Abstract
Pulmonary hypertension (PAH) is one of the more serious complications of Chronic kidney disease (CKD), and its exact pathogenesis has not been clarified. As an upstream proinflammatory factor, macrophage migration inhibitor (MIF) is involved in the occurrence and development of many diseases. This study aimed to detect the relationship between serum MIF and PAH in non-dialysis CKD patients. A total of 382 non-dialysis CKD patients were enrolled in this study. Bio-Plex cytokine assay was used to detect MIF. CKD patients were divided into the PAH group and non-PAH group according to echocardiographic results. Relative risk was determined by logistic regression analysis. The pulmonary artery pressure in the CKD group was higher than that in the control group (
Introduction
Pulmonary hypertension (PAH), as a chronic disease with the long course, many complications, poor prognosis, and high mortality, is characterized by increasing resistance of pulmonary artery, which can easily lead to progressive and irreversible remodeling of pulmonary vessels and death due to right heart failure. Previous studies have shown that chronic kidney disease (CKD), especially in patients with end-stage renal disease (ESRD), has a high incidence of PAH and mortality.1,2 So far, there is no effective treatment for CKD patients with PAH.3,4 The progression of CKD can lead to inflammation and oxidative stress. 5 As the degree of kidney failure increases, the antioxidant system gradually deteriorates. 6 Inflammation and oxidative stress are the most critical defense mechanisms against infection, but without proper regulation, they may trigger many harmful effects, such as excessive production of cytokines and an increase in proinflammatory mediators. 7 A series of studies have shown that inflammatory markers are associated with CKD-related complications, such as malnutrition, heart failure and increased CKD mortality.8,9 Macrophage migration inhibitory factor (MIF) is an upstream pro-inflammatory cytokine, and MIF-173CC genotype expression is up-regulated in ESRD patients. 10 MIF is involved in the occurrence and development of many diseases, such as tumors, atherosclerosis, autoimmune diseases and so on.11–14 Studies15,16 have shown that MIF may also have organ protective effects under certain conditions, especially in acute ischemia / reperfusion injury. Recent research 17 shows that MIF can also directly promote the proliferation of pathological glomerular cells and the progress of proliferative glomerulonephritis. In contrast, in acute and chronic non immune kidney diseases, MIF has a strong cytoprotective effect on renal tubular cells, which limits the severity and progress of the disease. Due to the dual disease-specific role of MIF in renal diseases, 17 this study specifically studied the relationship between renal function and pulmonary artery pressure in the population of eastern China (Jiangsu Province), focusing on the correlation between MIF and pulmonary artery pressure in the context of renal dysfunction.
Methods
Subjects
According to our pre-experiment, the PAH incidence in CKD patients was about 15%, and among non-PAH CKD patients, those with high serum MIF accounted for 12%. Set the odds ratio (OR) as 2, α as 0.05 (bilateral), β as 0.1, and the sample size was calculated in the PASS software. In this observational study, from March 2016 to October 2018, 382 newly diagnosed non-dialysis CKD patients in the Affiliated Hospital of Nantong University were enrolled as CKD group, including 212 males and 170 females, aged (51.19 ± 14.26) years. To increase the statistical power of this study, 50 healthy people in the same period were selected as the control group, 28 males and 22 females, aged (52.09 ± 18.32) years.
Inclusion criteria and exclusion criteria
Patients in the observation group were selected based on the criteria of improving the CKD assessment and management clinical practice guidelines issued by the Global Kidney Disease Prognostic Organization (KDIGO). 18 All patients were over 18 years old. Exclusion criteria: patients with infection or trauma within 1 month, acute cardiovascular disease within 1 month, thoracic or pulmonary disease, congenital heart disease, connective tissue diseases. The control group excluded cardiovascular and cerebrovascular diseases, diabetes and other systemic diseases. This study was approved by the Ethics Committee of the Affiliated Hospital of Nantong University. All patients or their families have signed the informed consent.
Detection indicators
Peripheral venous blood was collected on an empty stomach in the morning. (1) Blood routine: red blood cell (RBC) and hemoglobin (Hb) were detected by automatic hematology analyzer (SYSMEX XE2100). (2) Blood biochemistry: Albumin (ALB), urea nitrogen (BUN), creatinine (Cr), uric acid (UA), and cysteine C (CysC) were detected by ROCHE COBAS 8000 automatic electro chemiluminescent immunoassay and its supporting reagents. Glomerular filtration rate (GFR) was estimated by the simplified Modification of Diet in Renal Disease formula. 19 (3) Detection of MIF: When fasting in the morning, 2 ml of the patient’s peripheral vein EDTA anticoagulated blood was taken, placed at room temperature for 1 h, then placed in a refrigerator at 4°C for 1 h, centrifuged at 3000 rpm for 10 min, and the supernatant was frozen and stored in the refrigerator for testing. MIF was detected using a BioPlex human cytokine multiplex kit (Bio-Rad, CA, USA) by liquid phase chip technology. (4) Echocardiographic: the tricuspid regurgitation velocity was measured by two-dimensional and M-mode echocardiography using GE Vivid 7 color Doppler ultrasound diagnostic instrument (probe frequency 2.5 MHz). PAP (mmHg) = 4 × (maximum tricuspid regurgitation velocity) 2 + 10 mmHg (right atrial pressure estimation), 20 PAP (⩾ 40 mmHg) was diagnosed as PAH. 21
Statistical analysis
Results
General clinical data
There was no significant difference in gender, age, diastolic blood pressure (DBP), RBC and ALB between the CKD and control groups (
General clinical data of the two groups.

CKD: chronic kidney disease; SBP: systolic pressure; DBP: diastolic blood pressure; Hb: hemoglobin; RBC: red blood cell; ALB: albumin; Cr: creatinine; BUN: urea nitrogen; CysC: cystatin C: GFR: glomerularfiltrationrate.
Compared with the control group,
Comparison of pulmonary artery pressure
The incidence of PAH in the CKD group was 15.18% (58/382), while no PAH occurred in the control group (0/50). The pulmonary artery pressure in the CKD group was higher than that in the control group, and there was a significant difference between the two groups (
Serum MIF levels in the two groups
The serum MIF level of the CKD group was 786.23 (50.97–5457.00) pg/ml, which was higher than that of the control group 300.13 (45.02–1908.52) pg/ml, and there was significant difference between them (

Comparison of pulmonary artery pressure. (a) The pulmonary artery pressure in the CKD group was higher than that in the control group (
Correlation analysis of serum MIF with clinical indicators
Spearman rank correlation analysis showed that serum MIF levels in the observation group were positively correlated with BUN (
Spearman correlation analysis of MIF level and clinical indicators.
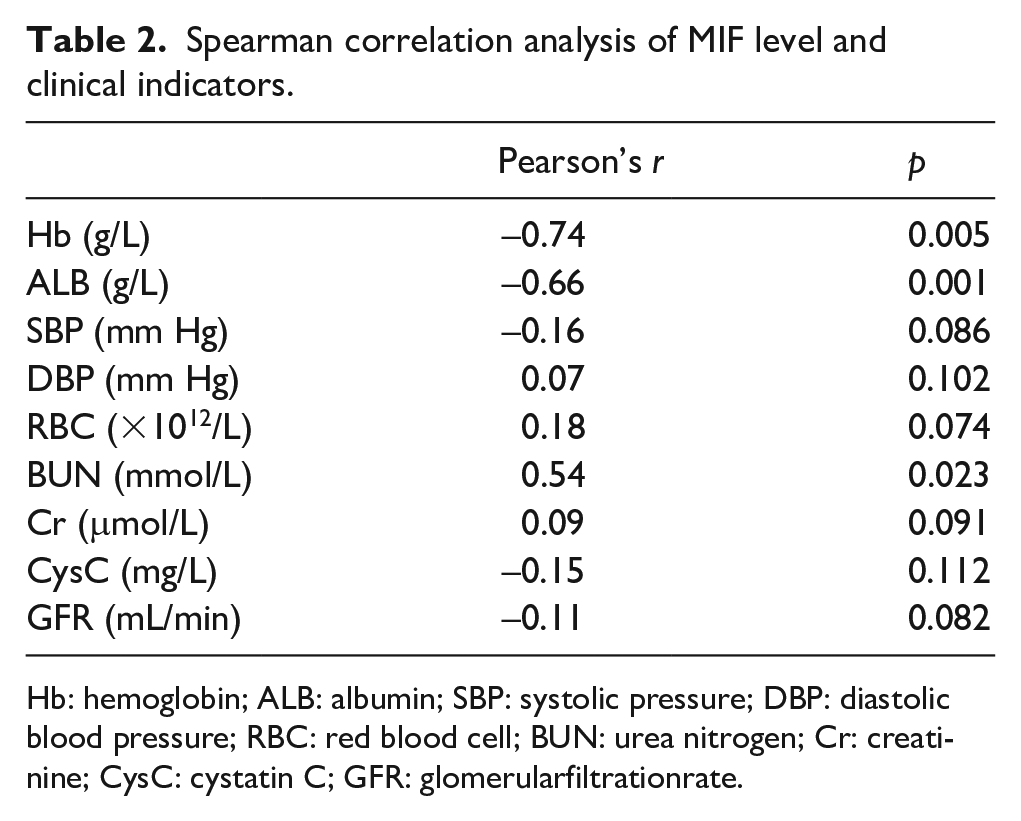
Hb: hemoglobin; ALB: albumin; SBP: systolic pressure; DBP: diastolic blood pressure; RBC: red blood cell; BUN: urea nitrogen; Cr: creatinine; CysC: cystatin C; GFR: glomerularfiltrationrate.
Comparison of serum MIF levels between the two groups
CKD Patients were divided into PAH group (
Mann-whitney test of PAH and related indicators.
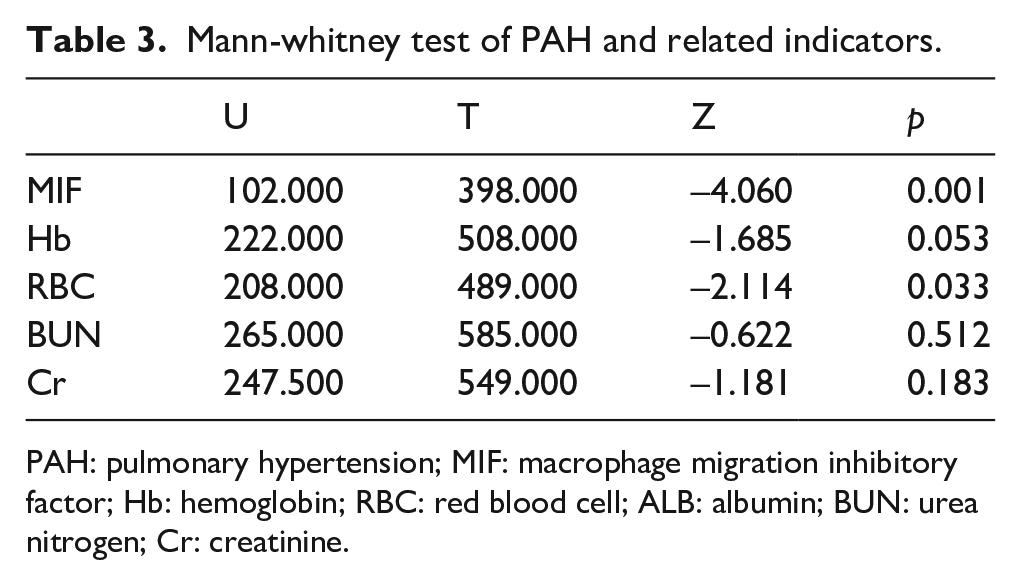
PAH: pulmonary hypertension; MIF: macrophage migration inhibitory factor; Hb: hemoglobin; RBC: red blood cell; ALB: albumin; BUN: urea nitrogen; Cr: creatinine.
Multivariate logistic regression analysis
Referring to previous literature, in our study, serum MIF < 1100 pg / ml was considered normal, and serum MIF > 1100 pg/ml was considered abnormal.
22
With PAH as the dependent variable and Sex, Age, BP, MIF, Hb, RBC, ALB, BUN, and Cr as independent variables, the logistic regression model was established. The results showed that MIF entered the regression model with statistical significance (
Discussion
When pulmonary artery pressure increases gradually under the influence of various factors, it will lead to the increase of right ventricular afterload, myocardial remodeling, right ventricular hypertrophy, which will lead to right heart and whole heart failure, and even death. 21 In the present study, we have demonstrated that 15.18% (58/382) of non-dialysis CKD patients were complicated with PAH, and the pulmonary artery pressure was higher in the CKD group than in the control group. Thus, these results suggest that CKD patients have a higher incidence of PAH and increased pulmonary arterial pressure. Our subgroup analysis showed that pulmonary artery pressure was higher in the stages 4 to 5 CKD group than in the stages 1 to 3 CKD group. Suggesting that as CKD progressed, pulmonary arterial pressure rised and the incidence of PAH increased. Our results from the univariate correlation analysis showed that pulmonary artery pressure was negatively correlated with GFR in CKD patients. Therefore, we can postulate that pulmonary arterial pressure was correlated with the deterioration of renal function, and the smaller the GFR, the more obvious the increase of pulmonary arterial pressure. Havlucu et al. reported that 39.1% of the 23 non-dialysis CKD patients had increased pulmonary arterial pressure (PAP ⩾ 35 mmHg), which also confirmed the trend of increased pulmonary arterial pressure in non-dialysis CKD patients. 23 However, the prevalence of PAH in this study was higher than in our study, which may be due to the small number of cases in this study and the lower diagnostic criteria for PAH.
MIF is a pluripotent cytokine that does not belong to any cytokine family. It can attract macrophages and T lymphocytes to infiltrate and accumulate in inflammation and enhance their phagocytic function.24,25 It was shown in the present study that MIF in CKD patients was higher than that in healthy controls. This indicates that MIF may be closely associated with renal disease in these patients. 26 The causes of microinflammation in non-dialysis CKD patients may be the following: The clearance of pro-inflammatory cytokines is reduced, resulting in the accumulation of pro-inflammatory cytokines and their metabolites in vivo. The weakening of the intestinal barrier aggravates the absorption of endotoxin in the intestine, which leads to the release of inflammatory mediators by activating the complement system and inflammatory cells.27,28 Unlike previous studies, 29 we found that PAH in non-dialysis CKD patients was significantly positively correlated with MIF, regardless of age, gender, blood pressure, etc. The results address that MIF in CKD patients were significantly higher than those in the control group. Further, we demonstrated serum MIF was significantly correlated with PAH. It suggests that high serum MIF is an important risk factor for PAH and may increase the incidence of PAH in patients with non-dialysis CKD. Our results suggest that high serum MIF is an important risk factor for PAH, which may increase the risk of PAH occurrence in non-dialysis CKD patients. It was found that the inflammatory response is significantly reduced after inhibition or neutralization of MIF.30,31 The transmembrane protein receptor CD74+ in MIF molecule can bind to its extracellular part with a high affinity, thus activating the intracellular signal transduction pathway, causing the synthesis and release of various cell proliferation and inflammatory mediators, and exerting various physiological effects. Anti-cd74+ antibody (LN2 or MB741) can significantly inhibit the above action pathway of MIF. 32 Our findings suggest that MIF plays an important role in the development of PAH in non-dialysis CKD patients. It can be inferred that blocking the effect of MIF in non-dialysis CKD patients may reduce the degree of PAH. Our study also suffers from some limitations. The specific mechanism of MIF affecting PAH needs further study.
Conclusion
In summary, our data indicate that serum MIF levels are positively correlated with pulmonary artery pressure in non-dialysis CKD patients, suggesting that serum MIF may be involved in the occurrence and development of pulmonary hypertension in CKD patients, and blocking the role of MIF in CKD patients may reduce the degree of PAH and PAH-related mortality.
Footnotes
Authors’ contributions
Jianhua Wu designed the study and drafted the manuscript. Naifeng Guo participated in data collection. Xiaolan Chen performed statistical analyses. All authors critically revised the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from *ETHICS COMMITTEE OF AFFILIATED HOSPITAL OF NANTONG UNIVERSITY (2016K030)*.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Nantong technology projects grants (MS12017002-2)
Informed consent
Written informed consent was obtained from all subjects before the study.
Trial registration
This randomized clinical trial was not registered because * It is an observational clinical study, not an interventional one *.
