Abstract
Background
The aim of this meta-analysis was to evaluate the efficacy and safety of peri-articular injection of tranexamic acid (TXA) during total knee arthroplasty (TKA) from clinical controlled trials.
Method
Eligible scientific articles published prior to October 2021 were retrieved from the PubMed, Springer, ScienceDirect and Cochrane Library databases. The statistical analysis was performed with RevMan 5.1.
Result
2 RCTs and 3 non-RCTs met the inclusion criteria. Meta-analysis showed significant differences in terms of hemoglobin reduction (MD = −1.04, 95% CI: −1.33 to −.76, P < .00001), total blood loss (MD = −342.80.70, 95% CI: −437.52 to −248.08, P < .00001), drainage volume (MD = −297.24, 95% CI: −497.26 to −97.23, P = .004) and blood transfusion rate (OR = .30, 95% CI: .14 to .62, P = .001) were found in the control group. No postoperative infection and deep venous thrombosis were found between 2 groups.
Conclusion
Peri-articular injection of TXA can effectively decrease perioperative blood loss and blood transfusion rate without increasing the incidence of postoperative complications during TKA.
Introduction
Total knee arthroplasty (TKA) is a successful treatment to relieve pain and improve postoperative knee function. 1 However, substantial blood loss is associated with the surgical procedure, ranging from 1000 mL to 2000 mL. 2 It has been reported that 10-38% of patients require blood transfusion after TKA. 3 Allogenic blood transfusions result in the risk of adverse effects and increase medical cost. 4
Tranexamic acid (TXA) is a synthetic amino acid derivative that promotes hemostasis by competitively blocking the lysine-binding site of plasminogen. 5 To date, administration of TXA has been well established in the literature to reduces blood loss and transfusion requirements during TKA.6,7 Recently, a new local administration method of TXA, periarticular injection (PAI), was introduced. Mao et al 8 reported that the direct injection of TXA solution into the soft tissue around the joint cavity that is vulnerable to postoperative bleeding is expected to decrease bleeding more effectively. Several studies9-12 have reported that PAI of TXA effectively reduces blood loss and allogenic blood transfusions during TKA. However, whether PAI is safe and efficient in TKA remains controversial. Therefore, we performed a meta-analysis with a large sample to evaluate the efficacy and safety of PAI of TXA in TKA from clinical controlled trials.
Materials and Methods
Search Strategy
This meta-analysis was conducted in accordance with the relevant Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. Potentially relevant scientific articles published from the inception of the electronic databases to October 2021 were retrieved from the PubMed, Springer, ScienceDirect and Cochrane Library databases. A final check that no relevant articles were missed was conducted by searching manually all references of included studies and by preforming citation tracking on the included articles. There were no restrictions on language of publication. The keywords used for the search were “total knee arthroplasty”, “tranexamic acid” and “peri-articular”.
Inclusion Criteria
Studies were considered eligible for inclusion if they met the following criteria: (1) the patients were treated with primary TKA; (2) the experimental group was treated via the PAI of TXA and the control group was treated with placebo or null; (4) the clinical outcomes included hemoglobin reduction, total blood loss, postoperative drainage volume, blood transfusion requirements and postoperative complications; and (5) the study was a published comparative trial, including randomized controlled trials (RCTs) and non-RCTs. Two independent reviewers screened the titles and abstracts of all studies identified by the search strategy. A third reviewer resolved any disagreements.
Exclusive Criteria
We excluded articles that were (1) duplicate articles or articles having the same patients, content and results; (2) case reports, conference reports, theoretical research, systematic reviews, meta-analyses, expert comments, and economic analyses; and (3) studies whose outcomes that were not relevant.
Data Extraction
Predefined data collection form was developed to extract data from the eligible studies by 2 independent reviewers. The following information was extracted: the author’s name, publication date, the intervention measures, patient demographics and the outcome measures. Other relevant parameters were also extracted from the individual studies. Corresponding authors of included studies were contacted via email for relevant information if the available data were insufficient.
Quality Assessment
The methodological quality of the RCTs was assessed by the modification of the generic evaluation tool described in the Cochrane handbook for systematic reviews of interventions. 13 The methodological quality of the non-RCTs was evaluated using the methodological index for nonrandomized studies (MINORS). 14
Data Analysis and Statistical Methods
The statistical analysis was performed using Review Manager 5.1 (The Cochrane Collaboration, Oxford, United Kingdom). Heterogeneity of the mean difference across studies was checked using the chi-squared test and I2 statistic. If significant (P < .1 or I2 > 50%), a random effects model was used to estimate the overall effect sizes and a sensitivity analysis was performed to investigate the potential sources of heterogeneity. Otherwise, fixed-effects model was adopted. The continuous data are expressed as mean differences (MDs) and 95% confidence intervals (CIs). The odds ratio (OR) and 95% CIs were calculated for the dichotomous data.
Results
Search Results
A total of 53 studies were identified as potentially relevant literature reports. There were no additional studies identified through other sources. After carefully scanning the titles, 48 studies were excluded. Eventually, 3 non-RCTs and 2 RCTs and were eligible for data extraction and meta-analysis. The process of study selection was showed in Figure 1. Flowchart of the study selection process.
Characteristics of the Included Studies
Characteristics of included studies.

TXA: tranexamic acid, C: control, DVT: deep venous thrombosis, LMWH: low molecular weight heparin, NS: not state.
Risk of Bias Assessment
The methodological quality of the RCTs were displayed in Figure 2. The inclusion and exclusion criteria were clearly described in all RCTs. All included RCTs reported methodology for randomization, concealment of allocation and blinding. Unclear bias was not reported due to incomplete outcome data or selective outcomes. All included non-RCTs were evaluated by the MINORS, and their scores ranged from 20 to 22 (Table 2). The summary of bias risk of randomized controlled trials. Quality assessment for non-randomized trials.

Outcomes of the Meta-analysis
Hemoglobin reduction
Hemoglobin reduction was documented in 3 studies. Present meta-analysis demonstrated that the hemoglobin drop in the PAI group was lower than that in the control group (MD = −1.04, 95% CI: −1.33 to −.76; P < .00001; Figure 3). Forest plot showing hemoglobin drop.
Total Blood Loss
Four studies stated total blood loss. Present meta-analysis demonstrated that total blood loss in the PAI group was lower than that in the control group (MD = −342.80, 95% CI: −437.52 to −248.08; P < .00001; Figure 4). Forest plot showing total blood loss.
Drainage Volume
Drainage volume was assessed in 2 studies. Present meta-analysis demonstrated that the drainage volume in the PAI group was lower than that in the control group (MD = −297.24, 95% CI: −497.26 to −97.23; P = .004; Figure 5). Forest plot showing drainage volume.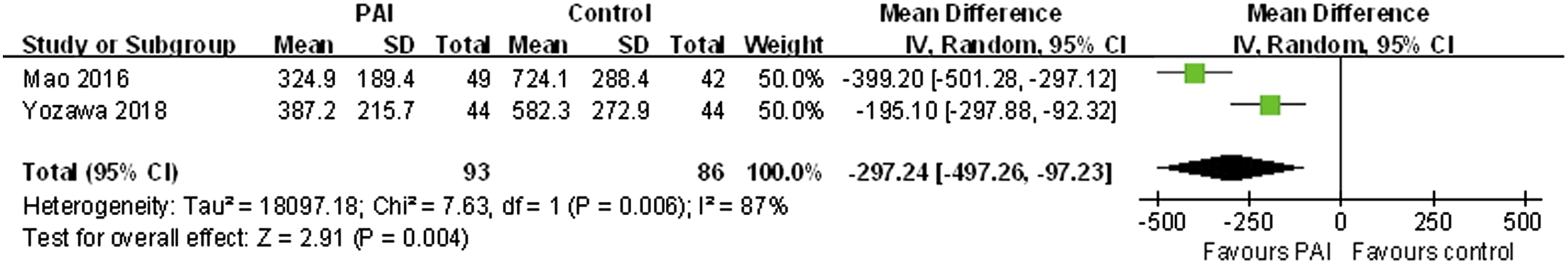
Blood Transfusion Rate
Three studies reported postoperative blood transfusion rate. Present meta-analysis demonstrated that the blood transfusion rate in the PAI group was lower than that in the control group (OR = .30, 95% CI: .14 to .62; P = .001; Figure 6). Forest plot showing blood transfusion requirement.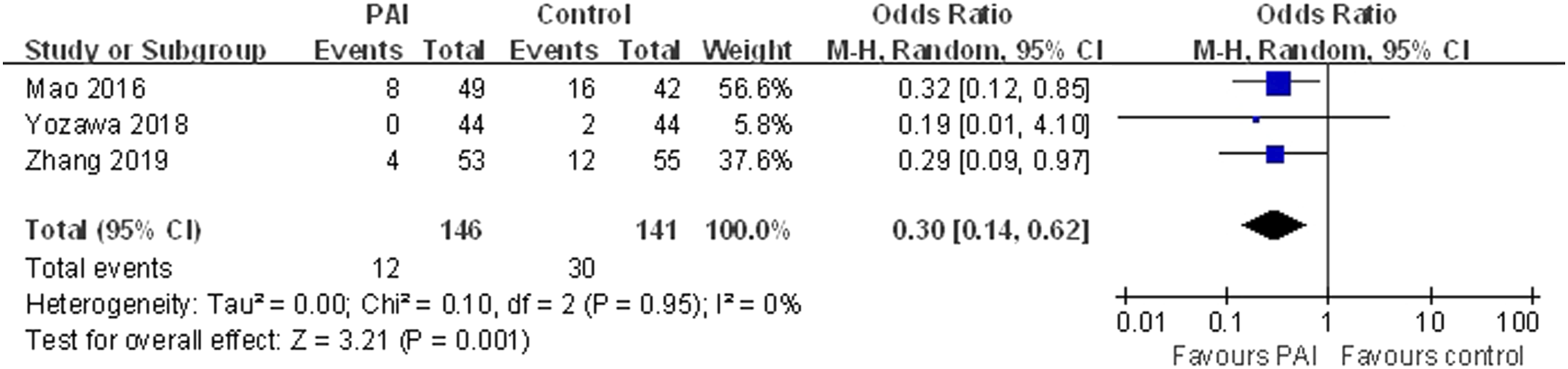
Other Outcomes
All included studies reported that none of the patients had symptomatic DVT postoperatively. Four of included studies reported that there was no infection in 2 groups.
Discussion
The mot importing finding of current meta-analysis was that PAI of TXA during TKA would reduce total blood loss, hemoglobin reduction, blood transfusion rate and drainage volume without increasing risk incidence of infection or DVT. Furthermore, no TXA-related adverse effect was discovered. To our knowledge, the present study is the first quantitative meta-analysis to evaluate the efficacy and safety of PAI of TXA during TKA.
Reducing perioperative blood loss during TKA is 1 of the most important goals today to accelerate patients’ recovery. TXA was reported to effectively reduces blood loss and transfusion requirements during TKA in intravenous or topical administration.6,7 PAI allow TXA solution directly injected into the soft tissue around the joint cavity that is vulnerable to postoperative bleeding. 8 Besiris et al 15 found that PAI of TXA after TKA is superior to reduce hemoglobin reduction, transfusion rate and length of hospital stay when compared with IAI route. Present meta-analysis indicated that PAI of TXA could significantly decrease the drainage volume and total blood loss.
Postoperative anemia is related to a longer length of hospital stay, wound complications, poor functional recovery and even death, especially for geriatric patients. 16 It has been reported that 10-38% of patients require blood transfusion after primary TKA to correct postoperative anemia. 3 A randomized controlled trial study by Pinsornsak et al 11 compared hemoglobin reduction between patients who received PAI of TXA or not during TKA. They observed that the PAI of TXA group had significantly lower hemoglobin reduction than the control group. Mao et al 8 and Yozawa et al 9 reported similar results. The pooled results demonstrated significant differences in the transfusion rate and hemoglobin reduction between the TXA group and the control group.
DVT is a common complication after TKA which may develop to pulmonary embolism and even death. 17 Although administration of TXA in TKA has been proven does not increase the risk for DVT, the effects of TXA on thromboembolic events and mortality remain uncertain. 18 None of DVT was reported in all the included studies. Pinsornsak et al 11 reported that PAI of TXA resulted in significantly lower serum TXA levels postoperatively when compared with others route. They concluded that PAI of TXA may be safer to high risk patients for thromboembolic events.
The limitations of present meta-analysis include the relatively low numbers for sample size. In addition, another limitation is the lack of high-quality evidence in several articles. Furthermore, we found several results with heterogeneity. However, we cannot perform further analysis by subgroup analysis due to insufficient data on this topic.
Conclusion
Peri-articular injection of TXA can effectively reduce perioperative blood loss and blood transfusion rate without increasing the incidence of postoperative complications during TKA.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Tianjin Health Science and Technology Project (RC20120, ZC20096).
Authors’ Note
YL, DS, PT, ZJL, GJX, and XF contributed to conception and design of this study. Study selection and data extraction of the finally included studies were done by YL and DS, PT and XF independently assessed the methodological quality of each included study. YL and DS, PT, ZJL, GJX, and XF contributed to preparation of the manuscript. The final version of the article was approved by all the authors.
