Abstract
Introduction
Complex diabetes-related foot wounds are at high risk of infection and subsequent major amputation unless healed expediently. Biodegradable Temporising Matrix (BTM) is a synthetic matrix that facilitates the organisation of the extracellular matrix, resulting in a neodermis layer over these difficult-to-heal areas. The aim of this study was to evaluate the efficacy of using BTM in the reconstruction of challenging diabetic foot wounds.
Methods
Eighteen patients with complex diabetic foot wounds (exposed tendon, fascia, joint, bone), or chronic ulcers at high shear stress locations had BTM applied. Indications for BTM application were high shear stress location (66.6%), exposed bone (16.6%), exposed fascia (5.6%), exposed tendon (5.6%) and chronic non-healing wound (5.6%). The time to complete healing, infection rate and incidence of subsequent wound breakdown was analysed.
Discussion
Thirteen of 18 patients completed the BTM treatment regime with all these patients achieving complete wound healing at a median time of 13 weeks. One patient had partial treatment with BTM and four patients were withdrawn from the study following BTM application. The rate of infection and re-ulceration were both 15.4%.
Conclusion
This is the first prospective cohort pilot study evaluating the use of BTM for complex diabetic foot wounds. BTM demonstrates potential in healing uninfected, non-ischaemic diabetic foot wounds with exposed deep structures and chronic wounds subject to high shear stress. The re-ulceration and infection rates were relatively low for this high-risk population. BTM may also offer promise as an alternative to free flaps.
Lay Summary
The prevalence of diabetes and its complications, including foot ulcers and wounds, have significantly increased worldwide over the last 40 years. Increasingly patients are admitted to hospital for antibiotics, debridements and subsequent amputations from these wounds. Complex diabetes-associated wounds are those at highest risk of these complications or necessitating more extensive, complex operations such as free flaps. These wounds may have exposed deep structures, be at risk of high shear stress or be chronic non-healing wounds.
Temporisers are a type of material which integrates into the wound and promotes in-growth of tissue, ideal for healing over these difficult to heal areas. Biodegradable Temporising Matrix (BTM) is a synthetic temporising matrix which has demonstrated positive outcomes in facilitating healing in burns and plastics wounds, but its effectiveness in diabetic foot wounds has not yet been proven. This is the first prospective cohort pilot study evaluating the use of BTM for complex diabetic foot wounds. BTM demonstrates potential in healing uninfected, non-ischaemic complex diabetic foot wounds and potentially avoiding more complex operations.
Introduction
Over the last four decades, the prevalence of diabetes and its complications, including foot disease, have significantly increased worldwide. 1 An estimated 360 million people will have diabetes by 2030, in stark contrast to the 30 million people with diabetes in 1985.2,3 Approximately 25% of patients with diabetes develop a foot ulcer in their lifetime. 4 Diabetes-related foot ulcers (DFU) are the most common reason for hospitalisation in patients with diabetes, with more than 50% of DFUs becoming infected. 5 Due to the underlying multifactorial barriers to achieving healing in the diabetic foot, an estimated 25% of DFUs remain unhealed at one year.6–8 Our group recently found that diabetes was implicated in 79% of minor amputations and 61% of major amputations performed in Australia and New Zealand between 2010 and 2015. 9 DFU substantially impacts an individual's quality of life with emotional and physical distress combined with economic loss, particularly in patients with chronic DFUs and those who progress to minor and major limb amputations.10,11
For complex diabetes-related foot wounds such as those with exposed tendon, fascia, joint or bone, healing can often be difficult to achieve over these structures. These wounds often develop following extensive debridement for diabetes-related foot infection. Once these structures are exposed, their structural integrity and functionality (e.g. desiccation of tendons) degrades and they become a potential nidus for infection (bone exposed for osteomyelitis) with high risk of rapid spread (e.g. ascending via tendon sheaths). As a result, complex DFUs are 11 times more likely to progress to midfoot or major limb amputation than fully granulated ulcers. 12 Moreover, some patients may be too high risk for anaesthetic or may refuse major limb amputation, and these wounds may persist as chronic wounds.
Chronic DFUs and wounds exposed to high shear stress and recurrent breakdown are also at risk of becoming infected. In diabetes, abnormal extracellular matrix (ECM) remodelling in wound healing occurs secondary to the formation of advanced glycation end products (AGEs). 13 This results in increased ECM degradation, production of non-soluble fibrils and a poorly-organised, yet ‘mature’, ECM network contributing to chronic wound formation. 14 Obtaining tissue coverage over these wounds is essential to decrease the risk of these complications. Split skin graft (SSG) application is often performed to obtain tissue coverage over large wound areas. However, split skin grafts can result in failure of the graft to take and/or may have the tendency to re-ulcerate due to a disorganised wound bed and abnormal biomechanical foot pressure. 15 They are also generally contraindicated for tissue coverage over exposed tendon, joint and bone and also over ulcers exposed to major shear stress or weight-bearing. Free flaps are an option for closure of these wounds, but in complex DFUs in patients with significant comorbidities and those with tibial artery disease, a simpler solution would be advantageous.16,17
Dermal regenerative templates have been used in DFU soft tissue replacement since 1994. 18 Examples of dermal regenerative templates include Integra (Integra Life Sciences Corp., Plainsboro, NJ), AlloPatch Pliable (MTF Biologics, Edison, NJ) and DermACELL (Stryker, Kalamazoo, MI). These templates act as structural support for ECM maturation with cellular response modulation and promote revascularisation and cell regeneration. 19 Some templates also provide a physical barrier for wound tissue coverage and prevent evaporative water loss. 20 They increase the rate of healing in patients with uninfected, non-ischaemic DFUs, as well as reconstructing complex diabetes-related foot wounds.18,21–23 However, the use of acellular dermal matrices is limited in clinical practice due to the relatively high costs of the products. 24 In addition, there is a theoretical increased risk of infection, given the biological composition of these collagen-based dermal matrices. 25 For these reasons there has been an increased interest in the development of inexpensive, synthetic dermal matrices which do not confer the same risk of infection. 26
Innovation
NovoSorb® Biodegradable Temporising Matrix

Illustration of BTM structure. (a) Sealing membrane: temporary, non-biodegradable layer sealing the wound, reducing moisture loss and acting as a barrier to external pathogens. (b) Adhesive bonding layer between sealing membrane and foam. (c) NovoSorb
Clinical problem addressed
BTM was first clinically utilised in 2012 in burns surgery due to its ability to provide temporary wound closure, prevent wound contraction, integrate to prove structural integrity to the dermis and improve cosmesis compared to split skin graft alone. 26 Subsequently it has been demonstrated to be effective at reconstructing complex burn injuries.27,28 Its use has extended to plastic surgery for free flap harvest donor sites, soft tissue trauma injuries and following extensive debridement for necrotising fasciitis.29–33 Following necrotising fasciitis debridement, the wound remains at risk of residual recurrent infection. However, several case series have demonstrated the maintenance of BTM's structural integrity and the ability for underlying infection to be expressed through perforations within the material for these cases.32,33 Often diabetic foot wounds are debrided for infection which can be extensive and may similarly pose a significant risk of residual or recurrent infection.
To date, there have been no studies evaluating the use of BTM in diabetic foot wounds. The aim of this study was to evaluate the efficacy of using BTM in the reconstruction of complex diabetic foot wounds, chronic DFUs and wounds subject to high shear stress.
Materials and methods
Patients
A prospective pilot study was conducted with ethics approval from Central Adelaide Local Health Network Human Research Ethics Committee (HREC/18/CALHN/468) and registered with the Australian and New Zealand Clinical Trial Registry (Registration No. ACTRN12618000608268p). Documented written informed consent was obtained from all participants.
Patients with diabetic foot wounds were enrolled from multidisciplinary diabetic foot clinics at the Royal Adelaide Hospital, Queen Elizabeth Hospital and Lyell McEwin Hospital, or whilst they were admitted under the Vascular Surgery unit at the Royal Adelaide Hospital. Patients were recruited from June 2019 to December 2020.
The inclusion criteria for this study were: adult diabetic patients with complex deep wounds (i.e. with exposed tendon, muscle, fascia, joint or bone exposed); chronic non-healing ulcers and; wounds deemed at risk of not healing due to high shear stress. The exclusion criteria were: arterial insufficiency (absent pedal pulses, or toe pressures <30 mmHg); active local wound infection, or evidence of osteomyelitis on imaging tests and; the inability to attend regular outpatient clinics (e.g. patients living in rural and remote locations).
Patient demographics, wound characteristics, Wound, Ischemia, and foot Infection (WIfI) system score 34 and peri- and post-operative details were collected prospectively. The Society for Vascular Surgery Lower Extremity Guidelines Committee developed the WIfI score which categorises the severity of each of the three major risk factors leading to an overall amputation risk estimation. The main outcome in this study was to determine if wound healing closure could be achieved with BTM. The time to achieve wound healing, subsequent wound breakdown following healing and wound infection were considered secondary outcomes.
Eighteen patients were recruited according to the treating physician's assessment and treated with BTM during the study period. One patient received partial treatment with BTM as the site become infected requiring BTM delamination early but completed follow up. Four patients were subsequently withdrawn from the study following BTM application: two patients were found to have residual osteomyelitis with an unsalvageable limb; one patient's infra-inguinal bypass occluded with subsequent arterial insufficiency and required a ray amputation; and another patient declined to continue participation in the trial due to the COVID-19 pandemic.
Surgical technique
For all patients, a pre-operative bacterial swab culture was obtained from the wound site. All BTM applications were performed in an operating theatre with an anaethetist present. They performed a regional ankle block with the anaesthetic type and dosage determined by the patient's co-morbidities, weight and degree of pre-existing peripheral neuropathy. A single dose of intravenous antibiotic based on microbial sensitivities was given prior to application of BTM. If no bacteria were grown, Cephazolin was given as per hospital protocol. The wound bed was scrubbed with a Medisponge® impregnated with chlorhexidine, followed by povidone-iodine for surgical site preparation.
The wound edge was sharply debrided using a standard surgical blade and the wound bed was lightly debrided using a metal ruler. In cases with visible bone in the wound bed, a drill was used to canulate into the medulla to provide an angiogenic marrow source for subsequent granulation tissue development. The wound was irrigated with povidone-iodine solution. BTM is supplied fenestrated, but additional perforations were made were made to the BTM to allow for easier drainage should a haematoma or fluid collection develop post-operatively. BTM was inset into the wound defect under slight tension using quilting staples or sutures to the wound edges to maximise adherence to the wound bed. For large flat wounds, Acticoat® (Smith and Nephew, Hull, UK) was used, Aquacel Ag® (ConvaTec, Princeton, NJ, USA) was used for wounds with moderate exudate, and negative pressure wound therapy (Vacuum Assisted Closure (VAC® [KCI, San Antonio, TX, USA]) at 75 mmHg for wounds with high exudate. A seven-day course of oral antibiotics was given post-operatively based on the pre-operative bacterial swab. Patients were non-weightbearing on the operated limb for five days to optimise BTM adherence to the wound bed, the wound was reviewed for adequate integration of BTM and patients were subsequently discharged if deemed suitable. To accommodate the appropriate weight-bearing status, orthotic and podiatric input was sought early for suitable footwear to minimise the pressure loading and shear stress over the BTM site.
Dressings were changed by community nursing staff two or three times weekly based on their wound bed characteristics and clinical judgement, and reviews were performed weekly in the vascular outpatient clinic. Cotton-tipped applicators were rolled gently over the BTM towards the fenestrations at each review to remove any fluid that had collected between the BTM and wound bed surface in order to promote BTM integration. BTM integration and vascularisation was assessed by the observation of the development of a red-salmon colour of the BTM and capillary refill on digital pressure.
Delamination occurred once the BTM foam was integrated. If integration failed to occur by six weeks, the BTM remained in situ for a further one–three weeks to allow further time for integration. In the vascular outpatient clinic, the staples were removed and the plastic sealing membrane of BTM was peeled off using forceps. The wound bed was refreshed using a curette to remove any fibrin and biofilm. Depending on the size and depth of the wound, patients were selected to commence either: a suitable conventional dressing regime for smaller (<5 cm2) or deep (≥3 mm depth) wounds until the wound fully healed, or a split skin graft for larger (≥5 cm2) flat wound deficits. SSGs were not used in small wounds due to the increased risk, cost of the operation and inpatient admission required with relatively limited benefit in expedited wound healing in comparison to its use in large wound. Similar to the secondary dressings for BTM, either Acticoat® or Aquacel Ag® were used for the conventional dressings. Split skin grafts proceeded as an elective procedure two weeks following BTM delamination. The skin grafts were harvested at 0.010″–0.012″ thickness using a Zimmer dermatome (Zimmer Surgical, Inc, Dover, OH) meshed at 1:1.5, and dressed with Jelonet (Smith and Nephew, Hull, UK) or with negative pressure wound therapy. Skin grafts were assessed for take at Day 5 post-operatively.
Management of wound infections
Infection at the BTM site was defined by the Society of Vascular Surgery's Wound, Ischemia, and foot Infection (WIfI) classification system. 34 Mild infection was defined by the presence of at least two of the following symptoms: local swelling/induration, erythema 0.5–2 cm around the ulcer, local tenderness/pain, local warmth or purulent discharge. Moderate infection was the presence of local infection (as described in mild infection) with erythema >2 cm, or involving deeper structures (e.g. osteomyelitis, abscess, fasciitis) with no evidence of systemic inflammatory response. Initial antibiotic therapy was based on pre-operative wound swabs, and was appropriately guided by subsequent wound culture and sensitivity results. Mild infection was managed with a course of oral antibiotics. Moderate infection was managed with intravenous antibiotics followed by oral antibiotics once the infection had clinically improved.
Results
This pilot study included 18 patients with a median age of 56 years (range 31–86 years old) and 16 were male (Table 1). All patients had Type II diabetes, with a median HbA1c of 9.5% (range 7.2–13.4%). One patient had Stage 4 chronic kidney disease (CKD) on peritoneal dialysis.
Patient demographics.

CKD, chronic kidney disease; WIfI, wound, ischemia and foot infection.
Initially 18 patients were recruited for the study and had BTM applied, but five patients were had their BTM delaminated removed earlier than study protocol and did not complete the full treatment course. One patient received partial treatment as the BTM site became infected and was delaminated early but completed the follow up period. Four patients were formally withdrawn from the study as they underwent further amputation which was not related to BTM failure or were unable to complete follow up. Two patients were discovered to have underlying osteomyelitis, with the limb unable to be salvaged, and both patients progressed to a below knee amputation. One patient developed an occluded infra-inguinal arterial bypass with arterial insufficiency whilst BTM was in situ requiring revascularisation procedures and subsequent ray amputation. One patient declined to continue participation in the trial due to the COVID-19 pandemic with the policy of their residential aged care facility mandating a period of quarantine following attendance at a hospital outpatient clinic.
The majority of wound defects resulted from diabetic foot infection debridement (77.8%) and amputations (16.7%) with one case of a chronic non-healing ulcer (5.5%) (Table 2). The decision to use BTM was based on the treating surgeon's preference with the indications for BTM being (i) areas at risk for high shear stress and breakdown (66.6%), (ii) exposed bone (16.6%), (iii) exposed tendon (5.6%), (iv) exposed dorsal fascia (Figure 2) (5.6%) and (v) chronic non-healing wounds with tendon on view (5.6%). One third of cases (33.3%) involved exposed fascia, tendon and bone, where direct split skin grafting was inappropriate. For the 13 patients who completed BTM treatment and were not excluded from the study, the BTM sealing membrane remained in situ on the wound bed site for a median time of six weeks (range 2.5–9 weeks). Four of these patients had a VAC® applied whilst BTM was in situ, with the remaining nine patients’ wounds dressed with Acticoat® or Aquacel Ag®.

Subject 2: A 59-year-old male with the dorsal fascia with underlying tendon on view following debridement for a diabetic foot infection (a) initial appearance of the wound following debridement, (b) appearance of the BTM at seven weeks in situ, (c) following BTM delamination at the same review and left to heal by secondary intention with conventional dressings, and (d) six months post BTM delamination.
Wound bed characteristics and BTM details.
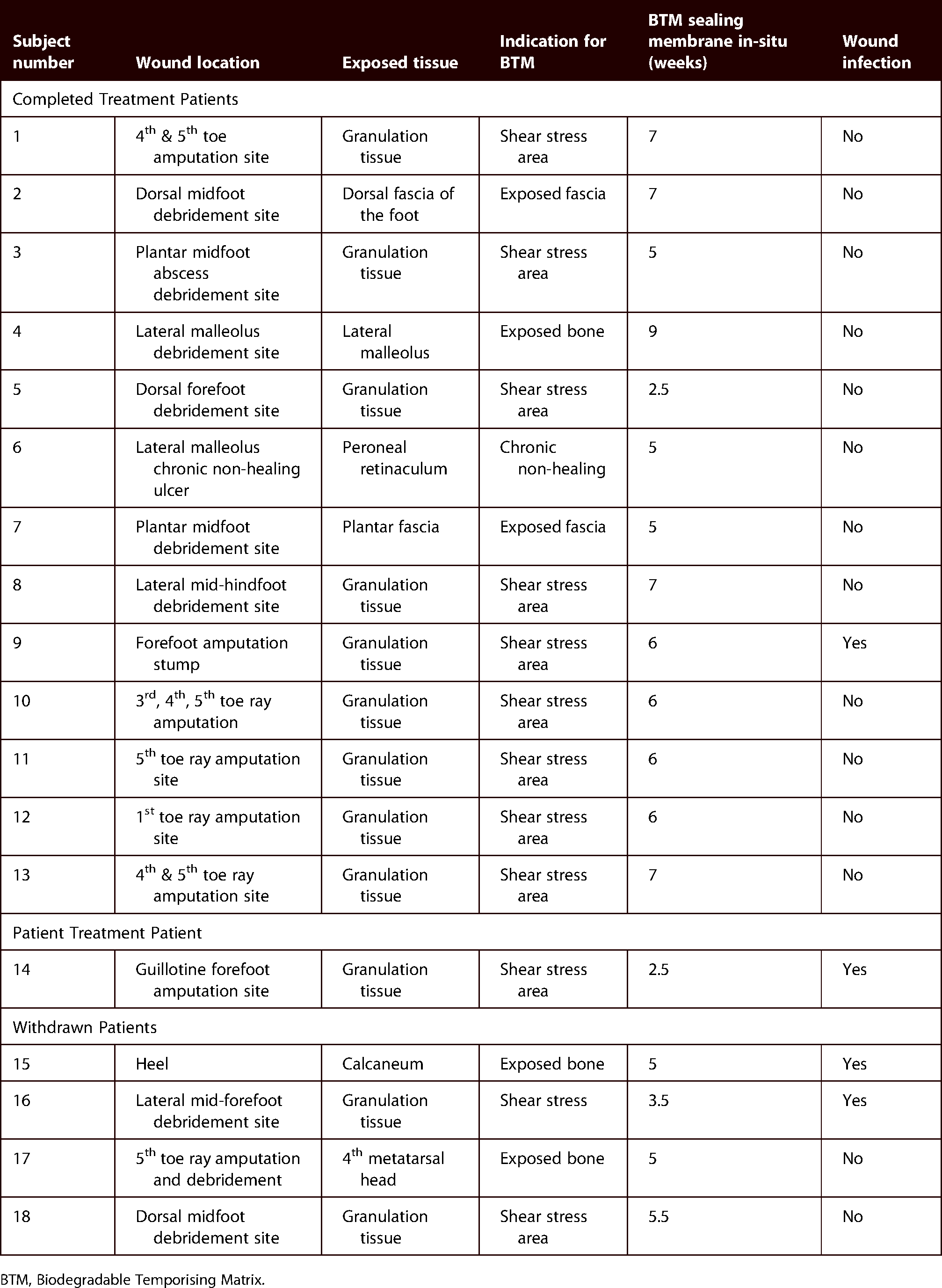
BTM, Biodegradable Temporising Matrix.
All of the patients who completed BTM treatment achieved complete wound healing, with a median duration of 13 weeks (range 8–41 weeks) post BTM application, with 84.6% of patients remaining healed with no subsequent wound breakdown by the end of their follow-up period.
Early delamination occurred for Patient 5 (Figure 3) because the BTM had fully integrated with granulations extending through the perforations in the BTM, indicating it was ready for split skin graft and healed without event. Late delamination occurred for Patient 4 (Figure 4) where their BTM remained in situ for nine weeks for due to a very large and complex wound deficit, which required more time for granulation tissue to develop but successfully proceed to SSG application. 23.1% of patients proceeded to split skin graft application and 76.9% continued with conventional dressings (Table 3).

Subject 5: A 51-year-old male with BTM in situ for two and a half weeks on the dorsum of the foot. (a) Where staples penetrate the BTM, granulation has extended through. The rapid growth of granulation tissue reflected accelerated BTM integration and prompted early delamination. (b) 12 weeks post BTM delamination and nine weeks post SSG application.

Subject 4: A 31-year-old male required extensive debridement over the lateral aspect of his ankle for a diabetic foot infection. (a) The lateral malleolus with the tendinous insertions and ligaments on view, (b) BTM was secured using staples surrounding the lateral malleolus, (c) BTM in situ for nine weeks pre-delamination, and (d) post-delamination, and (e) the result 10 weeks following SSG application.
BTM results.
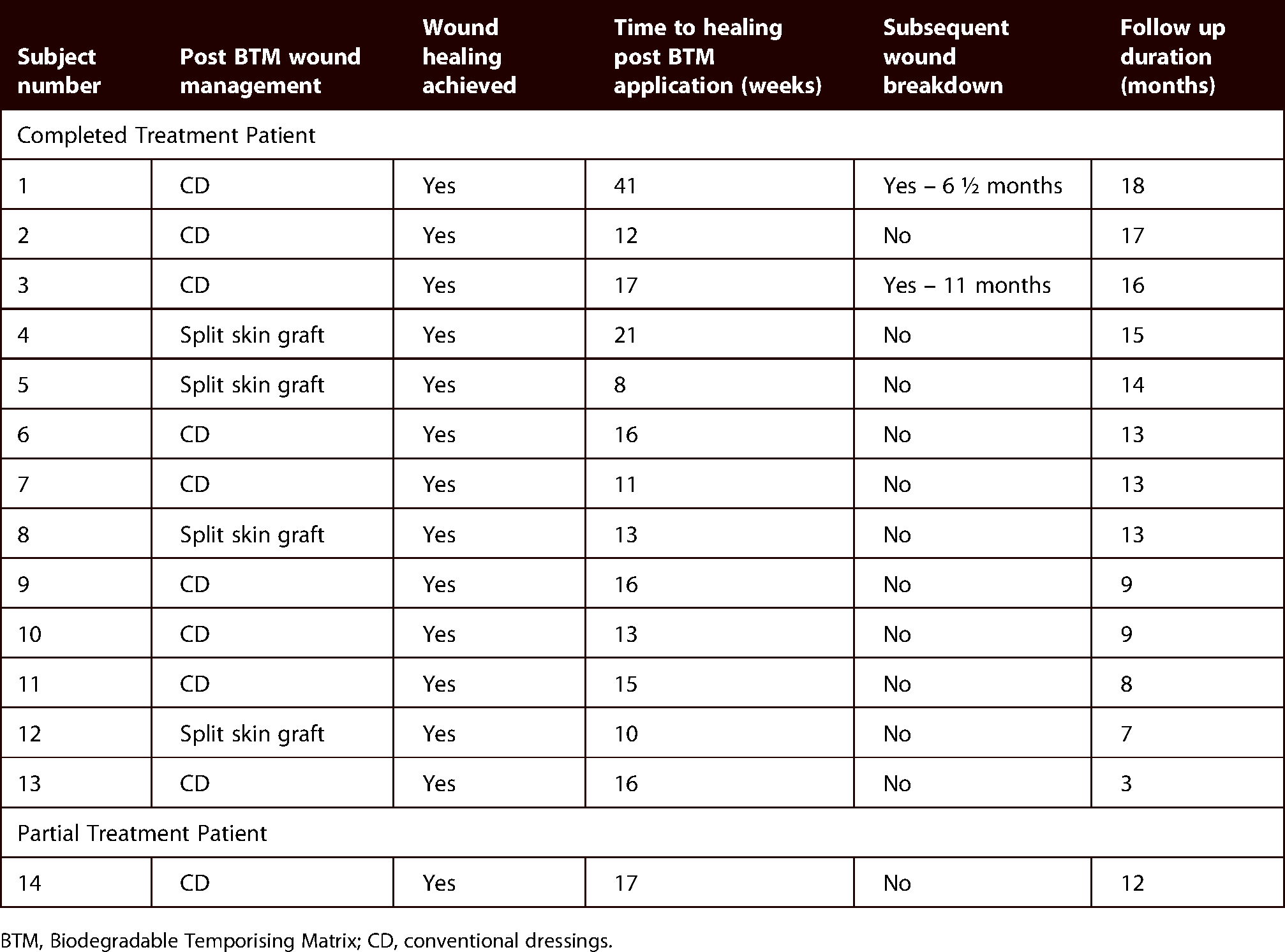
BTM, Biodegradable Temporising Matrix; CD, conventional dressings.
Patient 1 was noted to have a significantly delayed healing (achieved at 41 weeks) and subsequent wound breakdown occurring six and a half months later, which was attributed to the individual's non-compliance with offloading, footwear and wound management. Patient 3 had a Charcot's arthropathy involving with tarsometatarsal joints with midfoot collapse; while the BTM was able to heal the drained abscess and debridement site, the wound re-ulcerated 11 months later at the same site. This was secondary to the ongoing weightbearing bony prominence from the biomechanical deformity despite offloading with orthotics.
Patient 8 healed his wound three months following BTM application. Four months later, he was found to have Magnetic resonance imaging (MRI) established residual metatarsal osteomyelitis following the original mid-hindfoot debridement procedure which erupted in a new ulcer and abscess formation from the underlying infection. As a result, the patient proceeded with a Below knee amputation (BKA).
Patients 9 and 14 developed BTM wound site infections, which both developed 14 days post BTM application. Patient 9 developed a mild infection and completed a 10-day course of oral amoxicillin/clavulanic acid and 14 day course of oral ciprofloxacin targeting a wound swab culture demonstrating a mixed growth of coliforms and Pseudomonas species. The infection resolved and the BTM remained in situ and progressed to integration. Patient 14 (Figure 5) developed a moderate infection with a pre-operative wound swab demonstrating methicillin-resistant Staphylococcus aureus (MRSA). The patient was treated with four days of IV vancomycin and regular wound toilet with a Medisponge® impregnated with chlorhexidine and betadine irrigation. Following this treatment, the infection improved with resolution of the erythema, but the BTM was no longer adherent against the wound bed. The BTM was deemed not salvageable and it was removed in its entirety and deemed as partially completing treatment with BTM in situ for 18 days. The patient completed a 10-day course of oral amoxicillin/clavulanic acid as subsequent wound swab culture grew methicillin-susceptible Staphylococcus aureus (MSSA). Wound healing was achieved by secondary intention.

Subject 14: A 49-year-old male who had a guillotine forefoot amputation for diabetic foot infection sepsis control. (a) Prior to BTM application, (b) 14 days following BTM application, moderate infection developed with erythema extending ≥2 cm from the wound margin treated which was managed with intravenous antibiotics and dressings, and (c) following four days of treatment the BTM become completely non-adherent to the wound bed. (d) The infection has resolved and the wound healed with conventional dressings.
Discussion
Due to the complex pathogenesis of diabetic foot ulcers, they are prone to chronicity, wound breakdown and severe infections that can lead to exposure of deeper structures following debridement. This may result in wound beds which are less robust, ungraftable with direct split skin graft or cause significant contour abnormalities.
We have demonstrated the use of BTM in 18 patients, with 13 patients completing BTM treatment of whom all achieved healing of their diabetic foot wounds. Patients with diabetic foot wounds have variable presentations and co-morbidities contributing to the development of their disease, and our patients in the study reflected this with variable glycaemic control, wound location and wound bed tissue composition.
Patients with extensive tissue loss and exposure of deep structures such as tendon and bone were successfully reconstructed and achieved wound closure. Patient 4 had a lateral malleolus ulcer with tendons and ligaments on view (Figure 4) and was thus at very high risk of infection and major limb amputation. Free flap repair was an option, but BTM offered a good alternative as it is a relatively quick and easy procedure with simple postoperative care. Free flap reconstruction would be considered in young (<50 years old) patients with excellent lower limb perfusion (toe pressures >100mmHg and strong pedal pulses) without significant co-morbidities. Free flaps require longer anaesthetic time which is undesirable in the very comorbid and sick patient. They can also be bulky, making footwear application difficult, even after multiple revisions for thinning, and are contraindicated when significant tibial artery disease is present, whereas BTM offers durable cover with a better contour. BTM would be of particular benefit for rural hospitals, services without access to plastic surgery services or patients in whom free flap construction would be contra-indicated.
The rate of infection whilst BTM was in situ was 15.4%. Jia et al. found that in patients presenting with non-infected diabetic foot ulcers the incidence of subsequent diabetic foot infection was 40.1%. 35 Moreover, patients with deep diabetic foot ulcers were more likely to develop infections. 36 However, our observed low rate of infection was likely significantly impacted by the antibiotics given in the perioperative period for BTM application and frequent wound care and clinic review. For mild infections, we found BTM could remain in situ by expressing infected fluid out through the fenestrations in addition to standard wound infection management. High exudate can cause disruption of the integration of BTM with the wound bed. Potentially if more aggressive exudate management and dressing changes had occurred for Patient 14, it is possible that the BTM may have been salvageable.
Two of the patients (15.4%) re-ulcerated at the BTM site due to non-compliance with offloading and high biomechanical stress as discussed earlier. Pre-clinical trials examining BTM implanted into porcine models have demonstrated that the neodermis is principally composed of fibroblasts, keratinocytes, and blood vessels. 26 As a result, the BTM scaffolding is designed to promote shear force resistance and offers wound stability.26,37 Although there was a relatively low re-ulceration rate in this study, it was unclear if this theoretical advantage of BTM translated into increased robustness of the wound bed. In wounds which healed by secondary intention after BTM delamination, we observed that the wounds epithelised at a relatively quick rate: for example, Patient 2's wound initially had dorsal fascia on view, with BTM in situ for seven weeks, then five weeks following BTM delamination the wound had healed.
It is difficult to evaluate in this study if the speed of integration and healing was affected by Negative Pressure Wound Therapy (NPWT) compared to Acticoat®. This is because the wounds with NPWT applied had greater depth and resultant exudate which was the most significant barrier to BTM integration. Similarly for the risk of infection, the cohort is too small to comment on if there is a statistically significant reduction in infection with NPWT. However, it was observed in the two patients who developed an infection that both had been dressed with Acticoat®.
Limitations
This pilot study is constrained by a number of limitations. In addition to the small number of patients, the lack of a control group denies us the ability to compare BTM to standard care. Ideally, patients would be randomised to standard wound dressing care including NPWT or BTM. However, this is difficult to achieve due to the diverse wound profiles and patient co-morbidities in the diabetic foot ulcer population. Further studies examining the histological changes in the neodermis in patients with diabetic foot wounds would be useful to compare to the pre-clinical trial group. This would verify whether BTM increases the robustness of the wound bed and accelerates wound epithelisation in this patient population.
Eighteen patients were originally recruited to this study with the application of BTM and four patients were subsequently withdrawn. This reflects the challenges of real-world diabetic foot wound management. For digital and forefoot amputations, there needs to be a balance between removing macroscopically infected tissue but also maintaining a functional foot with the risk of leaving residual microscopic osteomyelitis. Up to 60% of patients have been found to have residual osteomyelitis based on culture and/or pathology from samples of clinically uninfected bone taken by the treating surgeon intraoperatively. 38 Revascularisation procedures for tissue loss due to chronic limb-threatening ischaemia are essential to achieve wound healing and for BTM integration. However, graft occlusion is a known complication with infra-inguinal bypass patency rates of 74–90% at three years.39,40
The logistics of outpatient follow-up was a significant barrier to patient recruitment in our study. A large proportion of our patient cohort live in outer metropolitan or rural areas for which the distance, time and cost for them to attend weekly follow up at our metropolitan hospital made their involvement impractical. As we have become more proficient with the use of BTM in this study, it is feasible that we could commence follow-up of these patients via videoconferencing to avoid patient travel for regular review. Our institution is establishing a BTM in DFU registry with potential Quality of Life (QoL) measures and cost-benefit analysis comparing in-person review, telehealth review and overall assessment of BTM in the future.
Conclusion
BTM is a good management adjunct to utilise for wound healing in a variety of diabetic foot wounds. In particular, it has been demonstrated to be a simple reconstructive option for diabetic foot wounds with exposed deep structures for patients who are at risk of limb loss in whom direct split skin grafting would not be suitable or durable. It has low rates of re-ulceration and infection. BTM may be salvageable despite infection. Following BTM application, wound healing can be achieved by either split skin grafting or by secondary intention based on the suitability of the wound.
Footnotes
Acknowledgments
The authors thank Dr Linday Damkat-Thomas and Dr Patrick Coghlan who provided clinical support for the use of NovoSorb® BTM. We express our gratitude to Wendy McInnes and Amanda McDonald for their nursing expertise and Ruth Battersby for data management. PolyNovo Biomaterials Pty Ltd provided the NovoSorb® BTM product in kind. PolyNovo Biomaterials Pty Ltd were not involved in any aspects of the study design or preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JG developed Biodegradable Temporising Matrix (BTM) between 2004 and 2016 in collaboration with PolyNovo Biomaterials Pty Ltd. He has no current affiliation with PolyNovo Pty Ltd, and does not receive any fee for consultation, presentation, travel or promotion. JG is a sharehold in Polynovo Pty Ltd. MW is a medical advisory to PolyNovo Biomaterials Pty Ltd, on an annual retainer and fee for service. He is a shareholder in PolyNovo Pty Ltd. None of the other authors have anything to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Foundation for Surgery Research Scholarship from the Royal Australasian College of Surgeons.
How to cite this article
Kuang B, Guilherme P, Cowled P, Fitridge R, Greenwood J, Wagstaff M and Dawson J. Use of Biodegradable Temporising Matrix (BTM) in the reconstruction of diabetic foot wounds: A pilot study. Scars, Burns & Healing, Volume 8, 2022. DOI: 10.1177/20595131221122272.
