Abstract
The lower potency of low dose of carboplatin often requires combination with other drugs to improve its efficacy. Newer and more potent carboplatin-based combination therapies are investigated for treatment. We investigated whether paclitaxel, carboplatin, and Angelica gigas Nakai (AGN) affect viability of H460 cells by MTT assay. Western blot analysis was used to measure the expression of various modulators, such as p-Stat3, p-Akt, and p-Erk. Paclitaxel, carboplatin, and AGN affected the viability of H460 cells. Paclitaxel, carboplatin, and AGN suppressed p-Akt, p-Erk, and p-Stat3 expression. AGN combined with carboplatin significantly decreased c-Jun, HIF-1α, and VEGF levels. AGN combined with carboplatin significantly increased p21 and p27 levels and suppressed cyclin D1 and cyclin E levels. AGN combined with carboplatin-induced apoptosis by increasing Bax and cleavage of caspase and Parp level and by suppressing Bcl-2 level. Our results clearly demonstrate that AGN combined with carboplatin could be a useful compound for treating lung cancer.
Background
The incidence rate of lung cancer has been increasing and one of the leading causes of cancer-related death worldwide. In the United States, there are 526,510 men and women living a history of lung cancer, and an additional 224,390 cases will be diagnosed in 2016. Also, an overall 5-year survival rate of non-small lung cancer is 20% in the worldwide.1–3 The primary types of lung cancer are small-cell lung cancer (SCLC) and non-small cell lung cancer (NSCLC). The majority of lung cancers (85%) are NSCLC which includes squamous cell carcinoma, adenocarcinoma, and large cell carcinoma. 4 NSCLC is a malignant cancer with poor prognosis.5–7
Paclitaxel-based chemotherapy is a standard first-line treatment for NSCLC patients because paclitaxel has been proved to have excellent antitumor properties against lung cancer. 8 Paclitaxel inhibits cell proliferation inducing microtubule polymerization with mitotic cell arrest at the metaphase/anaphase. Paclitaxel is an antimicrotubule agent that stabilizes microtubules by preventing depolymerization. 9 Nevertheless, paclitaxel is accompanied by the appearance of severe side effects such as myelosuppression, cardiotoxicity, and neurotoxicity.10,11 Many international guidelines recommend the use of carboplatin composed of a platinum backbone. 12 However, carboplatin is associated with myelosuppression. 13 Hence, the lower potency of low dose of carboplatin often requires combination with other drugs to improve its efficacy. Newer and more potent carboplatin-based combination therapies are needed for treatment. Recently, chemotherapy of lung cancer had advances. However, further investigation is required to identify novel therapeutic agents to treat lung cancer and reduce side effect. Factors that contribute to lung cancer insensitivity to current chemotherapeutic agents are drug resistance, 14 cigarette smoking, 15 and DeoxyriboNucleic Acid (DNA) topoisomerase II alpha expression. 16 Recently, paclitaxel, erlotinib, bevacizumab–taxol, docetaxel, and gemcitabine, or photodynamic therapy is being considered for combination therapy to combat side effect.17–23 Combination chemotherapy is alternative attempt to combat side effect in cancer cells. Combining two chemotherapeutic agents can result in a more effective response.24,25
Angelica gigas Nakai (AGN) is a biennial or herbaceous perennial plant and grows in relatively moist soil. AGN is known as Dang-gui in Korean. AGN are used in traditional medicine for the treatment of cancer, inflammation, anemia, pain, infection, and articular rheumatism.26–30 AGN has chemical components including decursin, ferulic acid, and nodakenin. Its major compound exerted antitumor activity by apoptosis induction or angiogenesis inhibition in various cancer cells, including prostate, bladder, leukemia, and colon.31–37 These results indicate that AGN may be good candidate for the control of cancer and beneficial in the treatment of human inflammation.
In the present study, we investigated whether carboplatin combination with AGN induces cell cycle arrest and apoptosis in H460. Therefore, AGN could be useful agents for treating lung cancer.
Materials and methods
Reagents and antibodies
AGN was supplied by Han-Poong Pharm Co., Ltd (Jeonju, Republic of Korea). Carboplatin, decursin, Dimethyl sulfoxide (DMSO), 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), and paclitaxel were purchased from Sigma-Aldrich (St. Louis, MO, USA). AGN powder was dissolved in distilled water. Carboplatin powder was dissolved in distilled water. Decursin and paclitaxel powder was dissolved in DMSO. The antibodies against Glyceraldehyde 3-phosphate dehydrogenase (GAPDH), phospho-Akt, phospho-Stat3 (Tyr705), total Akt, and total Stat3 were obtained from Cell Signaling (Danvers, MA, USA). The antibodies against Actin, Bcl-2, phospho-Erk, and total Erk were obtained from Santa Cruz Biotechnology (Dallas, Texas, USA). The tubulin antibody was obtained from Sigma-Aldrich.
Cell culture
H460 human lung cancer cells obtained from the American Type Culture Collection (ATCC) were maintained in RPMI 1640 supplemented with 10% heat-inactivated fetal bovine serum (Invitrogen, Carlsbad, CA, USA) and 100 U/mL antibiotics–antimycotics (Invitrogen). Cells were maintained at 37°C in a humidified incubator with 5% CO2.
Cell viability assay
Cell viability was measured using the MTT assay. Cells were plated in 96-well flat bottom tissue culture plates at a density of 3 × 103 cells/well and incubated for 24 h. Cells were cultured for an additional 24 h with paclitaxel (0.5–20 μg/mL) or carboplatin (1–100 μg/mL) or AGN (50–500 μg/mL). After incubation, MTT reagents (0.5 mg/mL) were added to each well, and the plates were incubated in the dark at 37°C for another 2 h. The medium was removed, formazan was dissolved in 100% DMSO, and the optical density was measured at 570 nm using an enzyme-linked immunosorbent assay (ELISA) plate reader.
Western blot analysis
Cells were harvested, incubated in one volume of lysis buffer (50 mM Tris–Cl, pH 7.4, 1% NP-40, 0.25% sodium deoxycholate, 0.1% Sodium dodecyl sulfate (SDS), 150 mM NaCl, 1 mM ethylenediaminetetraacetic acid (EDTA), and protease inhibitor) for 20 min, and centrifuged at 13,000 r/min and 4°C for 20 min. Aliquots containing 20 μg of protein were separated by SDS-polyacrylamide gel electrophoresis using 8%–12% gels and transferred to nitrocellulose membranes (Protran nitrocellulose membrane, Whatman, UK). Membranes were blocked with 5% nonfat milk and probed with specific primary antibodies. Membranes were then incubated with horseradish peroxidase-conjugated secondary Immunoglobulin G (IgG) antibody (Calbiochem, San Diego, CA, USA) and visualized using an enhanced chemiluminescence detection system (Amersham ECL Kit, Amersham Pharmacia Biotech Inc., Piscataway, NJ, USA).
Reverse transcription polymerase chain reaction (RT-PCR)
RiboNucleic Acid (RNA) was isolated using an Easy-blue RNA Extraction Kit (iNtRON Biotech, Republic of Korea). In brief, we harvested H460 cells and 1 mL of R&A-BLUE solution was added to each. Following this, 200 μL of chloroform was added to the lysate and then vigorously vortexed for 15 s. Then, the lysate was centrifuged at 13,000 r/min for 10 min at 4°C. We then transferred the appropriate volume of the aqueous phase into a clean tube, added 400 μL of isopropanol, and mixed the solution thoroughly by inverting the tube 6–7 times. After centrifuging the tube at 13,000 r/min for 10 min, the supernatant was carefully removed without disturbing the pellet. Then, 1 mL of 75% ethanol was added, and the solution was thoroughly mixed by inverting the tube 4–5 times. The mixture was then centrifuged for 1 min at room temperature, and the supernatant was carefully discarded without disturbing the pellet. Finally, the remaining RNA pellet was dried and then dissolved in 20–50 μL of RNase-free water. The concentration of the isolated RNA was determined using a NanoDrop ND-1000 spectrophotometer (NanoDrop Technologies Inc., Wilmington, USA). We treated DNase to each sample. Two micrograms of total cellular RNA from each sample was reverse-transcribed using a comolementary DNA (cDNA) synthesis kit (TaKaRa, Otsu, Shiga, Japan). Polymerase Chain Reaction (PCR) was conducted in a 20 μL reaction mixture consisting of a DNA template, 10 pM of each gene-specific primer, 10× Taq buffer, 2.5 mM Deoxynucleotide (dNTP) mixture, and 1 unit of Taq DNA polymerase (TaKaRa). PCR was performed using the specific primers listed in Table 1.
The sequence of PCR primers.

Glyceraldehyde 3-phosphate dehydrogenase (GAPDH), Hypoxia-inducible factor 1-alpha (HIF-1α), Polymerase Chain Reaction (PCR) and Vascular endothelial growth factor (VEGF).
Colony formation assay
The H460 cells were plated into six-well culture plates at a density of 3 × 103 cells/well. After 24 h, cells were cultured for an additional 24 h in the absence (control) or presence of carboplatin (10 μg/mL) and/or AGN (200 μg/mL) or decursin (20 μM), and cultured for 10 days to allow colony formation. Colonies were stained with 0.1% crystal violet (Amresco, Solon, OH, USA) in 50% methanol and 10% glacial acetic acid for counting.
Combination effect analysis and statistical analysis
Combinational effects were analyzed with CompuSyn software. 38 All quantitative data derived from this study were analyzed statistically. The results were expressed as the mean ± Standard error of the mean (SEM). Statistical significance at P < 0.05, P < 0.01, and P < 0.001 has been given respective symbols in the figures. All statistical analyses were performed using PRISM software (GraphPad Software Inc., La Jolla, CA, USA).
Results
Effect of AGN, paclitaxel, and carboplatin on H460 cell viability
We investigated whether AGN, paclitaxel, and carboplatin affected the viability of H460 cells. For that purpose, H460 cells were treated with different concentrations of AGN (50, 100, 200, and 500 μg/mL) and paclitaxel (0.5, 1, 2, 5, 10, and 20 μg/mL) and carboplatin (1, 2, 5, 10, 25, 50, and 100 μg/mL) for 24, 48, and 72 h. Cell viability was then measured by MTT assay. We found that AGN (200 and 500 μg/mL) significantly suppressed growth of H460 cells (Figure 1(a)). Also we found that Decursin, a major AGN compound significantly suppressed growth of H460 cells (Figure 1(b)). Also we found that paclitaxel significantly suppressed growth of H460 cells in a low dose (Figure 1(c)). Likewise, we found that carboplatin (50 μg/mL) significantly suppressed growth of H460 cells (Figure 1(d)). We found that distilled water, DMSO, and 30% EtOH did not induce cell death (Figure 1(e)). Taken together, we found that about time-dependent decrease in cell viability observed in response to AGN, Decursin, paclitaxel, and carboplatin.

Effect of AGN, paclitaxel, and carboplatin on H460 cell viability. H460 cells were treated with different concentrations of AGN (a), Decursin (b), paclitaxel (c), carboplatin (d), and DMSO, 30%EtOH, PBS (e) for 24, 48, and 72 h. Cell viability was then measured using the MTT assay. Data are presented as mean ± SEM. *P < 0.05, **P < 0.01, and ***P < 0.001 as compared to non-stimulated cells.
Paclitaxel, carboplatin, and AGN suppressed Erk and Stat3 expression in H460 cells
We investigated whether paclitaxel, carboplatin, and AGN inhibit Akt, Erk, and Stat3 signaling in H460 cells. Activated-Akt, activated-Erk, and phospho-Stat3 increase migration and invasion in cancer cells. Thus, Akt, Erk, and Stat3 expression levels have a critical role in cancer therapy.39–41 For this experiment, we treated H460 cells with paclitaxel (0.5, 2 μg/mL) or carboplatin (10, 50 μg/mL) or AGN (200, 500 μg/mL) and performed western blot analyses. We found that paclitaxel, carboplatin, and AGN decreased the levels of p-Erk and p-Stat3 in H460 cells (Figure 2). High dose of carboplatin and AGN exhibited more effect than low dose. However, high dose of drug exhibits various side effects. Several lung cancer studies demonstrated that carboplatin regimen was associated with a drug resistance and adverse side effects.42,43
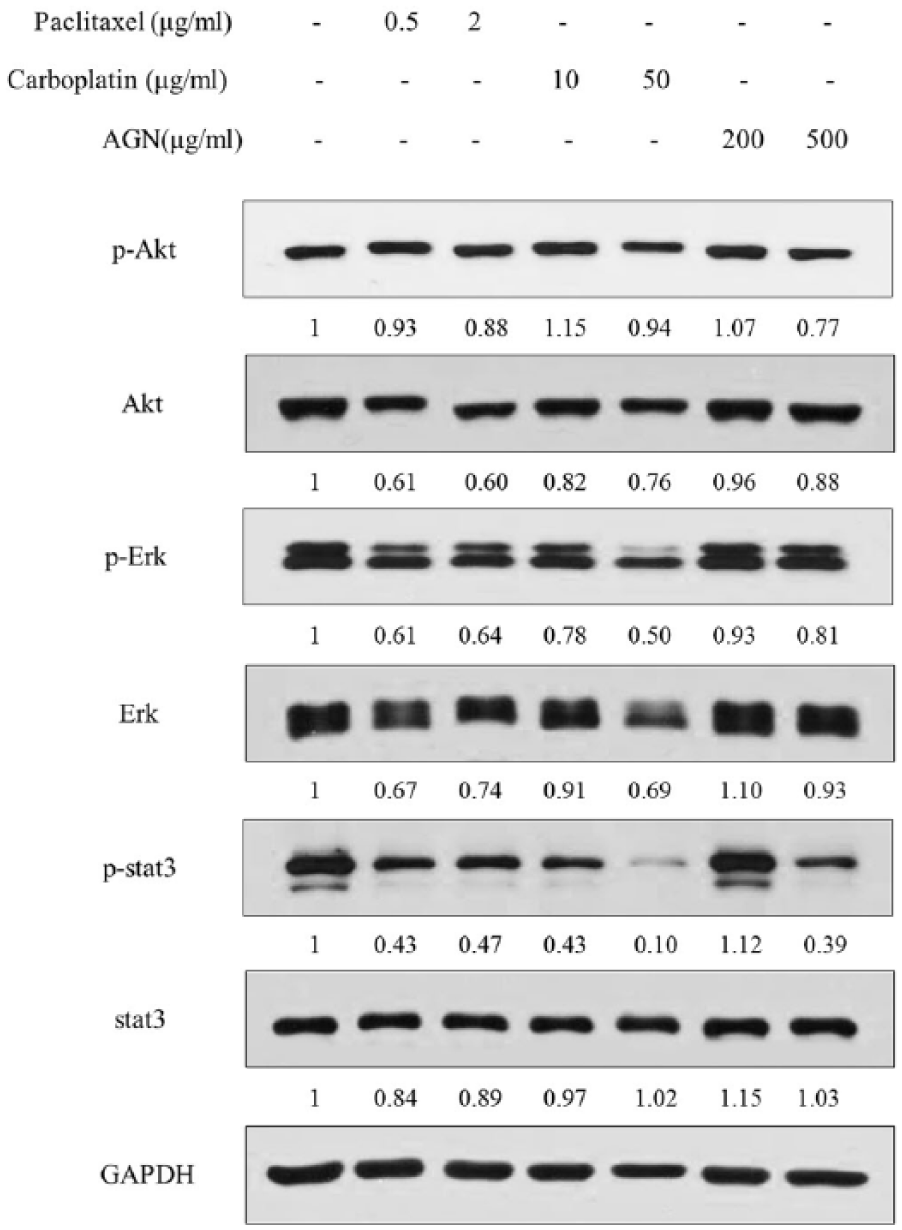
Carboplatin, paclitaxel, and AGN suppressed the expression of p-AKT, p-Stat3 in H460 cancer cells. H460 cells were treated with different concentrations of carboplatin, paclitaxel, and AGN for 24 h. Whole-cell lysates were analyzed by western blot. GAPDH was used as an internal control.
The combined effect of carboplatin and AGN on H460 cell viability
We investigated whether the combination of paclitaxel, carboplatin, and AGN affected the viability of H460 cells. For that purpose, we treated H460 cells with AGN (50, 100, 200 μg/mL) in the presence of paclitaxel (0.5 μg/mL) or carboplatin (10 μg/mL). Cell viability data from MTT assays were then analyzed by CompuSyn software to test a synergistic effect. 38 A combination of AGN with carboplatin showed the synergism. We found that AGN combined with paclitaxel suppressed cell growth, but it does not exhibit synergy effect (Figure 3(a)). We found that AGN combined with carboplatin significantly suppressed cell growth more than carboplatin alone (Figure 3(b)).

Effects of combined treatment with paclitaxel and AGN or carboplatin and AGN on H460 cell viability. H460 cells were treated with different concentrations of AGN, paclitaxel (0.5 μg/mL) (a) or carboplatin (10 μg/mL) (b) for 24, 48, and 72 h. Cell viability was then measured using the MTT assay. The effect is cell viability and CI is combination index. Synergistic effects were analyzed with CompuSyn software. Data are presented as mean ± SEM. *P < 0.05, **P < 0.01, and ***P < 0.001 as compared to non-stimulated cells.
The combination of AGN with carboplatin-inhibited angiogenesis and migration by suppressing Akt, Erk, p38, Stat3, HIF-1α, and VEGF expression in H460 cells
We investigated whether AGN combined with carboplatin-inhibited angiogenesis and migration by suppressing Akt, c-Jun, Erk, p38, Stat3, Hypoxia-inducible factor 1-alpha (HIF-1α), and Vascular endothelial growth factor (VEGF) in H460 cells. The c-Jun and HIF-1 are associated with cell migration, and VEGF is a key regulator of tumor angiogenesis. 44 We found that AGN combined with carboplatin significantly decreased c-Jun, HIF-1α, and VEGF levels by suppressing p-Akt, p-Erk, p-p38, and p-Stat3 levels more than AGN or carboplatin alone (Figure 4(a)).

Combined treatment with carboplatin and AGN suppressed cyclin D1, p-Akt, p-Erk, p-p38, and p-Stat3 expression in H460 cells. H460 cells were treated with carboplatin (10 μg/mL) and AGN (200 μg/mL) for 72 h. Then, whole cell lysates were analyzed by western blot with anti-actin, anti-Akt, anti-cyclin D1, anti-Erk, anti-p38, anti-Stat3, anti-p-Akt, anti-p-Erk, anti-p-p38, and anti-p-Stat3 antibodies. c-Jun, cyclin D1, cyclin E, GAPDH, HIF-1α, p21, p27, and VEGF levels mRNA expression were measured by RT-PCR ((a), (b)). Actin or GAPDH was used as an internal control.
The combination of AGN with carboplatin-induced cell cycle arrest by increasing p21 and p27 levels and suppressing cyclin D1 and cyclin E levels in H460 cells
We investigated whether AGN combined with carboplatin-induced cell cycle arrest by increasing p21 and p27 levels and suppressing cyclin D1 and cyclin E levels in H460 cells. We found that AGN combined with carboplatin significantly increased p21 and p27 levels and suppressed cyclin D1 and cyclin E levels more than AGN or carboplatin (Figure 4(b)).
The combination of AGN with carboplatin-induced apoptosis by increasing Bax and Parp levels and suppressing Bcl-2 level in H460 cells
We investigated whether AGN combined with carboplatin-induced apoptosis by increasing Bax and cleavage of caspase and Parp level and suppressing Bcl-2 level in H460 cells. We found that AGN combined with carboplatin significantly increased Bax, cleavage caspase 9, cleavage caspase 8, cleavage caspase 3, and Parp levels and suppressed Bcl-2 levels more than AGN or carboplatin alone (Figure 5(a) and (b)). Also we investigated whether AGN with carboplatin exhibited cell death by apoptosis. We found that AGN or carboplatin or AGN combined with carboplatin regulated Z-Vad (Figure 5(c)).

Combined treatment with carboplatin and AGN induce apoptosis in H460 cells. H460 cells were treated with carboplatin (10 μg/mL) and AGN (200 μg/mL) for 72 h. Then, whole cell lysates were analyzed by western blot with anti-Bax, anti-Bcl-2, anti-cleavage Caspase 3, anti-cleavage Caspase 8, anti-cleavage caspase 9, anti-Parp, and anti-tubulin antibodies (a). Bax and Bcl-2 mRNA expression was measured by RT-PCR (b). Cell viability was measured using MTT assay (c). Tubulin or GAPDH was used as an internal control. Data are presented as mean ± SEM. *P < 0.05, **P < 0.01, and ***P < 0.001 as compared to non-stimulated cells.
Synergistic effect of carboplatin with AGN or carboplatin with decursin inhibits colony formation in H460 cells
We investigated whether synergistic effect of AGN or Decursin combined with carboplatin inhibited colony formation in H460 (Figure 6). We predicted that synergistic effect of carboplatin with AGN or carboplatin with decursin inhibits colony formation in H460 cells. Synergistic effect of carboplatin with AGN (200 μg/mL) or carboplatin with decursin (20 μM) effectively inhibited the colony formation of H460 cells after 10 days (Figure 7).
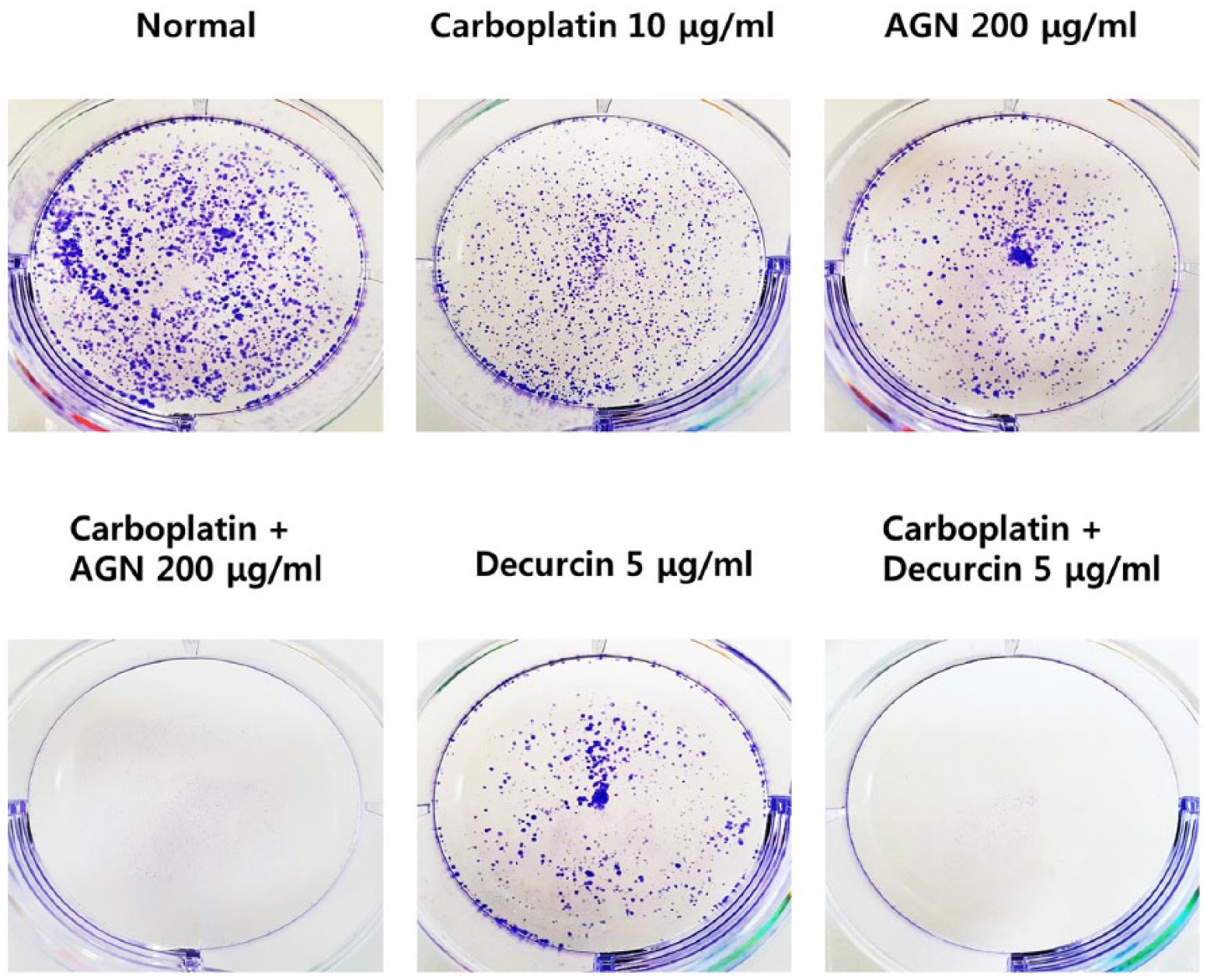
Synergistic effect of carboplatin with AGN or carboplatin with decursin inhibits colony formation in H460 cells. H460 cells were exposed to carboplatin and/or AGN (200 μg/mL) or decursin (20 μM) for 10 days.

Pathway diagram of the mechanism of inhibition of Akt, Erk, and Stat3 activation. Activated of Akt, Mek/Erk Mapk signaling, and Stat3 signaling induce cell cycle. Combined treatment with carboplatin and AGN inhibited Akt, Mek/Erk, and Stat3 activation and suppressed cell cycle.
Discussion
In this study, we found that AGN combined with low dose of carboplatin decreased cell proliferation by inhibiting Akt, Erk, and Stat3 signaling in the H460 human lung cancer cells.
Recently, lung cancer death rates declined due to reduced tobacco use because of increased awareness of the health hazards of smoking. But even yet, lung cancer, including SCLC and NSCLC, has the high incidence of malignancy and is primary reason of tumor-related deaths. 45 The Stat family is composed of Stats 1, 2, 3, 4, 5a, 5b, and 6. The Stat3 signaling pathway activation is involved in the tumor occurrence, cell proliferation, survival invasion, angiogenesis, and metastasis46–49 in many malignant tumor types including liver cancer,50,51 pancreatic cancer,52,53 ovarian cancer, 54 colorectal cancer, 55 and prostate cancer. 56
Major chemical component of AGN is decursin, constituting 3.3%–5.9% of its dry matter. 57 Decursin induces G1 cell cycle arrest and caspase-mediated apoptosis in prostate, leukemia, and breast adenocarcinoma cells.31,58–61 Doxorubicin combination with decursin in AGN induces apoptosis of ovarian cancer cells via blocking the expression of P-glycoprotein. 62
We investigated whether paclitaxel, carboplatin, and AGN affected the viability of H460 cells. We found that paclitaxel, carboplatin, and AGN significantly suppressed cell growth in H460 cells.
Effective therapy of lung cancer inhibited the Stat3 signaling pathway. 63 The breast cancer exhibited an induced upregulation of activated Stat3 in anti-cancer drug resistance. 64 Also, the Akt and Mapk/Erk pathway has been known to play a key role in proliferation and angiogenesis in several cancers. Akt activation is associated with breast cancer and poor clinical outcomes. 65 We investigated whether paclitaxel, carboplatin, and AGN inhibit AKT, Erk, and Stat3 signaling in H460 cells. We found that paclitaxel, carboplatin, and AGN decreased the levels of p-Erk and p-Stat3 in H460 cells. High dose of carboplatin and AGN exhibited more effect than low dose. However, high dose of drug exhibited various side effects such as anti-cancer drug resistance and high toxicity.42,43,64 Therefore, we investigated whether the combination of low dose of paclitaxel, low dose of carboplatin, and low dose of AGN affected the viability of H460 cells. We found that AGN combined with paclitaxel suppressed cell growth, but it does not exhibit synergy effect. We found that AGN combined with carboplatin significantly suppressed cell growth more than AGN or carboplatin alone. We investigated whether AGN combined with carboplatin inhibited angiogenesis and migration by suppressing Akt, c-Jun, Erk, p38, Stat3, HIF-1α, and VEGF in H460 cells. We found that AGN combined with carboplatin significantly decreased c-Jun, HIF-1α, and VEGF levels by suppressing p-Akt, p-Erk, p-p38, and p-Stat3 levels more than AGN or carboplatin alone. Also, we found that AGN combined with carboplatin significantly induced cell cycle arrest via increasing p21 and p27 levels and suppressing cyclin D1 and cyclin E levels. We found that AGN combined with carboplatin-induced apoptosis by increasing Bax and cleavage of caspase and Parp level and suppressing Bcl-2 level in H460 cells. Also, these cell death was accompanied by cell inhibition of colony formation. Because these AKT, Erk, and Stat3 pathways are implicated in angiogenesis and migration in cancer, AGN combined with carboplatin appears to inhibit these pathways and induced apoptosis (Figure 7). Taken together, our results suggest that AGN combined with carboplatin inhibited AKT, Erk, and Stat3 in H460 cell, thereby suppressing the angiogenesis and migration. Our present study clearly demonstrates that AGN combined with carboplatin inhibited Akt, Erk, and Stat3 activity more than AGN or carboplatin alone in H460 cells. Also AGN combined with carboplatin-induced apoptosis in H460 cells. These results clearly demonstrate that AGN combined with carboplatin could be used as a compound for treating human lung cancer.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This research was supported by a grant from the Korean Medicine R&D Project of the Ministry of Health and Welfare (HI11C2110, B110043, HI12C1889, B120014, and HI14D1109). This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (grant number 2015R1D1A4A01019557).
