Abstract
Introduction
Pain management in patients with hip fractures can be challenging. Poor pain control is associated with reduced mobility and increased morbidity. Inadequate analgesia in patients with dementia is a concern. After using several different alternatives, transdermal buprenorphine was chosen as a standardised approach for analgesia in patients with fragility fracture in our hospital. There is limited evidence on the use of buprenorphine in this population. Our aim was to investigate the safety and effectiveness of transdermal buprenorphine in patients with hip fractures.
Methods
A review of consecutive patients presenting with a hip fracture from June 2018 to December 2018 was conducted using medical records. Our primary outcome was the incidence of complications as a consequence of transdermal buprenorphine. Our secondary outcome was adequate analgesia measured by reviewing the requirement for analgesia during the first week following the patient’s admission. Analgesia demands were considered adequate if patients required less than 20 mg of oral morphine in total during the first week following injury.
Results
In total, 148 patients presented with a hip fracture during the study period. 128 patients had documented evidence of buprenorphine patch application. Complete data was available for the primary outcome of complications in all cases. Data was available for the secondary outcome in 124 patients. Buprenorphine was discontinued in 24 patients (19%), most commonly due to due to concerns about contribution to hypoactive delirium (9%), and when strong analgesia was no longer required (4%). There were no severe complications. Adequate analgesia was achieved using this regime in 68% patients. 38 patients (32%) required more than 20 mg of oral morphine sulphate solution in the first week post-admission.
Conclusion
This series suggests that transdermal buprenorphine is safe and effective in the management of pain following a neck of femur fragility fracture.
Introduction
Hip fracture is a common injury with more than 67,000 occurring in the United Kingdom each year. 1 Patients presenting with fragility fractures commonly have cognitive impairment, multi-comorbidity and polypharmacy. 2 Pain management in this population can be particularly challenging. Poor pain control is associated with reduced mobility and an increased risk of morbidity, including pneumonia, delirium, pressure ulcers and venous thromboembolism. 3
Current guidelines from the National Institute for Health and Care Excellence (NICE) advocate regular paracetamol, PRN opioids, and consideration of nerve blocks. 4 Non-steroidal anti-inflammatory drugs are not recommended by NICE for this patient group. 4 Updated guidelines from the Association of Anaesthetists of Great Britain and Ireland also support the use of peripheral nerve blocks. 5 However, these require specific expertise and are usually administered as a single injection lasting around 12 hours. As such, they are useful in the pre-operative period and as part of the anaesthetic but have limited use post-operatively. Continuous infusion catheters have limited evidence and are not yet recommended. 5
Systemic opioids are recommended by NICE, but these may be associated with multiple undesirable side effects including nausea and vomiting, constipation, delirium and respiratory depression. 4 These occur more frequently in the older population and can compound pre-existing cognitive impairment, making assessment of pain more difficult. Neither the NICE nor the Association of Anaesthetists of Great Britain and Ireland guidelines mention the use of transdermal buprenorphine for patients with a fractured neck of femur but this approach is increasingly being used.4,5
Buprenorphine is a synthetic opioid which acts as a partial agonist at mu opioid receptors, and an antagonist at kappa and delta receptors. 6 It can be delivered via several routes; intravenous, sublingual or transdermal, and its high lipid solubility means it has good skin penetration. 7 Buprenorphine is 75–100 times more potent than morphine and is well tolerated at high doses.8,9 The partial agonist activity gives buprenorphine a ceiling effect for respiratory depression but not for analgesia, reducing the risk of this potentially fatal side effect when compared with other opioid agonists such as fentanyl. 10
Several transdermal formulations exist; our preference is for Butrans® which is licenced for 7 day application. 7 It has an onset of action of 18–24 hours and a time to peak plasma concentration of 3 days. 7 The use of buprenorphine in chronic pain and malignancy is well established but there is limited evidence on its use in older patients following a neck of femur fracture. Our aim was to investigate the safety and effectiveness of transdermal buprenorphine for the management of post-operative pain in an older population of patients with neck of femur fracture.
Method
Data Collection.

All patients in our institution are offered a fascio-iliaca compartment nerve block in the emergency department unless contraindicated. Our standardised analgesia protocol includes regular paracetamol 500 mg–1 g (depending on estimated weight) every 6 hours and a buprenorphine 5mcg/hr patch applied pre-operatively. Oramorph 2.5 mg as required up to every 4 hours is also prescribed. If patients are already taking regular Opioids/high dose morphine then our standardised approach is not applied and an individualised approach is employed.
Breakthrough analgesia requirements were recorded for the first week post-admission. Our primary outcome was the incidence of complications as a consequence of buprenorphine patches in this patient population. The secondary outcome was the requirement for post-operative breakthrough analgesia in the first week post-admission.
An ordered logistic regression model was used to determine whether the use of regular pre-admission analgesia was associated with the amount of breakthrough oramorph required after adjusting for the use of a femoral nerve block (y/n) or spinal anaesthetic (y/n), patient age, and the implantation of a dynamic hip screw (DHS) (y/n). A DHS has been shown to be associated with increased analgesia requirements compared to other procedures for hip fracture management. 11
Analyses were performed using STATA 16 statistical software. P < .05 was considered significant
Results
148 patients were managed for a hip fracture during the 6-month study period. Four patients were excluded from the analysis; 2 patients received conservative management, 1 patient died prior to surgery and 1 patient was transferred to another hospital for surgery. Four generally fit and active patients who received a total hip replacement were not given topical buprenorphine. These patients followed the Enhanced Recovery Protocol for elective Total Hip Replacement which uses a standardised anaesthetic and oral oxycodone. Written drug chart documentation was unavailable through our paper medical records system in 12 patients.
In total, 128 patients were documented to have received a buprenorphine patch during the study period. Documentation was complete in all patients for the primary outcome of the incidence of complications due to the patch. The mean patient age was 86.9 years (range 60–99, SD 9.2). The majority of patients were American Society of Anaesthesiologists (ASA) 3 (59%). The procedures patients underwent included hemiarthroplasty, 64; femoral nail, 36; dynamic hip screw, 17; and total hip replacement, 11. The distribution of ASA is demonstrated in Figure 1. Patient American Society of Anaesthesiologists score.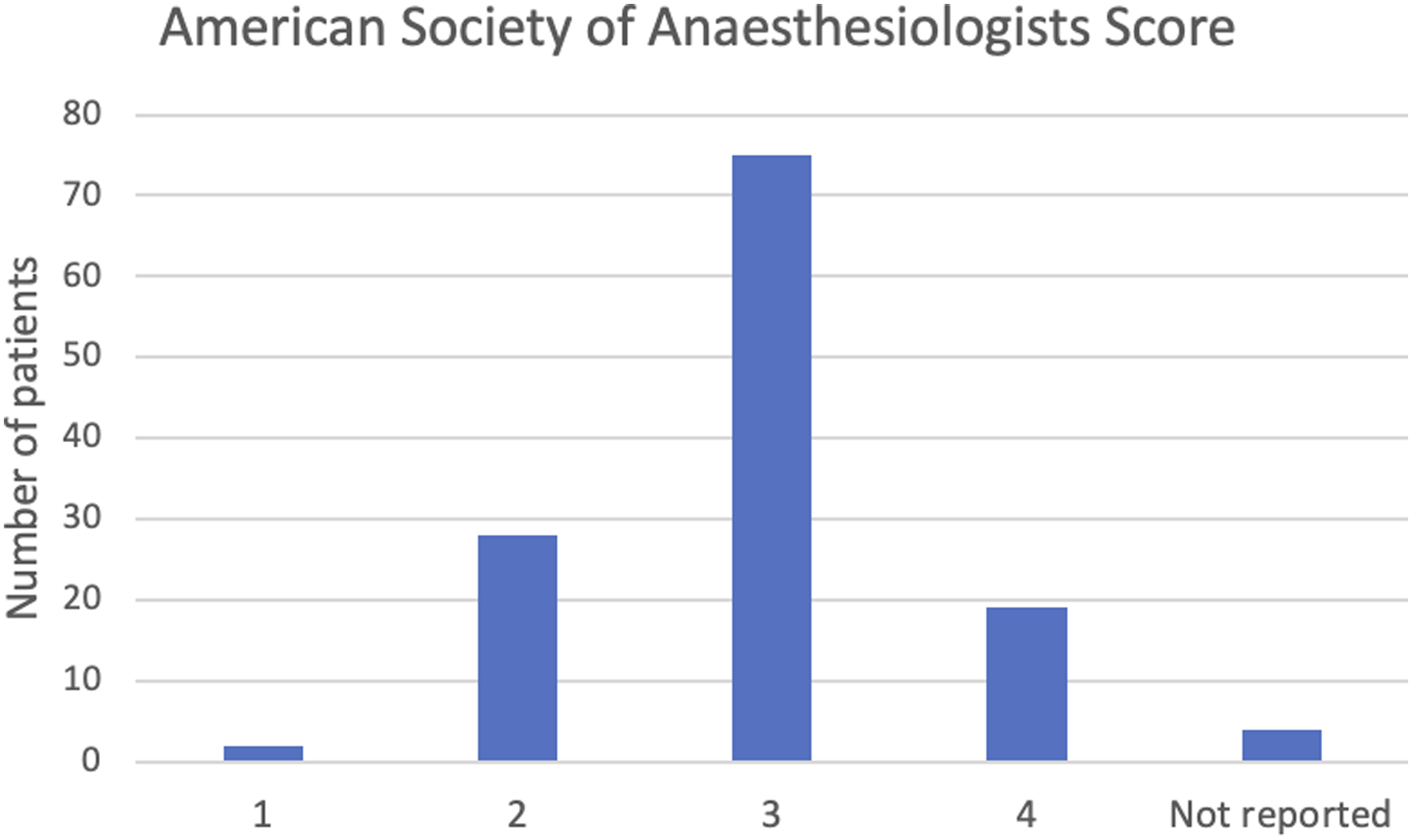
Data on post-admission analgesia requirements were available in 124 patients. 34 (27.4%) of these patients were taking regular analgesia prior to admission, 17 (13.7%) underwent a dynamic hip screw, 54 (43.6%) had a femoral nerve block and 69 (55.7%) received a spinal anaesthetic.
The total amount of analgesia required by each patient in the week following admission is shown in Figure 2. Breakthrough oramorph analgesia requirements 1 week post-admission.
The buprenorphine patch was removed in 24 patients (19%). In 11 of these patients, there was concern that it was contributing to hypoactive delirium. Other reasons included patients not requiring strong analgesia (5 patients) or that pain was not controlled and significant doses of breakthrough oramorph were required likely rendering transdermal buprenorphine ineffective (1 patient). Other side effects were rare: nausea (1 patient), and possible contribution to urinary retention (1 patient). Further reasons included patient request (1 patient), one patient was commenced on end of life medication, and the patch was removed in one patient who suffered bilateral pulmonary emboli. Other unspecified causes accounted for the 2 remaining cases. Our logistic regression model demonstrated no significant association between the amount of breakthrough analgesia required and whether the patient was taking regular analgesia on admission (P = .20), adjusted for the following variables; use of a femoral nerve block, spinal anaesthetic, a DHS implant, or patient age. The full results are available in the Online Appendix.
Data from the National Hip Fracture Database demonstrated outcomes including post-operative delirium and mortality for our trust being well below national average and day 1 mobilisation being better than average. 1
Discussion
Good pain management is a fundamental component of care in patients with a neck of femur fracture. Adequate analgesia is necessary to facilitate early mobilisation which is associated with a reduction in mortality. 12 Careful dosing is particularly important in this population and pain control can be challenging.
Contrary to oral or intermittent parenteral analgesia, transdermal preparations provide steady and continuous drug delivery, resulting in near constant plasma concentrations. 7 This may provide an advantage in those with cognitive impairment or post-operative delirium who are unable to ask for analgesia possibly leading to under-treatment of pain. A previous study in adults over 65 years with a hip fracture compared routine intravenous paracetamol combined with further analgesia as required, to a second group where all patients received analgesia as required. 13 Patients given intravenous paracetamol routinely had a shorter hospital stay, greater engagement with rehabilitation and a higher rate of discharge home. 13 This highlights the potential value of routine, pre-emptive analgesia. Further advantages of the transdermal route include ease of use for the patient or carer, greater adherence to treatment regimens and usability in situations where older patients are unable to tolerate or unwilling to swallow oral medications. 6
Nursing time is valuable, hospital wards are busy environments and staff are under constant time pressure. 14 Nurses spend approximately 19% of their time on medication-related tasks, during which time they are frequently interrupted. 14 Dispensing of multiple additional medications increases the number of important tasks a nurse must perform, and the risk of error. Using a buprenorphine patch for 7 days may reduce the total volume of medication dispensed and therefore minimise the risk of drug errors occurring elsewhere.
Buprenorphine can be given safely to patients with renal impairment; it is metabolised and excreted primarily via the liver. 6 This is beneficial in this patient group as the Association of Anaesthetists of Great Britain and Ireland estimate that 40% of hip fracture patients have renal dysfunction and acute kidney injury may occur in 24% of patients.5,15 The low incidence of adverse events in our study has been shown in other studies in different populations.16-18 However as an opioid analgesia, all patients should be carefully monitored for the risk of overdose. In our practice the patch is removed on discharge (or at 2 weeks) to prevent them inadvertently remaining on the buprenorphine for extended periods unless there is a clear need. Topical buprenorphine relies on adequate skin adhesion and accelerated opioid release may occur if the patch is heated externally; this is also true of transdermal fentanyl patches.19,20 Transdermal patches are removed if a patient becomes unwell with fever as this can result in increased absorption.
A small randomised study compared transdermal buprenorphine to oral tramadol in post-operative pain following hip fracture surgery. 18 Pain scores both were significantly lower in the patients who received buprenorphine for 7 days, starting 24 hours post-procedure. All the patients receiving regular oral tramadol needed rescue analgesia, compared to 68% of patients in the buprenorphine group. No patients in either group developed significant sedation or respiratory depression. Of note, the mean age in this study was lower than the average age of a hip fracture patient in the United Kingdom, and only 2 of the 50 patients studied underwent dynamic hip screw surgery.
Another randomised trial of 188 patients reported lower pain scores with buprenorphine patches compared to paracetamol and tramadol following elective knee replacements. 16 The patch was applied at the end of surgery in the intervention study group. All patients received a spinal anaesthetic with a femoral nerve catheter inserted at the end of surgery and a continuous infusion of ropivacaine administered for the first 48 hours, after which point the patients were mobilised. Topical buprenorphine resulted in higher patient satisfaction scores and a lower incidence of adverse events. One patient developed respiratory depression requiring discontinuation of the buprenorphine. Similar results have been shown in patients following lumbar discectomy. 17 More recently, Li and colleagues published a protocol for a randomised trial of buprenorphine compared to paracetamol and tramadol following total hip arthroplasty. 21
To our knowledge this study includes the largest series of patients who received transdermal buprenorphine following a fracture of the proximal femur. We included all patients with a range of ages and levels of frailty who underwent several different procedures using different combinations of anaesthetic and peripheral nerve blocks.
This study is retrospective and there is a risk of incomplete or inaccurate documentation. However, a complete dataset was available for over 90% of patients. We have included patients from a single centre; the age and ASA scores in our patient population are broadly similar to the majority of patients with a neck of femur fracture. We have not included information on analgesia requirements following discharge from hospital when the patch is removed.
Conclusion
This large series suggests that topical buprenorphine can be used safely in patients with a neck of femur fragility fracture. Patch removal was required in less than 20% of patients, and no serious complications occurred. Transdermal buprenorphine is a safe, reliable and cost-effective method of managing pain in an older population following a neck of femur fracture.
Supplemental Material
sj-pdf-1-gos-10.1177_21514593211070260 – Supplemental Material for Transdermal Buprenorphine for Pain Management Following a Neck of Femur Fracture
Supplemental Material, sj-pdf-1-gos-10.1177_21514593211070260 for Transdermal Buprenorphine for Pain Management Following a Neck of Femur Fracture by Andrew Davies, Jane Murray, Pardis Zalmay, Ewan Ross, Shumaila Dar, Helen Wilson in Geriatric Orthopaedic Surgery and Rehabilitation
Footnotes
Author Contributions
A Davies – Study design, analysis of results, writing manuscript.
J Murray – Analysis of results, writing manuscript.
P Zalmay – Data collection, writing manuscript.
E Ross – Data collection, editing of manuscript.
S Dar – Data collection, study design, editing of manuscript.
H Wilson – Study design, writing manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The work was approved by the Institution’s audit and research department; audit reference number 774.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
