Abstract
Introduction:
The community-based management of type 2 diabetes in China remains suboptimal. Incorporating family support presents a promising approach to enhancing facilitators and addressing barriers to effective diabetes self-management, particularly in the Chinese context where familial relationships and collective family responsibility play a central role in health-related decision-making and daily life. We aimed to validate a family support-based self-management behavioral intervention program, Systematic evaluation, core education, self-directed planning, triple feedback, and develop a habit program (SeCe-STRIVE). The program comprises 5 modules: systematic evaluation, core education, self-directed planning, triple feedback, and habit development.
Research Design and Methods:
We conducted a community-based, single-center, parallel randomized controlled trial in the Xietu community of Xuhui District, Shanghai. Eligible patients were randomly assigned to either the intervention or control group. Participants in the intervention group received the SeCe-STRIVE health management program based on family support, while those in the control group received the community-based follow-up management program for type 2 diabetes. The primary outcome measured was the total self-management behavior score and its changes across various dimensions from baseline. Secondary outcomes included family support, family function, diabetes knowledge, quality of life, self-efficacy, and biochemical markers. Post-intervention changes in both primary and secondary outcomes relative to baseline were analyzed using analysis of covariance.
Results:
The study ultimately included 113 participants in the intervention group and 112 in the control group. The intervention group’s total self-management behavior score improved by 12.74 (95% CI = 10.07, 15.40, P < .001) points more than the control group. Across all dimensions of self-management behaviors, the intervention group showed greater improvement, including diet, exercise, blood glucose monitoring, foot care, and medication adherence. Participants with low family support exhibited greater improvements in self-management behaviors compared to those with high family support. The intervention group demonstrated significant improvements in family support, family function, diabetes knowledge, and self-efficacy. Although quality of life and biochemical indicators of glycated hemoglobin, fasting blood glucose, and blood lipids showed a decreasing trend, the differences were not statistically significant.
Conclusions:
Compared to the control group, patients with type 2 diabetes who received the SeCe-STRIVE program intervention showed significant improvements in self-management behaviors, accompanied by enhancements in family support, family functioning, and self-efficacy. Further large-scale, multi-center trials with longer follow-up periods are needed to confirm these results.
Trial Registration:
ChiCTR, ChiCTR2200060416. Registered 01 June 2022, https://www.chictr.org.cn/showproj.html?proj=167086
Keywords
Introduction
China has the highest number of diabetes cases globally, and this figure continues to rise. 1 The prevalence of diabetes among Chinese adults aged 20 to 79 years is projected to increase from 8.2% to 9.7% between 2020 and 2030. 2 China has been committed to establishing a community-based primary healthcare system that provides patients with essential, low-cost healthcare services. 3 To enhance the management of type 2 diabetes mellitus (T2DM), the Chinese government introduced a family doctor contracting system. This initiative aims to improve diabetes control, reduce complications, and alleviate the healthcare burden by providing continuous treatment through contracted family doctors. 4 However, the community-based management of T2DM in China remains suboptimal due to various challenges. Community health service centers are responsible for managing multiple chronic diseases, making it difficult to focus solely on diabetes management. Additionally, there are significant disparities in basic healthcare capacity and health service levels across different regions and between urban and rural areas.5,6
The Association of Diabetes Care and Education Specialists (ADCES) highlights 7 key diabetes self-care practices that can improve health outcomes: healthy coping, healthy eating, being active, taking medication, monitoring, problem solving, and reducing risks. 7 Support from family and social networks is crucial in sustaining these lifestyle changes. 8 Increasing evidence suggests that family members are more effective in supporting individuals with diabetes when they are actively involved in diabetes management tasks, such as medication adherence and home blood glucose monitoring.9 -12 A randomized controlled trial in Brazil by Gomes et al 13 demonstrated a greater reduction in glycosylated hemoglobin (HbA1c) in the intervention group with family carer involvement compared to the control group. Similarly, Wichit et al 14 conducted a randomized controlled trial in Thailand, which showed that a family-oriented program improved self-efficacy and self-management in individuals with diabetes, with higher self-management scores correlating with lower HbA1c levels and enhanced quality of life. However, a 12-month controlled clinical trial in Chile by García-Huidobro et al 15 did not find a significant association between family-based intervention and HbA1c reduction, potentially due to incomplete implementation, low statistical power, and confounding variables across groups. However, research on family supported self-management program is limited in China. Moreover, most previous studies have overlooked the consideration of how family support influences patients’ self-management behaviors through specific pathways when designing intervention plans.
Family environments and relationships can be complex, as are the ways family members contribute to promoting self-management practices. Interventions aimed at enhancing family support must be tailored to an individual’s specific circumstances, family dynamics, and sociocultural context. Zhu et al 16 examined the self-management behaviors of T2DM patients in Shanghai, China, and concluded that family support significantly impacts these behaviors. By analyzing factors at both the individual and family levels, they identified the pathways through which family support influences self-management: family function → family support → self-management behaviors → quality of life. These findings suggest that enhancing family function can foster greater family support, which in turn improves self-management behaviors in T2DM patients. Building on this role mechanism model, we developed a self-management behavior intervention program for T2DM patients based on family support.
While family-based interventions have shown promise in enhancing diabetes management globally, few studies have validated such interventions in China, particularly in urban community settings. This study aims to evaluate the short-term efficacy of the Systematic evaluation, core education, self-directed planning, triple feedback, and develop a habit program (SeCe-STRIVE program) in improving self-management behaviors and explore its impact on quality of life and family dynamics.
Methods
Design and Participants
We conducted a community-based, single-center, parallel randomized controlled trial in the Xietu community of Xuhui District, Shanghai. Participants were enrolled based on the following inclusion criteria: a diagnosis of T2DM according to the Guideline for the prevention and treatment of T2DM in China (2020 edition); more than 1 year of residence in Xietu Street, Xuhui District, Shanghai; full cognitive and behavioral abilities; voluntary informed consent to participate; and having a regular family member or primary caregiver living with them. Exclusion criteria included: inability to cooperate due to cognitive impairment (eg, Alzheimer’s disease), severe physical function impairment, severe audiovisual impairments, conditions that could result in noncompliance or loss of follow-up, and current or recent (within the last 30 days) participation in other related research projects. Patients were contacted and recruited by their pre-enrolled family doctors before the study. The study received ethical approval from the Ethics Committee of Zhongshan Hospital, Fudan University (Approval Number: B2022-055).
Randomization, Allocation, and Concealment
Eligible participants were randomly assigned to either the trial or control group. Block randomization with a block size of 4 was used. A statistician, who was not involved in the trial, generated the blinded randomization sequence using SAS 9.4 software. Allocation concealment was ensured through opaque envelopes, and family doctors were assigned to the intervention and control groups in a 1:1 ratio.
Sample Size
We employed a 2-group, equal sample size, parallel randomized controlled trial design, with the change in self-management behavior in patients with T2DM after a 3-month intervention as the primary outcome. The sample size was calculated using the formula:
Intervention Description
Participants randomly assigned to the intervention group received the family support-based SeCe-STRIVE health management program, while those in the control group received the community-based routine follow-up management program for individuals with T2DM. The SeCe-STRIVE program comprises 5 modules: systematic evaluation, core education, self-directed planning, triple feedback, and develop a habit (Figures 1 and 2). Nine intervention teams were formed, each consisting of trained family doctors, public health doctors, and general nurses. Each team included a family doctor paired with either a nurse or a public health doctor, and they were responsible for managing a group of 12 to 13 diabetic patients and their families. The intervention was implemented in 3 phases. In the first phase, the intervention team provided patients and their families with knowledge and skills related to diabetes, while also establishing a support system. The second phase focused on the active involvement and decision-making of the family members, helping them to enhance the patient’s self-management behaviors by implementing the care plan. In the third phase, the family doctor team evaluated the self-management behaviors of both the patient and their family, as well as the family support system, and made targeted improvements based on identified issues.

Theoretical model of the SeCe-STRIVE Health Management Program.

Implementation process of the SeCe-STRIVE Health Management Program.
The community-based routine follow-up management program for individuals with T2DM involves establishing a follow-up plan based on the patient’s glycemic control and comorbidities. This includes determining the frequency and method of follow-up, the specific content of the follow-up, and the record of follow-up information. The entire follow-up process is managed by the family doctor’s assistant, who handles the patient’s consultations. If the assistant is unable to address a patient’s concerns, the family doctor provides further guidance.
Data Collection and Outcomes
The intervention was conducted over a 3-month period, with comprehensive data collection performed at baseline and at the end of the intervention using the survey section of the case report form for both the intervention and control groups. Additionally, participants in the intervention group completed the Family Support Behavior Implementation Plan form and the Patient Self-Management Behavior Scale.17,18 Data collection for the trial group occurred weekly during the first month, bi-weekly in the second month, and every 4 weeks in the third month, for a total of 7 measurements.
The primary outcome was assessed using the total self-management behavior score and the change in each dimension compared to baseline. Secondary outcomes included family support and its dimensions, family function and its dimensions, diabetes knowledge score, quality of life, self-efficacy, body mass index (BMI), HbA1c, postprandial blood glucose, fasting blood glucose, total cholesterol, triglycerides, high-density lipoprotein cholesterol (HDL-C), and low-density lipoprotein cholesterol (LDL-C).
We employed the Family Adaptability and Cohesion Evaluation Scales (FACES III) to assess the adaptability and cohesion of the family. 19 To evaluate family support, we utilized the Multidimensional Diabetes Questionnaire (MDQ). 17 The Summary of Diabetes Self-Care Activities Assessment (SDSCA) was used to measure diabetes self-management behaviors. 18 Quality of life was assessed using the EuroQol 5 Dimensional Questionnaire (EQ-5D), 20 while diabetes knowledge was evaluated through the Michigan Diabetes Knowledge Test (DKT). 21 Finally, self-efficacy was measured using the Self-efficacy for Diabetes (SED) scale. 22
Statistical Analyses
According to the intention-to-treat (ITT) principle, subjects were analyzed based on their randomized group assignments, irrespective of the intervention they actually received. The primary dataset for analysis was the full analysis set (FAS), which included subjects who underwent at least 1 intervention and had a post-intervention efficacy assessment.
Counts were expressed as frequencies (percentages), while continuous measures were presented as means with standard deviations
Since the primary and secondary outcomes measure post-intervention changes relative to baseline, analyses of covariance (ANCOVA) were applied, with the baseline level as the covariate.
In the intervention group, changes in self-management behavior scores and in each dimension over time were analyzed using repeated measures analyses of variance (ANOVA). The data were tested for the assumption of sphericity; if this assumption was met, the results of the univariate ANOVA were used. If the assumption of sphericity was not met, results from both multivariate and univariate ANOVA were reported.
We further conducted a post-hoc exploratory analysis. Using the critical value of the total family support score from a previous cross-sectional study,
16
we divided the intervention group into 2 subgroups: a low family support group (total family support score <31.5) and a high family support group (total family support score ≥31.5). Measurements were expressed as
Missing data for the primary outcome were handled using multiple imputation, while secondary outcomes were analyzed using complete case analysis.
We performed the data analysis using SPSS 25.0, with a 2-sided P value of <.05 as significance level.
Results
Participants
Recruitment began on April 28, 2023, the intervention started on May 18, 2023, and the intervention lasted for 3 months. During the study, 243 individuals underwent the screening assessment. After excluding those who did not meet the inclusion criteria and those unwilling to participate, 226 subjects were randomly assigned to groups on a 1:1 basis. One patient in the control group withdrew after randomization, without receiving any intervention, due to personal reasons. The final sample included 113 participants in the test group and 112 in the control group (Figure 3). The mean age in the intervention group was 70.87 years, with 54 participants (47.8%) being male. In the control group, the mean age was 72.52 years, with 57 participants (50.9%) being male (Table 1).
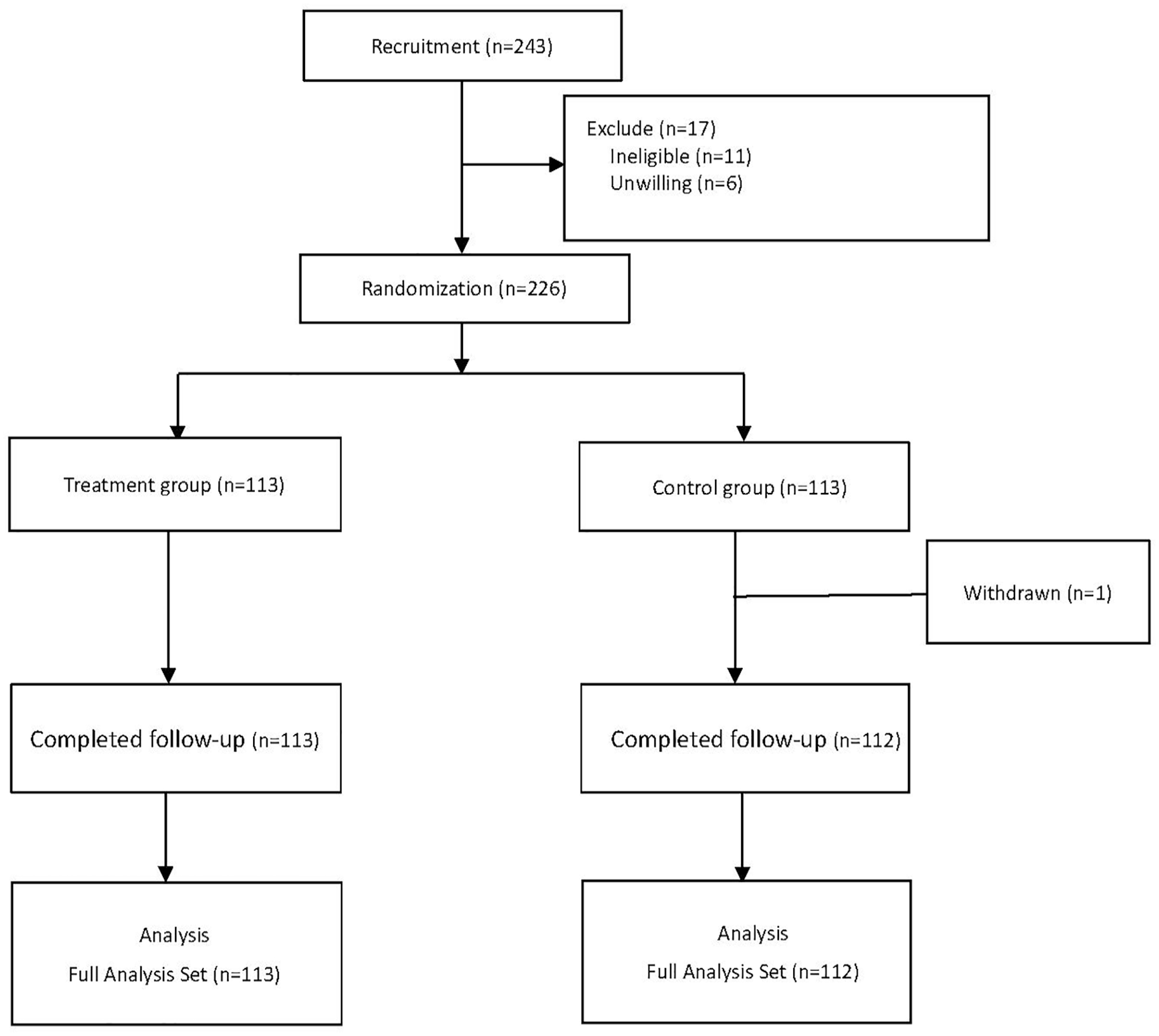
Flow chart of individuals with type 2 diabetes participating in the study.
Baseline Characteristics of Individuals Randomized to Either the Intervention Group or Control Group.

Abbreviations: BMI, body mass index; HbA1c, glycosylated hemoglobin; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol.
Measured by Summary of Diabetes Self-Care Activities Assessment (SDSCA), scale range 0 to 77. SDSCA consists of 11 items (4 diet, 2 exercise, 2 blood glucose monitoring, 2 foot care, 1 medication adherence).
Measured by Multidimensional Diabetes Questionnaire (MDQ), scale range 0 to 72. MDQ consists of 12 items (8 Positive reinforcement behaviors, 4 Negative reinforcement behaviors).
Measured by Family Adaptability and Cohesion Evaluation Scales (FACES III), scale range 20 to 100. FACES III consists of 20 items (10 cohesion and 10 adaptability).
Measured by Diabetes Knowledge Test, scale range 0 to 100.
Measured by Self-efficacy for Diabetes, scale range 8 to 80.
Measured by EuroQol 5 Dimensional Questionnaire, scale range 5 to 15.
Primary Outcome
Compared to the control group, the total self-management behavior score in the intervention group was significantly higher by 12.74 points (95% CI = 10.07, 15.40, P < .001). Across all 5 dimensions of self-management behavior, the intervention group showed significantly greater improvements than the control group: diet improved by 2.77 points (95% CI = 1.86, 3.69, P < .001), exercise by 1.87 points (95% CI = 1.05, 2.69, P < .001), blood glucose monitoring by 2.57 points (95% CI = 1.66, 3.48, P < .001), foot care by 2.57 points (95% CI = 3.92, 5.80, P < .001), and medication by 0.61 points (95% CI = 0.15, 1.06, P = .009; Table 2).
Test of Differences in Primary Outcome Between Intervention Group and Control Group.
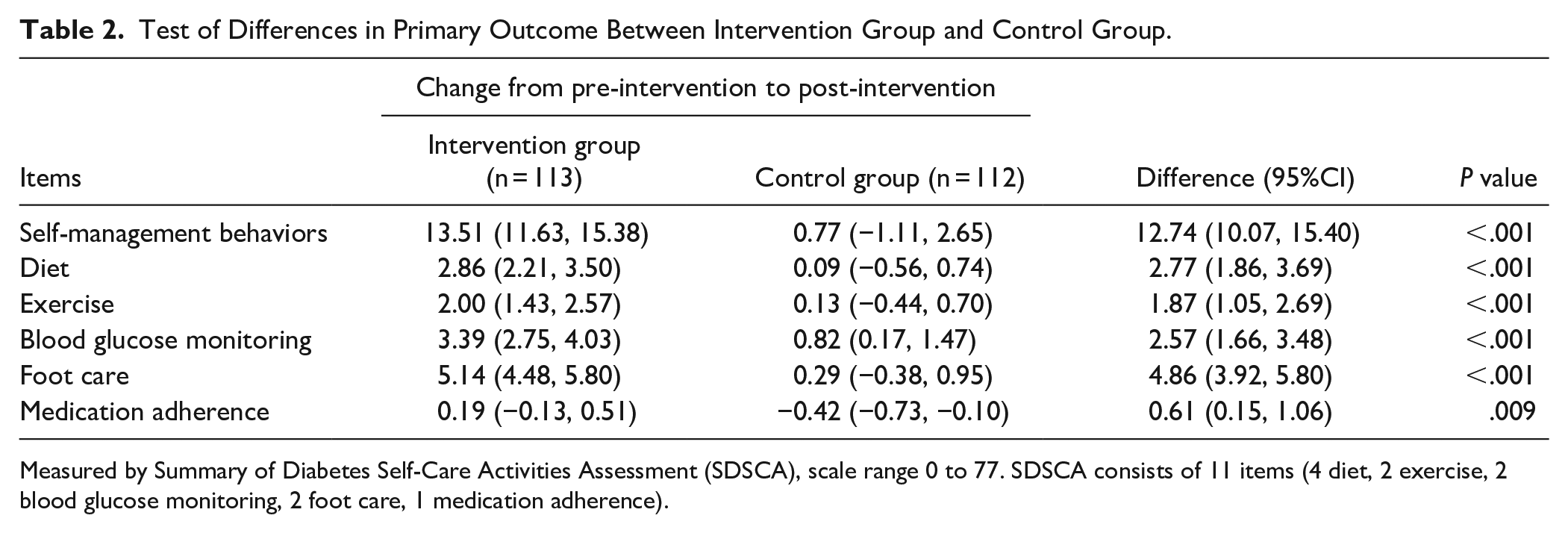
Measured by Summary of Diabetes Self-Care Activities Assessment (SDSCA), scale range 0 to 77. SDSCA consists of 11 items (4 diet, 2 exercise, 2 blood glucose monitoring, 2 foot care, 1 medication adherence).
Secondary Outcome
In terms of family support, after 3 months of intervention, the total family support score in the intervention group was significantly higher than that in the control group by 22.72 points (95% CI = 19.96, 25.48, P < .001). The positive reinforcing behaviors dimension significantly improved by 19.12 points (95% CI = 16.79, 21.45, P < .001), while the negative reinforcing behaviors dimension increased by 3.55 points (95% CI = 2.24, 4.86, P < .001). Regarding family function, the intervention group significantly improved by 13.19 points (95% CI = 10.53, 15.85, P < .001) more than the control group in total score, 6.46 points (95% CI = 5.18, 7.74, P < .001) more in cohesion, and 6.65 points (95% CI = 5.05, 8.26, P < .001) more in adaptability. Additionally, the intervention group showed a greater improvement of 18.44 points (95% CI = 14.66, 22.22, P < .001) in diabetes knowledge and a significant increase of 14.51 points (95% CI = 11.88, 17.15, P < .001) in self-efficacy. No significant difference was observed in the change in quality of life between the intervention and control groups, with a difference of 0.19 points (95% CI = −0.06, 0.44, P = .268). Similarly, there was no significant difference in the change in BMI, with a difference of −0.14 points (95% CI = −0.94, 0.67, P = .268) between the 2 groups.
In terms of biochemical indicators, after the intervention, the 2-h postprandial blood glucose level in the intervention group significantly decreased by 0.58 mmol/L (95% CI = −1.10, −0.06, P = .03) more than in the control group. Although the biochemical indicators of glycated hemoglobin, fasting blood glucose, and blood lipids showed a decreasing trend, the differences were not statistically significant. Glycated hemoglobin in the intervention group decreased by 0.21% more than in the control group (95% CI = −0.47, 0.06, P = .124), and fasting blood glucose decreased by 0.02 mmol/L more than in the control group (95% CI = −1.10, −0.06, P = .97). For blood lipids, triglycerides, total cholesterol, and LDL-C in the intervention group decreased slightly compared to the control group, with reductions of 0.10 mmol/L (95% CI = −0.34, 0.14, P = .4), 0.01 mmol/L (95% CI = −0.33, 0.31, P = .960), and 0.01 mmol/L (95% CI = −0.25, 0.23, P = .911), respectively. HDL-C increased by 0.12 mmol/L (95% CI = −0.01, 0.24, P = .058), though none of these differences reached statistical significance (Table 3).
Test of Differences in Secondary Outcome Between Intervention Group and Control Group.

Abbreviations: BMI: body mass index. HbA1c: glycosylated hemoglobin. HDL-C: high-density lipoprotein cholesterol. LDL-C: low-density lipoprotein cholesterol.
Measured by Multidimensional Diabetes Questionnaire (MDQ), scale range 0 to 72. MDQ consists of 12 items (8 Positive reinforcement behaviors, 4 Negative reinforcement behaviors).
Measured by Family Adaptability and Cohesion Evaluation Scales (FACES III), scale range 20 to 100. FACES III consists of 20 items (10 cohesion, 10 adaptability).
Measured by Diabetes Knowledge Test, scale range 0 to 100.
Measured by Self-efficacy for Diabetes, scale range 8 to 80.
Measured by EuroQol 5 Dimensional Questionnaire, scale range 5 to 15.
Changes in Self-Management Behaviors Over Time in the Intervention Group
In addition to assessments at baseline and at the end of the 3-month intervention, self-management behaviors of patients in the intervention group were continuously measured at the second, third, fourth, sixth, eighth, and twelfth weeks of the intervention.
For self-management behaviors and the scores of each dimension, including the total self-management behavior score, diet score, exercise score, blood glucose monitoring score, foot care score, and medication adherence score, none met the assumption of sphericity. The result of multivariate ANOVA (P < .001) indicated statistically significant differences in all indicators across different time points. Further pairwise comparisons revealed that the differences in mean values gradually increased over time, and were statistically significant at all time points compared to the first week (P < .05). The trends in mean value changes across time showed a linearly improvement (P < .05; Figure 4).

Self-management behaviors and changes in dimensions in patients with type 2 diabetes mellitus.
Changes in Self-Management Behaviors Across Family Support Groups in the Intervention Group
After the 3-month intervention, the results of the independent samples t-test on the pre-post differences in self-management behaviors scores between the high family support group and the low family support group revealed significant differences in the changes between the 2 groups of patients with T2DM. The difference in the total self-management behavior score was 13.40 points (95% CI = 6.00, 20.81, P < .001). For specific dimensions, the dietary score difference was 4.44 points (95% CI = 1.98, 6.91, P < .001), the exercise score difference was 2.42 points (95% CI = 0.02, 4.82, P = .048), and the foot care score difference was 4.50 points (95% CI = 1.85, 7.15, P < .001). In all these aspects, the low family support group showed greater improvements than the high family support group (Table 4).
Test of Differences in Self-management Behaviors Across Family Support Groups in Intervention Group.

Measured by Summary of Diabetes Self-Care Activities Assessment (SDSCA), scale range 0 to 77. SDSCA consists of 11 items (4 diet, 2 exercise, 2 blood glucose monitoring, 2 foot care, 1 medication adherence).
Discussion
This study employed a randomized controlled trial design, involving 225 patients with T2DM mellitus who were randomly allocated to either the intervention or control group. The intervention group received a 3-month family support-based SeCe-STRIVE health management intervention, while the control group underwent the community-based routine follow-up management program. Through comparisons of self-management behaviors, blood glucose levels, quality of life, and health outcomes between the 2 groups, we found that the SeCe-STRIVE program significantly improved self-management behaviors in patients with T2DM. These findings suggest that the family support program effectively helps patients establish and maintain consistent, structured, and evidence-based self-management practices, which contribute to better blood glucose control, prevention of diabetes-related complications, and improvements in quality of life and overall health.
Our findings are consistent with previous studies regarding the role of family interventions in diabetes management.11,14,23,24 A 9-month follow-up randomized controlled trial (RCT) involving 196 T2DM patients in Thailand demonstrated that family-involvement interventions are beneficial for diabetes management, particularly when spouses or women serve as caregivers. 11 Another single-blinded RCT conducted in rural Thailand (including 140 T2DM patients) found that family-oriented programs significantly improved patients’ self-efficacy and self-management capabilities. 14 An RCT in Western Ethiopia (including 152 T2DM patients) showed that a culturally tailored, family-supported, community-based diabetes self-management education and support program positively affected patients’ HbA1c and triglyceride levels. 23 Based on the mechanism model of family support’s impact on self-management behaviors in T2DM patients, our developed intervention program also demonstrated positive effects on diabetes self-management behaviors. These findings collectively emphasize the importance of incorporating family into diabetes management programs.
Additionally, the SeCe-STRIVE program significantly improved both family support and family function in patients. For family support, the intervention group showed an increase in the total score, positive reinforcement behaviors, and a reduction in negative reinforcement behaviors. This indicates that the intervention encouraged family members to engage more in behaviors such as helping, monitoring, encouraging, and praising the patient, as well as supporting tasks like preparing medication and reminding the patient to exercise regularly. 25 In terms of family function, the intervention led to improvements in overall function, cohesion, and adaptability. The family-based intervention fostered greater care and interaction among family members. 26
Our protocol is grounded in a model of the mechanism of action influencing self-management behaviors in patients with T2DM, derived from a previous study. 16 This study validates the mechanism of the family support system and offers foundational insights for future intervention strategies. While past research on family-supported self-management has primarily concentrated on patient knowledge and glycemic control outcomes, 27 this study extends its focus to include patient self-efficacy and quality of life, in addition to knowledge and biochemical indicators. This broader approach helps provide a more comprehensive understanding of patient outcomes.
In this study, patients in the intervention group demonstrated a significant increase in their knowledge of foot care, blood glucose monitoring, and dietary management for T2DM. Their self-efficacy levels also improved significantly more than those in the control group, which aligns with findings from previous studies.28 -32 However, while there was an upward trend in the quality of life among patients in the intervention group, no significant difference was observed compared to the control group. This may be because quality of life typically reflects a patient’s long-term life status, making it harder to observe substantial changes over a short period of time. 33 To better understand the sustained effects of the intervention, future studies should incorporate longer-term follow-up periods. In addition, specific modifications to the intervention could be explored to enhance outcomes. For example, incorporating mental health components, such as stress management and psychological counseling, may further improve quality of life.
The results revealed that the reduction in 2-h postprandial blood glucose was more significant in the intervention group compared to the control group. Although biochemical markers such as glycated hemoglobin, fasting blood glucose, and lipid levels showed a downward trend, no statistically significant differences were found. This may be attributed to the relatively short intervention duration in this study. A systematic review by Chrvala et al 34 found that the most substantial reductions in HbA1c occurred in studies with follow-up periods ranging from 6 to 12 months, particularly in those where healthcare professionals had more than 10 h of contact with patients. Given the progressive and chronic nature of T2DM, this downward trend could still be considered a positive outcome, as it may help delay chronic complications. 35 Moreover, the sample size was not calculated based on secondary outcome measures, and the current sample size may be insufficient for detecting significant differences in these outcomes. Future studies could explore extending the intervention duration and increasing the study sample size to potentially achieve more pronounced effects. Another possible reason for the lack of significant improvement after a short-term intervention is that patients enrolled in family doctor contract services were relatively healthy at baseline.
Lipid levels are associated with diet, exercise habits, and genetic factors. 36 In the intervention group of this study, patients’ diets and exercise routines were modified through a comprehensive family-supported intervention. After the intervention, total cholesterol, triglycerides, and LDL-C in the test group showed greater reductions compared to the control group, while HDL cholesterol exhibited an upward trend. Regarding changes in patients’ self-management behavior scores, except for a decline observed between the fourth and sixth weeks, all other time points showed a steady increase, demonstrating the positive impact of the family-based SeCe-STRIVE health management program on self-management behaviors. The decline seen from the fourth to sixth weeks could be attributed to the 2-week interval in assessments, compared to the weekly intervals at other time points. This gap may have led to a temporary slackening of self-management behaviors during that period. Notably, neither the total score nor the dimension scores plateaued by week 12, indicating that patients’ self-management behaviors had further potential for improvement. Future studies could consider longer interventions or extended follow-up periods to explore the trajectory of these changes and determine when patients’ self-management behaviors stabilize.
In this study, baseline total self-management behavior scores differed significantly between the groups with high and low family support. After receiving the family support-based SeCe-STRIVE health management program, the group with low family support exhibited a more substantial increase in total self-management behavior scores and improvements in diet, exercise, and foot care dimensions compared to the group with high family support. This indicates that patients with low family support and their families derive greater benefits from the SeCe-STRIVE intervention program.
This study was conducted in Xietu Street, Xuhui District, Shanghai, China. Consistent with findings from several other studies,14,23,37 our results indicate that a diabetes management program incorporating family support significantly improved self-management behaviors in patients with T2DM. To reduce selective bias, opaque envelopes were used to ensure allocation concealment. However, this study still has some limitations. First, potential limitations in generalizability should be acknowledged, as demographic factors, lifestyle habits, and socio-cultural contexts may differ across regions. Future studies should consider expanding to diverse geographic locations and populations to validate and enhance the applicability of these findings. Second, our study lacked blinding during implementation, and since participants in both the intervention and control groups lived in the same community, contamination of the control group may have occurred. To mitigate this bias, we emphasized confidentiality in the informed consent form. Although the intervention was benign and any contamination likely led to an underestimation of the true effect, our study still demonstrated a significant improvement in self-management behaviors in the intervention group compared to the control group.
Conclusion
The study demonstrated that the SeCe-STRIVE health management program, based on family support, was effective in enhancing self-management behaviors among patients with T2DM. Significant improvements were observed in family support, family function, self-efficacy, and diabetes perceptions. Notably, the program had a more pronounced impact on the self-management behaviors of patients with low family support, highlighting the need for future interventions to focus specifically on this group. Regarding the sustainability of the intervention’s effects, it remains unclear whether the systematic family support model requires a 1-time reinforcement or ongoing regular reinforcement similar to medication taking. The current findings are inconclusive, necessitating further multi-center studies and long-term follow-up to validate and refine this intervention approach.
To promote and encourage the integration of family support into diabetes management, healthcare providers should actively involve family members in consultations and provide educational resources, while governments and organizations should develop policies that incentivize family involvement, such as financial subsidies or community recognition programs. Future research should explore the cost-effectiveness of this program by calculating the incremental cost-effectiveness of improving self-management behavior, assessing glycemic control rates, per capita costs, and annual medical expenses after implementing the program, and comparing these outcomes with the conventional approach.
Supplemental Material
sj-docx-1-jpc-10.1177_21501319251330384 – Supplemental material for Effectiveness of a Family-Based Self-Management Intervention for Type 2 Diabetes Patients Receiving Family Doctor Contract Services: A Community-Based Randomized Controlled Trial
Supplemental material, sj-docx-1-jpc-10.1177_21501319251330384 for Effectiveness of a Family-Based Self-Management Intervention for Type 2 Diabetes Patients Receiving Family Doctor Contract Services: A Community-Based Randomized Controlled Trial by Lan Zhu, Jing Wang, Zhigang Pan, Wei Zhang, Jing Tang, Hua Yan, Fulai Shen and Yi Shen in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
We would like to extend our sincere gratitude to all the participants for their valuable contributions to this study. Additionally, we are deeply thankful to the Xietu community for their support.
Author Contributions
Lan Zhu led the conception and design of the study, participated in data analysis and interpretation, and drafted the manuscript. Jing Wang contributed to methodology, data interpretation and drafted manuscript. Wei Zhang contributed to methodology, data analysis, and manuscript revisions; Zhigang Pan contributed to the study conception, design, and manuscript revisions. Fulai Shen and Yi Shen participated in study design, data analysis, and data visualization. Hua Yan and Jing Tang contributed to study design and coordinated data collection and cleaning. All authors read and approved the final manuscript.
Ethical Considerations
The study received ethical approval from the Ethics Committee of Zhongshan Hospital, Fudan University (Approval Number: B2022-055).
Consent to Participate
All participants provided informed consent before participating in the study.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Scientific Research Project of Science and Technology Commission of Xuhui District, Shanghai (Grant # SHXH202111). The First National High-Level Medical Talent Program (Grant # NHFPC [2024] No. 41) Shanghai Health System Key Support Discipline Plan (Grant # 2023ZDFC0401).
Data Availability
Data sharing is not applicable due to confidentiality policies.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
