Abstract
There is a significant overlap between intestinal lymphoproliferative disorders (LPDs) and inflammatory conditions of the intestine, including inflammatory bowel disease (IBD), in clinical, endoscopic, or histologic appearance, leading to diagnostic challenges. We report two cases of monomorphic epitheliotropic intestinal T-cell lymphoma (MEITL) that were initially considered to be ulcerative colitis (UC) and lymphocytic colitis, respectively. Both patients presented with diarrhea and abdominal pain. Colonoscopy revealed hyperemia and scattered shallow ulcerations in the colon, without obvious mass. Microscopically, the first case exhibited features of active chronic colitis. The second case exhibited features mimicking lymphocytic colitis and ileitis. Correct diagnosis of lymphoma was established when the cases were received as consultation, with additional ancillary tests performed. Being familiar with the endoscopic features and unusual histologic patterns of MEITL described here is critical for prompt diagnosis and timely treatment, which may be conductive to a better prognosis.
Keywords
Introduction
Monomorphic epitheliotropic intestinal T-cell lymphoma (MEITL), previously known as Type II enteropathy-associated T-cell lymphoma (EATL), is a rare and aggressive peripheral T-cell lymphoma (PTCL) that arises from intestinal intraepithelial T lymphocytes. 1 It predominantly affects Asian populations and is not associated with celiac disease. MEITL mainly involves the small bowel, particularly jejunum and ileum. Rarely, the duodenum, stomach, colon, and extraintestinal sites may be involved as well. 2 Endoscopically, the lesions are characterized by multiple raised and ulcerative masses or nodules. Microscopically, the tumor typically consisted of dense monomorphic, small- to medium-sized lymphocytes, with pale cytoplasm, and round and hyperchromatic nuclei, with epithelial tropism. It usually lacks an inflammatory background or necrosis. 3 There is no villous atrophy in small bowel mucosa away from the tumor. Immunohistochemically, the tumor cells are CD3+, CD5–, CD4–, CD8+, CD56+, CD103+/–, CD30–, MATK+, and EBER–; about 80% of cases show T-cell receptor (TCR)-γ and TCR-δ rearrangement. 4
Although the diagnostic features are straightforward, most patients are not diagnosed until they reach an advanced stage, thus the prognosis is poor, given the aggressiveness of the disease. Early recognition of clinical and pathologic features may help the diagnosis to be made earlier. In this report, we describe features of MEITL that may tend to overlap with those of intestinal inflammatory disorders including IBD, and potentially lead to misdiagnosis, based on two challenging cases. In both cases, the clinical and endoscopic findings were not highly suggestive of lymphoma. Features that may help in the correct diagnosis of this rare type of lymphoma will be discussed in detail, which may be helpful for similar cases in the future.
Materials and methods
The clinical history, patient demographic information, and clinical examination including endoscopic findings and related tests were obtained from the patients’ charts. Slides were examined by two expert pathologists. Immunohistochemical stains were performed with antibodies against CD20, CD79a, CD3, CD2, CD4, CD5, CD8, CD56, CD30, CD10, Ki-67, Granzyme B, CK, CK20, and c-Myc which were obtained from ZSGB-BIO or LEICA BIOSYSTEMS, China. All the antibodies were in prediluted form and immunohistochemical (IHC) analysis was performed using an automated machine Leica Bond Max. In situ hybridization of EBV early RNA (EBER) was also performed via Leica Bond Max. Molecular analysis for rearrangement of the TCR in case 2 was performed with the polymerase chain reaction (PCR) according to the established protocols.
Report of cases and workup
Case 1
The patient was a 58-year-old man who presented with a 3-month history of abdominal pain, diarrhea, and weight loss at an outside hospital (OSH). He also had urinary frequency, urgency, dysuria, and other discomforts. A colonoscopy performed at OSH revealed prominent congestion and edema, loss of vascular pattern, and multiple moth-eaten ulcers with purulent exudates involving the entire colon. Multiple biopsies were obtained and were interpreted as ulcerative pan-colitis. There was no response to treatment for ulcerative colitis (UC). Two months later, he was transferred to our hospital with the working diagnosis of UC and renal disease.
A repeat colonoscopy showed no significant abnormality of the ileum, but scattered irregular ulcers with exudates throughout the colon. The background colonic mucosa was granular with “fuzzy” vasculature. The clinical impression was UC. Rectal biopsies were obtained.
Microscopically, the biopsies showed mucosal architectural distortion with ulcerations. There was infiltration of the surface and crypt epithelium, and the lamina propria, by small- to medium-sized atypical lymphocytes. These cells had scant cytoplasm, round or slightly irregular nuclei, with coarse chromatin pattern, and inconspicuous nucleoli. Immunohistochemically, the cells were CD2(+), CD3(+), CD30(–), CD4(–), CD5(–), CD56(+), CD8(+), granzyme B(+), and negative for CD20. The Ki-67 index was 70%. Immunostaining with cytokeratin revealed lymphoepithelial lesions. In situ hybridization for EBER was negative. A diagnosis of MEITL was thus rendered (Figure 1). Review of the original biopsies from the OSH revealed crypt loss and increased mononuclear cell infiltration in the lamina propria. Some crypt changes initially interpreted as cryptitis in fact were intraepithelial lymphocytes instead of neutrophils, similar to the findings from the repeat biopsy. Due to the new diagnosis of MEITL, a positron emission tomography (PET) scan was performed, which showed irregular thickening of the colon and rectum. Since the patient also complained of discomfort in the upper abdomen, an esophagogastroduodenoscopy (EGD) was performed, which revealed multiple erosions in the stomach, with the largest area measuring 1.5 cm in diameter. A gastric body mucosa biopsy showed obscured fundic glands with intraepithelial infiltration by small- to medium-sized lymphoid cells (Figure 2). The patient was treated with one course of Cyclophosphamide, Doxorubicin, Vincristine and Prednisolone (CHOP) regimen with initial improvement of symptoms, but later developed bone marrow suppression and died.
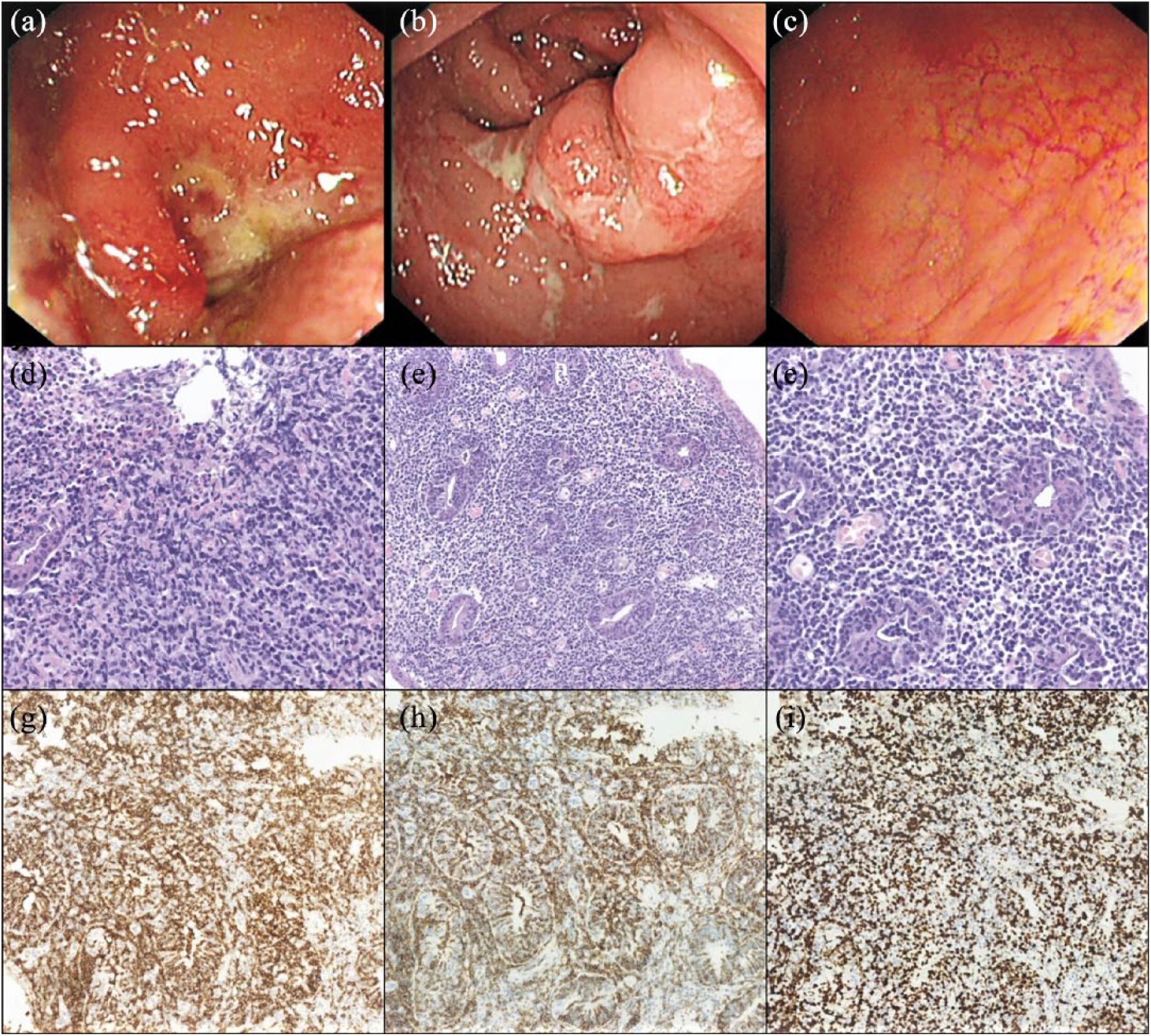
Colonoscopy with biopsies for case 1. (a) Ulcers in the descending colon. (b) Ulcers in the sigmoid. (c) Mild congestion and edema in the rectum. (d) In contrast to the relatively mild endoscopic findings of the rectum, histologically the infiltration is marked by dense atypical lymphoid cells with loss of crypts. (e) Low power view of the rectal lymphomatous infiltrate. (f) Higher power view of (e). Immunohistochemically the tumor cells are positive for (g) CD8, (h) CD56, (i) with high Ki-67 index.

Esophagogastroduodenoscopy and biopsies of case 1. (a) Erosions in the gastric body. (b) Narrow band image of the gastric body. (c) Biopsy of the body mucosa showing marked atrophy. (d) High power view showing intraepithelial infiltrate by lymphomatous cells.
Case 2
The patient was a 64-year-old woman who presented with a 5-year history of abdominal pain and diarrhea. The explosive diarrhea and abdominal cramps were self-limited at first. An EGD at an OSH revealed normal gastric mucosa negative for
The patient underwent repeat in-house EGD and colonoscopy. It showed normal esophageal, gastric body, and antral mucosa. Diffuse mucosal changes characterized by hypertrophy, congestion, and granularity were found in the duodenal bulb and in the second portion. The terminal ileum was normal, and a localized area of mildly erythematous mucosa was seen in the sigmoid colon. Histologic examination of the biopsies revealed intraepithelial lymphocytosis (IEL) involving the terminal ileal mucosa. There is extensive intraepithelial and lamina propria infiltration of monotonous medium-sized lymphoid cells with pale cytoplasm, in both random colon biopsies and the sigmoid biopsies (Figure 3). Immunohistochemically, the cells are positive for CD3, CD8, and CD56, but negative for CD20 and c-Myc. Stain for CK20 highlighted extensive epitheliotropism of lymphocytes. The findings are consistent with persistent MEITL.

Histologic findings in repeated biopsies of case 2. Infiltration by atypical lymphoid cells in (a) stomach, (b) duodenum, and (c) rectum. (d) Higher power view of (c), showing intraepithelial infiltration by monotonous medium-sized lymphoid cells with pale cytoplasm. (e) Immunostain for CK20 highlighting lymphoepithelial lesion. In addition, the tumor cells are strongly positive for (f) CD3, (g) CD8, and (h) CD56.
The patient received chemotherapy but with no improvement and developed severe complications such as fever and hyponatremia. She passed away a month later.
Discussion
MEITL is an aggressive form of T-cell lymphoma with poor prognosis. Even with new development in the understanding of the pathogenesis, such as findings of activation of the JAK-STAT and G-protein-coupled receptor signaling pathways, 5 there is still no efficient therapeutic intervention except for stem cell transplant.
Few publications are available regarding the endoscopic features of MEITL. In a report of four cases, innumerable fine mucosal granules and diffuse thickening of the mucosa with semicircular shallow ulcerations were described in the small bowel and nonspecific changes in colon. 6 The endoscopic appearance in our first case differed from the above, with scattered shallow ulcerations with superficial exudates throughout the colon, within a background of rough mucosa with loss of vascular pattern. This pattern likely caused the initial consideration of UC. As for case 2, the in-house endoscopy revealed essentially normal gastric and colonic mucosa, except for erythematous changes in the sigmoid. Combined with the microscopic findings of increased IEL, it is understandable why the initial diagnosis of “lymphocytic colitis and ileitis” was made. Although focal shallow ulcerations may have suggested an alternate diagnosis, erosions or ulcerations can occur in some patients with microscopic colitis who took nonsteroidal anti-inflammatory drugs (NSAIDs). 7
Recognizing IEL is crucial for diagnosing a variety of gastrointestinal disorders such as celiac disease, microscopic colitis, celiac-like drug-induced enteropathy, EATL and MEITL.8,9 Finding more than 20 intraepithelial lymphocytes per 100 epithelial cells is accepted as the minimum criterion for abnormal IEL. 7 Lymphoma with IELs usually have much greater density than inflammatory disorders. In case 1, IELs involving the crypts were mistakenly seen as “cryptitis.” Combined with the marked crypt architectural abnormalities and loss, and inflammatory infiltration in lamina propria, a diagnosis of UC was erroneously made. In case 2, although IELs were correctly identified, the density of infiltration far exceeding that seen in lymphocytic colitis was not appreciated, not mentioning the evident cytologic atypia. Another pitfall encountered in case 2 was the distinction between MEITL and PTCL not otherwise specified (PTCL-NOS) with cytotoxic phenotype. The most important features distinguishing MEITL from other types of T-cell lymphoma are the monomorphic cell shapes, epitheliotropic pattern, and immunopositivity for CD8 and CD56.
EATL, intestinal NK/T-cell lymphoma, and indolent T-cell lymphoproliferative disease should be considered in differential diagnosis of MEITL. Although no association has been established between MEITL and celiac disease, recent reports have suggested that a variant of celiac disease may precede rare cases of MEITL. 10 EBER is of most help in differentiating NK/T-cell lymphoma from MEITL, as it is positive in the former and negative in the latter. As for indolent T-cell lymphoproliferative disorder (LPD), it exhibits a chronic indolent clinical course. And the infiltrating lymphocytes are mature small lymphocytes negative for CD56 and are nondestructive, with a proliferative index of 5%–10%. 11
In summary, we described two cases of MEITL that were incorrectly diagnosed initially due to a combination of inexperience in endoscopic and histologic features of the disease, and its overlap with inflammatory disorders in endoscopic and microscopic manifestations. Careful review of endoscopic images and biopsy slides with a high level of suspicion for MEITL is critical when encountering biopsies that show findings similar to UC or microscopic colitis, but with unusually prominent IEL. Repeat biopsy may be necessary in some cases, including biopsies from the duodenum and stomach.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
