.14,15 Until now, the efficacy and safety of topical TXA compared to IV TXA for controlling blood loss and transfusions in TKA still remains controversial. Therefore, the new strategy of TXA administration, the combined regimen, has been explored. 16 The present study revealed that compared with the IV or topical application of TXA, a total dose of 2 g combined IV and topical application of TXA significantly reduced total blood loss, hidden blood loss, and Hb drop without increased risks of transfusion requirement and adverse events in primary TKA.
Though there are many studies on combined IV and topical TXA in TKA, the methods and effects are still controversial. A prospective randomized controlled double-blinded trial conducted by Prakash et al. indicated that a combination of preoperative and postoperative TXA injections of 10 mg/kg with 1.5 g of topical TXA is most effective to decrease postoperative bleeding and requirement of transfusion in unilateral TKA without increasing any risk of complications compared with a single use of IV or topical application of TXA. 17 However, in another randomized clinical trial conducted by Song et al, 18 patients in the combined group received the same dose as Prakash et al.’s study, 17 but did not observe a significant difference in the blood loss compared with a single administration of IV or topical group. The reason for these conflicting results is still unclear. Thus, more studies are required to explore the routes and efficacy of combined administration of IV and Topical TXA.
The treatment regimen proposed in the study was based on a literature review and our experience. A lot of studies selected a 10 mg/kg or 1 g dose of TXA for IV use and 1-3 g for topical use. Some authors recommended that the dose of topically administered TXA >2 g play its role in reducing blood loss and the transfusion rate.19-21 However, this recommendation was meant for a single-TXA-use strategy (IV or topical) and is not suitable for our combined strategy. Based on our preliminary study, a total of 2 g TXA was used with 1 g for IV administration and 1 g for topical use. It is demonstrated that the combined therapy is safe and could provide enough efficacy to use this lower dose, rather than the maximum dose of 3-4 g in previous studies, for the combined TXA strategy.
Hidden losses are reported to be approximately 40%–50% of total losses,17,22 which was consistent with our series for the IV group (43.9% of total loss). However, the hidden losses for the topical group were 36.9% and 38.9% for the combined group, indicating that the intraarticular administration was effective in reducing hidden losses. These hidden losses are thought to be due to bleeding from tissues and hemolysis, and a direct high concentration of drug in tissues may have decreased this part of bleeding resulting in lower hidden losses. A study conducted by Prakash et al. also revealed that intraarticular administration could reduce postoperative hidden blood loss. 17
Limitations
There are some limitations of this study. Firstly, the present study has not addressed the subjective knee function score of the patients after TKA, as the main purpose of this study was to evaluate the ideal mode of administration of the drug. In addition, although no thrombotic events were observed, our study was not powered for such clinical outcomes. Last, we also did not include patients who underwent simultaneous bilateral TKA in the present study, and therefore, our conclusions may not apply to these patients.
In conclusion, combined administration of IV and topical TXA is the most effective approach to decrease postoperative total blood loss and Hb drop in the treatment of TKA without increasing any risk of complications.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
