Abstract
The main alterations of osteoarthritis are degenerative lesions of articular cartilage and secondary hyperostosis. Despite the unclear pathogenic mechanism, experimental studies have confirmed that the incidence of osteoarthritis is closely associated with inflammation-related substances. In this study, we explored the significance of serum GPX2 levels in patients with osteoarthritis and the mechanism of GPX2 in the anti-inflammation effects in osteoarthritis. As a result, serum GPX2 level was down-regulated and there was a negative correlation between GPX2 levels and IL-1β levels in patients with osteoarthritis. Over-expression of GPX2 decreased the inflammatory levels and down-regulation of GPX2 increased inflammatory levels in vitro. In contrast, GPX2 combined and regulated STAT3 protein expression. Over-expression of GPX2 promoted ubiquitination-STAT3 protein expression to decrease p-STAT3 protein expression in vitro. The inhibition of STAT3 attenuated the anti-inflammation effects of GPX2 in osteoarthritis. Thus, we proposed that GPX2 was down-regulated in patients with osteoarthritis and GPX2 played vital roles in osteoarthritis for clinical diagnosis or therapy.
Introduction
Osteoarthritis is one of the most common joint diseases affecting human health, which is a chronic and degenerative osteoarthropathy caused by local knee injury, inflammation or chronic strain, subsequently resulting in knee cartilage degeneration and movement disorders. 1 The incidence of osteoarthritis is approximately 10% and 13% in females and males, respectively, which generally occurs in the elderly over 60 years old. 2 The main manifestations are joint pain and inflexibility. Studies have shown that chronic pain is closely correlated with aging and inflammation.3,4 Cytokines are small molecular proteins and peptides that can regulate cellular functions, secreted by cells involved in immune and inflammatory responses.3,5 Some scholars consider that certain types of cytokines are associated with pain, anxiety and fatigue3,6.
Cytokines are widely present in various types of tissues of the body, playing important roles in regulating the occurrence and development of multiple types of diseases at molecular levels. 7 The occurrence and development of osteoarthritis originate from changes in gene expression and stimulation of reactive oxygen species (ROS), thereby changing the metabolism and bone resorption of joint components, which increases the levels of proinflammatory cytokines in the knee joints. 8 Some studies have revealed that the loss of joint function is associated with the high concentration of proinflammatory cytokines in serum and tissues.9,10
Five types of glutathione peroxidase (GPx) (GPx1, GPx2, GPx3, GPx4, and GPx6) have been found in humans, and four types of PGx (GPx1, GPx2, GPx3, and GPx4) have been found in rodents. 11 GPX2, a type of GPX, is selectively expressed in the gastrointestinal tract, liver and breast tissues, which is the main enzyme catalyzing reduction and eliminating ROS of the lipid hydroperoxide ingested by the digestive system.12,13 Relevant studies have indicated that knockdown of GPX2 is more sensitive to H2O2-induced apoptosis and can inhibit the growth of tumor cells.13,14 GPX2 exerts diverse functions, including anti-oxidation, improving immunity and disease resistance, anti-aging, preventing cardiovascular diseases and certain types of cancers, preventing endemic diseases and affecting animal reproduction. 14 Osteoarthritis is known to exert major socioeconomic burden, which is only expected to increase in a rapidly aging society. As there is currently no cure for osteoarthritis, the aim of this study to identify novel therapeutic targets and biomarkers is important. We explored that the significance of serum GPX2 levels in patients with osteoarthritis and the mechanism of GPX2 in the anti-inflammation effects in osteoarthritis.
Material and methods
Study design for clinical research
Demographic characteristics of included patients.

qPCR validation and Microarray experiments
RNA was reverse transcribed into cDNA using a PrimeScript OneStep RT-qPCR kit. cDNA was then transcribed using SuperScript III First strand cDNA system (Invitrogen). The detailed RT-qPCR procedure is described as follows: 95°C for 10 min; followed by 40 cycles of 95°C for 30 s, 55°C for 30 s, 72°C for 15 s, 99°C for 10 s, 59°C for 35 s, and 95°C for 5 s; and then cooled to 4°C. The relative expression levels were calculated with the 2−ΔΔCq method.
Total RNA was extracted from serum samples, and the amount of RNA was quantified by use of NanoDrop 1000. Total RNA of each sample was used for reverse transcription using an Invitrogen SuperScript double stranded cDNA synthesis kit. Double stranded cDNA was executed with a NimbleGen one-color DNA labeling kit and then executed for array hybridization using NimbleGen hybridization system and washing with the NimbleGen wash buffer kit. Axon GenePix 4000B microarray scanner (Molecular Devices) was used for scanning.
Animal experiment for vivo model
All experiments were approved by Animal Care and Use Committee of First Teaching Hospital of Tianjin University. All C57BL/6 mice were fed in a 25 ± 1°C temperature and 50 ± 5% humidity with a 12 h light/dark cyclic schedule and free access to standard diet. The medial anterior meniscotibial ligament of the right knee was cut using a surgical scissor in the patellar tendon in the middle and tendon of the tibial plateau. Number of model mice = 10, number of sham mice = 10, and number of GPX2 = 10. Model mice were injected with 1 μg/3 days/mice for 2 weeks.
Histological analysis
The bone tissues were fixed with 4% paraformaldehyde for 24 h. Tissue samples were embedded in paraffin, and the 5 μm thick sections were stained with Safranin O and Fast Green. Tissue samples were obtained using fluorescence microscopy (Nikon Eclipse TE2000-U, Japan).
Osteoarthritis was assessed using Mankin OA Scoring System: 0–11, changes in articular cartilage structure; 0–8, safranin O staining; 0–2, hypertrophic chondrocytes.
Enzyme-linked immunosorbent assay
The serum IL-1β level was quantified using an IL-1β kits and absorbance were read at a 450 nm wavelength using a microplate reader (Model 3550; Thermo Fisher Scientific, Inc.). Cell levels of TGF-β, TNF-α, IL-1β, IL-6, and IL-18 was quantified using TGF-β, TNF-α, IL-1β, IL-6, and IL-18 kits and absorbance were read at a 450 nm wavelength using a microplate reader (Model 3550; Thermo Fisher Scientific, Inc.).
Cell culture for vitro model and cell transfection
THP-1 cell is purchased from ATCC (Porton Down, Salisbury, UK) and cultured in RPMI 1640 (HyClone; GE Healthcare Life Sciences, UT, USA) containing 10% heat-inactivated fetal calf serum (Gibco; Thermo Fisher Scientific, Inc.). Cells are stimulated by 100 nM PMA for 48 h into macrophage-like cells. GPX2, si-gpx2, and negative mimics were purchased from Genepharma Co., Ltd. (Shanghai, China) and transfected into macrophage-like cells using Lipofectamine 2000 (Invitrogen, USA). After 48 h, macrophage-like cells was induced with LPS (500 ng/mL) for 4 h.
Western blotting
Total proteins of cell samples were by using RIPA lysis buffer and protease inhibitor cocktail (Beijing Solarbio Science Technology Co., Ltd., Beijing, China). The protein concentration was determined by using BCA Kit (Beyotime). 50 μg protein samples were loaded to 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and then transferred onto polyvinyl difluoride (PVDF, Thermo Scientific™, USA) membranes. Membranes was incubated with GPX2, STAT3, p-STAT3, and GAPDH at 4°C overnight after blocking with 5% non-fat milk in tris-buffered saline with 0.1% tween 20 (TBST). The membrane was washed with TBST and then incubated with anti-rabbit secondary antibody (1:5000) for 2 h at room temperature. Immunoreactive bands were visualized using the ECL kit (Thermo Scientific™, USA) and integrated density of the bands was quantified by Quantity One software (Bio-Rad).
Statistical analysis
Data were analyzed using SPSS version 17.0 (SPSS, IBM, Chicago, IL, USA, N = 3). The level of significance (p) was set at 0.05. The two-tailed unpaired Student’s t-tests were used for comparisons of the two groups. One-way analysis of variance followed by Tukey’s post hoc test was used for multiple groups.
Results
Clinical significance of serum GPX2 levels in patients with osteoarthritis
First, we determined the serum levels of GPX2 in OA and healthy population. Compared with healthy normal group, the mRNA expression of GPX2 was decreased while the serum level of IL-1β was increased in patients with late stage of OA (Figure 1(a) and (b)). GPX2 levels were negatively correlated with IL-1β levels in patients with osteoarthritis (Figure 1(c)). ROC analysis showed that the serum GPX2 levels could be used to screen osteoarthritis patients from healthy normal, with an ROC curve area of 0.9167 (Figure 1(d)). In mice with osteoarthritis, both mRNA and protein levels of GPX2 in bone tissue were suppressed (Figure 1(e)–(g)). Clinical significance of serum GPX2 levels in patients with osteoarthritis. MRNA expression and serum levels of GPX2 in patients with osteoarthritis (a and b), there was a negative correlation between GPX2 levels and IL-1β in patients with osteoarthritis (c), ROC analysis (d); GPX2 mRNA and protein expression in bone tissue of mice with osteoarthritis (e and f). Normal, healthy normal group; Osteoarthritis, patients with osteoarthritis; 2 grade OA, patients with 2 grade osteoarthritis; 3 grade OA, patients with 3 grade osteoarthritis; 4 grade OA, patients with 4 grade osteoarthritis. Patients with knee OA (N = 24) and healthy normal (N = 24). Number of mice = 6. **p < 0.01 compared with healthy normal group.
GPX2 regulated inflammation levels in in vitro model of osteoarthritis
over-expression of GPX2 decreased the levels of TGF-β, TNF-α, IL-1β, IL-6, and INF-γ, and induced IL-10 cell levels in vitro model of osteoarthritis (Figure 2(a)–(g)). si-gpx2 plasmid reduced GPX2 expression, promoted the cellular levels of TGF-β, TNF-α, IL-1β, IL-6, and INF-γ, and suppressed the cellular level of IL-10 in in vitro model of osteoarthritis (Figure 2(h)–(n)). GPX2 regulated inflammation levels in vitro model of osteoarthritis. GPX2 expression (a), TGF-β, TNF-α, IL-1β, IL-6, INF-γ, and IL-10 (b––g) by over-expression of GPX2; GPX2 expression (h), TGF-β, TNF-α, IL-1β, IL-6, INF-γ, and IL-10 (i––n) by down-regulation of GPX2; Negative, negative mimics group; GPX2, over-expression of GPX2 group; Si-NS, si-negative mimics group; Si-GPX2, down-regulation of GPX2 group. Number of experiment = 3. **p < 0.01 compared with negative mimics or si-negative mimics group.
GPX2 presented osteoarthritis in mouse model
The study assessed the function of GPX2 in mouse model of osteoarthritis. GPX2 reduced OARSI score, presented bone cell damage, decreased the levels of TGF-β, TNF-α, IL-1β, IL-6 and INF-γ, and increased IL-10 level in mouse model of osteoarthritis (Figure 3(a)–(h)). Moreover, we examined the mechanism of GPX2 in model of osteoarthritis using microarray analysis (Figure 3(i)). GPX2 suppressed the protein expression of p-STAT3 in bone tissue of mice with osteoarthritis (Figure 3(j)–(f)). GPX2 presents osteoarthritis in mice model. OARSI score (a), bone cell damage (HE, b), TGF-β, TNF-α, IL-1β, IL-6, INF-γ and IL-10 levels (c–h) in mice model of osteoarthritis; The mechanism of GPX2 in model of osteoarthritis using microarray analysis (i), p-STAT3 protein expression in bone tissue of mice with osteoarthritis (j and k). OA, mice with osteoarthritis; OA+GPX2, mice of osteoarthritis with GPX2 protein. Number of mice = 6. **p < 0.01 compared with mice with osteoarthritis group.
GPX2 combined and regulated STAT3 protein expression
We evaluated the mechanism of GPX2 on inflammation of osteoarthritis. Over-expression of GPX2 induced the protein expression of GPX2, and suppressed that of p-STAT3 in vitro (Figure 4(a)–(b)). Over-expression of GPX2 suppressed p-STAT3 expression in vitro (Figure 4(c)). IP assay showed that GPX2 combined STAT3 protein expression (Figure 4(d)). Then, over-expression of GPX2 promoted Ubiquitination-STAT3 protein expression in vitro (Figure 4(e)). Down-regulation of GPX2 suppressed the protein expression of GPX2, and induced that of p-STAT3 in vitro (Figure 4(f)–(g)). GPX2 combined and regulated STAT3 protein expression. GPX2 and p-STAT3 protein expression (a and b) by over-expression of GPX2; GPX2 and p-STAT3 expression using immunofluorescence (c), IP (d), Ubiquitination-STAT3 expression (e); GPX2 and p-STAT3 protein expression (f and g) by down-regulation of GPX2. Negative, negative mimics group; GPX2, over-expression of GPX2 group; Si-NS, si-negative mimics group; Si-GPX2, down-regulation of GPX2 group. Number of experiment = 3. **p < 0.01 compared with negative mimics group.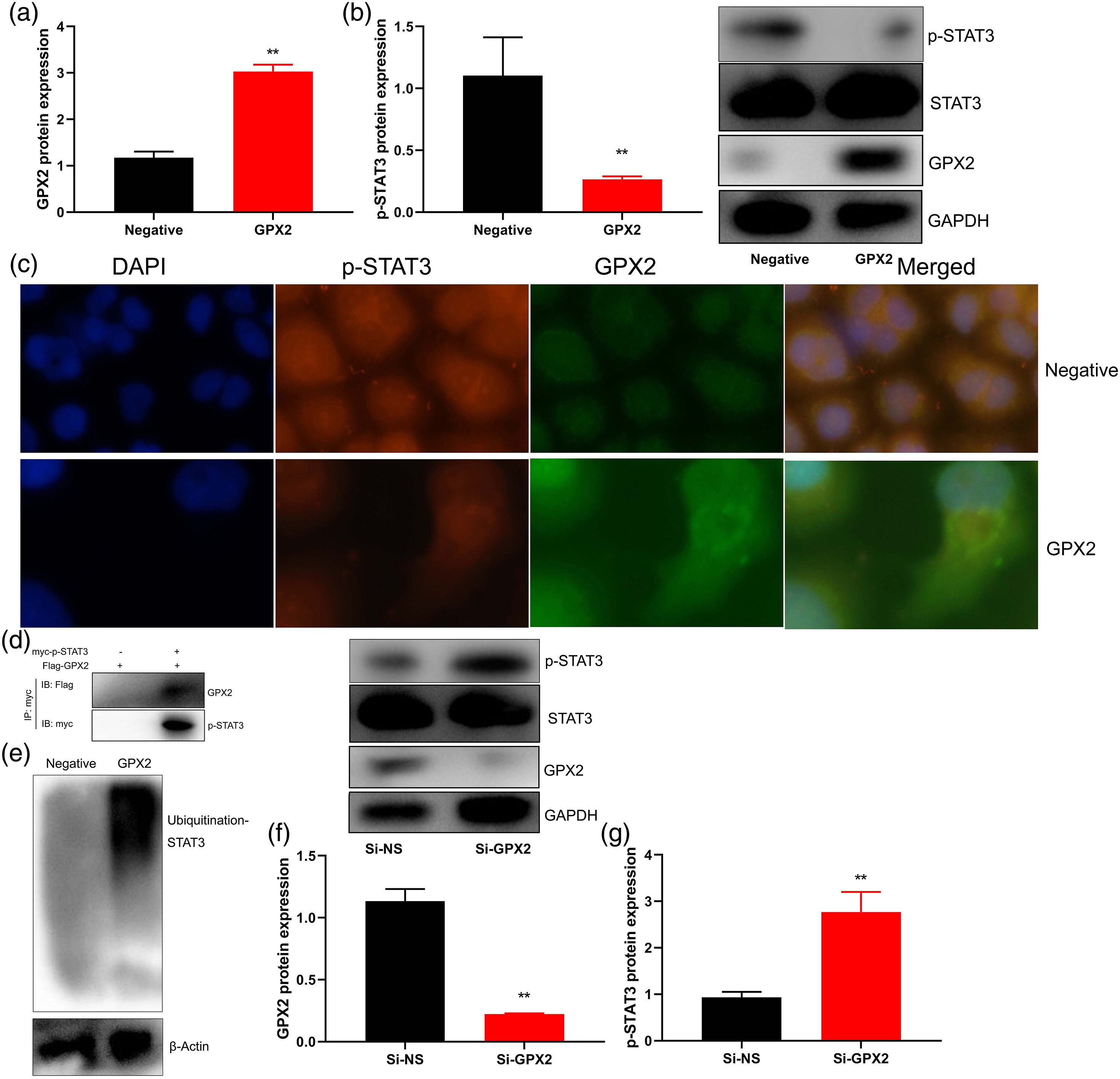
The inhibition of STAT3 reduced the anti-inflammation effects of si-GPX2 in osteoarthritis.
We further evaluated the role of STAT3 in the anti-inflammation effects of GPX2 in osteoarthritis. The administration of STAT3 inhibitor (5 μM, Alantolactone) reduced the anti-inflammation effects of si-GPX2 on the induction of p-STAT3 protein expression, and the inhibition of TGF-β, IL-1β, and IL-6 levels in in vitro model of osteoarthritis (Figure 5). The inhibition of STAT3 reduced the anti-inflammation effects of GPX2 in osteoarthritis. p-STAT3 protein expression (a), TGF-β, TNF-α, IL-1β, IL-6, INF-γ, and IL-10 levels (b, c, d, and e). Negative, negative mimics group; SI-GPX2, down-regulation of GPX2 group; si-GPX2+STAT3i, down-regulation of GPX2 and STAT3 inhibitor group. Number of experiment = 3. **p < 0.01 compared with negative mimics group, ***p < 0.01 compared with down-regulation of GPX2 group.
Discussion
The incidence of osteoarthritis increases with age, which also has an increasing impact on the health and quality of life of middle-aged and elderly population. 15 With the aging of society, the incidence of osteoarthritis is getting higher and higher. 16 According to domestic statistics, the incidence of osteoarthritis is 75% among elderly people over 65 years old. 17 Osteoarthritis is a degenerative disease of articular cartilage, whose etiology mainly includes mechanical and biological factors. 18 The pathological process of osteoarthritis is complicated, and many links have not been completely understood. Here, we showed novel data that mRNA expression and serum levels of GPX2 in patients with osteoarthritis were reduced. Over-expression of GPX2 reduced inflammation levels in in vitro model of osteoarthritis. Krehl et al. showed that GPx2 decreased inflammation and tumor multiplicity. 19 The sample size selected in this study was not calculated, so we only collected 24 patients in this experiment. It was one limitation for this study and we will collect more sample size in further experiment to confirm our conclusion.
The main pathological change of osteoarthritis is degeneration of cartilage, and cartilage degeneration is closely associated with inflammation. 20 Moreover, inflammation can promote the degeneration of cartilage, and degeneration of cartilage can, in turn, stimulate the development of inflammation. 21 Inflammation can destruct chondrocytes and extracellular matrix through cytokines. 22 Cytokines have been currently found to participate in the pathological process of cartilage degeneration by mediating multiple signaling pathways, including STAT3 signaling pathway, MAPK signaling pathway, Wnt/β-catenin signaling pathway, etc. 23 This study showed that GPX2 combined and regulated STAT3 protein expression. Over-expression of GPX2 suppressed p-STAT3 protein expression and promoted Ubiquitination-STAT3 protein expression in vitro model of osteoarthritis. Dittrich et al. reported that GPX2 protects from allergen-induced airway inflammation in mice. 24 This paper only used one cell lines THP-1 cell for vitro model, which is a limitations of the study, and we will used more vitro models in further experiment and execute more models to check our conclusion.
The STAT3 signaling pathway is widely involved in the biological process of the body, playing an important role in regulating cell proliferation, differentiation, apoptosis and inflammation mainly via cytokines to play the corresponding biological effects. 23 The STAT3 signaling pathway is closely associated with the pathogenesis of osteoarthritis. 25 The detection of the expression levels of relevant proteins in the joint fluid, synovium and articular cartilage of osteoarthritis patients can determine its correlation with osteoarthritis.26–28 The study concerning the role of STAT3 signaling pathway in the pathogenesis of osteoarthritis would shed novel lights on the treatment of osteoarthritis. In addition, we found that the inhibition of STAT3 reduced the anti-inflammation effects of si-GPX2 in osteoarthritis. Hiller et al. showed that GPx2 mediated STAT in acute colitis. 29 Therefore, in the future, it will be important to study the signal transduction and pathogenic mechanisms of GPX2/STAT3 in order to find a new therapeutic target for osteoarthritis therapy
Conclusion
In summary, for the first time, we found that the mRNA expression and serum levels of GPX2 in patients with osteoarthritis were down-regulated and over-expression of GPX2 reduced inflammation levels in vitro model of osteoarthritis by the inhibition of STAT3 function. More importantly for clinical significance, by targeting STAT3, GPX2 could be used as a potential biomarker for osteoarthritis patients.
Footnotes
Acknowledgements
The authors thank Experimental center of Tianjin University.
Declaration of conflicting interests
The authors declare no conflicts of interest related to this report.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by China’s Natural Science Foundation (81503671).
Ethics approval
Ethical approval for this study was obtained from the ethics committee of the First Teaching Hospital of Tianjin University of TCM(2FT-526).
Informed consent
Written informed consent was obtained from all subjects before the study.
