Abstract
Introduction
Hip fractures cause significant morbidity and mortality for geriatric patients, and incidence is increasing as the population ages. Following a primary hip fracture, up to 20% may suffer a contralateral hip fracture within 5 years despite fracture risk reduction measures, including fall prevention and osteoporosis pharmacologic treatment. The aim of this study is to assess whether insertion of the Femoral Neck System (Depuy Synthes, West Chester, PA) into the contralateral proximal femur may strengthen the bone and decrease the incidence of contralateral hip fractures.
Materials and Methods
ScanIP, an image processing software was used to produce 3-dimensional models of a cadaver femur with the implanted device. Models were meshed and exported to Abaqus for finite element analysis to evaluate the device’s ability to reduce stress in the proximal femur. Results were analyzed for element-wise volume and von-Mises stresses.
Results
The implant reduced peak stress and bone failure at all levels of bone quality. Specifically in osteoporotic bone, the implant decreased peak stress by 27%, proximal femur trabecular bone failure by 5% and cortical bone failure by 100% in the femoral neck.
Conclusions
Our results from computer generated finite element analyses indicate that the Femoral Neck System may strengthen an osteoporotic proximal femur in the event of a lateral fall. Further investigation with expanded finite element analysis and cadaveric biomechanical studies are needed to validate these results.
Introduction
The estimated annual incidence of geriatric hip fractures globally is expected to reach 6.3 million by 2050 1 reflecting the increased longevity of the population and decrease in bone mineral density observed with aging.2-4 Hip fractures cause significant morbidity and mortality. Recent studies have shown that the risk of a contralateral hip fracture following primary hip fracture ranged from 3.5% - 20% with increased mortality of up to 64% and loss of 27 disability-adjusted life years (DALYs) per 1000 individuals.5,6 These studies demonstrate the susceptibility of the contralateral proximal femur in the setting of hip fracture and the importance of contralateral hip fracture prevention.
Primary prevention for hip fractures centers around pharmacotherapy. In addition to calcium and vitamin D fortification, antiresorptive agents, such as bisphosphonates (BPs), are the most common treatment option. However, use of these drugs is challenged by suboptimal patient adherence and “paradoxical side effects”, such as atypical femoral fractures and avascular necrosis of the jaw.7-11 Although BPs demonstrated increases in bone mineral density (BMD) and reduction in fracture risk,7,12 recent work suggests that pharmacologic treatment with BPs lacks efficacy, showing that 175 women would need to be treated with BPs for 3 years before preventing 1 hip fracture. 13
Prevention using non-pharmacologic means has also been examined. Wearable external hip pads attenuate forces that the hip experiences in the event of a fall and have been studied for their use in prevention of hip fracture. However, prior studies have only demonstrated a “marginally statistically significant reduction in incidence of hip fracture” coupled with poor patient adherence. 14
Femoroplasty using polymethyl methacrylate (PMMA) bone cement has also been studied to reduce contralateral hip fractures. Injection of PMMA into the femoral head has shown promising biomechanical results, with increased fracture load of up to 82% and increased energy to fracture of up to 188%. However, femoroplasty has been associated with several risks including thermal tissue necrosis, fat embolism, and an increased occurrence of subtrochanteric and atypical femoral shaft fractures.15-18
Augmentation of the proximal femur with the combination of PMMA and metal implants has also shown promising potential in reducing the risk of femoral neck and trochanteric fracture.3,18 Limitations of this approach may include increased complication of revision surgery if the patient were to fracture around the devices, or if the devices should fail. Additionally, significant mismatch between the material properties of the construct and the bone may act as a stress riser or induce stress shielding, leading to weakening of surrounding bone and increasing the risk of device failure under repetitive physiologic loading. 18
We studied the off label use of the Femoral Neck System ® (DePuy Synthes, West Chester, PA) for prophylactic fixation. This device was chosen due to its minimally invasive profile, ease of use, and comparable performance for fracture fixation to a sliding hip screw construct. 19 Additionally, this device has a low risk profile and may improve upon some of the limitations of prior studies. 18 The aim of the current study is to use finite element analysis (FEA) to evaluate the ability of the implant to reduce stress and thereby strengthen the proximal femur. We hypothesize that models with the implant will display lower femoral neck stress compared to finite element models without the implant.
Methods
Device Description
The components of the Femoral Neck System (FNS) and their function are shown in Figure 1.
20
The FNS is indicated for fixation of femoral neck fractures (basilar, transcervical, and subcapital) in adults. All components are made of TAN (Ti-6Al-7Nb) and the device is available with bolt lengths from 75 mm to 130 mm with plates that have 1 or 2 holes for 5 mm locking screws. The anti-rotation screw integrates into the bolt and locks in place. For this study, we used the single hole plate, 90 mm anti-rotation screw, 90 mm bolt, and a 44 mm bicortical locking screw. After implantation, 8.7 mm of bone remained between the bolt and the subchondral bone of the proximal femur (yellow arrow, Figure 1). Components of FNS and their respective functions.
Model Generation
A computed tomography (CT) scan of a 68-year-old Caucasian female donor was obtained. The scan had an in-plane pixel size of .6895 × .6895 mm with a .9 mm slice thickness and voxel resolution of 512 × 512 × 230. The height and mass of donor were estimated using the femoral characteristics derived from the CT scan.21,22 The CT scan was imported into Simpleware ScanIP (Synopsys Inc, Mountain View, CA) module, an image processing software package. The left femur was isolated and coarsened to .7 mm pixels to facilitate computational analysis. Further processing was done to close holes, fill cavities, remove islands and smooth the resultant mask using a recursive gaussian filter.
The CT scan did not provide quality image data of the joint cartilage in the patient’s native acetabulum. Therefore, a cuboid acetabulum was generated using the 3D editing feature to preserve the biomechanics of the joint and allow for uniform loading of the femoral head during a lateral fall. Stereolithography files of the components of the FNS were imported as surfaces. The components were mated using the register datasets and group objects features and then positioned in the femur in accordance with surgical technique using the position and orientation features. The implant positions were confirmed by an orthopedic surgeon fellowship trained in orthopedic trauma (ML).
The image data was then cropped around the femur and implant using the shrink wrap feature and the resample tool was used to change image resolution to .25 mm in order to preserve finer details in the implant. The FNS surface object was converted to a smoothed, manifold object mask. The shape fitting and 3D editing features were used to remove bone from areas of the femur that would be removed during surgery. The resultant femur and implant masks were filtered to remove islands and fill gaps between the 2 masks.
Two models were created in ScanIP – a control model with only the femur and the acetabulum and a model that also included the implant (Figure 2). Mesh convergence analyses showed that the peak stress converged from 67 MPa to 62 MPa for mesh sizes of −40 to −20. A mesh size of −30 was chosen as this was approximately in the middle of the range and would still capture fine details in the models without prolonging runtime. Although quadratic elements are known to be more accurate, linear tetrahedra elements were used for this study because their shape is more suitable to capture the small features of the device. Additionally, initial analyses comparing the 2 element types showed minimal variation in the results. Fully bonded contact pairs were established between the femur and acetabulum as well as the femur and FNS. Material properties were input in SI (mm) units for the acetabulum and FNS as described in Table 1. Material properties of the femur at 3 levels of bone quality – healthy, osteopenic and osteoporotic – were derived from literature23-25 and models were exported to Abaqus (Dassault Systemes, Vélizy-Villacoublay, France) at each level of bone quality. The average apparent wet densities were .94 g/cm3, .87 g/cm3 and .77 g/cm3 for healthy, osteopenic, and osteoporotic bone, respectively. We defined cortical bone as any element with an apparent density greater than 1 g/cm3.
26
Scan IP rendering of the models used in this study. The left image depicts the model that includes the FNS implant. The right image depicts the control model with the femur and acetabulum only. Material Properties Assigned to the Acetabulum and FNS Implant in Scan IP.

Lateral Fall Simulation in Abaqus
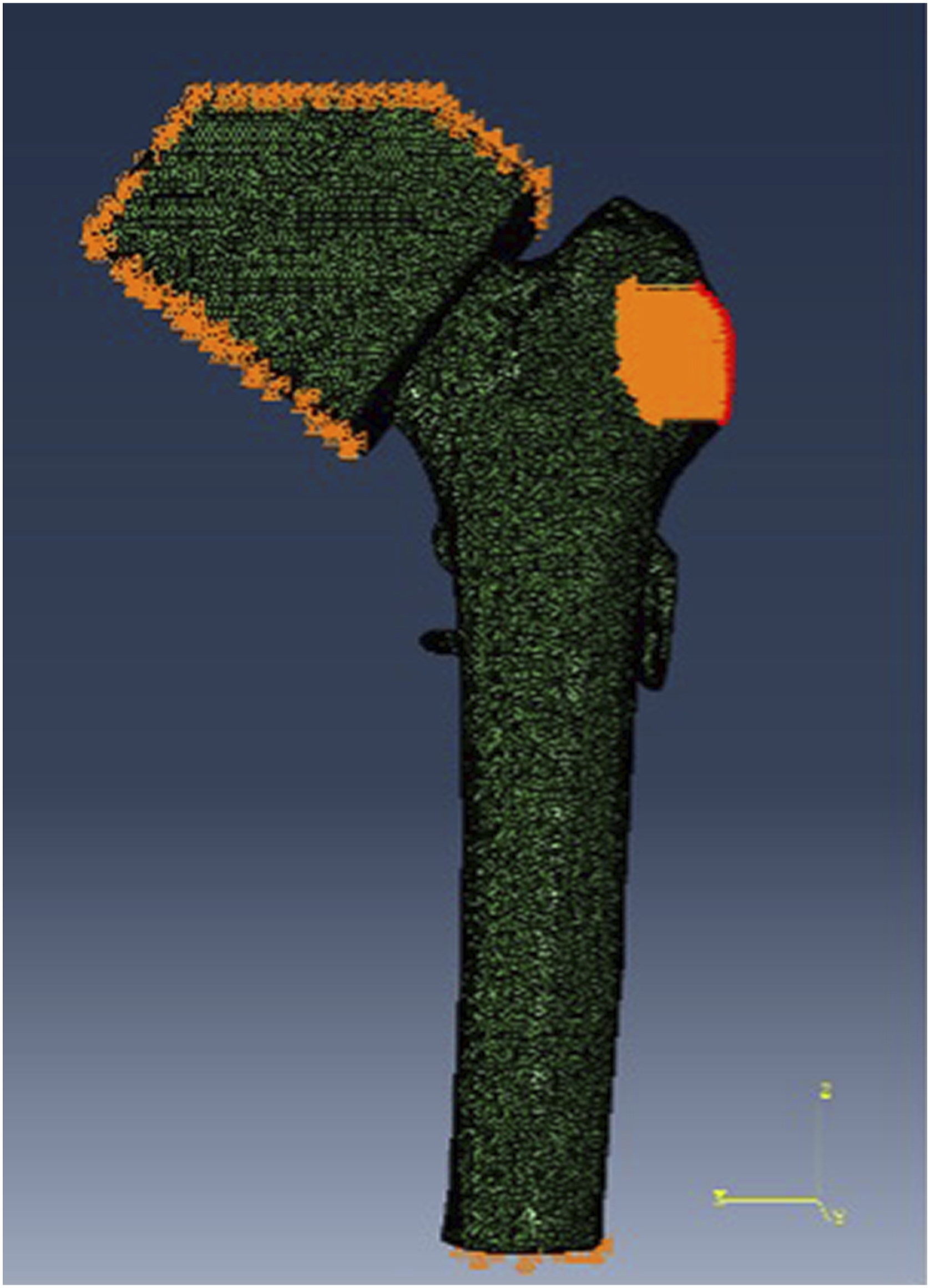
Six models were imported into Abaqus and a lateral fall simulation was prepared. As shown in Figure 3, boundary conditions with zero degrees of freedom were imposed on the distal surface of the femur as well as sides of the acetabulum. A force was loaded onto the lateral surface of the greater trochanter of the femur (Figure 3). Using the patient’s weight (w) in Newtons and height (h) in centimeters, the equation below from Yoshikawa et al
27
was used to estimate the magnitude of the force experienced in a lateral fall. Abaqus simulation showing a lateral fall with a force placed on the greater trochanter and boundary conditions placed on a representative acetabulum and the distal end of the femur.
The field output requests were set to evaluate both element-wise volume (EVOL) and von Mises stresses (S, Mises).
Fracture Risk and Volumetric Failure Analyses
The results generated by Abaqus for element-wise volume and von Mises stress were exported and analyzed using custom MATLAB codes. Failure was defined as any element that experienced stress greater than its ultimate tensile strength (UTS), which was calculated for each element using the ash density (⍴, g/cm3) obtained from a previously established correlation with the apparent wet density.28,29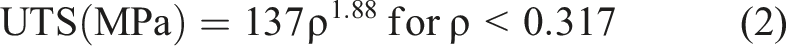

For each element, the ratio of von Mises stress to its respective UTS was calculated. If this value was greater than 1, the element failed. Then, the fracture risk (FRP) for each model was calculated as the ratio of failed elements to total elements in the model.
30

Similarly, the volumetric failure (VFP) was defined as the ratio of volume of failed elements to the total volume of all elements in the model.
Location of Failed Elements
For each of the 7 material groups of bone elements, a display group was created in the results module of Abaqus that showed only the elements that surpassed their UTS. The failing elements were highlighted in each material group, which allowed for visualization of the general location of failed elements within the femur. Additionally, this also identified elements that failed in trabecular bone and cortical bone separately.
Statistical Analyses
Data analyses were performed using R Project for Statistical Computing (version R-4.0.4). 31 At each level of bone quality (healthy, osteopenic, and osteoporotic), the number of elements that failed was compared to the number of elements that did not fail for the whole model using the chi-squared test. Similarly, the number of elements of cortical bone that failed was compared to the number of elements of cortical bone that did not fail using Fisher’s Exact Test. Statistical significance was defined as P < .05. Data are represented as the Chi-Squared value, P-value (X 2 , P-value) or the Fisher’s Exact P-value.
Results
Fracture Risk and Volumetric Failure Analyses
Volume That is Spared from Failure Due to the Presence of the FNS in the Model.

Results of Fracture Risk and Volumetric Analyses. VFP was Explored for the Overall Model as Well as in Trabecular and Cortical Bone Separately in Order to Have a Better Understanding of the Spatial Impact of the Failed Elements on these Bone Compartments.


Visualization of failed elements. The blue line represents the UTS; elements in red that surpass this line have failed. The 7 groups on the figures represent the 7 unique material groups generated by ScanIP with different BMD. For healthy and osteopenic bone quality, the 3 rightmost groups on the graph represent cortical bone. For osteoporotic bone, the 2 rightmost groups on the graph represent cortical bone.
Chi Squared Comparisons Showing the of Element Failure on Presence of the Device in the Model.

Location and Volume of Failed Cortical Elements
Prior studies have reported fractures following prophylactic fixation.
18
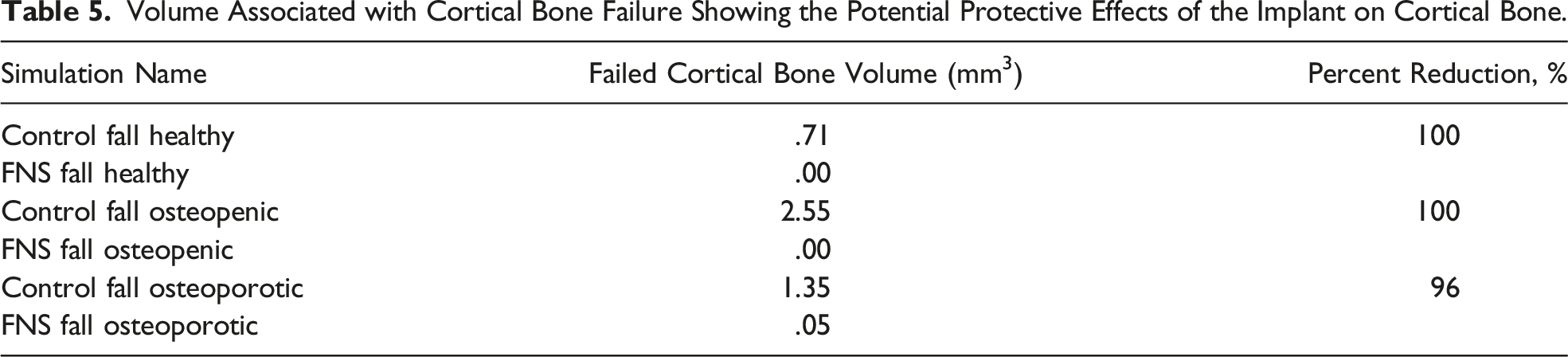
Therefore, we evaluated the location of cortical bone failure within the femur. Control models demonstrated cortical bone failure in the femoral neck (Figures 5A-5C), while cortical bone failure in models with the implant occurred in the femoral diaphysis, around the distal locking screw of the plate (Figure 5D). The implant reduced the volume (Table 5) of cortical bone failure by 100% in healthy and osteopenic bone and by 96% in osteoporotic bone. In order to determine if there was a relationship between cortical bone element failure and presence of the device, a Fisher’s Exact Test was performed for healthy (P = .49), osteopenic (P = .055) and osteoporotic (P = 1) bone in place of Chi-squared because the frequency of cortical bone failure was <5 elements in all models. Representative images from models with failing elements in cortical bone. (A) Control Fall Healthy. (B) Control Fall Osteopenic. (C) Control Fall Osteoporotic. (D) FNS Fall Osteoporotic. Volume Associated with Cortical Bone Failure Showing the Potential Protective Effects of the Implant on Cortical Bone.
Discussion
Individuals who suffer a primary hip fracture are at risk of fracturing the contralateral hip. Current pharmacologic treatment options may lower this risk but may not be suitable or efficacious for all patients. The goal of this study was to use FEA to evaluate the ability of the FNS to reduce peak stress and fracture risk in the proximal femur in the event of a fall.
Peak stress, fracture risk and volume of failing elements were all reduced in models with the implant. These results demonstrate that the implant may strengthen the proximal femur and may improve the bone’s resistance to fracture at all levels of bone quality. Additionally, Chi-Squared analyses of fracture risk were statistically significant suggesting that the extent of trabecular bone failure was dependent on presence of the device. Our results are consistent with other studies that have investigated the use of similar metal implants for this purpose.3,18,32
Most bone failure occurred in trabecular bone. Figure 4 does show elements of cortical bone failure in models A, C, E and F, however, Table 3 shows that the volume of these failing elements represents a very small amount of cortical bone. In healthy and osteopenic bone, Figure 4 shows some cortical bone failure in control models and lack of cortical bone failure in models with the implant suggesting the implant may spare cortical bone from failure. However, cortical bone failure results were not significant. The P-values from Fisher’s Exact Tests were >.05, suggesting that there was no relationship between cortical bone failure and presence of the device. This may be due to the small observation frequency. Therefore, further testing with a larger sample size is warranted to further evaluate this relationship.
A criticism of using metal implants for prophylactic fixation is that they can act as stress risers and may not prevent fracture, but merely shift the location of a fracture from the femoral neck to the femoral diaphysis. 18 In this study, we found that although implantation of the FNS lowered peak stress, it also introduced more variation in peak stress that was inversely related to bone quality. This may suggest that the device could be acting as a stress riser. Additionally, models without the implant displayed cortical bone failure in the femoral neck, while models with the implant displayed either no cortical bone failure or failure in the femoral diaphysis. This result suggests that implantation of the device may shift the location of cortical bone failure from the femoral neck to the proximal femoral diaphysis. This is of particular concern with the FNS as cortical bone failure in the subtrochanteric region is more challenging to reduce and stabilize. 33 The volume of element failure in the femoral diaphysis was less than 0.1 mm3. Thus, although the FNS may shift the location of cortical bone failure to the femoral diaphysis, the volume of affected bone may be insufficient to cause a fracture at the site of the locking screws. The FNS is also not designed to dissipate forces and potential alterations in the design of the FNS might reflect this as an important factor. These could include nonlocking screw options and longer plates.
This study should be considered in light of several limitations. Only 1 bone model was used and thus our results are unimpacted by natural variations in human anatomy. As such, conclusions drawn from this work may not be generalized to the population at large. Our findings are best understood as a foundation to support the need for further study of orthopedic implants as prophylactic treatments. Additionally, the donor of our bone model was a 68-year-old woman, and we did not have data from a DEXA scan. Thus, the baseline bone quality of our model is not known. Hip fractures also tend to occur in those above 68 years of age so future evaluations should include older specimens so that the results may be more applicable to the population of interest. This analysis also simplifies proximal femur loading and the degrees of freedom in femoral movement by studying the femur in isolation of the pelvis. Our models are not able to localize failure and do not account for the effects of micro-architecture on prediction of bone failure. Our definition of failure as any element that experiences stress greater than its ultimate strength may be an oversimplified estimate. Finally, similar studies have reported conflicting results between their FEA and cadaveric biomechanical studies.18,34 Further work is needed to validate our models with biomechanical data.
The consideration of prophylactic stabilization of the proximal femur cannot be taken lightly. If performed at the same surgical setting the anesthetic burden to the patient must be considered. In addition, the operating room costs and time will nearly be doubled. Despite this predictable benefit to the prevention of proximal femur fractures, the associated risk might not be acceptable for all patients.
Conclusions
In conclusion, this study demonstrates the potential of the DePuy Synthes FNS to strengthen the proximal femur and serve as prophylactic fixation to prevent a contralateral hip fracture. The results suggest the device may improve the ability of the femur to resist fracture by lowering the peak stress and fracture risk in the proximal femur during a lateral fall across all levels of bone quality. The results also suggest that implantation of the FNS may spare cortical bone from failure during a lateral fall and may shift the burden of cortical bone failure from the femoral neck to the femoral diaphysis. Further work in biomechanical studies are needed to validate these models before consideration for clinical use.
Footnotes
Acknowledgments
This study acknowledges with great thanks Dr R. Dana Carpenter, Associate Professor at University of Colorado Denver, for providing the calibrated femur model and DePuy Synthes for providing the computer aided design files to model the Femoral Neck System implant.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Julia N. LaMonica (N), Brian Rhee (N), Michael Leslie (N), Steven M. Tommasini (N), Daniel Wiznia (3B-Intellijoint Surgical).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the CTSA Grant UL1 TR001863 from the National Center for Advancing Translational Science (NCATS), a component of the National Institutes of Health (NIH), and NIH Roadmap for Medical Research. Its contents are solely the responsibility of the authors and do not necessarily represent the official view of NIH.
Clinical Significance
Evaluation of a metal implant for contralateral prophylactic femoral neck fracture prevention in the setting of primary hip fracture.
