Abstract
Tuberculous wound therapy is a major challenge in clinical practice, due to the protracted disease course, high recurrence rate, and an unclear pathogenesis. We explored the expression patterns of Beclin1 and LC3B in tuberculous wound tissues in human tuberculous chronic wound and normal tissues was assayed by immunohistochemistry. Rat models of tuberculous wounding were induced by the Bacillus Calmette-Guerin (BCG) method. Beclin1 and LC3B protein expression in human tuberculous wound tissues differed from that of normal skin and non-tuberculous chronic wound tissues.In rat tuberculous wound tissues, expression of Beclin1 and LC3B mRNA time-dependently changed post-infection. Abnormal fluctuation of autophagy protein in the development of tuberculosis wound could be one of the causes for the repeated occurrence and protracted disease course of the tuberculous wound.
Introduction
Tuberculosis (TB) is among the 10 leading causes of mortality across the world, and is the first leading cause of death on infection by a single pathogen. In 2017, 10 million patients were newly diagnosed with TB, most of whom were from developing countries. It is estimated that newly diagnosed TB patients in China accounts for 9% of all the newly diagnosed TB patients globally. 1 In 2016, over 0.78 million of TB patients were reported in China, among whom 5% had presented with extrapulmonary tuberculosis. 2 The presentation of extrapulmonary tuberculosis 3 could induce damage of surrounding soft tissues in about 50% of the patients, which finally resulted in formation of a “tuberculous wound.” 4 Due to the lack of specific clinical manifestations, 5 while the pathogeneses are unique and complex, and remain unclear today, the clinical treatment of the tuberculous wound has emerged as a significant challenge. 6 The rates of missing diagnosis and misdiagnosis are relatively high. 4 Therefore, further investigation of the pathogeneses of the tuberculous wound is necessary.
Most previous studies on the pathogeneses of Mycobacterium tuberculosis had focused on pulmonary TB. The current opinions suggested that after phagocytosis by macrophages, Mycobacterium tuberculosis could still exist in phagosomes, which consequently inhibited the fusion of the phagosome-lysosome complex and enabled its evasion from killing by the host. By contrast, Mycobacterium tuberculosis could also disrupt the membrane of phagosomes to translocate to the cytoplasm. 7
Two major mechanisms, namely the proteasome system and autophagy, are available in eukaryotes for the degradation of proteins. 8 The structural characteristics and components of Mycobacterium tuberculosis could inhibit the maturation and acidification of the phagolysosome. 9 In vitro studies have demonstrated that autophagy could inhibit the survival of BCG and Mycobacterium tuberculosis in macrophages. 10 Beclin1 is the homolog of Atg6 (autophagy protein in yeast) in mammals, 11 and is essential for autophagy and the maturation of phagosomes after Mycobacterium tuberculosis is phagocytosed. 10 AMP-activated protein kinase (AMPK) mediated phosphorylation of Beclin-1 can activate autophagy to promote the maturation of autophagosomes. 12 Metformin can induce AMPK-related autophagy with resultant improvement in the treatment efficacy of diabetes patients with Mycobacterium tuberculosis infections. 13 LC3B is the homolog of yeast Atg8 in mammals, and is the specific component of the autophagosome membrane. 8 In addition, LC3B is the only highly specific biomarker of the autophagosome to date. 14
Although only very few studies have been conducted to investigate the tuberculous wound, relatively fewer still have explored the pathogeneses.
Previous studies conducted by our team have found that there were several similarities between the tuberculous wound and pulmonary TB. 15 Therefore, we hypothesized that the abnormal expression of autophagy-related proteins Beclin1 and LC3B could be among key mechanisms involved in the tuberculous wound. To verify this hypothesis, this study measured the Beclin1 and LC3B proteins in different human wound tissues (tuberculous wound, chronic wound, and normal skin), and explored the expression of autophagy proteins at different times after infection. In addition, autophagy inducer rapamycin (RAPA) and autophagy inhibitor 3-methyladenine (3MA) were used in rat models of the tuberculous wound, and then the differences in the protein expression levels of Beclin1 and LC3B in local tissues could be investigated.
The study aimed to demonstrate that the abnormal expression of the autophagy-related proteins Beclin1 and LC3B could be among key mechanisms that play important roles in the development of the tuberculous wound.
Materials and methods
This experiment complies with the regulations of Shanxi Medical University and the country on the management and use of animal experiments. The experimental animals are provided by the Laboratory Animal Center of Shanxi Medical University. License number: SCXK (Jin) 2015-0001.The human tissue sections in this study are a retrospective study and are limited by the number of patients. The animal experiment is limited by the number of animals. The minimum sample size n = 3 is selected for the non-main observation objects, and the maximum sample size n = 6 allowed by the conditions is used for the main observation objects. Paraffin-embedded human samples (n = 11 for tuberculosis wound tissues; n = 5 for normal skin tissues; and n = 6 for chronic wound tissues) were collected from clinical practices and were included in this study.
Culture of Bacillus Calmette-Guerin
Bacillus Calmette-Guerin (BCG; ATCC, USA) that was stored at −80°C was placed at room temperature (23°C) and thawed on ice. Then BCG the bacterial solution was added to 7H9 culture medium (BD, USA) with OADC enrichment broth (BD, USA) in a biosafety cabinet. Then the flask was placed on the 37°C constant temperature orbital shaker at 80 rpm. The OD of the bacterial solution was measured after colony formation according to the methods described previously. 16 Then the bacterial solution was centrifuged at 12,000 rpm for 20 min, and then diluted to predefined concentrations for further use.
Animal model
First,s ubcutaneous injection of Freund’s complete adjuvant (FCA, 0.2 mL/rat; Sigma, USA) was conducted for sensitization in 12 male SD rats after a 3 weeks adaptation period. Then subcutaneous injections of BCG (0.2 mL/rat; concentration of bacterial solution was 1 × 107/mL) was conducted at 3 weeks after sensitization. Rats were divided into the following groups: 8 days group (n = 3); 15 days group (n = 3); 32 days group (n = 3); 43 days group (n = 3). The day of the BCG injection was defined as 0-day of infection. Then three rats were killed at 8-, 15-, 32-, and 43-days post-infection each to collect the required specimens.

Second, Subcutaneous injection of FCA (0.2 mL/rat; Sigma) sensitized 23 male SD rats after 3 weeks of adaptation. Then a subcutaneous BCG injection (0.2 mL/rat; concentration of bacterial solution was 1 × 107/mL) was conducted at 1 week after sensitization. Rats were divided into the following groups: Normal group (n = 3) (normal feeding without any treatment). BCG group (n = 5), the day of BCG injection was defined as 0-day of infection. For rats in the BCG group, no other intervention was provided post-BCG injection. BCG + RAPA (Solarbio, Beijing, China) group (n = 6), the first intraperitoneal injection of RAPA (3 mg/kg) was also conducted at 0-day, after which, RAPA were administrated every other day. BCG + 3MA (Solarbio, Beijing, China) group (n = 6), the first intraperitoneal injection of 3MA (3 mg/kg) was also conducted at 0-day, after which, 3MA were administrated every other day. BCG + Starvation group (n = 3), for rats in the starvation group, animals were starved for 48 h after BCG injection to induce drastic body weight reduction, and then animals were fed regularly. Animals were euthanized 7-days post-infection to collect the test specimens, and specimens from the three male SD rats not receiving any treatments as the normal group.

Acid-fast staining
The tissues were fixed, embedded, sliced, and dewaxed, then stained by carbol fuchsin (Solarbio, Beijing, China) at room temperature (23°C) for 30 min. The slides were washed until the liquid was colorless, and methylene blue counter-staining liquid (Solarbio, Beijing, China) was added for further staining over a 1 min period. The slides were washed again until the liquid was colorless, and then dehydration, clearing, and mounting was conducted.
HE staining
The tissues were fixed, embedded, sliced, and dewaxed; following which, specimens were stained by hematoxylin for 30 min. Next, slides were washed by running water, treated by 1% hydrochloric acid-alcohol for several seconds, then washed by running water again to allow the color change from red to blue. The slides were stained by eosin for 5 min, and then dehydration, clearing, and mounting was conducted.
Fluorescent quantitative PCR (QPCR)
The animal tissues were added to the Trizol (Ambion, USA) grinding tube to extract total RNA, which was reverse transcribed to cDNA by using the PrimeScript™RT reagent Kit (Takara, China). Primers for GAPDH, LC3B, and Beclin1 were designed and synthesized (Sangon Biotech, Shanghai, China). Then qPCR was conducted using the TB Green® Premix Ex Taq™II kit (Takara China). GAPDH was used as the internal reference control to assess the expression of LC3B and Beclin1. Bio-Rad CFX Manager 3.0 software was used for the analysis, and the expression was estimated by using the 2−ΔΔCt fold-expression equation.

Immunohistochemistry and mean optical density (OD) analysis
The tissues were fixed, embedded, sliced, and dewaxed; following which, citrate antigen retrieval solution (pH 6.0) was used for antigen retrieval (moderate heat for 8 min was applied until specimens were boiling, at which point the temperature was sustained for several minutes, then moderated to a low heat for 7 min, and then finally cooled to room temperature (23°C)). After incubation in 3% hydrogen peroxide for 25 min, the slides were blocked with 10% normal goat serum (Solarbio, Beijing, China) at room temperature (23°C) for 30 min. After the serum was removed, primary antibody (LC3BB, 1:400 diluted, Wanleibio, China; Beclin1, 1:200 diluted, Wanleibio, China) was added and incubated overnight at 4°C, and then restored to normal temperature at room temperature (23°C) for 30 min. The slides were washed three times in PBS (5 min/wash), and secondary antibody (HRP-goat-anti-rabbit IgG, Jackson ImmunoResearch, USA) was added and incubated at room temperature (23°C) for 50 min. The slides were washed three times in PBS (5 min/wash), then DAB developing solution (MXB, China) was added under the microscope for controlled development, and hematoxylin (Solarbio, Beijing, China) was added for counter-staining. The slides were washed with running water, treated by 1% hydrochloric acid-alcohol, washed by running water again to allow the color to change from red to blue, followed by dehydration, clearing, and mounting. After immuno-histochemistry, seven random visual fields were selected in each paraffin section, for which the photos were taken, and the mean OD was calculated. Image-pro plus software was used to analyze the percentage of positive proteins in the overall visual field, which was used for the estimation of the relative expression of the target protein.
Western blot
After the proteins were extracted from the tissues, the BCA method was used for the quantification of the protein concentration. Polyacrylamide gel (concentration of the stacking gel was 5%, and concentrations of the separation gels were 15% and 11%, respectively) was prepared, and 20 μL protein solution (40 μg protein) was loaded into each well. After the electrophoresis was completed, a PVDS membrane (Millipore, USA) was used for the transmembrane. After blocking, primary antibody for Beclin1 (1:500 diluted; Wanleibio, China), LC3A/B (1:500 diluted; Wanleibio, China), or β-actin (1:1000 diluted; Wanleibio, China) was added and incubated at 4°C overnight. Then secondary goat-anti-rabbit IgG-HRP (1:5000 diluted; Wanleibio, China) was then added and incubated at 37°C for 45 min. ECL was added to induce the chemiluminescence of substrates, and then the antibodies were separated and blocked. Gel-Pro-Analyzer software was used to analyze the gray-scale of the target strip, and β-actin was used as an internal reference to calculate the relative expression levels of the target proteins.
Statistical analysis
Graphpad prism7 software was used for statistical analysis of the data. The data were described by the means and standard division (mean ± SD), while one-way analysis of variance (ANOVA) was used for comparisons among different groups and to obtain the corresponding alpha p-values.
Results
The levels of Beclin1 and LC3B protein in the human tuberculous wound tissues were different from the normal skin and chronic wound tissues
HE staining (Figure 1) showed that the cells in normal skin tissues were regularly assembled (Figure 1(a)). The cells in the chronic wound tissues were irregularly assembled, with infiltration by inflammatory cells, and aggregation of the cells also found (Figure 1(b)). While for the tuberculous wound tissues, showing cell nuclei fusion and multinuclear giant cells (Figure 1(c)).
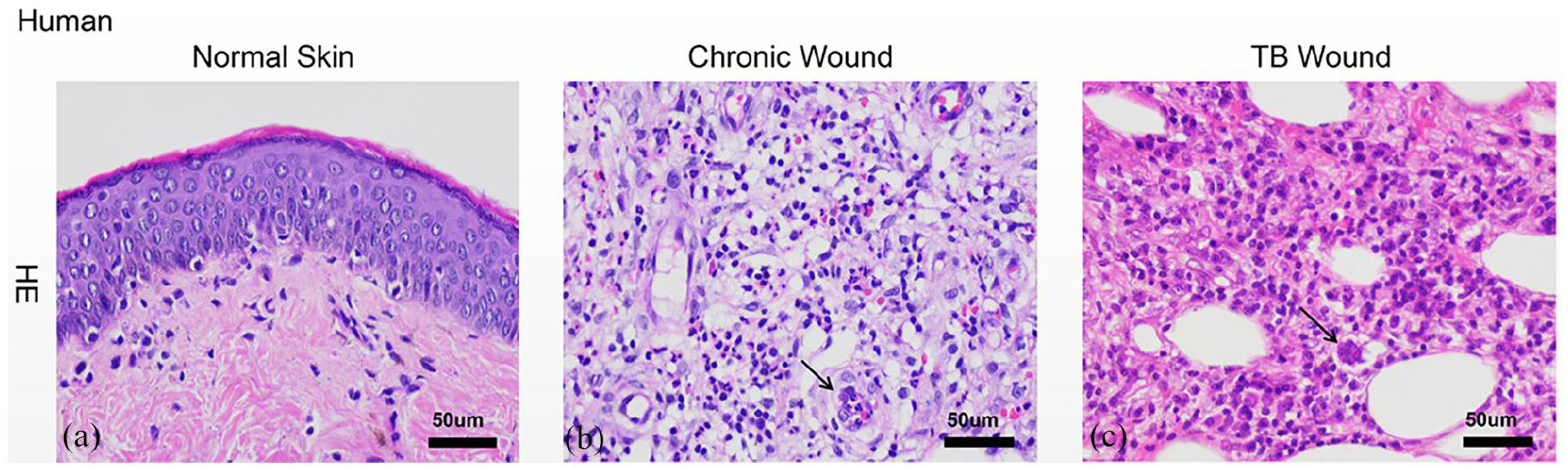
HE staining of paraffin-embedded human tissue of different wound types: (a) HE staining of normal skin tissues that the cells were regularly assembled, (b) HE staining of chronic wound tissues that the cells were irregularly assembled, with infiltration by inflammatory cells, and aggregation of the cells also found, and (c) HE staining of tuberculous wound tissues that the cell nuclei fusion and multinuclear giant cells.
Immunohistochemistry showed that the Beclin1 and LC3B proteins were located mainly around the cell nuclei for subcutaneous tissues (Figure 2). The mean OD values for Beclin1 protein was 0.00439 ± 0.003826, 0.004048 ± 0.003678, and 0.005583 ± 0.004853 in normal skin tissues, chronic wound tissues, and tuberculous wound tissues, respectively. In addition, the mean OD values for LC3B protein was 0.02804 ± 0.02836, 0.01008 ± 0.007779, and 0.01667 ± 0.02283 in normal skin tissues, chronic wound tissues, and tuberculous wound tissues, respectively.The expression of LC3B protein in tuberculous wound tissues are higher than chronic wound tissues (2-F and G).

Immunohistochemistry and mean OD analysis of Beclin1 and LC3B proteins in different types of human wound tissues: (a) immunohistochemistry of Beclin1 protein in normal skin tissues, (b) immunohistochemistry of Beclin1 protein in chronic wound tissues, (c) immunohistochemistry of Beclin1 protein in tuberculous wound tissues, (d) mean OD analysis of Beclin1 protein in different groups, (e) immunohistochemistry of LC3B protein in normal skin tissues, (f) immunohistochemistry of LC3B protein in chronic wound tissues, (g) immunohistochemistry of LC3B protein in tuberculous wound tissues, and (h) mean OD analysis of the LC3B protein in different groups.
Rat models of tuberculous wound induced by BCG
The conditions of the infected skin tissues were assessed at 43-days of infection (Figure 3). Rupture of the bulge at the injection site was found at 32-days of injection, the surface of the abscess showed a volcano appearance, and the section of the abscess showed the formation of small subcutaneous abscess cavities that connected to the external environment (Figure 3(a)). The outflowing purulent secretion was cheese-like, strip shaped, and the abscess had characteristics of a tuberculous abscess (Figure 3(b)). Afterwards, HE staining was conducted for tissues at different times of infection. At 32-days post-infection, large areas of cell destruction were found at the area infiltrated by bacterial aggregation (Figure 3(c)). The acid-fast staining of the infected tissues showed rod-shaped bacteria were clearly identified (Figure 3(d)).
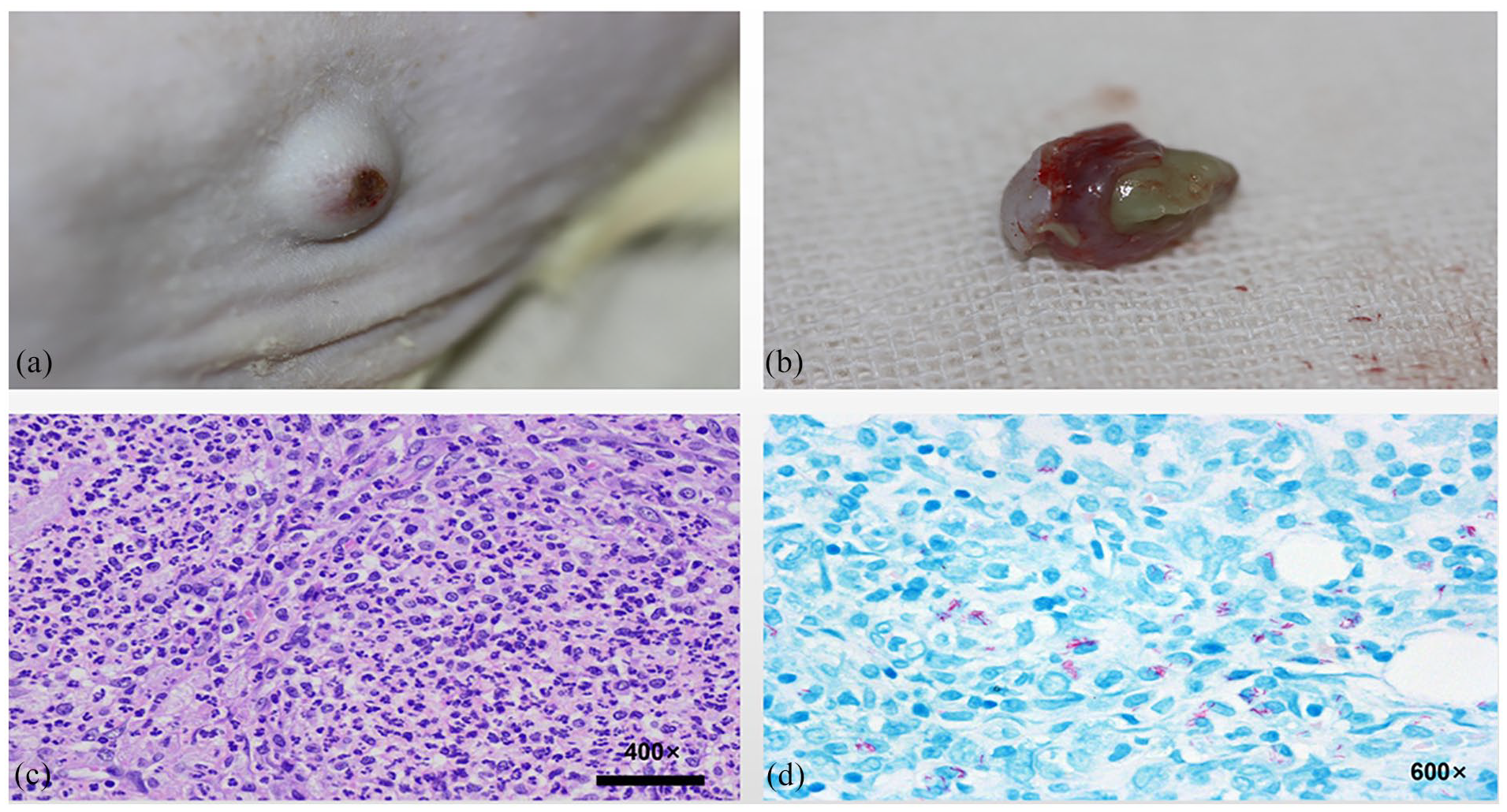
(a) Appearance of the wound at 43-days post- infection, (b) showing outflowing of a purulent secretion from the infected lesion, (c) HE staining at 43-days post-infection, and (d) (43-days), acid-fast staining of the tuberculous wound tissues.
Transcription and expression of autophagy-related proteins Beclin1 and LC3B in the infected tissues of rat models
Immunohistochemistry (Figure 4) showed that only very few Beclin1 and LC3B proteins were expressed in normal rat skin tissues, which were seen mainly in dermal tissues, while hardly any such proteins were found in subcutaneous tissues (Figure 4(a) and (f)). High levels of Beclin1 and LC3B proteins were found in the surrounding cell nuclei at 8-days post-infection (Figure 4(b) and (g)). In addition, high levels of Beclin1 protein were found surrounding the cell nuclei at 15-days of infection (Figure 4(d)), while the level of LC3B protein decreased evidently despite obvious cell aggregation (Figure 4(h)). Only very low level of Beclin1 and LC3B proteins were found in aggregated large cellular densities at 32-days of infection (Figure 4(d) and (i)). Increased Beclin1 protein surrounding the cell nuclei was found again at 43-days post-infection (Figure 4(e)), while the level of LC3B did not increase evidently (Figure 4(j)).
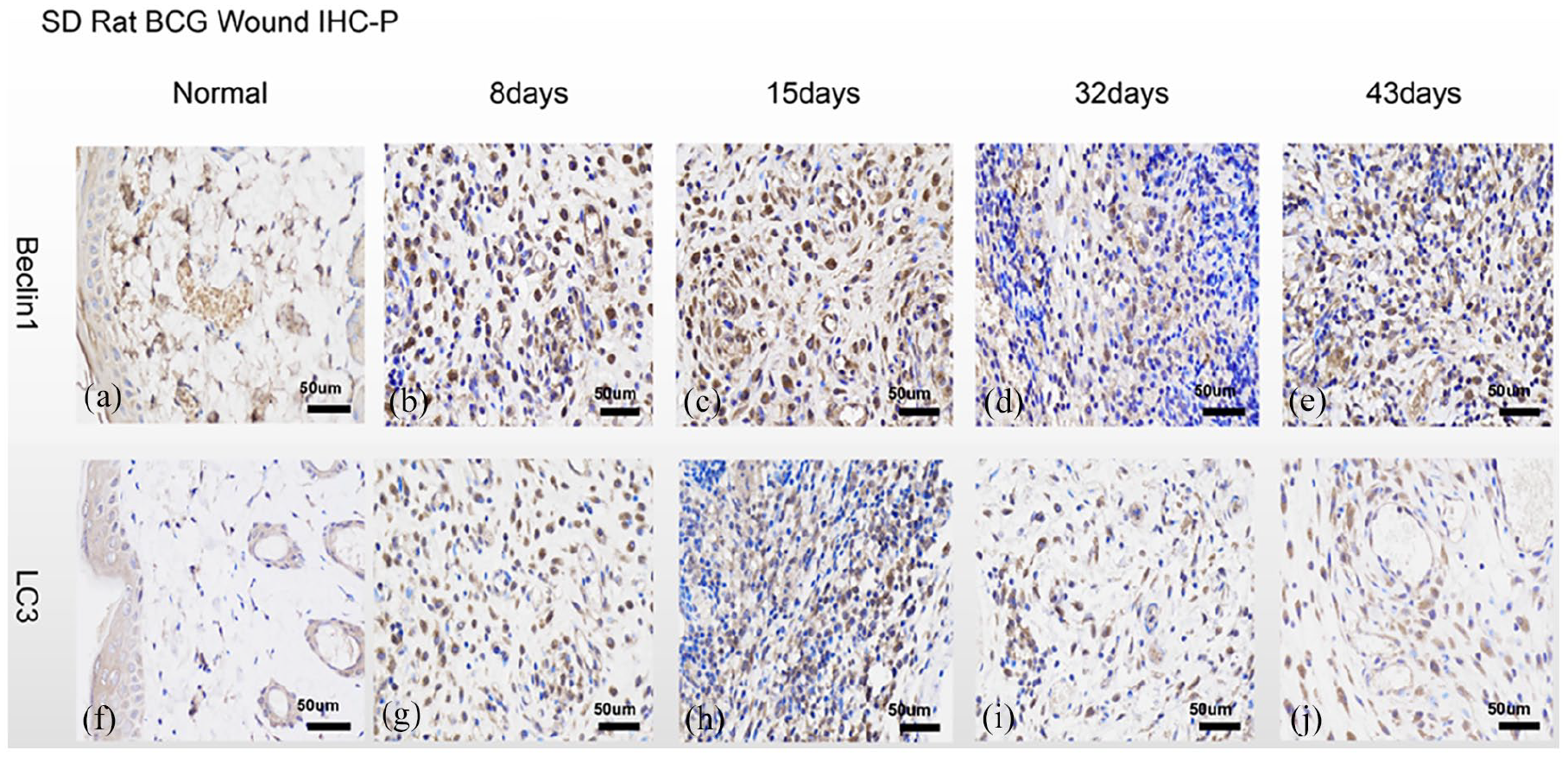
Immunohistochemistry of Beclin1 and LC3B proteins at different times post-infection in the rat model: (a) immunohistochemistry of Beclin1 protein in normal rat skin tissues, (b) immunohistochemistry of Beclin1 protein at 8-days of infection, (c) immuno-histochemistry of Beclin1 protein at 15-days of infection, (d) immunohistochemistry of Beclin1 protein at 32-days of infection, (e) immunohistochemistry of Beclin1 protein at 43-days of infection, (f) immunohistochemistry of LC3B protein in the normal rat skin tissues, (g) immuno-histochemistry of LC3B protein at 8-days of infection, (h) immunohistochemistry of LC3B protein at 15-days of infection, (i) immunohistochemistry of LC3B protein at 32-days of infection, and (j) immunohistochemistry of LC3B protein at 43-days post-infection.
The mRNA levels of Beclin1 and LC3B in the infected tissues at different times post-infection are shown in Figure 6(a) and (e). In brief, the mRNA levels of Beclin1 increased gradually following infection (Figure 5(a)), which was 0.1476 ± 0.01075 at 8-days of infection, 1.415 ± 0.2032 at 15-days of infection, which then peaked at 32-days of infection (3.154 ± 0.3812), and then started to decrease. The level at 43-days post-infection was 2.035 ± 2.121. Similarly, the mRNA level of LC3B also increased gradually following infection (Figure 5(e)), which was 1.123 ± 0.2631 at 8-days of infection, 2.973 ± 0.6744 at 15-days of infection, and then peaked at 32-days of infection (3.617 ± 1.754), and decreased by 43-days of infection (1.763 ± 1.64). The mean OD analysis (Figure 5(b) and (f)) showed that the relative expression levels of Beclin1 and LC3B in normal skin tissues was 0.003123 ± 0.0005632 and 0.003605 ± 0.00136, respectively. At 8-days of infection, the levels of Beclin1 (0.04614 ± 0.02157) and LC3B (0.03767 ± 0.01112) were both higher than in normal skin tissues (p < 0.05; one-way ANOVA). At 15-days of infection, the level of Beclin1 protein (0.05042 ± 0.01903) in the infected tissues remained higher than in normal skin tissues (p < 0.01; one-way ANOVA), while the LC3B protein level (0.02661 ± 0.02341) decreased slightly. Beclin1 protein (0.0163 ± 0.005093) and LC3B protein (0.02429 ± 0.007145) expression levels were both at lowest levels by 32-days post-infection. However, the levels of Beclin1 protein (0.04136 ± 0.01417) and LC3B protein (0.03241 ± 0.01372) at 43-days of infection were higher than were seen by 32-days of infection. In addition, the Beclin1 level was significantly different from the levels found in normal skin tissues (p < 0.05; one-way ANOVA).Western blotting (Figure 5(c) and (g)) showed that changes in Beclin1 protein expression were in agreement with the immunohistochemistry findings (Figure 5(b) and (d)). The Beclin1 protein at 8-days of infection was 0.41 ± 0.3412, which reached a peak at 15-days post-infection (0.8933 ± 0.2281), and decreased by 32-days of infection (0.5467 ± 0.2155), but increased again by 43-days of infection (1.093 ± 0.2196). The ratio of LC3B/LC3A indicates the activity of autophagy (Figure 6(h)). The LC3B/LC3A ratio was 0.7767 ± 0.2706 at 8-days of infection, which achieved a peak ratio by 15-days post-infection (1.423 ± 0.5398), and had decreased by 32-days post-infection (0.5067 ± 0.1914), and had increased again at 43-days post-infection (0.8 ± 0.1732). In this study, immunohistochemistry showed that the LC3B protein level decreased at 15-days of infection (Figure 5(f)); however, grayscale analysis showed that the LC3B/LC3A ratio at 15-days of infection had peaked. The mRNA levels of Beclin1 and LC3B during infection had increased gradually and peaked at 32-days post-infection. While the protein levels of Beclin1 and LC3B also increased gradually but peaked at 15-days post-infection, and reached their lowest levels at 32-days post-infection. The autophagy activity at 43-days of infection was higher than was found at 32-days of infection.

Quantitative analysis of Beclin1 and LC3B mRNA, mean OD analysis, and Western blot assay of the tissues at different times post-infection in the rat model: (a) quantitative analysis of Beclin1 mRNA in tissues at different times post-infection, (b) mean OD analysis of Beclin1 protein in tissues at different times post-infection, which was significantly different at 8-days (p < 0.05), 15-days (p < 0.01), and at 43-days post-infection (p < 0.05) as compared normal skin tissues, (c) Western blotting of Beclin1 protein in tissues at different times post-infection, (d) grayscale analysis of Beclin1 protein in tissues at different times post-infection, (e) quantitative analysis of LC3B mRNA in tissues at different times post-infection, (f) mean OD analysis of the LC3B protein in tissues at different times post-infection, which was significantly different at 8-days of infection (p < 0.05) as compared normal skin tissues, (g) Western blotting of LC3B/LC3A proteins in tissues at different times post-infection, and (h) grayscale analysis of LC3B/LC3A proteins in tissues at different times post-infection.
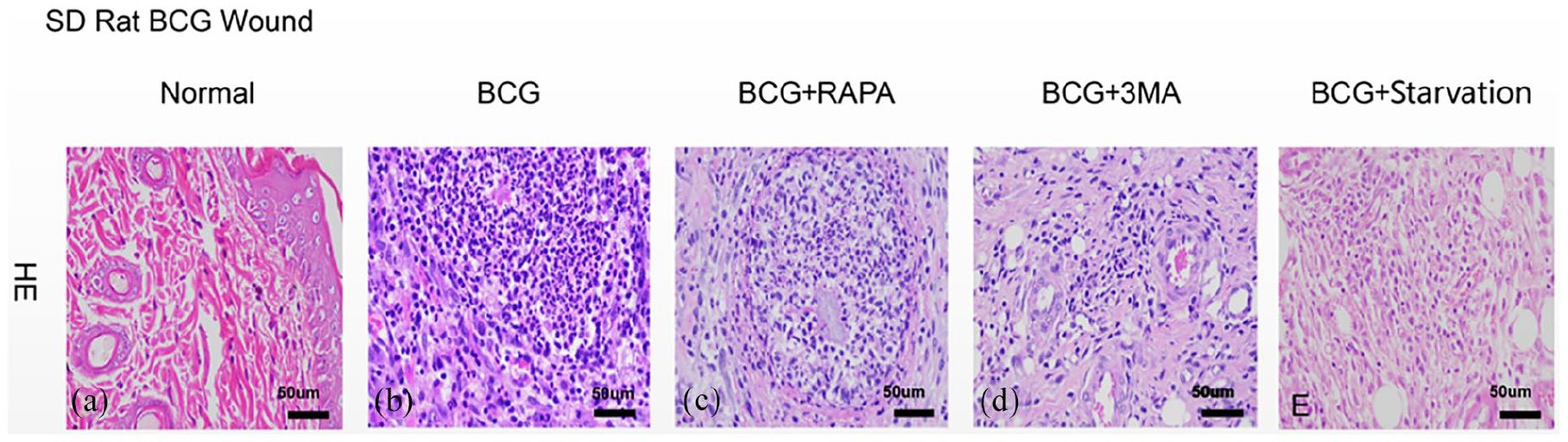
HE tissue staining in rat models after different treatments: (a) HE staining of normal rat skin tissues, (b) HE staining of tissues in the BCG group, (c) HE staining of tissues in the BCG + RAPA group, (d) HE staining of tissues in the BCG + 3MA group, and (e) HE staining of tissues in the BCG + Starvation group.
Influence of RAPA and 3MA on the transcriptional expression of Beclin1 and LC3B in the tuberculous wound tissues of rat models
HE staining (Figure 6) showed that the cells in normal rat skin tissues were regularly assembled, while non-inflammatory cell infiltration was found (Figure 6(a)). In contrast, necrotic lesions were found in the BCG and BCG + RAPA groups, while no cells were found in the center of the necrotic lesions. However, the necrotic lesions were surrounded by high cell densities, of which the nuclei were dividing and showed fragmentation (Figure 6(b) and (c)). Infiltration of inflammatory cells was also found in both the BCG + 3MA and BCG + Starvation groups (Figure 6(d) and (e)).
Immunohistochemistry (Figure 7) showed that only low levels of Beclin1 and LC3B proteins were found in normal rat skin tissues (Figure 7(a) and (f)), while high levels of Beclin1 protein was found surrounding the nuclei of cells in the BCG, BCG + RAPA, and BCG + 3MA groups (Figure 7(b)–(d)). By contrast, Beclin1 protein levels in the BCG + Starvation group were lower than were found in the BCG group (Figure 7(e)). The LC3B level in the BCG group was higher than was found in the other groups (Figure 7(g)).

Immunohistochemistry of Beclin1 and LC3B proteins differentially treated rat models. Immunohistochemistry of Beclin1 in normal rat skin tissues (a); the BCG group (b); the BCG + RAPA group (c); the BCG + 3MA group (d); the BCG + Starvation group (e); and in normal rat skin tissues (f). Immunohistochemistry of LC3B in the BCG group (g); the BCG + RAPA group (h); the BCG + 3MA group (i); and the BCG + Starvation group (j).
The quantitative analysis of Beclin1 mRNA in the lesions of different groups (Figure 8(a)) showed that the Beclin1 mRNA level was 0.9162 ± 0.6581, 0.8789 ± 0.7338, and 1.574 ± 1.2 in the BCG, BCG + RAPA, and BCG + 3MA groups, respectively. The Beclin1 mRNA level in the BCG and BCG + RAPA groups was not significantly different, while the level in the BCG + 3MA group was significantly higher than found in the BCG and BCG + RAPA groups. Quantitative analysis of LC3B mRNA in the lesions of different groups (Figure 8(e)) showed that LC3B mRNA levels were 1.122 ± 0.5263, 0.6973 ± 0.2156, and 1.069 ± 0.5991 in the BCG, BCG + RAPA, and BCG + 3MA groups, respectively. LC3B mRNA levels in the BCG and BCG + 3MA groups were similar, while the levels in the BCG + RAPA group were significantly lower than were found in the BCG and BCG + 3MA groups. Mean OD analysis (Figure 8(b) and (f)) showed that the relative levels of Beclin1 and LC3B were 0.003123 ± 0.0005632 and 0.003605 ± 0.00136 in normal skin tissues, and were 0.03747 ± 0.02522 and 0.02624 ± 0.01404 in the BCG group, which were significantly different between both groups (p < 0.05; one-way ANOVA). The expression of Beclin1 protein was similar among the BCG + 3MA group (0.02679 ± 0.0164), BCG + RAPA group (0.03003 ± 0.007981), and BCG + Starvation group (0.02813 ± 0.006291). The expression of LC3B protein in the BCG + 3MA group (0.007489 ± 0.003152) was significantly lower than in the BCG + RAPA (0.01441 ± 0.01467) and the BCG + Starvation groups (0.0148 ± 0.005684). Western blot (Figure 8(d) and (h)) assay showed that the relative expression levels of Beclin1 was 0.515 ± 0.2132 in the BCG + 3MA group, which was higher than found in the BCG (0.302 ± 0.1252) and BCG + RAPA groups (0.4217 ± 0.2638), which agreed with the findings for mRNA analysis. The LC3B/LC3A level was 0.538 ± 0.1942, 0.4117 ± 0.2895, and 0.49 ± 0.2559 in the BCG, BCG+RAPA, and BCG+3MA groups, respectively.
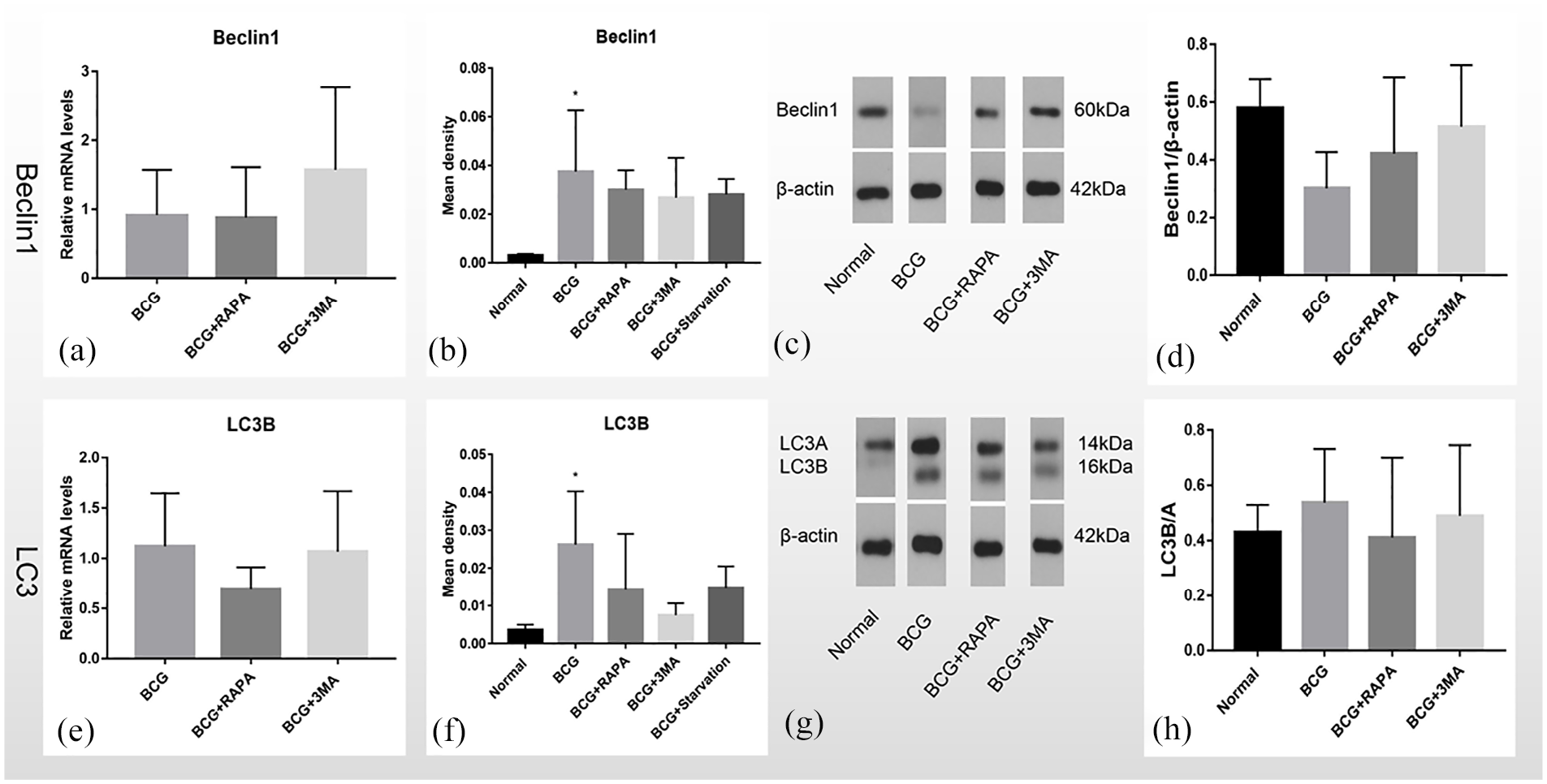
Quantitative analysis of Beclin1 and LC3B mRNA, mean OD analysis, and Western blot of tissues from the differentially treated rat models: (a) quantitative analysis of Beclin1 mRNA in different groups; and (b) mean OD analysis of Beclin1 protein in different groups. Levels in the BCG group were significantly different from the levels found in the normal group (p < 0.05), (c) Western blot of Beclin1 protein in different groups, (d) grayscale analysis of Beclin1 protein in different groups, (e) quantitative analysis of LC3B mRNA in different groups, (f) mean OD analysis of LC3B protein in different groups. The level in the BCG group was significantly different from that found for the normal group (p < 0.05), (g) Western blot assay of LC3B/LC3A protein in different groups, and (h) showing grayscale analysis of the LC3B/LC3A protein in different groups.
Discussion
Animal models of tuberculous wound
Animal models could offer a good platform for various scientific research activities. Previous studies 17 have reported the successful induction of models in various animals, such as the rabbit,18,19 mouse, 20 rat, 15 zebrafish, 21 and monkey. 22 Our team has already successfully induced rat models of a tuberculous wound by using viable BCG (1 × 107 CFU/site). 15 Previous studies 23 have shown that skin TB in rabbits induced by viable BCG peaked and liquefied at 11-days post-injection, while skin TB in rabbits induced by inactivated BCG relieved slowly and healed by 35-days post-injection. 24 The mean time of the formation of a tuberculous wound in zebrafish induced by Mycobacterium marinum was 59 days. 21 In contrast, local swelling was still found at 15-days after injection in rat models of tuberculous wound induced by BCG (1 × 106 CFU). 15
In this study, local abscess with positive results in acid-fast staining were found in rat models, which confirmed that rat models of tuberculous wound were successfully induced. Some prior studies have suggested that light-emitting diode fluorescence microscopy (LED-FM) in TB patients might offer an optimal replacement for Ziehl-Neelsen acid-fast staining. 25 However, acid-fast staining is the most universally used method for the diagnosis of TB for more than a century, 26 and thus this method was also used in this study. Herein, 2 × 106/mL BCG was injected to each site, while no signs of acute inflammation, such as local swelling, were observed. We speculated that such a manifestation was not caused by the low injection dose of BCG, but was caused instead by the fact that BCG was injected to deep subcutaneous sites, thus the abscess was also relatively deep and the rupture was also relatively difficult, and thus relatively difficult for an inflammatory response to appear at superficial skin tissues.
Pathogeneses of tuberculous wound
One of the characteristics of autophagy is dynamic regulation, and thus under normal conditions, autophagic activities in cells are mainly relatively low. 27 LC3B-I is located in the cytoplasm, while LC3B-II is a membrane-binding protein. LC3B-II is the first mammalian protein that specifically binds to the autophagosome membrane. 8 Quantitative analysis of LC3B has shown that the mycobacterium-containing organelles are autophagosomes. 10 In addition, previous studies have demonstrated that the increasing rate of LC3B-II is associated with the rate of autophagosome formation. Wortmannin and 3MA are two autophagy inhibitors that can inhibit the increase in LC3B-II expression that is induced by starvation. By contrast, some other autophagy-inducing drugs that include Vinblastine and Bafilomycin A1, are quite potent at increasing the levels of LC3B-II, which might thus reflect the number of autophagosomes. 8 However, some opinions have also suggested that it is a common misunderstanding to believe that an increase in the expression levels of the LC3B-related autophagosome indicates an increase in autophagy. 27 Since a relatively high proportion of LC3B is degraded after lysosomal fusion, the factors extending the time of autophagic stimulation might provoke a further decrease in LC3B, which could be misinterpreted as reduced autophagic activity. 28 Therefore, measuring LC3B at some single time-point might not sufficiently indicate the status of autophagy. 27
The findings in this currently reported study showed that only very few Beclin1 and LC3B proteins were expressed in normal rat skin tissues. Specifically, these proteins were mainly expressed in the dermis, while hardly any such proteins were found in subcutaneous tissues. In contrast, high expression of Beclin1and LC3B was found at 8-days post-infection, suggesting that the autophagy system was activated after infection with Mycobacterium tuberculosis. We speculated that the advanced decrease in protein expression levels (as compared decreases in mRNA levels) might have been due to the increased degradation of such proteins after participating in autophagy.
The LC3B/LC3A ratio at 15-days post-infection was the highest in this study. The LC3B/LC3A ratio has been considered a key criterion for the assessment of autophagic activity. The findings in this study showed that the autophagic level at 15-days post-infection had peaked, which led to a drastic consumption of proteins, which thus resulted in an advanced decrease in protein levels. The autophagy level at 43-days post-infection was higher than at 32-days post-infection, and with the higher autophagy level, the consumption of autophagy-related proteins Beclin1 and LC3B was also higher. However, at which time the mRNA levels of Beclin1 and LC3B were decreased was a question we wished to explore. Since there is a dynamic balance between the synthesis and consumption of autophagy-related proteins during the control of an infection, we speculated that promoting the synthesis of such proteins and increasing the autophagic activity could potentially favor the control of an infection. The Beclin1 and LC3B proteins, as well as the LC3B/LC3A ratio were reduced at 32-days post-infection, but were then increased again in this study. In contrast, the mRNA levels of Beclin1 and LC3B were highest at 32-days post-infection, but then decreased again. Such fluctuation could be one of the causes for the repeated occurrence and protracted disease course of the tuberculous wound.
Influences of the autophagy inhibitor on tuberculous wounds
Both 3MA and Wortmannin8,29 could inhibit the formation of autophagosomes by inhibiting PI3K. 28 The PI3 kinase inhibitor (3MA and Wortmannin) mainly block the formation of autophagosomes at an early phase, which could thus abrogate the acidification of mycobacterium-containing phagosomes induced by starvation. 30 Treating the infected macrophages with RAPA could upregulate the acidification of mycobacterium-containing phagosomes. 30 Bafilomycin A1 and chloroquine could inhibit the acidification of lysosomes to block autophagic degradation. 28 In addition, Vinblastine and Bafilomycin A1 could also inhibit the fusion of autophagosomes with lysosomes. 31 Treating cultured cells with Wortmannin or 3MA could completely abrogate an increase in LC3B-II that was induced by starvation. 8 3MA could reverse the bactericidal effects of macrophages when explored during in vitro cellular experiments. 10 In vitro studies have shown that when using toxic Mycobacterium tuberculosis H37Rv to infect macrophages, conventional autophagy inhibition could still abrogate the autophagy that was induced by starvation or RAPA, and thus reduce the viability of H37Rv. 10 3MA is a specific autophagy inhibitor that affects the formation of autophagosomes. 32 The findings in this study showed that treating rat models with 3MA could increase the transcription of Beclin1 mRNA in infected tissues. The protein expression of Beclin1 in the BCG + 3MA group was significantly higher than in the BCG and BCG + RAPA groups, indicating that 3MA induced Beclin1 protein and mRNA accumulation, which might be associated with the accumulation of pre-substances caused by 3MA-mediated inhibition of autophagy. Autophagy was needed to eliminate Mycobacterium tuberculosis in tuberculous wound tissues; the accumulation of the pre-substances without activation of autophagy was apparently unbeneficial to the healing process of tuberculous wounds.
Influences of autophagy inducer on tuberculous wounds
Mammalian target of rapamycin (mTOR), which is a serine/threonine kinase, is a major regulator of cellular metabolism. 33 It has been demonstrated that mTOR is a major negative regulator of autophagy. 28 The activity of mTOR is inhibited when nutrient substances are sufficient, while the activity of mTOR is inhibited upon starvation, which in turn activates the autophagic pathway, this process also serves to promote cell survival under unfavorable conditions. 34
Starvation could promote autophagy. Numerous studies have demonstrated that autophagy is induced in various tissues and organs when food is insufficient. 35 The findings in this study showed that the protein expression of Beclin1 and LC3B in the BCG + RAPA and BCG + Starvation groups was not significantly different, indicating that RAPA and starvation had comparable effects on autophagy. mTOR could inhibit the enzymes that are involved in the autophagy pathway to inhibit autophagy. 36 Treating cultured cells with RAPA could reduce the survival of the Mycobacterium tuberculosis in cells 30 ; therefore, utilizing the mTOR pathway might serve as a potential approach to improve the effects of anti-TB treatment. 37 The findings in the animal models that were treated with RAPA in this study showed that RAPA blocks transcription of LC3B mRNA. The protein expression of Beclin1 in the BCG + Starvation group was lower than was found in the BCG group, which could be due to the fact that starvation increased autophagic activity and therefore upregulated the consumption of Beclin1. The grayscale value of LC3B/LC3A in the BCG, BCG + RAPA, and the BCG + 3MA groups was not significantly different, suggesting that autophagic activity at the early stage was not significantly different, although the tendency was in agreement with changes seen for LC3B mRNA expression. The continuous promotion of autophagy by RAPA increased autophagic substances, and thus promote decreased protein expression of Beclin1 and LC3B as compared the non-treated group. There is a dynamic balance between the synthesis and degradation of autophagy-related proteins in tuberculosis wounds. It would better favor the control of tuberculosis wounds if the synthesis of autophagy-related proteins were increased when promoting autophagy. RAPA treatment could dose-dependently reduce the viability of mycobacterium 10 ; however, more studies are required to adequately explore whether increasing the dose of RAPA could favor the treatment of tuberculosis wounds.
Clinical significance of autophagy
Host directed therapy represents a novel strategy for treating TB, which aims to enhance the endogenous host immune system to combat an infection with Mycobacterium tuberculosis, while autophagy is one of the targets in this treatment strategy.38,39 To date, several autophagy-enhancing drugs have already been approved. 40 Everolimus, 41 a RAPA homolog, is a new mTOR inhibitor that could be used to treat Mycobacterium tuberculosis infection. 36 Gefitinib could also reduce the load of Mycobacterium tuberculosis in macrophages of murine lung tissues. 42 Statins can promote autophagosome maturation to reduce Mycobacterium tuberculosis load in human macrophages and in mice. 43 Regulating autophagy could be a potentially new target for treating diseases. 44
However, some studies have suggested that several uncertainties should be ruled out when using autophagy as a therapeutic target. 28 For instance, it has been demonstrated that some pathogens could block the downstream pathway of autophagy, while just increasing autophagy could lead to an accumulation of toxic by-products. 28 It has been demonstrated that autophagy plays a role in various common bacterial comorbidities in diseases such as HIV infection, tumorigenesis, and diabetes. Therefore, the effects of such drugs on drug comorbidities must be considered. 28 In addition, other studies have demonstrated that in cases of co-infection of macrophages with HIV and Mycobacterium tuberculosis, promoting autophagy by RAPA could increase intracellular survival of Mycobacterium tuberculosis. 45 It has also been suggested that further investigation of pathogen-evading mechanisms are also needed when discussing treatment methods. 28 The findings in this study showed that with an increase in post-infection tuberculosis wounds, the levels of the autophagy-related proteins Beclin1 and LC3B were found to first be increased and then subsequently decreased. By promoting or inhibiting autophagy, the protein expression of Beclin1 and LC3B could be disrupted in the early stages of an infection in animal models. These findings demonstrated that interfering with autophagy to influence the capability of the infected tissues to eliminate Mycobacterium tuberculosis could be a potential approach for treating tuberculosis wounds.
Limitations of the study
Due to time restrictions, only relatively low numbers of paraffin-embedded human samples (n = 11 for tuberculosis wound tissues; n = 5 for normal skin tissues; and n = 6 for chronic wound tissues) were collected from clinical practices and were included in this study. In addition, changes in protein expression were only assessed at one time point in the treatment groups. Such limitations could influence the robustness and/or reliability of the obtained results, which could of course be addressed in future studies. In our future studies, the other processes in autophagy of tuberculosis wounds will be investigated to more comprehensively elucidate the pathogeneses of tuberculosis wounds.
Conclusion
Abnormal fluctuation of autophagy protein in the development of tuberculosis wound could be one of the causes for the repeated occurrence and protracted disease course of the tuberculous wound.
Footnotes
Acknowledgements
We should like to thank Zhou Yu from the Department of Plastic and Reconstructive Surgery of Xijing Hospital of the Air Force Medical University helps in experimental techniques study and Lin Chen helps provide the normal human paraffin-embedded tissue from the Department of Plastic and Reconstructive Surgery of Xijing Hospital of the Air Force Medical University; Yueyun Ma and Liang Chang from the Department of Clinical Laboratory Medicine of Xijing Hospital of the Air Force Medical University helps in experimental techniques study; and Professor Yinlan Bai and Jian Kang from the Department of Microbiology and Pathogen Biology of the Air Force Medical University provide the BCG bacteria; Zhen Liu and Chunjuan Chang and Fenghua Wang from the The Eighth Medical Center of Chinese PLA General Hospital helps provide the wound paraffin-embedded tissue of human tuberculosis.
Author contributions
Keqiang Zhou developed the initial concept and wrote the manuscript and performed the experiments and analyzed the data. Yingjun Su designed the experiments. Chiyu Jia supervised the study. All authors discussed the results and commented on the manuscript. All authors read and approved the final version of this submitted manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Starting Package of Xiang’an Hospital of Xiamen University (PM201809170010). The project was supported by the Natural Science Foundation of Fujian Province of China (No. 2019J01011).
Ethics approval
Ethical approval for this study was obtained from Shanxi Medical University Ethics Committee (Approval Number/ID: SCXK (Jin) 2015-0001).
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
