Abstract
Objective:
Assess whether maternal rural residence was an independent risk factor (RF) for spontaneous preterm birth (sPTB) at a Midwest tertiary care center.
Methods:
After excluding deliveries complicated by indications for induced preterm birth, there were 16 974 deliveries among 13 339 women between May 1, 2009, and May 31, 2023. Standard descriptive statistics and Generalized Linear Mixed Models were used to identify RFs for sPTB.
Results:
Of the 16 974 deliveries, 89.1% were full term, 28.8% were of rural mothers, and the mean maternal age was 29.6 years (y). 15.5% of rural deliveries were sPTBs versus only 9.1% of urban deliveries (P<.001). Significant independent RFs for sPTB included the extremes of maternal age (aOR, 2.14 for age 14 to <20 y and aOR, 3.37 for age ≥40 y) compared to age 20-<30 y, rural vs. urban residence (aOR, 1.56), maternal smoking (aOR, 2.47), transfer from an outside hospital (aOR, 671.3), and female sex of the baby (aOR, 0.79). With the transferred deliveries excluded these factors remained significant with similar aORs and Asian race was an additional significant protective factor (aOR, 0.57). Body mass index, race, ethnicity, type of medical insurance, and primigravidity were not significant RFs.
Conclusion:
Rural residence, extremes of maternal age, maternal smoking, female newborn sex, and transfer from an outside hospital were significant and independent RFs for sPTB.
Introduction
Preterm deliveries increase the risk of adverse developmental outcomes and death for newborns,1,2 raise healthcare costs, 3 and disproportionally affect people of minority race. 4 An early delivery, or a delivery at <37 weeks gestation, is termed preterm birth (PTB). In 2020, there were 13.4 million PTBs globally, or 9.9% of total births. 2 The U.S. PTB rate is close to the global average at 10.4%, despite the U.S. being a developed nation, and spending 17.6% of the gross domestic product on healthcare.4,5
There are many known causes of PTB, including cervical insufficiency, infection, and maternal or fetal conditions necessitating induction of labor, but the majority of PTB occur without known cause. 6 PTB without known etiology comprise a subset of PTBs defined as spontaneous preterm birth (sPTB), or PTB without induction. 6 sPTB occurs in 40% to 50% of all PTB cases. 6 There are many proposed pathways that may contribute to the development of sPTB without known causation, such as genetic implications, 7 immunologic conditions, 8 as well as exposure to environmental contaminants, 9 but the possible causes are multifactorial, numerous, and difficult to implement into clinical practice. Additionally, many sociodemographic risk factors (RFs) for PTB have been identified, some including low socioeconomic status, 6 high or low maternal age, stress, tobacco use, low body mass index, minority race, 10 and rural residence. 11 Despite this, the differences between sociodemographic RFs for sPTB specifically versus PTB, which includes induced PTB (iPTB), is unclear. 12 Additionally, many of the recent studies identifying the relevant RFs for PTB use national level data,13,14 which may miss the region specific conclusions that can be drawn, as it is known that the different pertinent RFs for PTB vary between regions of the U.S.15,16
At Iowa Health Care (IHC), a Midwest tertiary care center, a relevant potential RF for sPTB was rural residence, as IHC serves a significant rural population due to recent labor and delivery (L&D) and obstetric care unit closures. Rural locations are more vulnerable to adverse outcomes with childbirth, 11 and this was of importance to Iowa’s population as one third of Iowans were of rural residence. 17 The purpose of this study was to identify RFs for sPTB at IHC, a Midwestern, academic health care center, after excluding deliveries complicated by indications for iPTB, hypothesizing that rural residence would be independently associated with higher risk of sPTB.
Methods
The Iowa Intergenerational Health Knowledgebase (IHK), IRB # 201902830, 202101369, was used to obtain birth data. The IHK includes >67 000 pregnancies in the IHC electronic medical record (EMR) from May 2009 to May 2023. Only pregnancies delivered at IHC were included in the dataset (N = 29 620). Deliveries complicated by diagnoses which were indications for iPTB or induced abortion were excluded, including multiple gestations (n = 2399), 18 diabetes (gestational diabetes, Type 1 diabetes and Type 2 diabetes) (n = 4598), 18 chronic hypertension (n = 2678), 18 preeclampsia or eclampsia (n = 3351), 18 cardiomyopathy (n = 1136), 19 history of previous PTB (n = 3174), gestational age ≤20 weeks or ≥43 weeks (n = 339), if there was insufficient data available for the pregnancy (n = 95), or if the delivery was classified as a spontaneous or induced abortion (n = 375). Figure 1 shows the study flow diagram. Some deliveries qualified for more than 1 exclusion criteria. The final dataset included 16 974 deliveries among 13 339 mothers. Because women with indications for iPTB were excluded, the PTB included in the final dataset were assumed to be sPTBs.

Study flow diagram.
To analyze the relationship between rural residence and sPTB, Rural-Urban Commuting Area Codes (RUCA codes) were used to categorize zip codes of residence at the time of delivery into either rural or urban areas. 20 These codes were formulated using U.S. census data, which considered the population size and the degree of commuting the population engaged in. The codes ranged from 1 to 10, from most urban to most rural. RUCA codes 1 to 3 were considered urban, and RUCA codes 4 to 10 were considered rural, according to Health Resources and Services Administration definitions. 21
Many other RFs for PTB were included as independent variables in the multivariable model to account for potential confounding variables. Maternal age was included as a categorical variable to identify significant differences between age groups (14-<20 years (y), 20-<30 y, 30-<40 y, ≥40 y). 6 Body mass index (BMI) was also included as a categorical variable of ≥30 kg/m2 or <30 kg/m2 following U.S. Centers for Disease Control and Prevention definitions of obesity.6,22 Self-identified race was included as 4 categories, including White, Black or African American, Asian, and other/unknown race. 10 The other/unknown race group included races or other EMR inputs that had decreased prevalence in the sample, including American Indian or Alaska Native, multiple race, Hispanic (which was included in the EMR as a race option, although Hispanic was also listed as an ethnicity), unknown, other, refuse to answer, and no information. Ethnicity as an independent variable was defined as Hispanic or non-Hispanic. 10 Type of medical insurance was included as a proxy for socioeconomic status, 6 and the 3 categories included Medicaid, private, and other insurance. Sex of the newborn was included as a variable as male newborns are at increased risk for PTB, 23 with the categories of male and female in the model. Maternal tobacco smoking was a known RF and was included with 2 categories in the model, as current smoking and not currently smoking. 6 Primigravidity was included in the analysis as gravida 1 or gravida >1, as gravida 1 mothers are at increased risk for sPTB. 24 Lastly transfer to IHC from an outside hospital was included, as one of the indications for transfer to IHC is escalation of care for potential PTB.
Statistical Analysis
Standard descriptive statistics were used to characterize the study population. Generalized linear mixed models (SAS Proc GLIMMIX) were used to determine whether the dichotomous outcome (<37 weeks [sPTB] vs. ≥37 weeks [full term]) was associated with potential RFs. As deliveries were nested with the mother, maternal-specific random intercepts were included to account for the association of multiple deliveries within the same mother. The potential RFs included were maternal age, race, ethnicity, rural vs. urban residence, type of medical insurance, sex of the baby, maternal smoking, primigravidity, and delivery at IHC after transfer from an outside hospital. Such factors were first tested individually, then factors with a P value below 0.15 were included in a full multivariable model, and the backward stepwise method was used to remove variables one at a time until all variables in the model had a P value below 0.05. Interaction terms, rurality with age, rurality with smoking status, and rurality with transferred were examined and tested in the multivariable model to evaluate for confounding variables. None of the interaction terms were significant at P value below 0.05 level, so the interaction terms were not controlled for in the final multivariable model. Collinearity diagnostics were conducted using the Variance Inflation Factor (VIF) for all predictor variables included in the model. The VIF values were all below 2, indicating that multicollinearity is unlikely to be a significant issue. Patients with missing values were categorized as unknown or other or missing categories to retain all observations, ensuring no loss of potentially valuable data. In the models, we present both unadjusted and adjusted odds ratios (aORs) for each RF with a 95% confidence interval (CI). P values <.05 were considered statistically significant. All statistical analyses were performed using SAS, version 9.4 (SAS Institute).
Results
There were 16 974 spontaneous deliveries among 13 339 mothers from May 2009 to May 2023, which were not complicated by the excluded indications for iPTB or induced abortion.18,19 Of the 13 339 mothers in the study, the racial distribution was 71.8% White, 9.3% Black or African American, 6.4% Asian, 0.3% American Indian or Alaska Native, 0.2% Native Hawaiian or other Pacific Islander, 2.2% multiple race, and 1.7% other race or unknown (Table 1). 8.6% were Hispanic (Table 1). Of the 16 974 deliveries, 89.1% of deliveries were full term, 28.8% lived in rural areas, 26.7% had Medicaid insurance, 48.5% of the babies were female, 6.2% of the mothers were current smokers, 31.4% of the mothers were obese (BMI ≥ 30), 30.4% of the mothers were gravida 1, 3.9% were transferred to IHC from an outside hospital, and the mean maternal age was 29.6 years (y) (standard deviation, 5.4) (Table 1).
Maternal (n = 13 339) and Delivery (n = 16 974) Characteristics by Rural Versus Urban Maternal Residence.

Abbreviation: BMI, body mass index.
Figure 2 identifies how many sPTB occurred for each RUCA code. Although RUCA code 3 had the highest percentage of sPTB at 27.3% and RUCA code 9 had the lowest percentage of sPTB at 7.4%, these 2 categories made up a very small percentage of the total population, and did not significantly influence the overall trends. The next highest percentages of sPTB were with the rural RUCA codes 6 (20.0%), 5 (19.9%), and 4 (19.3 %). Overall, 15.5% of rural deliveries were sPTBs, compared to only 9.1% in the urban group (P < .001).

Percentage of sPTBs by RUCA Code.
Table 2 shows the statistically significant, independent RFs for sPTB in the multivariable model among all deliveries (n = 16 974), as well as the univariable model odds ratios. Statistically independent RFs for sPTB included the extremes of maternal age, 14-<20 y (aOR, 2.14; 95% CI, 1.46-3.14) and ≥40 y (aOR, 3.37; 95% CI, 2.04-5.56) using a reference age 20-<30 y, rural vs. urban residence (aOR, 1.56; 95% CI, 1.28-1.90), maternal smoking (aOR, 2.47; 95% CI, 1.77-3.44), and transfer of the mother to IHC for delivery (aOR, 671.3; CI, 247.3->999.9). Female sex of the baby (aOR, 0.79; 95% CI, 0.67-0.94) was protective. Race, ethnicity, obesity, health insurance type, and primigravidity were not independently associated with sPTB.
Generalized Linear Mixed Model of Factors Associated with sPTB (<37 weeks vs Full Term) (n = 16 974).
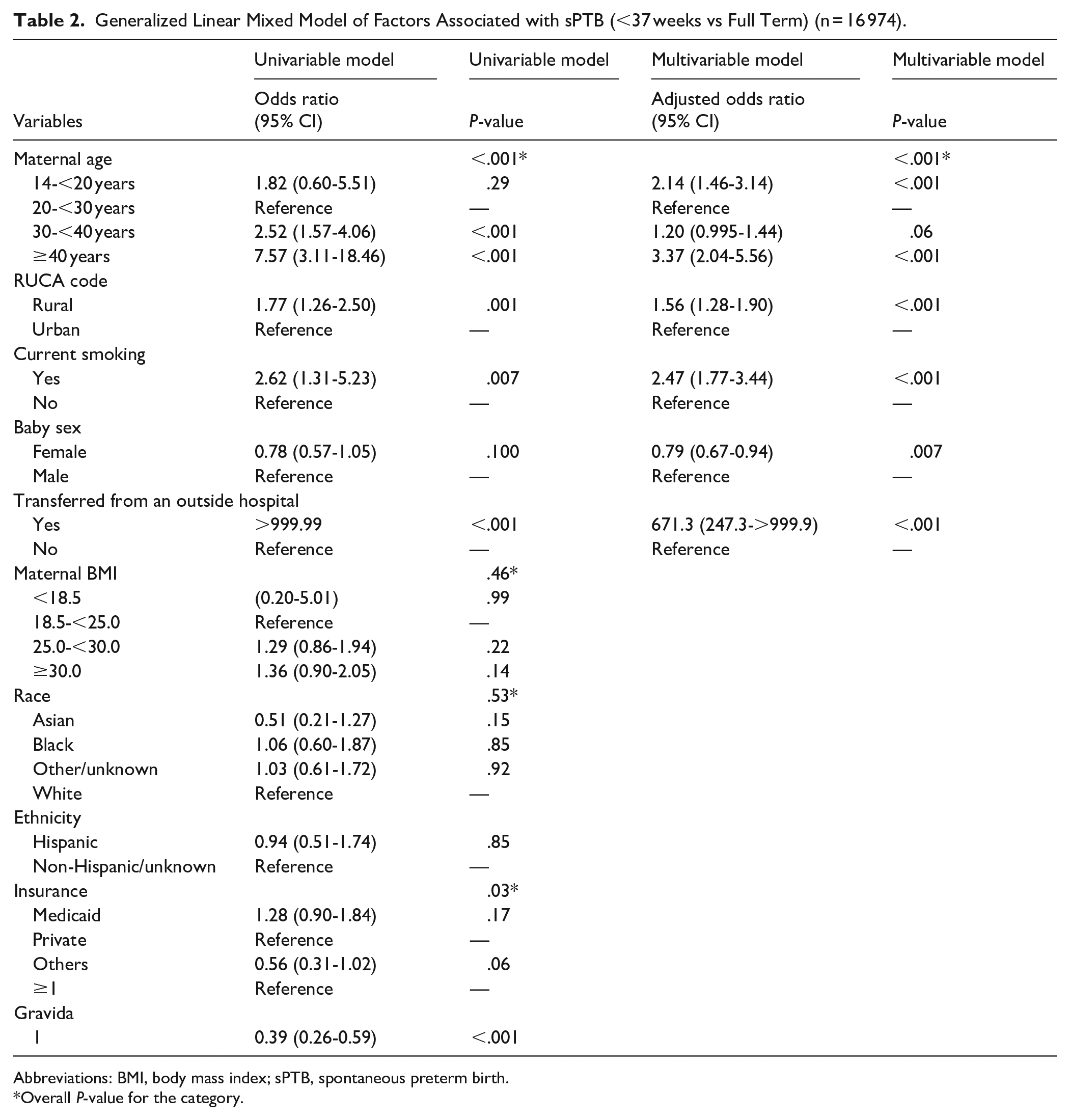
Abbreviations: BMI, body mass index; sPTB, spontaneous preterm birth.
Overall P-value for the category.
After excluding deliveries that were transferred to IHC (n = 668), there were 16 306 deliveries. An identical analysis was done, with the results displayed in Table 3. Independent RFs for sPTB were extremes of maternal age, 14-<20 y (aOR, 2.70; 95% CI, 1.72-4.24) and ≥40 y (aOR, 4.03; 95% CI, 2.27-7.15) using a reference age of 20-<30 y, rural residence vs. urban residence (aOR, 1.67; 95% CI, 1.31-2.09), and maternal smoking (aOR, 2.96; 95% CI, 2.00-4.38). Independent protective factors for sPTB were female sex of the baby (aOR 0.77; 95% CI, 0.64-0.93), and Asian maternal race using White race as a reference (aOR 0.57; 95% CI, 0.35-0.91).
Generalized Linear Mixed Model of Factors Associated With sPTB, With Deliveries That Were Transferred to IHC Excluded (<37 weeks vs Full Term) (n = 16 306).
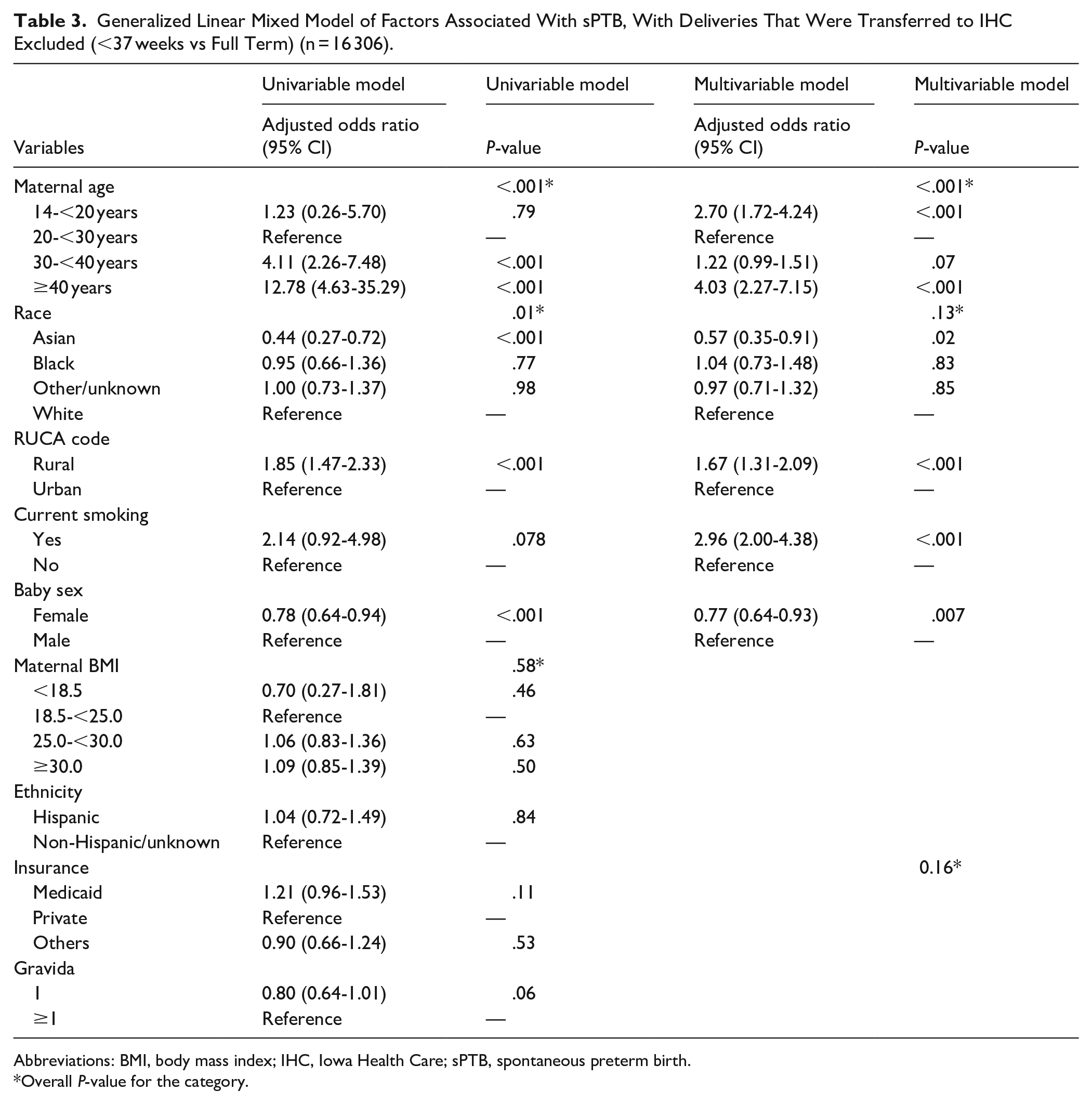
Abbreviations: BMI, body mass index; IHC, Iowa Health Care; sPTB, spontaneous preterm birth.
Overall P-value for the category.
Discussion
Rural Residence and sPTB
Rural residence was significantly and independently associated with increased risk of sPTB at IHC, whether transfers were included or excluded. When examined by individual RUCA codes, 3, 6, 5, and 4 were found to have the highest percentages of sPTB, at 27.3%, 20.0%, 19.9%, and 19.3%, respectively. These percentages of sPTB are difficult to interpret due to the small sample size in each of these RUCA codes. When women were dichotomized (rural vs. urban), sPTB was significantly increased among women from rural areas compared with women from urban areas. These results are consistent with other studies which found that rural communities had an increased the risk for PTB,13,25,26 and expand the current base of literature, as our study is the first to draw the conclusion that maternal rural residence is associated with increased risk of sPTB in comparison to maternal urban residence. Other studies have shown that rural residence is associated with PTB, but have not discriminated between sPTB and iPTB.13,25,26
This study is critically relevant to Iowans and potentially other Midwest residents living in rural areas with decreased access to prenatal and obstetrical care, as 43 out of 97 L&D units have closed across Iowa from 2000 to 2025, 27 leaving 33 out of 99 counties without L&D services. 28 As of 2025, 61% of 93 rural hospitals in Iowa do not provide L&D care. 29 This is associated with decreased quality of patient care, with a recent study finding that rural L&D unit closures are associated with decreased number of prenatal care visits in Iowa. 30 The greatly increased risk of sPTB among patients living in rural communities may be a result of this lack of access to prenatal care due closures of L&D units, as well as the stress that is associated with decreased access to prenatal care.31 -33 Furthermore, this limited healthcare access of rural communities will worsen with the Medicaid cuts that were recently passed by congress. 34
In addition, although we were not able to measure pesticide concentrations, maternal exposure to pesticides may be affecting our rural population as counties with increased pesticide use have an increased risk of PTB. 35 Increased incidence of pesticide drift, or accidental spray of pesticides on unintended areas, affect IHC’s more rural, adjacent counties at increased rates. 36 Increased exposure to other contaminants associated with increased risk for sPTB may also be relevant to rural population, such as polyfluoroalkyl substances 37 and nitrates. 9 The vast majority of the rural population in the U.S. uses groundwater sources for drinking water, 38 and groundwater is susceptible to increased levels of pesticides, 39 nitrates, 39 and polyfluoroalkyl substances. 40
Race and sPTB
We found no association with race and sPTB in this study. Current literature has found that mothers of minority race have increased incidence of PTB in the U.S. 10 More specifically to sPTB, a descriptive prospective study done in North Carolina, by Jesse et al, 41 among 260 women of low-income status and rural residence found that African American race was associated with increased risk of sPTB compared to White race, when controlling for other confounding variables. This study was able to exclude iPTB specifically and controlled for preeclampsia and chronic hypertension in their analysis.
Our contrasting results may be due to the robust exclusion criteria used in attempt to only include sPTB in our study. To include only sPTB, many deliveries among women who had chronic diseases were excluded because they were indications for iPTB. Racial minorities are disproportionately affected by these chronic diseases, including hypertension, 42 diabetes, 43 and preeclampsia. 44 This may have led to unintended exclusions of deliveries that were not induced, while disproportionately excluding mothers of minority race. Considering this, the racial findings of the current study suggest that the relationship between race and sPTB may not be direct but modulated by the relationship between race and other health conditions that impact pregnancy. This supports the understanding that race is a social construct and a social determinant of health. 45 Race does not have a biological basis in increasing risk for poor health outcomes. Additionally, the categories of race in the model were Asian, Black, White, and other/unknown. The other/unknown category includes races of low prevalence in the sample and unknown race, which does not allow for inclusion of all races in the model.
When the deliveries transferred to IHC were excluded, the same variables entered the model with similar aORs, with the addition of maternal Asian race, which was found to be a protective factor for sPTB (aOR, 0.57; 95% CI, 0.35-0.91), with a P-value of 0.02. This finding was less significant than the other factors associated with sPTB and may be significant only due to the large sample size and not be a clinically meaningful finding. Despite this, the literature is consistent with this result, with Dongarwar et al 46 finding that Chinese, Korean, Japanese, and Asian Indian mothers are at decreased risk of PTB when compared to White mothers. Our data did not have the granularity on specific Asian race categories noted by Dongarwar et al, and thus could not assess whether these subcategories were at decreased risk of sPTB.
Other RF and sPTB
The other significant RFs for sPTB found in our study, including extremes of maternal age and maternal smoking, were consistent with known risk factors for PTB, 6 although it was unclear in literature if these RFs were specific to sPTB as well. Additionally, female sex of the newborn was found to be a protective factor for sPTB and is a known protective factor of PTB. 23 Lastly, our study finds that transfer to IHC from an outside hospital is highly associated with sPTB. This is expected, as transfers are typically due to the need for escalation of care for both the mother and the fetus, one of the indications being a PTB and the need for a higher-level nursery. We acknowledge that an aOR of 671.3 is unusually high and may raise concerns about model stability or potential multicollinearity. To address this, we conducted a collinearity diagnostic using the Variance Inflation Factor (VIF) for all predictor variables included in the model. The VIF values were all below 2, indicating that multicollinearity was unlikely to be a significant issue. The high aOR reflected a strong and genuine association between those transferred to IHC and sPTB in our sample. Specifically, 81.3% of transferred deliveries were sPTBs, compared to only 8.1% in the non-transferred group.
Additionally, a separate analysis was done with the transferred deliveries excluded due to concern that the rural population’s risk of sPTB was being inflated with the assumption that term deliveries from outside hospitals were less likely to be transferred to IHC. The resulting significant RFs were similar to the RFs found in the original analysis, the only difference being that Asian race was a significantly protective factor of sPTB. The analyses suggest that despite the high aOR for those transferred to IHC for sPTB, the other significant RFs included in the model are independently and statistically significant.
Strengths and Limitations
Strengths of our study include the large sample size spanning over 14 years, access to extensive EMR data including many PTB RFs, the use of a multivariable model, the Midwest region specific results, and the focus on sPTB. Many studies evaluating rurality as a RF for PTB in the past used birth record data along with patient surveys and had limited access to variables,13,25,26 while the current study utilized all EMR data associated with pregnancies as well as the EMR data of the newborn. Second, the current study allowed for Midwest region specific conclusions, which is valuable, due to the wide variability in risk for PTB among U.S. states. 15 Many recent studies investigate rurality as a RF for PTB on a national level,13,14 but this does not allow for region specific conclusions necessary for informing regional policy and clinical practice. Our study allows for conclusions specific to the Midwest, although this may be viewed as also a limitation, as the results may not be applicable outside of the Midwest. Additionally, our study focused on sPTB specifically to identify RFs for sPTB with unknown causation, as opposed to including iPTB, like other studies assessing rurality as a RF for PTB.13,25,26 However, there could be misclassification bias, since it is possible that not all the preterm deliveries included in our study were sPTBs, as we did not have access to a variable stating if the PTB was induced or spontaneous. Instead, to include only sPTBs, pregnancies with diagnoses that were indications for iPTB 18 and induced abortion were excluded, 19 resulting in the possibility that some of the data set includes iPTB due to other indications that were not excluded, such as fetal intrauterine growth restriction. Furthermore, it is possible that not all the excluded high-risk pregnancies ended in induction, so sPTBs may have been excluded unintentionally.
Another limitation of this study was that our data was only as accurate as what was entered into the EMR. Additionally, not all known RFs for PTB were able to be tested in the study or were crudely measured due to data limitations. Maternal stress, 47 illicit drug use, 48 access to prenatal care, and environmental exposures were not included in the study, and maternal socioeconomic status 47 was estimated by type of maternal health insurance. Illicit drug use is only measured when there is suspicion of such. The race categories were also crudely formed, as the unknown/other race category included many different races of low prevalence of those races in the data. These approximations and omissions may have caused potential for confounding variables in the study, although interactions for confounding variables were tested and not found to enter the models as noted in the statistical analyses above.
Lastly, it is known that nationally, rural mothers of higher risk pregnancies are more likely to seek care in an urban area than rural mothers of lower risk pregnancies. 11 Because our data set did not include the denominator of all rural pregnancies in Iowa, it only included the deliveries occurring at IHC, it is possible that the rural pregnancies in our study are at higher risk of sPTB because the rural women traveling to our center may be inherently at higher risk. However, our study excluded many high-risk pregnancies in efforts to include only sPTB opposed to iPTB, and all of the adjacent counties to the county where IHC is located, except for 1 county, were either maternity care deserts, 28 or have had a L&D unit closure during the time span the data set included. 49
Conclusion
Our study identified significant and independent RFs for sPTB at our Midwestern tertiary care center, including rural residence, extremes of maternal age, and maternal smoking, while female sex of the newborn was a protective factor. The next step is to identify why our rural community is at increased risk. It may be due to the L&D closures across the state decreasing the quality of prenatal care, increased maternal stress, increased illicit substance use, immunologic factors, genetic contributions, increased exposure to pesticides or other contaminants, or a combination of these factors. The many proposed causes and RFs for sPTB are multifactorial, and each region of the U.S. (or furthermore, world) has their own uniquely relevant contributors. It is important for other regions to identify the independently significant sociodemographic RFs for sPTB in their communities as well, with the goal that all regions of the U.S. may decrease risk for sPTB through optimization of public health policy and targeted healthcare interventions for improved health of rural communities.
Footnotes
Acknowledgements
Department of Family and Community Medicine, The University of Iowa Roy J. and Lucille A. Carver College of Medicine, University of Iowa, Iowa City, IA; ICTS and Biomedical Informatics Group; Department of Obstetrics and Gynecology, The University of Iowa Roy J. and Lucille A. Carver College of Medicine University of Iowa, Iowa City, IA; The University of Iowa Roy J. and Lucille A. Carver College of Medicine; Intergenerational Health Knowledgebase; Women’s Health Tissue Repository Group; and Iowa Developmental Disability Research Center.
Ethical Considerations
This study received ethical approval from IRB-01 (approval #201902830) in 2019. This is an IRB-approved retrospective study, all patient information was de-identified and patient consent was not required. Patient data will not be shared with third parties.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Department of Family and Community Medicine, The University of Iowa Roy J. and Lucille A. Carver College of Medicine, University of Iowa, Iowa City, IA; Department of Obstetrics and Gynecology, The University of Iowa Roy J. and Lucille A. Carver College of Medicine, University of Iowa, Iowa City, IA; The University of Iowa Roy J. and Lucille A. Carver College of Medicine; UL1TR002537, UL1TR002537-S1, HD089940, P50HD10355601A1 (via Hawk-Intellectual and Developmental Disability Research Center) (to M.K.S.); and National Institutes of Health (NIH) CTSA UM1TR004403 (UI ICTS).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data may be available upon request.
