Abstract
Objective:
To evaluate whether there is an increase in vaccination rates of patients with diabetes mellitus who received guidance to update their vaccination schedules for influenza, hepatitis B, pneumonia, and tetanus.
Methodology:
A randomized controlled trial was conducted between December 2018 and November 2020. The sample consisted of 139 patients from the endocrinology service outpatient clinic of Santa Maria University Hospital was randomized into an intervention group (
Results:
The mean age of the subjects was 59.17 ± 12.91 years and 62.6% were female. No age differences were observed between genders and randomization groups (
Conclusion:
The orientation to update the vaccination schedule through telephone contact was effective in increasing vaccination rates for influenza, hepatitis B, pneumonia, and tetanus.
Trial registry:
RBR-92z99d2 https://ensaiosclinicos.gov.br/rg/RBR-92z99d2
Introduction
Diabetes mellitus (DM) is one of the most prevalent chronic diseases worldwide. It presents as hyperglycemia due to total or relative insulin insufficiency.1,2
The immunization of patients with DM is an important health protection strategy because of the potential to reduce morbidity and hospitalizations. The absolute risk of infection from any cause in patients with DM is 8% higher compared to the general population. But, when the rate of hospitalization for infectious disease is evaluated, it is twice as high, and mortality is about 90% higher. 3 The importance of vaccination as a form of disease prevention has even become a hot topic worldwide since 2020 due to the COVID-19 pandemic. 1
In Brazil, protocols establishing the recommended vaccines for diabetics are updated annually by the Brazilian Society of Immunizations (SBI). The Brazilian protocols have the same recommendation of the American Diabetes Association which recommends that patients with DM have the following vaccines updated for their age group: influenza, pneumococcal disease, hepatitis B, tetanus, herpes zoster (for patients, ⩾50 years old) and human papillomavirus (HPV) (for women, <26 years). The American Diabetes Association and SBI emphasize the importance of the influenza, tetanus, and hepatitis B vaccines. 4 Other vaccines are also suggested for this population according to the SBI, but it is believed that all were done in childhood, as recommended by the immunization schedule for children in our country. The suggested vaccines are Bacillus Calmette–Guérin (tuberculosis), rotavirus, poliomyelitis (inactivated or oral), hepatitis A, tetraviral (measles, mumps, rubella, chickenpox), yellow fever, and conjugated meningococcal (MenC or ACWY). In Brazil, the vaccines for influenza, pneumonia, triple bacterial (tetanus, diphtheria, and pertussis), hepatitis B, and HPV are considered the recommended vaccines for DM which are made available by both the Unified Health System (SUS), through Basic Health Units (BHUs) and Reference Centers for Special Immunobiologicals, and by the private network. 5 Of the suggested vaccines, all vaccines are also available at SUS.
Despite the availability of these vaccines in the SUS, 5 therefore free of charge for the population, there is no specific vaccination campaign for the population with DM, which is one of the barriers to vaccination. Added to this fact, there is often negligence on the part of physicians, who do not advise the patient on the importance of vaccination 6 or who do not provide the necessary prescription for this (in the case of the pneumococcal vaccine). 7 Some vaccine schedules are difficult to adhere to due to the number of doses and intervals between vaccine doses. 8 Together, these factors reduce vaccination rates in the DM patients and few studies have analyzed these rates in this population. 9 The telephone intervention proved to be an important tool to guide the population about the importance of vaccination, increasing adherence and therefore improving immunization rates. Studies analyzing this approach in our country were performed only in general populations, 10 and there was no study in DM population.
The aim of the study was to evaluate whether orientation through telephone contact for diabetic patients to update their vaccines would be an effective form of intervention.
Methodology
A randomized controlled trial was conducted from December 2018 to November 2020 at the University Hospital of Santa Maria of the Federal University of Santa Maria in Brazil.
Sample size calculation was performed using the online sample size calculator program (surveysystem.com/sscalc.htm), based on a pilot survey carried out by these authors with a population of 60 patients, a margin of error of 5% and 95% confidence interval. The minimum sample number was 52 patients (26 in each group).
All patients admitted to the hospital’s endocrinology service outpatient clinics diagnosed with DM were eligible. Patients who had a medical record of this diagnosis or who were taking insulin or oral antidiabetic medications were considered diabetic. Patients with a diagnosis of gestational DM or patients with any contraindication to vaccination (reported by the patient or recorded in the medical record, such as allergy to any of the components in the formula of any of the vaccines) were excluded.
A total of 228 patients were found to be suitable to participate in the study. As a result of this finding, it was decided to carry out a study with a larger sample size than that intended by the sample calculation performed. Therefore, a total of 228 patients were initially screened. In all, 23 patients refused to participate in the study. In all, 205 patients were randomized using the electronic system software Random Allocation 2.0 into two groups: the intervention group (IG) and control group (CG). There were 102 patients in the IG and 103 patients in the CG (Figure 1). The physicians who regularly follow these subjects in the hospital’s endocrinology Service outpatient clinic were blinded to the allocation group. All telephone contact was carried out by one of the researchers who was not blinded in relation to the allocation of subjects. When telephone contact with subject was not achieved, three more attempts were made daily, for a period of 4 days. Telephone contact (call 1) was made in the year 2019 for all patients included in the study, regardless of the randomization group. In this call, the informed consent form was read and explained, and verbal consent was requested. The subjects were advised that they would receive more than one call regarding vaccination, but the number of calls or the reasons were not informed. Therefore, they were blinded to their allocation group. In the call 1, a questionnaire (Supplemental Material) was applied to characterize and determine the demographic profile of the study population. The questionnaire was not validated, although it has been tested on all 60 subjects from the same pilot survey carried out by the authors used to calculate the sample size. Issues related to vaccination were understood by more than 94% of the pilot survey subjects.
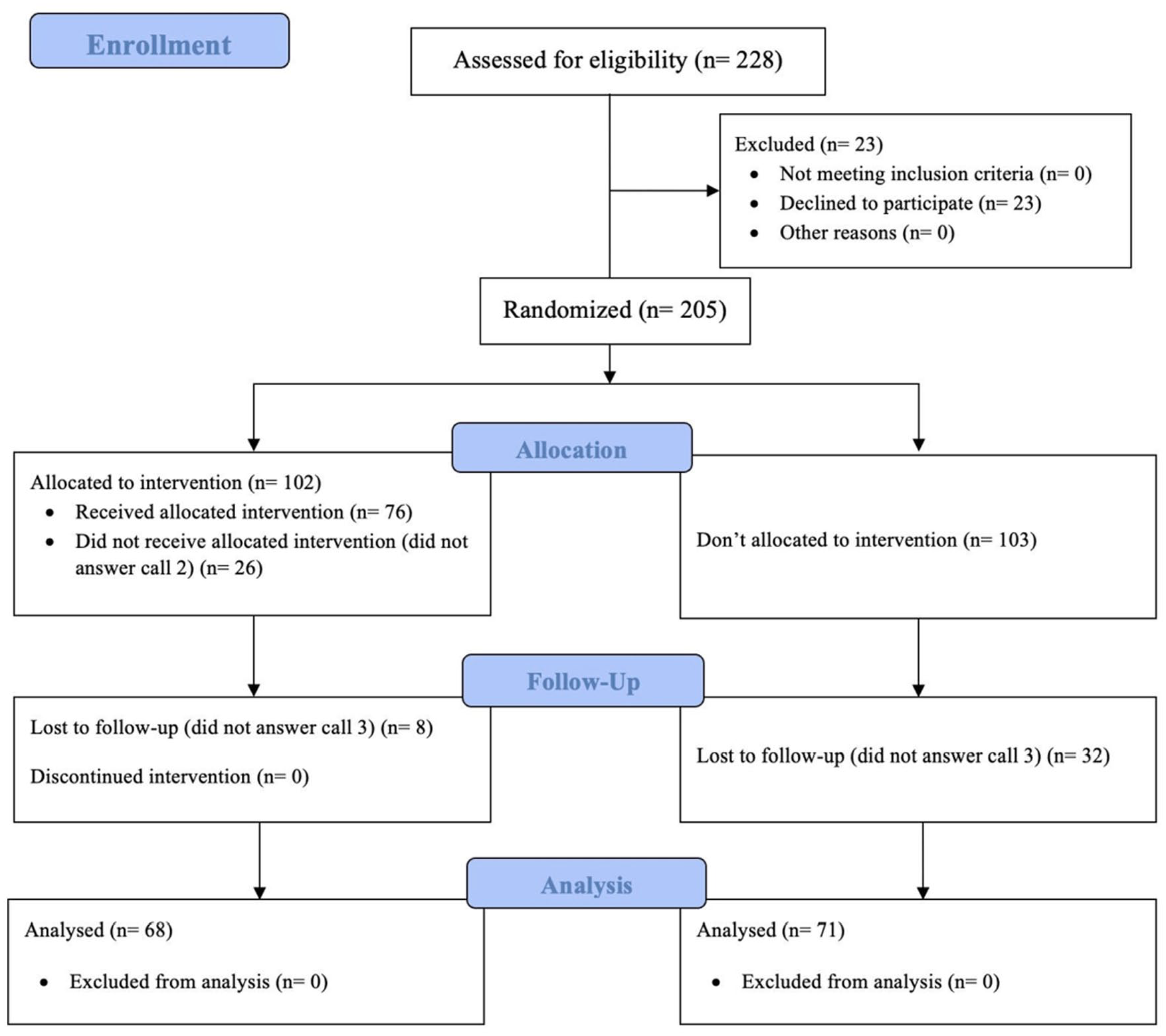
Flowchart of the study.
The following was carried out in the IG:
- Patients in this group received a phone call (call 2) prior to the 2020 influenza vaccine campaign to provide guidance on the need to update influenza, triple bacterial, pneumococcal, and hepatitis B vaccines.
- After the 2020 vaccination campaign, patients in this group received a new phone call (call 3) to assess whether the intervention resulted in increased vaccination rates for the IG.
The following was carried out in the CG:
- Patients in this group did not receive the phone call (call 2) prior to the 2020 influenza vaccine campaign and were not counseled on the need to update their vaccines.
- After the 2020 vaccination campaign, patients in this group also received a new phone call (call 3) to assess vaccination rates in the CG.
All patients included in the study after the inclusion and exclusion criteria received call 1 for demographic data collection and verbal informed consent. There were 102 patients in the IG and 103 patients in the CG. Of the 102 patients in the IG, only 76 of them answered and therefore were contacted (call 2), and in the last call (call 3), which was performed for both groups, 68 of 76 patients in the IG and 71 of 103 patients in the CG answered the phone call and were contacted. The study started with 205 subjects being randomized, as all of them agreed to participate. However, for unknown reasons, some subjects did not answer the remaining calls and had to be excluded from the study.
Outcomes
The vaccination rate in both randomized groups was accessed and the success rate of the intervention on vaccination rate of the IG was verified. It was realized after the 2020 vaccination campaign, by a phone call (call 3).
Statistical analysis
The analysis was performed by protocol. Only patients in whom telephone contact was possible in all phases of the randomized study were analyzed. Those in which there was a loss of follow-up were excluded. Statistical analysis was performed using the SPSS program version 18.0 (SPSS Inc., IBM Corporation, Armonk, NY, USA). The Kolmogorov–Smirnov test
11
was applied to verify the normal distribution of the variables. Normal quantitative variables were presented as means and standard deviations, and non-normal quantitative variables as medians and interquartile ranges. Qualitative variables were presented by their absolute and relative frequencies. Differences in the prevalence (distribution) of genders between the randomized groups were assessed using the chi-square test. ANOVA test was used to verify age differences between genders and the randomized groups and to verify the average difference of consultations carried out over a year with a general practitioner between randomized groups, and a Levene test was used to assess the homogeneity of the analyzed groups. McNemar’s test
12
was used to compare proportions between the randomization groups. The significance level adopted was
Results
Of the 228 patients initially screened, 205 were randomized and 139 were analyzed. In all, 87 (62.6%) patients were female, the overall mean age was 59.17 ± 12.91 years. No differences were observed in the prevalence of genders between randomization groups (
Considering patients comorbidities, 103 (74.1%) had systemic arterial hypertension, 58 (41.7%) patients were dyslipidemic, 42 (30.2%) patients had macrovascular disease, 32 (23%) patients were obese, 17 (12.2%) patients had diabetic retinopathy, 16 (11.5%) patients had chronic kidney disease, and 10 (7.2%) patients had diabetic neuropathy. It was also noteworthy that 85 (61.2%) patients reported having diseases other than those mentioned. In all, 77 (55.4%) subjects were non-smokers, 53 (38.1%) former smokers, and only 9 (6.5%) were smokers.
Regarding the evaluation of the proportion of vaccinated patients and the impacts of the intervention, it was observed that the IG showed a significant increase in the proportion of vaccinated individuals for all vaccines evaluated. In the CG, there was no significant increase for any vaccine. More data about this are shown in Table 1 and in Figures 2–5.
Results of the intervention.


Proportion of individuals vaccinated for pneumonia before and after the intervention: There was a significant increase in the proportion of individuals in the intervention group who received the pneumonia vaccine: 15 (22.1%) individuals were vaccinated before, and 20 (29.4%) individuals were vaccinated after (
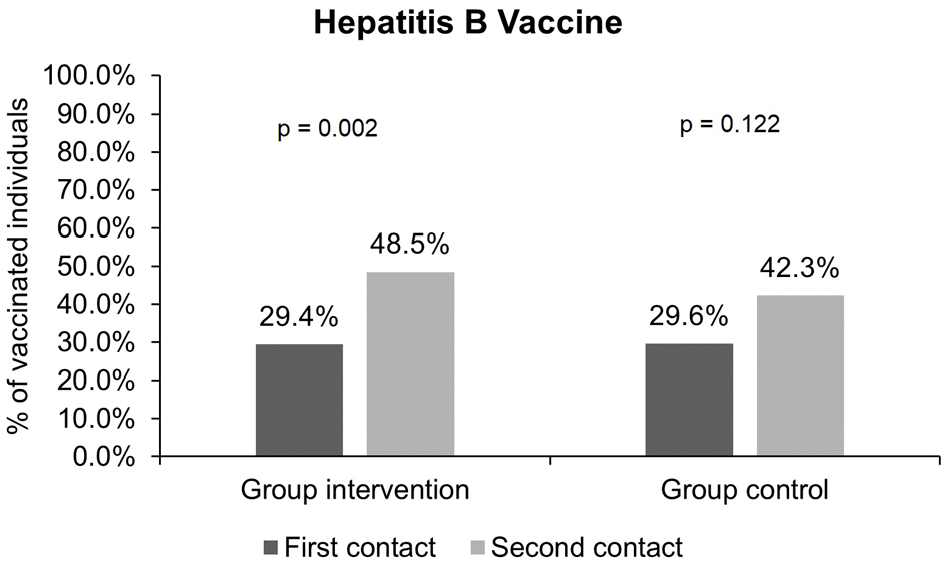
Proportion of individuals vaccinated for hepatitis B before and after the intervention: There was a significant increase in the proportion of individuals in the intervention group who received the hepatitis B vaccine: 20 (29.4%) individuals were vaccinated before, and 33 (48.5%) individuals were vaccinated after (
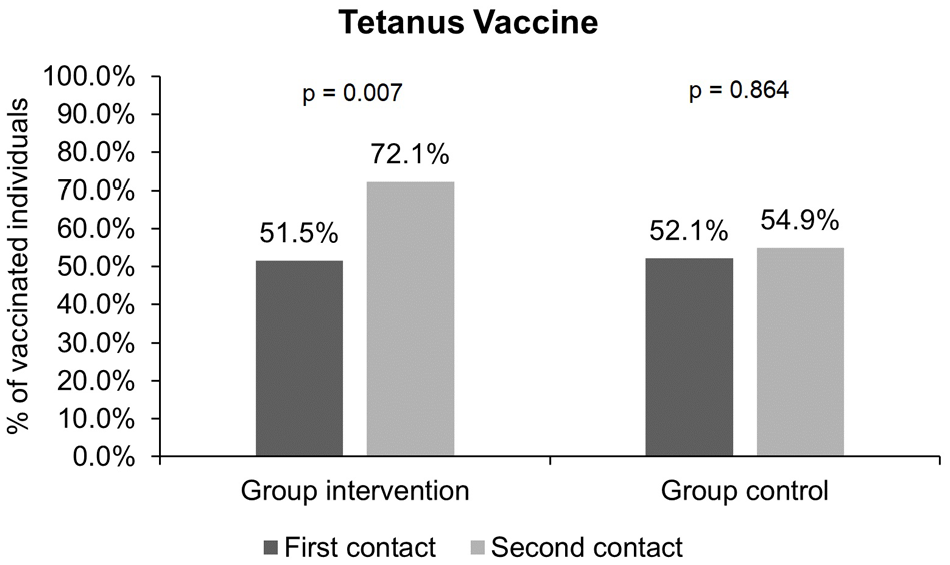
Proportion of individuals vaccinated for tetanus before and after the intervention: There was a significant increase in the proportion of individuals in the intervention group who received the tetanus vaccine: 35 (51.5%) individuals were vaccinated before, and 49 (72.1%) individuals were vaccinated after (

Proportion of individuals vaccinated for influenza before and after the intervention: There was a significant increase in the proportion of individuals in the intervention group who received the influenza vaccine: 54 (79.4%) individuals were vaccinated before, and 61 (89.7%) individuals were vaccinated after (
Tetanus vaccination was the one that most increased in the IG, as 51.5% of this group was vaccinated before and 72.1% was vaccinated after the intervention (
Discussion
It was observed in our study a significant increase in vaccination rates for influenza, pneumonia, hepatitis B, and triple bacterial in the IG. In the CG, there was an increase, but it was not statistically significant.
In Brazil, the vaccines evaluated are freely available from the Brazilian Health System for diabetic patients according to the following schemes. 5
The influenza vaccine must be administered annually during the vaccination campaign period. In the year 2020, the campaign was extended, lasting from March to August, and aimed to vaccinate at least 90% of eligible groups [carriers of chronic diseases (among them DM), elderly, health workers, teachers, pregnant and postpartum women, indigenous people, and children 6 months to 6 years old]. 13 The year 2020 was marked by the COVID-19 pandemic. The topic of vaccination was widely covered in the media due to the urgency of creating an effective and safe vaccine against the virus. It was encouraged worldwide by public agencies to vaccinate for influenza before the winter months to avoid a double pandemic and the collapse of health services.14,15 Even so, in some countries, there has been no increase in influenza vaccination rates. 16 In Rio Grande do Sul, however, while in 2019, 87.13% of people in the priority groups had the influenza vaccine, in 2020, the percentage of vaccinated individuals increased to 93.54%. Evaluating only diabetic patients, there were 81,426 individuals vaccinated in 2019 and 86,387 individuals in 2020. 17 In our study, although the percentage of vaccinated individuals was lower than that of the state, after the intervention (telephone call), the goal set by the Ministry of Health in vaccinating 90% of the priority groups was reached. 9 The influenza vaccine had a higher percentage of vaccinated subjects when compared to other vaccines, both before and after the intervention. The high vaccination rates are probably related to the fact that the influenza vaccine is widely publicized annually by the Ministry of Health through television advertisements, radio advertisements and campaigns in BHUs. It is believed that mass dissemination has been an effective measure to increase vaccine coverage. for influenza. However, even though it is already common knowledge, the intervention performed individually increased the prevalence of vaccinated.
In the Brazilian literature, studies on vaccination in diabetic patients are lacking. A 2015 review conducted in Campinas noted that 65.5% of diabetics were vaccinated against diphtheria/tetanus, 27.8% against pneumococcus, 27.5% against hepatitis B, and 14.5% against influenza. 9 The pneumococcal vaccine is available for adults in two forms: pneumococcal 23 valent (PCV23) and pneumococcal 13 valent (PCV13). 18 For patients who have no previous vaccination, the schedule is started with PCV13 and 2 months later PCV23 is administered. If the patient has already received one dose of PCV23 and has no previous dose of PCV13, an interval of 12 months is recommended for the administration of PCV13 and 5 years for the administration of the second dose of PCV23. 5 In a study conducted in Denmark, individuals with DM under 40 years of age had three times the risk of hospitalization for pneumonia compared to individuals without DM. 19 Pneumonia has financial implications for public services, since treatment often involves the use of antibiotics, exacerbates other pre-existing diseases, and may cause prolonged hospitalizations. 20 In our study, we observed that 29.4% of the patients in the IG was vaccinated after the phone call. A study conducted in São Paulo with diabetic patients found that, in 2003, the frequency of pneumonia vaccination was 17.9% (95% CI: 11.1–27.6), and in 2008, 13.2% (95% CI: 9.4–18.3), with a small increase in 2015, 26.1% (95% CI: 20.1–33.0). 9 Data from the Federal District point out that only 12.1% of patients with DM and pneumonia were vaccinated for pneumonia, 69.7% were not and 18.2% did not know about the existence of the vaccine. 7 The pneumococcal vaccine is dispensed by the SUS with medical prescription and with justification for patients in special situations, such as diabetics, because it is considered an intervention to reduce the morbidity and mortality of these individuals. The State Health Secretariats release the doses to the municipalities through the BHUs according to the demand.7,21 The fact that it requires a medical requisition may be a contributing factor to the low vaccination rates.
The hepatitis B vaccine is available in the public health system in a monovalent form and in a combined pentavalent form (adsorbed diphtheria, tetanus, pertussis,
The tetanus vaccine is available in the form of the adult-type double bacterial vaccine (dT). 22 Three doses of dT are indicated, with an interval of 1–2 months between the first and second doses and 1–2 months between the second and third doses. Patients who have all three doses should be administered boosters every 10 years. 22 The prevalence of vaccination in the IG after the phone call was 72.1%, while in a study with a Brazilian population conducted in 2017, the prevalence was 65.5%. 23
There is a lack of studies in the Brazilian literature that have performed interventions aimed at increasing vaccination coverage in diabetic patients. Evaluating intervention studies in other population subgroups, Krieger et al.,
10
in 2000, compared 102 elderly patients in the IG (consisting of a telephone call and educational brochure for influenza vaccination) with 91 patients in the CG and found a statistical difference in vaccination rates of the counseled patients (OR: 3.33, 95% CI: 1.79–6.22;
The effectiveness of the immune response after vaccination in diabetic patients is also a factor that should be addressed. Patients receiving mRNA vaccine, such as vaccination against SARS-CoV-2, have a significantly altered immune response, a blunted response, due to inadequate glycemic control. 26 Studies suggest that glycemic control is the key to a better immune response in diabetic patients after vaccination27,28 and that patients with changes in glycemic levels may be candidates for a booster dose of vaccines. 28 Short-term glycemic control through continuous glycemic control or chronic control with HbA1c rates < 7% seem to have an important impact on decreasing disease severity rates and infection rates in patients with COVID-19. 29 Marfella et al. 26 suggest that glycemic control after vaccination may improve and even restore the immune response to the vaccine. Although this entire discussion is based on vaccination against COVID-19, it is healthy to believe that the findings of these studies should be transposed and used in the same way in relation to the immune system response of diabetics to other vaccines. Therefore, glycemic control is imperative for an adequate immune response to vaccines.
This study has several limitations. In the year 2020, the COVID-19 pandemic rises and increased, and many activities and in-person consultations were suspended. As a result, in-person consultations and data collection were carried out by telephone calls, and self-report bias may have occurred among the research subjects. Subjects who have not been vaccinated may have confirmed that they have been vaccinated. Possibly because they thought they could be asked why they did not get vaccinated. Therefore, the information collected and presented in this study may be overestimated leading to a potentially important measurement bias. Allocation groups can have much lower rates of adherence to vaccination. However, at the height of the COVID-19 pandemic was taking place in our country, the study was carried out. Only in-person consultations or procedures were realized in our hospital for what was strictly necessary to avoid exposing subjects to unnecessary risks. Ideally, the study should have been conducted in face-to-face medical consultations, checking each patient’s vaccination card. The number of patients in the study was also hampered by the pandemic, and the sample could have been larger in non-pandemic times. Another limitation of the study was the 66 subjects who did not respond to any of the calls. This number, which represents 32.2% of the randomized population, can be considered high and would make a significant difference in the study, if these subjects had been included in their appropriate randomization groups and considered as non-immunized. However, as a significantly larger number of subjects with complete follow-up was achieved, more than twice the sample calculation, the researchers decided to carry out an analysis by protocol. Only subjects who answered all calls were included in the study and follow-up losses (these 66 subjects) were excluded. However, it also has strengths. This is one of the few studies conducted in our country that evaluated vaccination counseling and its impact on the quest to increase vaccination rates in diabetic patients.
Conclusion
Significant increases in vaccination rates in the IG subjects after telephone call proved to be a simple and effective educational and public health action because it only involves a professional for realization. Influenza had the highest rate of people vaccinated among the other vaccines. This demonstrates that telephone intervention combined with an already implemented national vaccination campaign significantly increases immunization rates. The non-significant increase in vaccination rates in CG demonstrates that these subjects did not receive adequate guidance from the professionals who care for them, whether in specialized outpatient clinic or in general medical care. Therefore, an education action for health professionals is imperative, so they realize the importance of providing vaccine orientation in diabetic population and begin to carry it out during routine medical appointments. Added to the guidance during consultation, telephone call would reinforce the need for vaccination in this population.
Supplemental Material
sj-docx-1-smo-10.1177_20503121231161193 – Supplemental material for Strategy to increase vaccination coverage in diabetic patients at a public tertiary university hospital: A randomized controlled trial
Supplemental material, sj-docx-1-smo-10.1177_20503121231161193 for Strategy to increase vaccination coverage in diabetic patients at a public tertiary university hospital: A randomized controlled trial by Giulia Limana Guerra, Fabio Lopes Pedro, Mateus Dornelles Severo, Giorgia Limana Guerra and Tiango Aguiar Ribeiro in SAGE Open Medicine
Footnotes
Author contributions
GLG and GLG have involved in data collection; GLG, FLP, MDS, GLG, and TAR have involved in writing the paper, revising the paper; TAR has involved in data analysis; and TAR and FLP have involved in orientation.
Availability of data and material
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
Ethical approval for this study was obtained from the Institutional Review Board of the Federal University of Santa Maria (UFSM) (approval number/ID 02024018.5.0000.5346). All participants were informed about the purpose of the study and informed consent was read on the first call to patients, doubts were clarified, and patients agreeing to participate provided verbal consent. The study followed the CONSORT 2010 guidelines and was registered in the Brazilian Registry of Clinical Trials (ReBEC) (RBR-92z99d2).
Verbal consent collected via telephone contact was approved by the Research Ethics Committee of our institution (Institutional Review Board of the Federal University of Santa Maria (UFSM)), as the study was carried out at the height of the COVID-19 pandemic. The Ethics Committee agreed that exposing the research subjects to a face-to-face consultation just to carry out the research was too great and an unnecessary risk. That is, coming to a tertiary hospital, which was seeing a great demand from patients in the city and region of the unified health system, patients affected by COVID-19, was a high risk of contracting the infection. Therefore, the Committee authorized the collection of consent for research via telephone, a verbal consent.
Informed consent
Verbal informed consent was obtained from all subjects before the study. All participants were informed about the purpose of the study and informed consent was read on the first call to patients, doubts were clarified, and patients agreeing to participate provided verbal consent. Verbal consent collected via telephone contact was approved by the Research Ethics Committee of our institution (INSTITUTIONAL REVIEW BOARD of the Federal University of Santa Maria (UFSM)), as the study was carried out at the height of the COVID-19 pandemic. The Ethics Committee agreed that exposing the research subjects to a face-to-face consultation just to carry out the research was too great and unnecessary a risk. That is, coming to a tertiary hospital, which was seeing a great demand from patients in the city and region of the unified health system, patients affected by COVID-19, was a high risk of contracting the infection. Therefore, the Committee authorized the collection of consent for research via telephone, a verbal consent.
Trial registration
Brazilian Registry of Clinical Trials (ReBEC) (RBR-92z99d2).
Consent for publication
All authors have reviewed the final version of this article before its submission and authorize submission and publication if approved.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
