Abstract
Chronic kidney disease (CKD), characterized by the progressive, irreversible loss of kidney function, has been a significant socioeconomic burden. Thus, identifying potential treatment targets and incipient biological risk indicators for CKD prediction is crucial. Recently several studies revealed that IL-10, IL-10RA, and IL-10RB genes were involved in the development of vascular complications in hypertension and stroke. The purpose of this study was to evaluate the association between CKD and IL-10, IL-10RA, and IL-10RB gene polymorphisms in the Korean population. Ninety-two CKD patients and 312 control subjects participated in the study. Blood samples were drawn for biochemical measurements and genetic polymorphism analysis. To analyze the genotypes of each single nucleotide polymorphism (SNP), PCR products were sequenced by a DNA analyzer. The four SNPs of the IL-10RA gene showed significant associations between CKD and the control group in the codominant1 (rs2228054, rs2228055, and rs9610, OR = 0.48, 95% CI = 0.28–0.82,
Introduction
Chronic Kidney Disease (CKD) is typically defined by continuous and often gradual reductions in kidney function that cannot be reversed. For evaluation of kidney function, the equations based on serum creatinine value, gender and ethnicity are used to calculate estimated glomerular filtration rate (eGFR). The stage of CKD is classified by eGFR classification and slope of eGFR is used one of surrogate indicator in clinical research. However, CKD dose not progress at the same rate in all patients, and frequently, CKD progression is vary in same disease, such as autosomal dominant polycytic kidney disease, glomerulonephritis, hypertension and even in diabetes.1–3 Despite the fact that genetic predisposition to CKD is proved, the risk alleles indicated by association researches are regarded as a minor contribution to overall complex disease risk. 4 CKD can be secondary to conditions, for instance, glomerulonephritis, hypertension or diabetes. In specific, patients with glomerulonephritis, hypertension or diabetes will not encourage progressive CKD that induces an investigation of genetic modification related to a susceptibility to renal illness or protection from the occurrence of complications.3,5 Interrelationship between CKD and genetic modifications has been reported. Specifically, suggestion of Caveolin-1 Single Nucleotide Polymorphism (SNP) as a genetic indicator in CKD, 6 association between CKD patients’ kidney survival and SNP in D-loop region of mitochondrial DNA, 7 and interaction between SNPs such as superoxide dismutase, glutathione peroxidase, peroxisome proliferator activated receptor gamma concerning ESRD. 8
Persistent, low-grade inflammation is considered important component in CKD progression and outcome. Interleukin-10 (IL-10), produced by various kinds of cells, is a cytokine that reduces inflammation. These immune suppression virtues range from restricting pro-inflammatory cytokine production, down-regulation costimulatory protein expression on antigen-presenting cells to limiting T cell proliferation. IL-10 receptor (IL-10R) which consists of two α subunits (encoded by IL-10RA) and two β subunits (encoded by IL-10RB), was bound to IL-10 and activates downstream signaling. It can also hinder the synthesis of pro-inflammatory cytokines, for instance, TNF alpha, IL-2, and IL-3. Together with IFN-γ receptor 2 (IFNGR2) on chromosome 21q22, IFN-α receptor 1 (IFNAR1) and 2 (IFNAR2), IL10RB is situated in a class II cytokine receptor gene cluster. 9
According to recent researches, association between IL-10 polymorphism and ischemic stroke has been found. 10 Also, the possibility of genes IL-10, IL-10RA, and IL-10RB involved in progressing hypertension in the risk of ischemic stroke, 11 and association between IL-10RB polymorphisms and diffuse cutaneous systemic sclerosis has been reported. 12
This study evaluated the association between CKD and IL-10, IL-10RA, and IL-10RB genes polymorphisms among Korean population.
Materials and methods
Subjects and clinical data
92 CKD patients (44 male, 51.82 ± 9.52 and 48 female, 48.21 ± 13.54 years) and 312 control subjects (157 male, 47.23 ± 10.5 years and 155 female, 46.21 ± 10.03 years) were participated in this research. The control subjects were selected based on the health check-up program results in Health Promotion Center, who showed no clinical evidence for cardiovascular diseases, dyslipidemia, diabetes mellitus, hypertension and cancer. Also eGFR of control subjects was greater than 60 ml/min/1.73 m2 without history of kidney disease or functional/structural abnormality of the kidney. Written informed consent was obtained from all subjects. The study was approved by the institutional review of board, Keimyung University Dongsan Medical Center.
Biochemical measurement
Laboratory evaluation were performed using blood samples obtained in the overnight fasting state. The samples were analyzed for biochemical indicators on the same day they were obtained. Blood samples were drawn for biochemical measurements such as C-reactive protein (CRP), WBC, creatinine, BUN, albumin, r-glutamine transaminase (r-GT), serum transaminase (SGPT), serum transaminase (SGOT), high-density lipoprotein (HDL) cholesterol, low-density lipoprotein (LDL) cholesterol, Total cholesterol (TC), triglyceride (TG), hemoglobin A1c(HbA1c) and plasma fasting blood sugar (FBS). The eGFR was calculated with CKD-EPI equation. 13
SNPs selection and genotyping
Blood samples for DNA extraction from all subject were collected in EDTA tube. Human genomic DNA was extracted from peripheral blood samples using the Nucleospin 96 Blood (Macherey Nagel, Germany).
Pipet 50 μL proteinase K and 400 μL blood to lysis block. Add 400 μL BQ1 to each, mix 5 times and incubate samples at least 15 minutes at room temperature. Recommendations are 20 minutes at 1500 rpm and shake at room temperature. Add 400 μL ethanol, mix at least 5 times and transfer crude lysates to blood binding plate. Binding genomic DNA to silica membrane. Centrifuge 600 rpm for 5 min. Wash silica membrane and leave blood binding plate. Add 1200 μL B5 to each of blood binding plate and elute highly pure genomic DNA, Add 400 μL elution buffer and store DNA at −20°C during genotyping assay.
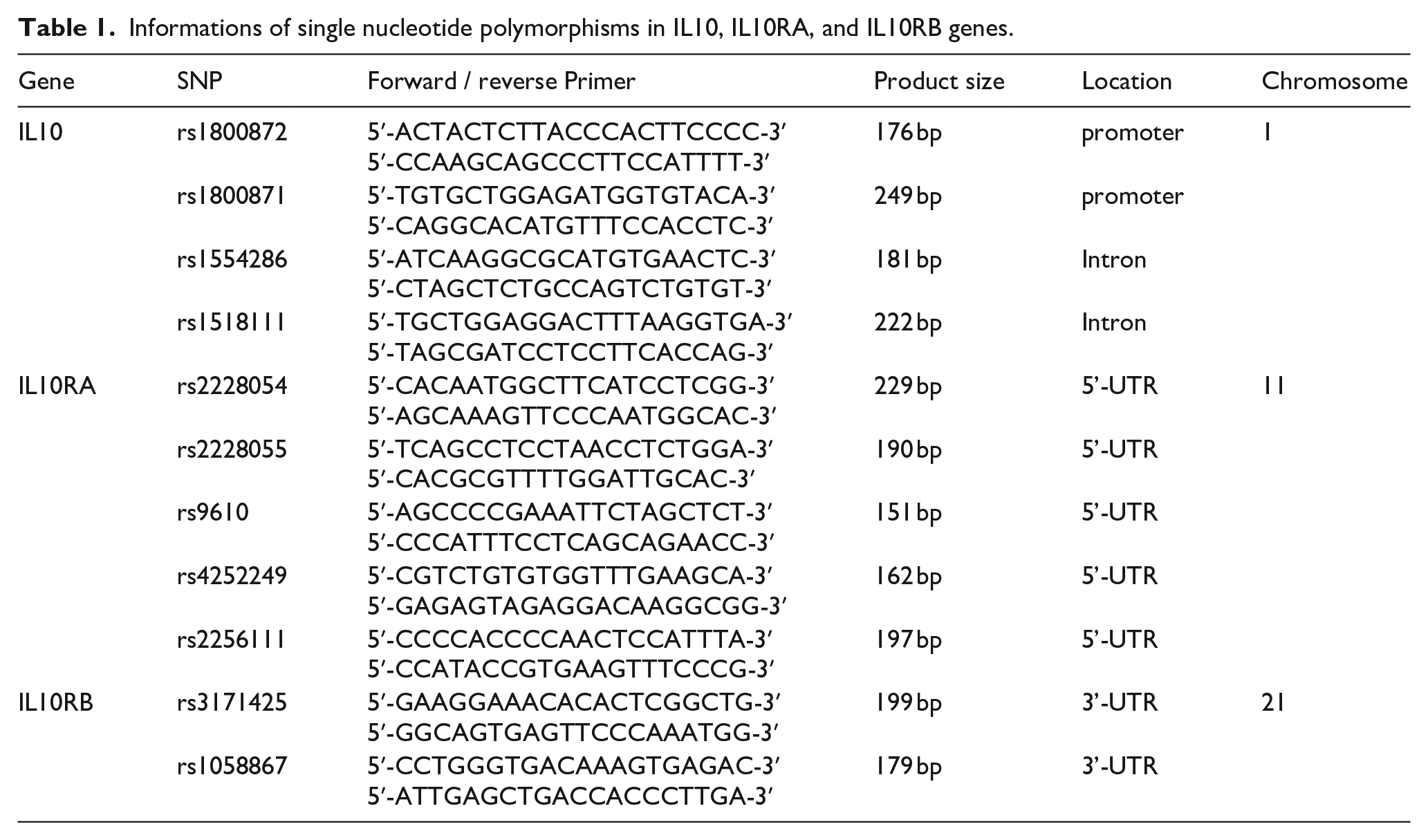
The 11 SNPs with greater than 0.1 heterozygosity of IL-10, IL-10RA, and IL-10RB genes were selected and genotyped by direct sequencing. The following primers for the 11 SNPs were used to amplifier Genomic DNA (Table 1). Polymerase chain reaction (PCR) conditions included 32 cycles at 92°C for 30 s, 60°C for 50 s, and 70°C for 40 s. PCR products were identified on 1.5% agarose gel by electrophoresis. To analyze the genotypes of each SNP, PCR products were sequenced by the DNA analyzer (ABI Prism 3730XL, Applied Biosystems, Foster City, CA, USA) and the genotypes were determined using SeqManII software (DNASTAR Inc., Madison, WI, USA).
Informations of single nucleotide polymorphisms in IL10, IL10RA, and IL10RB genes.
Statistical analysis
The Window version 23.0 of statistical package for the social sciences (SPSS) (SPSS Inc., Chicago, IL, USA) was used for analysis of statistics. The results were considered statistically significant at
Results
Demographic and clinical characteristics of study subjects
The demographics and clinical parameters of the study subjects are summarized in Table 2. Control group consists of 312 subjects including 157 males and 155 females (Mean age of 46.7 ± 10.3 years). The CKD group consists of total 92 adults and included 44 males and 48 females (Mean age of 49.9 ± 11.9 years). The distribution of gender among subjects was not significantly different between the two groups. The levels of body mass index (BMI), systolic blood pressure (SBP), diastolic blood pressure (DBP), FBS, TG, BUN, creatinine, uric acid, WBC, and CRP in the CKD group were significantly higher than those of the control group (
Demographic characteristics and clinical parameters for the study population.

Quantitative data are the mean ± SD: DM: diabetes mellitus; HTN: hypertension; CGN: chronic glomerular nephritis are shown as frequency (percentage). BMI: body mass index; HDL: high-density lipoprotein; LDL: low-density lipoprotein; eGFR: estimated glomerular filtration rate.
Logistic regression analysis of IL-10, IL-10RA, and IL-10RB polymorphisms between CKD and controls
The association between CKD and 11 SNPs was analyzed with multiple logistic regression and with adjustment for age, gender, BMI, and prevalence of hypertension, diabetes and dyslipidemia (Table 3). The rs2228054 (G/A), rs2228055 (A/G), rs9610 (A/G), and rs2256111 (G/A) of the IL-10RA gene showed a significant association between CKD and the control group in the codominant1 (rs2228054, rs2228055, and rs9610, OR = 0.48, 95% CI = 0.28–0.82,
Association of IL-10, IL-10RA, and IL-10RB polymorphisms with CKD.
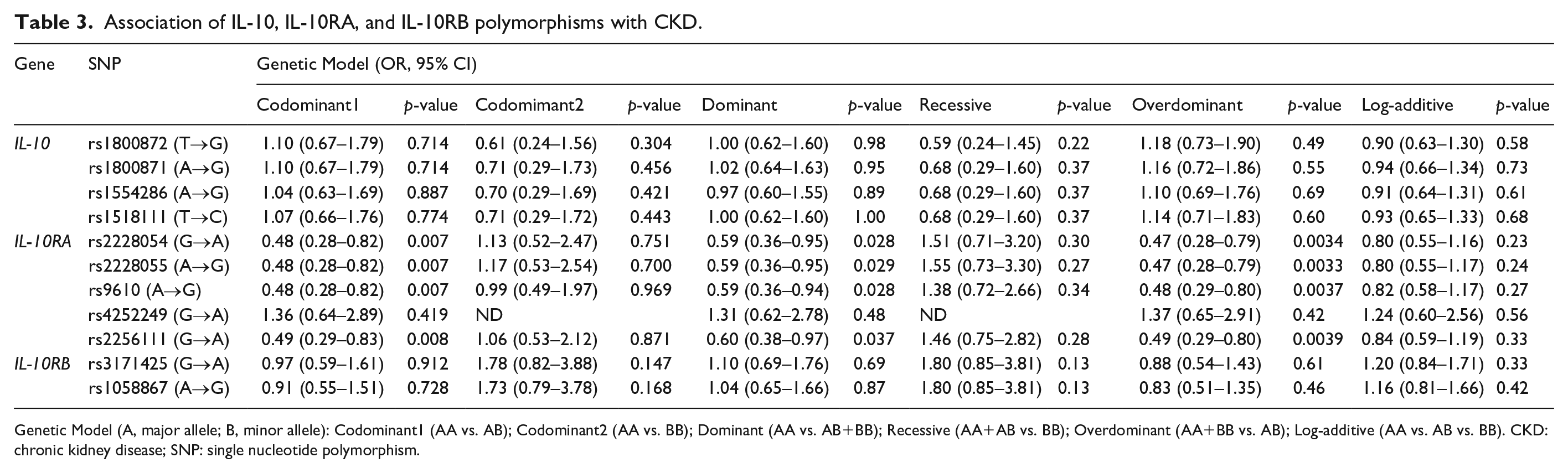
Genetic Model (A, major allele; B, minor allele): Codominant1 (AA vs. AB); Codominant2 (AA vs. BB); Dominant (AA vs. AB+BB); Recessive (AA+AB vs. BB); Overdominant (AA+BB vs. AB); Log-additive (AA vs. AB vs. BB). CKD: chronic kidney disease; SNP: single nucleotide polymorphism.
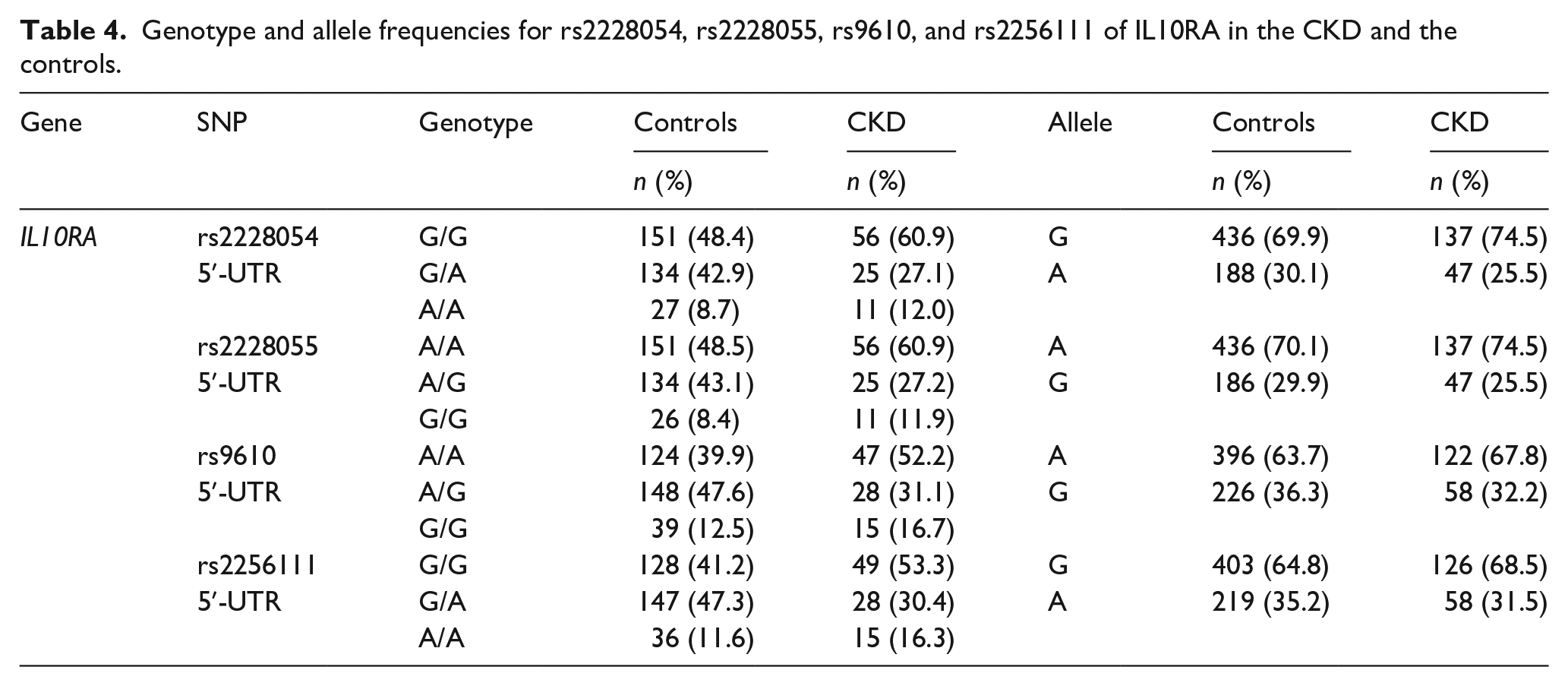
Genotype and allele frequencies for rs2228054, rs2228055, rs9610, and rs2256111 of IL10RA in the CKD and the controls.
Logistic regression analysis of IL-10, IL-10RA, and IL-10RB polymorphisms with clinical characteristics in CKD and controls
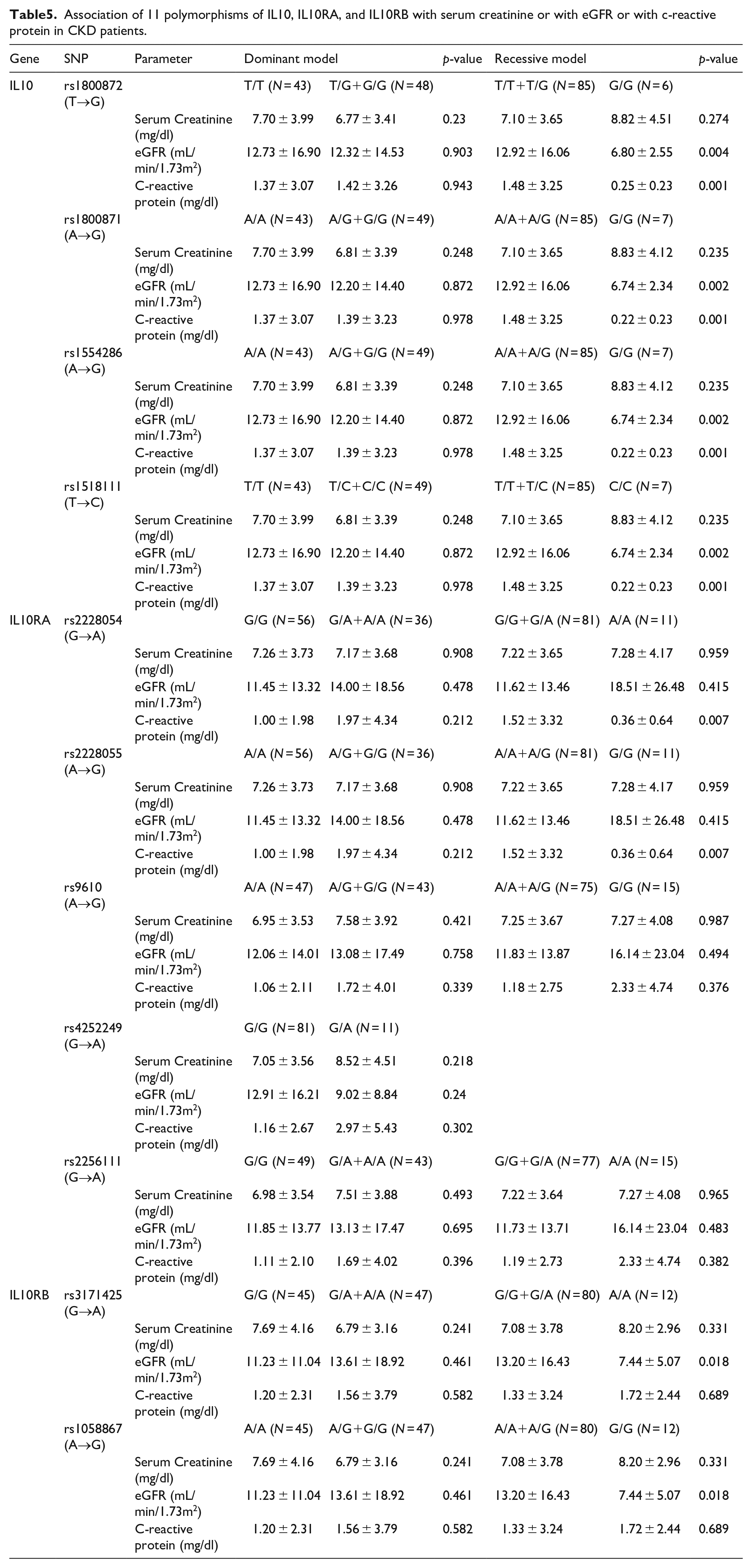
The association between 11 SNPs of IL-10, IL-10RA, and IL-10RB genes with clinical biochemical measurements (BUN, creatinine, eGFR, uric acid, total protein, albumin, WBC, and CRP) with adjustment of age, gender, BMI, and prevalence of hypertension, diabetes mellitus and dyslipidemia were evaluated. The rs1800872, rs1800871, rs1554286, and rs1518111 SNPs of IL-10 showed significant associations with the eGFR and CRP (recessive model), with the minor homozygote genotype of each SNP being associated with the lower eGFR and decreased CRP levels. The rs 2228054 and rs 2228055 SNPs of IL-10RA (recessive model) showed significant associations with the CRP levels. The rs3171425 and rs1058867 SNPs of IL-10RB were significantly associated with the eGFR (recessive model), with the minor homozygote genotype of each SNP being associated with the lower eGFR (Table 5).
Association of 11 polymorphisms of IL10, IL10RA, and IL10RB with serum creatinine or with eGFR or with c-reactive protein in CKD patients.
Haplotype analysis consisting polymorphisms in IL-10, IL-10RA, and IL-10RB Genes
In addition to SNP analyses, the haplotype was evaluated for association with CKD to determine whether the SNPs have greater predictive value when analyzed together. I estimated the LD block using a Haploview version 4.2. The LD block was made between two IL-10RB SNPs (rs3171425 and rs1058867, |D’| = 1.0,

The haplotypes (Consist of rs3171425 and rs1058867 in block1) in IL10RB, (Consist of rs4252249, rs2256111, rs2228054, rs2228055, and rs9610 in block2) in IL10RA, and (Consist of rs1554286, rs1518111, rs1800872 and rs1800871 in block3) in IL10, had frequencies greater than 0.5, and all haplotypes was used for association analysis.
Distribution of haplotypes for IL10, IL10RA, and IL10RB gene polymorphisms in CKD patients and Controls groups.
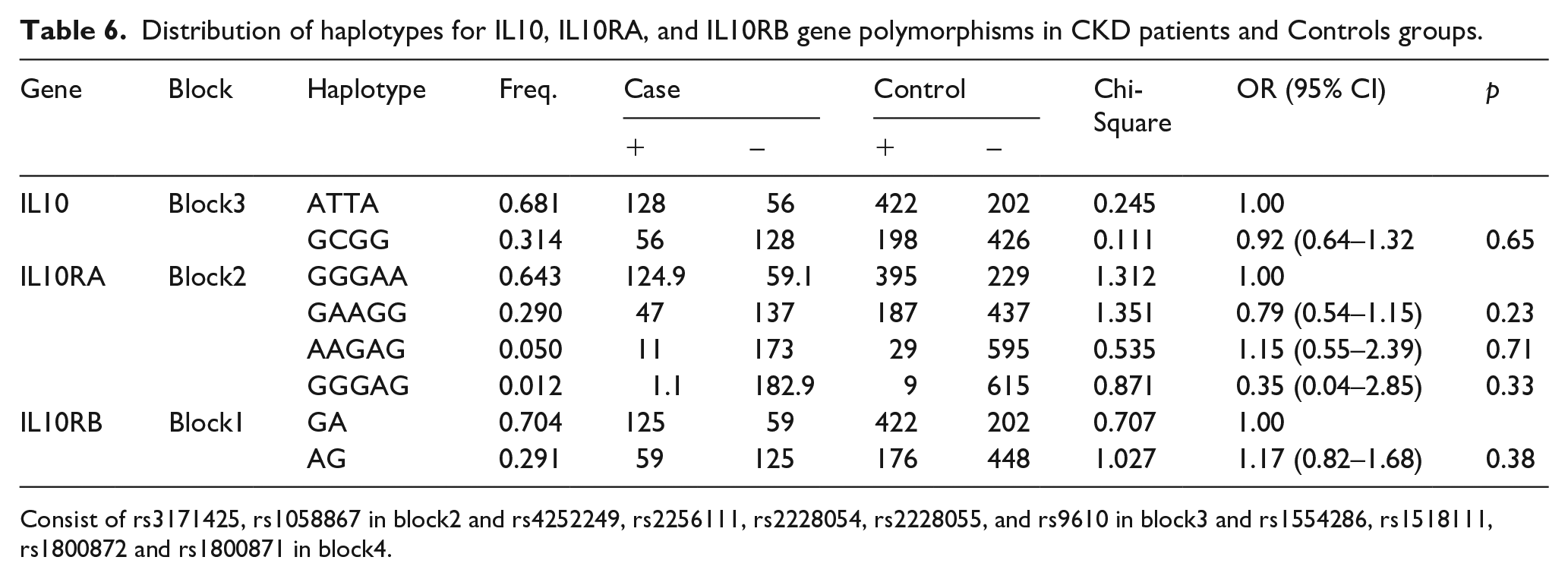
Consist of rs3171425, rs1058867 in block2 and rs4252249, rs2256111, rs2228054, rs2228055, and rs9610 in block3 and rs1554286, rs1518111, rs1800872 and rs1800871 in block4.
Discussion
In this study, the associations of IL-10, IL-10RA and IL-10RB with CKD were evaluated in Korean patients and healthy control. The study revealed that the rs2228054 (G/A), rs2228055 (A/G), rs9610 (A/G) and rs2256111 (G/A) of the IL-10RA gene were associated with CKD in codominant 1, domianat and overdominant model and SNPs of IL-10RA were associated with CRP level. IL-10 SNPs were associated with eGFR and CRP and SNPs of IL-10RB were associated with eGFR.
The delayed diagnosis of CKD can be happened since it typically has no symptoms, which means that there is a risk of further progression of CKD before it is detected. Therefore, the progression of CKD can be slowed or disrupted by early diagnosis and effective management of the disease. Around 10% of adults are affected by CKD specifically in older age groups and also CKD has a high connection with premature death as well as cardiovascular disease.14–16 In CKD patients, there are no specific symptoms in early stages and symptoms are non-specific even in later stages. Eventually, CKD patients can develop into End-Stage Renal Disease (ESRD) that calls for kidney replacement therapies which include from chronic dialysis to kidney transplantation. CKD and ESRD treatments indicate a significant socio-economic burden. 17 Therefore, identifying potential treatment targets and incipient biological risk indicators for CKD prediction is crucial.
Recently inflammation is considered one of important factors related to CKD progression and outcome. 18 Amdur RL et al. revealed inflammatory markers such as fibrinogen and TNF-α were associated with rapid loss of kidney function in patients with CKD in large cohort study. 19 Another large cross-sectional study showed that SNPs of IL-4 and IL-6 were associated with kidney function and CKD prevalence in Japanese population. 20
IL-10 is a multifunctional anti-inflammatory cytokine, mainly secreted by lymphocyte and monocyte. IL-10 interacts with interleukin 10 receptor. IL-10 receptor complex is composed of IL-10 receptor alpha (IL10RA) and beta (IL10RB). IL-10 first binds to IL-10RA, and induces a conformational change that enables IL-10RB to interact with IL-10/IL-10RA complex. 21 Polymorphisms of cytokine genes may influence gene transcription and cytokine secretion and therapy modulate the risk of progression of renal and cardiovascular diseases. 18 Association between IL10, IL10RA, and IL10RB polymorphism and several diseases have been reported. Polymorphism of IL10, IL10RA, and IL10RB genes may be contributed to the hypertension in the risk of ischemic stroke. 11 IL10RA polymorphisms (rs2228054 and rs2228055) were associated with very early-onset ulcerative colitis. 22 In the kidney, IL-10 is expressed in the mesangial cells and endothelial cells and it plays an important role in normal physiology and in the progression of chronic kidney disease. 23 IL-10 induces the proliferation of mesangial cell leading to structural intraglomerular and tubulointestitial changes, such as cellular hypertrophy, glomerular basement membrane thickening and mesangial matrix accumulation. These pathological changes were related to progression of CKD and ESRD. 23
Several studies have been shown that IL-10 was associated with various kidney diseases, glomerulonephropathies and diabetic nephropathy. IL-10 gene G-1082A polymorphism was an important marker of progression in Immunoglobulin A nephropahty and focal segmental glomerulosclerosis patients. 24 The study with white brazilians with type 2 diabetes, the −1082A>G polymorphisms in the IL-10 gene was associated with diabetic nephropathy. 25 And also, in the study with Chinese population, the IL-10 −1082G/A polymorphisms was found to be correlated with the diabetic nephropathy. 26 However, to our knowledge, no studies have been performed to evaluate the association between CKD and IL-10, IL-10RA, and IL-10RB genes polymorphism among Korean patients. In this study, IL-10RA SNPs were related with development of CKD but, IL-10RB polymorphisms were not associated with CKD.
The limitation of this study was small sample size used for comparison within CKD group, and did not included various etiology of CKD such as diabetic nephropathy, hypertension. But this study have performed for the first time genetic analysis of association between IL10RA and IL10RB and CKD.
Conclusion
Our results show that IL-10RA gene variants are associated with CKD development. Additionally, this study suggests that SNPs of IL-10RA were associated with CRP levels in CKD. Further investigations are necessary in large scale prospective CKD cohort study for determing the role of IL10RA polymorphisms.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Bisa Research Grant of Keimyung University in 2013.
Ethics approval
Ethical approval for this study was obtained from Keimyung University/Dongsan Hospital Institutional Review Board (IRB No 12-257).
Informed consent
Written informed consent was obtained from all subjects before the study.
