Abstract
Z-ligustilide (LIG) is the main bioactive compound of Danggui essential oil, which was reported to exert neuroprotective and anti-inflammatory effects. However, the underlying mechanism remains largely elusive. The present study aims to investigate the effect of LIG on oxygen-glucose deprivation/reoxygenation (OGD/R)-induced injury and whether Nod-like receptor protein 3 (NLRP3) inflammasome and related pyroptosis are targets for the treatment of LIG. The OGD/R model was established in BV-2 microglial cells to investigate the protective effect of LIG. Cell viability and the release of lactate dehydrogenase (LDH) were determined by cell counting assay kit 8 and the LDH release assay kit. Western blot and immunofluorescence staining were carried out to detect NLRP3 inflammasome activation and pyroptosis. Active caspase-1 and TdT-mediated dUTP nick end labeling (TUNEL) double positive cells were defined as pyroptosis population. Statistical comparison among multiple groups was carried out by one-way analysis of variance (ANOVA) followed by least significant difference (LSD) test. Compared with control cells, OGD/R impaired cell viability and induced the release of LDH in BV-2 microglial cells, which were associated with the activation of NLRP3 inflammasome as evidenced by increased expression of NLRP3 and the cleavage of caspase-1 and interleukin-1 beta (IL-1β). In parallel with NLRP3 inflammasome activation, OGD/R induced pyroptotic cell death, manifested by the cleavage of gasdermin D (GSDMD) and increased population of active caspase-1+/TUNEL+ cells. All these events were significantly attenuated by treatment with LIG, indicating that LIG significantly inhibited NLRP3 inflammasome activation and pyroptosis, and ameliorated OGD/R-induced cell injury. In conclusion, LIG protects BV-2 microglial cells against OGD/R-induced injury via inhibition of NLRP3 inflammasome and pyroptosis.
Introduction
Radix angelicae sinensis is the dried foot of
Microglia are the key innate immune cells and sensitive to the change of microenvironment in the central nervous system. 6 Activated microglia, which normally respond to neuronal damage or LPS stimuli in vitro, are known to release a number of proinflammatory cytokines such as IL-1β.7,8 Activation of microglia is a hallmark of brain pathology. Accumulating evidence implicated that microglia-mediated inflammatory response played an important role in the pathogenesis of neurodegenerative diseases, ischemic stroke, and chronic pain. 9
Inflammasomes are cytosolic multiprotein oligomers of the innate immune system responsible for the activation of inflammatory responses.10,11 The Nod-like receptor protein 3 (NLRP3) inflammasome is composed of NLRP3, the adaptor protein apoptosis-associated speck-like protein containing a caspase recruitment domain (ASC) and inflammatory caspase-1. NLRP3 inflammasome activation mediates the activation of caspase-1, which further catalyzed the maturation of IL-1β. NLRP3 inflammasome activation was crucial for mediating inflammatory response in the process of ischemic stroke.12 –14 Recent finding indicated that NLRP3 inflammasome was activated in response to oxygen-glucose deprivation/reoxygenation (OGD/R) injury in microglia. 15 Besides, caspase-1 after its activation triggered programmed necrosis termed pyroptosis. The defining features of pyroptosis consist of pore formation, osmotic swelling, and early loss of membrane integrity. 16 Gasdermin D (GSDMD) serves as a specific substrate of inflammatory caspase-1 and its N-terminal cleavage product (GSDMD-NT) was identified as the principal executor of pyroptosis by inducing the formation of membrane pores. Although pyroptosis was first associated with infectious diseases, emerging evidence showed the involvement of pyroptosis in alcoholic hepatitis, cerebral and renal ischemia-reperfusion injury.8,17,18 It was reported that OGD/R induced pyroptotic cell death of neurons. 19 Therefore, targeting the NLRP3 inflammasome and related pyroptosis may develop a novel therapeutic strategy to suppress the excessive inflammatory response of OGD/R injury.
The present study was conducted to investigate the anti-inflammatory effect of LIG in OGD/R-induced BV2 microglial cells and the potential mechanism involved.
Methods
Reagents
LIG (purity ⩾ 98%) was purchased from MedChemExpress (New Jersey, USA). LIG was dissolved in dimethyl sulfoxide (DMSO) for stock preparation, and the concentration of 0.05% DMSO was used as the control. LIG bears a typical electrophilic unsaturated lactone structure (Figure 1).

Chemical structure of Z-ligustilide.
Oxygen-glucose deprivation/reoxygenation
BV-2 microglial cell lines were obtained from China Center for Type Culture Collection (Wuhan, China). BV-2 cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) containing fetal bovine serum (FBS) (10%) (GIBCO, Carlsbad, CA, USA), penicillin (100 U/mL), and streptomycin (100 mg/L). BV-2 cells were incubated with 1, 2.5, 10 μmol/L of LIG (MedChemExpress), respectively, during the exposure of OGD/R injury. Briefly, the cells were cultured in DMEM medium without glucose (GIBCO) and placed inside a chamber with a premixed gas (0.2% O2, 94.8% N2, 5% CO2) for 3 h. Then BV-2 cells were sent back to basic DMEM medium supplemented with 25 mmol/L D-glucose (GIBCO) in a normoxic cell culture incubator for 24 h.
Cell viability and lactate dehydrogenase assays
After OGD/R for 24 h, cell viability was determined using cell counting assay kit 8 (CCK-8 Kit, Beyotime, Beijing, China). The absorbance was measured at 450 nm with a reference wavelength at 650 nm using a microplate reader (Thermo fisher scientific, MA, USA). Cell injury was determined using the lactate dehydrogenase (LDH) release assay kit (Beyotime, Beijing, China) according to the manufacturer’s instruction. The absorbance of samples was measured at 490 nm. The results are expressed as the percentage of LDH release relative to the control cells.
Analysis of cell pyroptosis
In brief, OGD/R-induced cells cultured chamber slides were fixed with 4% paraformaldehyde, blocked with 10% FBS and permeabilized with Triton X-100. Slides were then double immunofluorescent stained with TdT-mediated dUTP nick end labeling (TUNEL) (Beyotime, Beijing, China) and anti-active caspase-1 antibody (1:100, Merck Millipore, Darmstadt, Germany). After washing with phosphate buffer saline (PBS), slides were incubated with Alexa-488-labeled secondary antibody for 1 h at room temperature. After being mounted with DAPI-containing mounting solution, the slides were subjected to examinations using a confocal laser scanning microscope. Active caspase-1+/TUNEL+ cells were designated as pyroptotic cells.
Western blot
Protein samples were extracted from BV-2 cell lysates. Western blot procedures were carried out using standard protocol with specific antibodies against NLRP3 (1:1000, Adipogen, Liestal, Switzerland), caspase-1 (1:1000, Abcam, Cambridge, UK), IL-1β (1:1000, Cell Signaling Technology, MA, USA), and GSDMD (1:1000, Abcam, Cambridge, UK). Incubation with β-actin antibody (1:2000, Sigma, USA) was served as endogenous reference. The blots were visualized using a ChemiDoc™ Touch Imaging System (Bio Rad, CA, USA). Quantification of band intensity was carried out using Image J software (NIH, Bethesda, MD, USA).
Statistical analysis
Data are shown as the mean ± SD. All data were from at least three independent experiments. Statistical comparison among multiple groups was carried out by one-way analysis of variance (ANOVA) followed by least significant difference (LSD) test. Data were analyzed using Graphpad Prism 6.02 software. Differences were considered significant for
Results
Z-ligustilide protected BV-2 cells against OGD/R-induced injury
Compared with control cells, OGD/R significantly induced cell injury in BV-2 cells as evidenced by the decreased cell viability (

LIG attenuated OGD/R-induced cell injury in BV-2 microglial cells. BV-2 cells were treated with different concentrations of LIG and then exposed to OGD/R. (a) Cell viability and (b) LDH release in BV-2 microglial cells.
Z-Ligustilide suppressed OGD/R-induced NLRP3 inflammasome activation in BV-2 cells
To further evaluate the role of LIG in NLRP3 inflammasome activation, BV-2 microglial cells were subjected to OGD/R injury in the presence or absence of LIG. Our data showed that OGD/R significantly increased the expression of NLRP3 (

LIG suppressed OGD/R-induced NLRP3 inflammasome activation in BV-2 microglial cells: (a) Representative blots and (b-d) quantitative analysis showing the expression of NLRP3 and the cleavage of caspase-1 and IL-1β.
Z-Ligustilide suppressed OGD/R-induced pyroptosis in BV-2 cells
We then investigated the effects of LIG on pyroptosis in OGD/R microglial cells in the absence or presence of LIG. Our data showed that OGD/R significantly increased the expression of GSDMD (
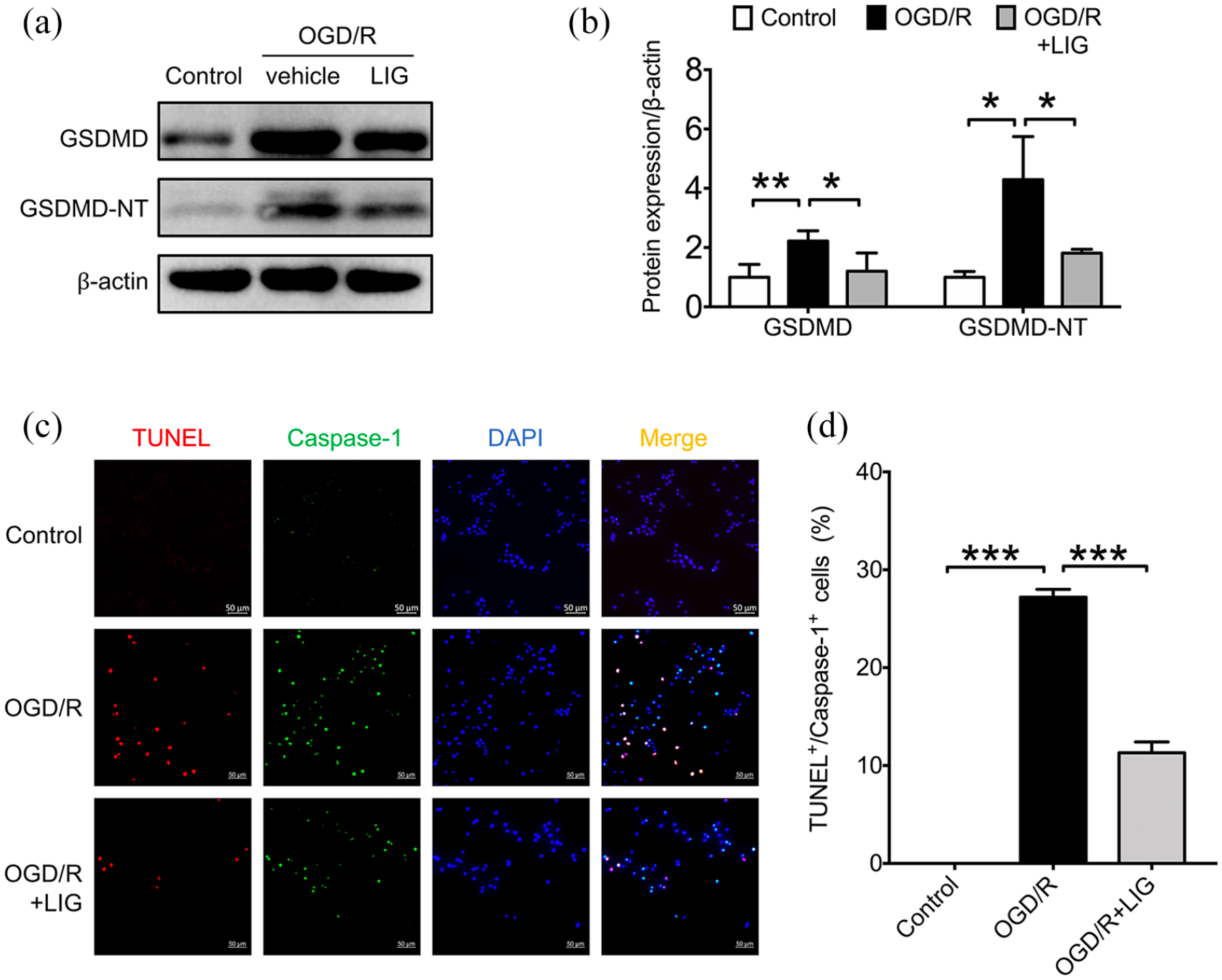
LIG suppressed OGD/R-induced pyroptosis in BV-2 microglial cells: (a) Representative blots and (b) quantitative analysis showing the expression and cleavage of GSDMD. (c) Representative confocal microscopic images showing the colocalization of TUNEL (red) with active caspase-1 (green) and (d) the percentage of active caspase-1+/TUNEL+ cells (pyroptotic cells).
Discussion
In this study, we identified that OGD/R-induced injury was associated with NLRP3 inflammasome activation and pyroptosis in microglia. Furthermore, we demonstrated for the first time that LIG protected BV-2 cells against OGD/R-induced cell injury by inhibiting NLRP3 inflammasome activation and related pyroptosis.
Microglia are the major resident immune cells in the brain. Microglia can be activated by either single or multiple exposures to LPS, hypoxia, brain injury, and so on. Emerging evidence showed that microglia activation triggered deleterious inflammatory reaction, resulting in excess production of inflammatory cytokines (such as IL-1β). 20 In the present study, the OGD/R model was established in BV-2 microglial cells to investigate the neuroprotective effects of LIG. OGD/R exposure remarkably impaired cell viability and increased LDH release in BV-2 microglial cells. It was reported that LIG protected PC12 cells, SHSY-5Y cells, and neurons from OGD/R-induced injury.21–23 Correspondingly, we uncovered the protective effect of LIG on OGD/R-induced microglial cell injury, indicating the therapeutic potential of LIG for ischemic stroke. However, the precise molecular mechanisms underlying the neuroprotective effect of LIG have not been fully elucidated.
Recent findings implicated that NLRP3 inflammasome activation was originally participated in the process of brain damage caused by neuroinflammation in ischemic stroke.24,25 Pringle et al. 26 reported that IL-1β strongly potentiated hypoxia-induced neuronal damage. Consistent with these findings, we found that NLRP3 inflammasome was markedly activated as evidenced by the cleavage of caspase-1 and IL-1β in OGD/R microglial cells. Previous study demonstrated that NLRP3 deficiency or inhibitor attenuated ischemic damage in vivo and OGD/R-induced cell injury in vitro.14,27 In addition, the suppression of NLRP3 inflammasome activation reduced OGD/R-induced neurovascular unit dysfunction in mouse brain microvascular endothelial cells. 28 Emerging evidence identified that treatment with LIG significantly inhibited proinflammatory cytokines IL-1β and TNF-α in experimental ovariectomized osteopenic rats and human osteoarthritis chondrocytes.5,29 In the present study, we demonstrated for the first time that treatment with LIG significantly inhibited OGD/R-induced NLRP3 inflammasome activation. These results indicated that the protective effect of LIG on OGD/R-injured microglial cells was associated with the inhibition of NLRP3 inflammasome.
The assembly of NLRP3 inflammasome triggers the autocleavage of pro-caspase-1 into active caspase-1. In addition, caspase-1 cleaves GSDMD to yield GSDMD-NT, which critically determined the fate of pyroptotic cell death.30,31 Poh et al. 32 reported that inflammasome contributed to pyroptotic cell death in microglia following cerebral ischemia. Previous studies also demonstrated that microglial cells and astrocytes suffered from pyroptotic death in OGD/R injury.33,34 Consistent with these findings, we found that OGD/R promoted the cleavage of GSDMD in BV-2 microglial cells, indicating the presence of pyroptosis.
Pyroptotic cells share some common features with apoptotic cells, including DNA fragmentation which can be labeled by TUNEL staining. 35 To identify pyroptosis in OGD/R-treated microglia, double-immunostaining of active caspase-1 and TUNEL was performed in our study. Our result showed that OGD/R induced about 27.2% of pyroptotic cells in BV-2 cells. We further disclosed that treatment with LIG significantly reduced OGD/R-induced microglial pyroptosis. Dai et al. 36 reported that inhibition of microglial pyroptosis attenuated inflammatory reaction in acute spinal cord injury in rats, as evidenced by decreased expression of NLRP3, ASC, caspase-1, and GSDMD and the release of IL-1β. Previous studies also reported that the inhibition of NLRP3-mediated microglial pyroptosis attenuated radiation-induced brain injury and cerebral ischemia-reperfusion injury.19,37 Our results indicated that LIG might have protective effect on cerebral ischemia-reperfusion injury. A limitation to the present study is that we have not provided therapeutic evidence of LIG for ischemic stroke in vivo. Further study should be needed to characterize the detailed influence of LIG on NLRP3 inflammasome and pyroptosis in ischemic stroke animal model.
Conclusion
The present study demonstrated for the first time that treatment with LIG ameliorated OGD/R-induced microglial cell injury by inhibiting NLRP3 inflammasome activation and pyroptosis, highlighting the therapeutic potential of LIG on diseases which are associated with NLRP3 inflammasome and microglial pyroptosis.
Footnotes
Author contributions
J.H.: data collection and interpretation, and writing-original draft preparation. J.W. and C.Z.: visualization and investigation. F.D.: methodology and software. S.L.: software and validation. H.T.: funding, conceptualization, writing-reviewing, and editing. All authors read and approved the final manuscript.
Availability of data and materials
The datasets used and/or analyzed in the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Guangdong province (grant number 2017A030311017), the National Natural Science Foundation of China (grant numbers 81873514, 81570394 and 81370371), and Guangxi Natural Science Foundation (2018GXNSFAA294102).
