Abstract
The aim of this study was to investigate the mechanisms of combination treatment with FK506 and natamycin on alleviating damage of the cornea in mouse model of fungal keratitis.
In this study, the mouse model of fungal keratitis was created by intrastromal injection with Fusarium solani or Aspergillus flavus. The mice received 5% natamycin eye drops 6–8 times a day, or the mice received 0.05% FK506 eye drops 2 times per day for 21 consecutive days. Corneal damage was evaluated by H&E staining. The protein expression levels of NLRP3 were detected by immunohistochemistry. Moreover, the markers of inflammasome activation including NLRP3, ASC, caspase-1, IL-1β, and IL-18 were detected by western blot.
Histopathological results showed increased corneal thickening, dense inflammatory cell infiltration, and loss of epithelial continuity in the corneas after fungal infection. In addition, NLRP3 positive signals were observed to be obviously increased in the corneas after A. flavus or F. solani infection compared to the control group. Furthermore, the NLRP3 inflammasome is induced by fungal infection, as evidenced by increased protein expression levels of NLRP3, ASC, caspase-1, and downstream cytokines, such as interleukin (IL)-1β and IL-18. However, the corneal damage was alleviated and the activation of the NLRP3 inflammasome was significantly inhibited by drug treatment. Besides, the treatment outcomes were better in combined treatment group than that in single-agent treatment group.
In conclusion, FK506 combined with natamycin alleviate fungi-induced corneal damage by suppressing NLRP3 inflammasome activation.
Introduction
Fungal keratitis is an important cause of blindness and visual impairment worldwide. 1 The incidence of fungal keratitis has increased in recent years, especially in contact lens wearers. 2 In China, the most common pathogens are Fusarium solani and Aspergillus flavus. 3 Current practice in treating fungal keratitis involves the topical antifungal drops, such as natamycin. 4 However, it has limitations, such as recurrent corneal epithelial erosions and chemosis. 5 To overcome these limitations, novel antifungal agents are being tried to improve outcomes after fungal infections.
Fungal infection may activate one or more members of the inflammasome family. 6 Among inflammasome family, the nucleotide-binding oligomerization domain (NOD)-like receptor containing pyrin domain 3 (NLRP3) inflammasome is induced upon exposure to pathogens. 7 Upon activation, the NLRP3 proteins lead to recruitment of the adapter apoptosis-associated speck-like protein containing a C-terminal caspase recruitment domain (ASC), in order to cause the activation of pro-caspase-1 into its cleaved form. 8 Caspase-1 plays a critical role in the maturation of IL-1β and IL-18 into active cytokines. 9 NLRP3 has a regulatory and beneficial role in herpetic stromal keratitis pathogenesis. 10 However, the role of NLRP3 inflammasome in fungal keratitis remains unclear.
Tacrolimus (FK506), a strong immunosuppressant drug, is isolated from Streptomyces tsukubaensis. 11 Topical administration of FK506 was effective in the treatment of many inflammatory skin diseases, such as eczema 12 and atopic dermatitis. 13 Moreover, it has been demonstrated that FK506 may inhibit the inflammation induced by fungi and alleviate the severity of corneal damage at an early stage of fungal keratitis. 14
In this study, we sought to investigate the mechanisms of combination treatment with FK506 and natamycin on alleviating damage of the cornea in mouse model of fungal keratitis.
Materials and methods
Animals
All the animal studies were approved by the Ethics Committee of Shangdong University, and animal experiments were performed in accordance with the Association for Research in Vision and Ophthalmology Statement for the Use of Animals in Ophthalmic and Vision Research. Female C57BL/6 mice (6- to 8-week old) were purchased from the Laboratory Animal Center of Shandong University (Jinan, Shandong, China).
Mouse model of Aspergillus and Fusarium keratitis
Aspergillus flavus (A. flavus) and Fusarium solani (F. solani) strains were cultured as described previously for 2–3 days. 15 Conidia were harvested and adjusted to a final concentration of 5 × 104 conidia/μL in phosphate-buffered saline (PBS). Mice were anesthetized with 10% chloral hydrate, and the corneal epithelium was abraded using a 30-gauge needle. Subsequently, 2 µL f 1 × 105 conidia in PBS was injected into the corneal stroma of the left eye of each animal using a 33-gauge Hamilton syringe. 16 The mice without fungal keratitis were used as control (n = 6).
Drug treatment and grouping
Forty-eight hours after inoculation, in the first experiment set, the mice infected with A. flavus were divided into 5 different treatment groups, 10 mice of each group: A. flavus group, saline-treated group, natamycin-treated group, FK506-treated group, and a combination of natamycin and FK506 groups.
In the second experiment set, the mice infected with F. solani were divided into 5 different treatment groups, 10 mice of each group: F. solani group, saline-treated group, natamycin-treated group, FK506-treated group, and a combination of natamycin and FK506 groups.
Mice in the saline group were treated with saline solution 6–8 times a day up to 21 days. The mice in the natamycin group were treated with 5% natamycin 6–8 times a day up to 21 days. The mice in the FK506 group received 0.05% FK506 solution 2 times per day for 21 consecutive days. In addition, the preparation of FK506 solution was performed as described previously. 17 And the mice in the natamycin + FK506 group were treated with 5% natamycin 6–8 times combined with 0.05% FK506 solution 2 times a day up to 21 days. The eye drop, each time 1 drop (25 μL), was administered to both eyes of the mice for 21 days. Then, the animals were sacrificed by isoflurane anesthesia followed by cervical dislocation. And their eyes were enucleated for pathologic examination.
Histology
For histology, the rat corneas were fixed in 4% formaldehyde and processed for paraffin embedding. Sections with a thickness of 5 µm were then cut and stained with hematoxylin and eosin (H&E; Sigma–Aldrich). Histology evaluation was performed under a Nikon Eclipse 50i microscope (Nikon Instruments).
Immunohistochemical staining
Immunohistochemical (IHC) staining was performed as described previously. 4 The corneas were excised from sacrificed mice and were fixed in 4% paraformaldehyde for 10 min. After blocked with 10 mM PBS, containing 2% bovine serum albumin (BSA), the sections were then incubated with primary antibodies: rabbit anti-mouse NLRP3 (1:100; Santa Cruz Biotechnology) overnight at 4°C, followed by the secondary antibody, biotinylated goat-anti-rabbit in IgG (1:100; eBiosciences) at 37°C for 1 h. For all slides, the immune reaction was demonstrated with DAB (Sigma) and examined by light microscopy.
Western blot analysis
Proteins of corneas were extracted from a radioimmunoprecipitation (RIPA) buffer supplemented with a protease inhibitor cocktail (Roche). Equal amounts of proteins (30 μg) were subjected to electrophoresis in 10% SDS-PAGE gels (Bio-Rad) and electro-transferred to nitrocellulose membranes. Subsequently, membranes were blocked with 5% nonfat milk and then incubated with primary antibodies overnight at 4°C. After washing with PBS containing 0.1% Tween 20, membranes were incubated with horseradish peroxidase-conjugated secondary antibodies. The bands were visualized with enhanced chemiluminescent (ECL; Thermo Scientific). β-actin was used as a loading control.
Results
Establishment of Aspergillus and Fusarium keratitis mouse model
To assess the histological changes in corneas after A. flavus or F. solani infection, H&E staining of corneal sections was performed. As shown in Figure 1(a), the eyes maintained the normal architecture of the cornea, preserving the epithelium, endothelium, and stroma in the control group. However, after 48 h of A. flavus or F. solani infection, the eyes showed disruption of the tissue architecture, presence of endothelial plaques, and increased corneal thickness. Moreover, the histopathological changes in the corneas in F. solani group were more serious than that in A. flavus group (Figure 1(a)). These results suggested that the mouse model of Aspergillus and Fusarium keratitis was established successfully.

Histological changes in corneas and the expression levels of NLRP3 in the corneas after A. flavus or F. solani infection for 48 h. (a) Representative pictures of H&E staining of corneas; magnification: ×40. (b) Immunohistochemistry analysis for NLRP3 expression in corneas; magnification: ×40.
A. flavus or F. solani infection induces NLRP3 inflammasome activation in corneas
To evaluate the activation of the NLRP3 inflammasome in corneas, we assessed the protein expression levels of NLRP3 by IHC staining. The results showed that NLRP3-positive signals were observed to be obviously increased in the corneas after A. flavus or F. solani infection compared to the control group. In addition, the expression levels of NLRP3 were higher in F. solani group than that in A. flavus group (Figure 1(b)).
Next, the markers of inflammasome activation including NLRP3, ASC, caspase-1, and the proinflammatory cytokines IL-1β and IL-18 were detected by western blot. As shown in Figure 2, protein expressions of NLRP3, ASC oligomerization (oligomer, trimer, dimer, and monomer forms), pro-caspase-1 (p45), cleaved caspase-1 (p10), pro-IL-1β, IL-1β, and IL-18 were markedly increased in corneas after A. flavus or F. solani infection compared to the control group. Moreover, the expression levels of NLRP3, ASC (oligomer, trimer, dimer, and monomer forms), cleaved caspase-1 (p10), IL-1β, and IL-18 were higher, while pro-caspase-1 (p45) and pro-IL-1β were lower in F. solani group than that in A. flavus group (Figure 2), indicating that A. flavus or F. solani infection triggered activation of the NLRP3 inflammasome in corneas.

A. flavus or F. solani infection triggered NLRP3 inflammasome activation in corneas. The mice were infected with A. flavus or F. solani for 48 h. The expression levels of NLRP3, ASC, caspase-1, IL-1β, and IL-18 in corneas were detected by western blot.
Combination treatment with FK506 and natamycin alleviates A. flavus-induced activation of the NLRP3 inflammasome
Histopathologic studies of corneas following 21 days of infection with A. flavus showed a large infiltrate of inflammatory cells and obvious edema in corneas, whereas there was much less inflammation and edema and decreased corneal thickness in corneas of mice with single-agent treatment or combination treatment (Figure 3(a)). And the tissue architecture was better in combination treatment group compared to that in single-agent treatment group.

Effects of FK506 combined with natamycin on the corneas infected with A. flavus by suppressing NLRP3 inflammasome activation. (a) Representative pictures of H&E staining of corneas; magnification: ×40. (b) Immunohistochemistry analysis for NLRP3 expression in corneas; magnification: ×40. (c) Expression levels of NLRP3, ASC, caspase-1, IL-1β, and IL-18 in corneas were detected by western blot.
To determine whether the drug treatment could alleviate A. flavus-induced activation of the NLRP3 inflammasome, we further investigated NLRP3 expression in corneas. The results showed that compared to A. flavus group, NLRP3 protein expression significantly decreased after FK506 or natamycin treatment or after FK506 combined with natamycin treatment (Figure 3(b)). Moreover, the expressions of NLRP3 were confirmed by western blot (Figure 3(c)). In addition, NLRP3 protein expression levels were lower by combined treatment compared to that by FK506 alone or natamycin alone (Figure 3(b) and (c)).
Similarly, protein expressions of ASC oligomerization (oligomer, trimer, dimer, and monomer forms), pro-caspase-1 (p45), cleaved caspase-1 (p10), pro-IL-1β, IL-1β, and IL-18 significantly decreased in corneas after treatment with FK506 alone or natamycin alone or combined treatment (Figure 3(c)). Furthermore, the protein expression levels of these molecules were lower in combined treatment group than that in single-agent treatment group.
Combination treatment with FK506 and natamycin alleviates F. solani-induced activation of the NLRP3 inflammasome
Subsequently, we tested the effect of this combination on the histopathologic changes of corneas after F. solani infection. As shown in Figure 4(a), corneas in mice with F. solani keratitis had dense inflammatory cell infiltration, extensive stromal destruction, and ulceration detected by H&E staining. However, these effects were blocked by drug treatment. Moreover, the effect was better by combination treatment compared to that by single-agent treatment.
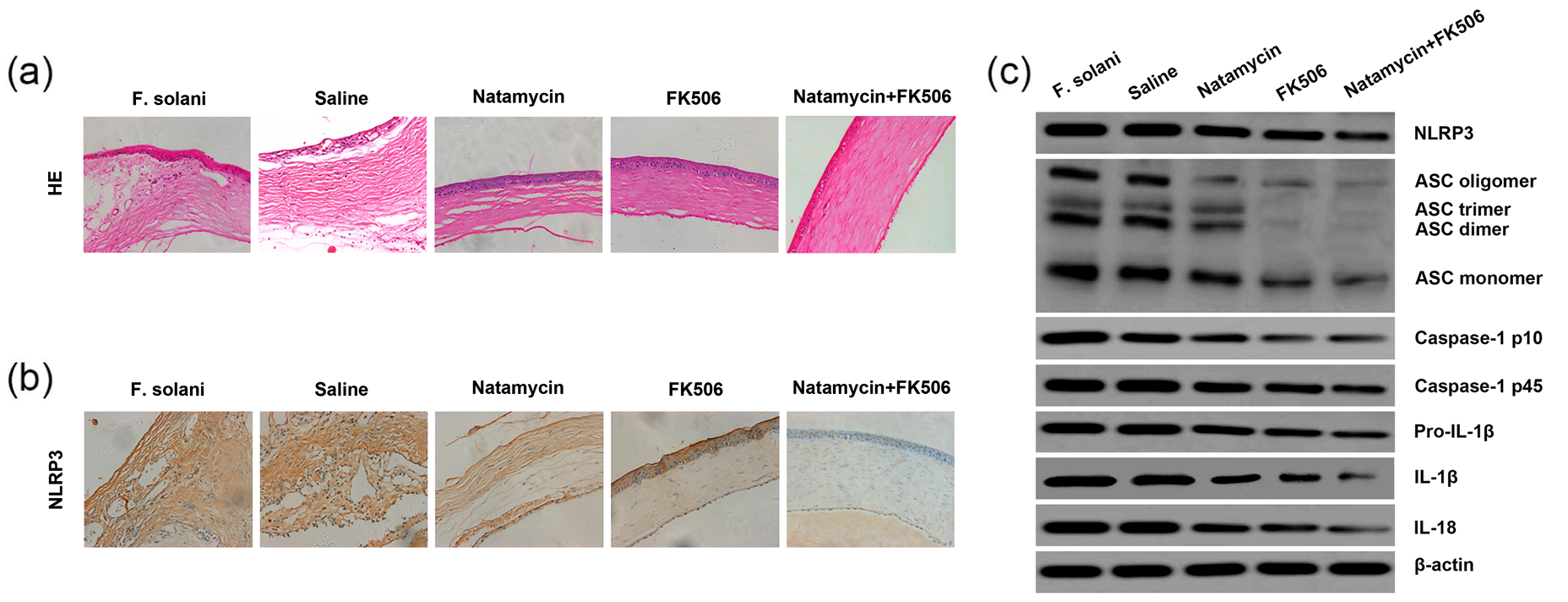
Effects of FK506 combined with natamycin on the corneas infected with F. solani by suppressing NLRP3 inflammasome activation. (a) Representative pictures of H&E staining of corneas; magnification: ×40. (b) Immunohistochemistry analysis for NLRP3 expression in corneas; magnification: ×40. (c) Expression levels of NLRP3, ASC, caspase-1, IL-1β, and IL-18 in corneas were detected by western blot.
NLRP3 protein expression levels decreased after FK506 or natamycin treatment alone, or after combined treatment with FK506 and natamycin. In addition, NLRP3 protein expression levels were lower in combined treatment group compared to that in FK506 alone or natamycin alone group (Figure 4(b) and (c)).
Next, we found that FK506 promotes natamycin-induced resistance to the activation of the NLRP3 inflammasome, as evidenced by lower protein expression levels of ASC (oligomer, trimer, dimer, and monomer forms), pro-caspase-1 (p45), cleaved caspase-1 (p10), pro-IL-1β, IL-1β, and IL-18 in corneas after combined treatment compared to that after single-agent treatment (Figure 4(c)).
Discussion
Aspergillus and Fusarium are major cause of visual impairment and blindness worldwide. 18 However, the available clinical drug treatments to these organisms in the cornea are still scarce. In this study, we perform a prespecified subgroup analyses of treatment within Fusarium keratitis, and separately, within Aspergillus keratitis, to assess the efficacy of FK506 and natamycin.
Natamycin is the only commercial antifungal drug in ophthalmic form. 19 It has a broad spectrum of activity against spoilage fungi. 20 It can bind irreversibly to ergosterol, disrupting the fungal cell membrane and cause a loss of solutes from the cytoplasm. 21 In addition, natamycin has been considered as the first line in the management of fungal keratitis. 22 However, it has been reported that natamycin has little corneal penetration and low efficiency. 23 Lin et al. 24 reported that almost 70% of patients with deep lesions of Fusarium keratitis do not respond to medical therapy alone. Thus, there is a need to evaluate other antifungals.
FK506, structurally similar to FK520 and rapamycin, exhibits potent immunosuppressive activity and strong antifungal activity. 25 Previous report demonstrated that FK506 is relatively active against Aspergillus fumigatus. 26 FK506 binds to FK506-binding proteins within T lymphocytes and inhibits calcineurin (cnaA) activity. 27 CnaA is important for Aspergillus fumigatus morphology, survival, and pathogenicity. 28 Therefore, a mutant Aspergillus fumigatus strain without the cnaA catalytic subunit shows physiological defects that lead to stunted growth. 29 Moreover, FK506 can inhibit the inflammation and diminish the severity of corneal damage at an early stage of fungal keratitis. 14 So we hypothesized that natamycin can be combined with FK506 in the treatments for fungal keratitis. Histopathologic studies of corneas showed that the treatment effects were better by combination treatment compared to that by FK506 alone or natamycin alone.
Inflammation is likely one of the most important causes of corneal damage after fungal infection. 30 And the process is highly dependent on the activation of NLRP3 inflammasomes. 31 The NLRP3 inflammasome consisted of the NLR protein NLRP3, the adapter ASC, and pro-caspase-1. 32 Activated caspase-1 cleaves the pro-IL-1β into the active form. 33 The fungi such as A. fumigatus are reported to be sensed by the NLRP3 inflammasome and to trigger the secretion of IL-1β from both mouse and human cells. 34 Karthikeyan et al. 35 found that a significant increase in IL-1β transcripts, as well as expression of the NLRP3 and ASC inflammasome proteins in human corneas infected with Aspergillus or Fusarium, suggests a role for inflammasomes in fungal keratitis. Our data were in agreement with the above studies. However, the enhanced expression levels of these molecules were blocked by the combination treatment with FK506 and natamycin, suggesting FK506 combined with natamycin inhibits the activation of NLRP3 inflammasomes.
Clinicians tend to use FK506 as an immunosuppressant due to its antifungal ability. 36 Topical FK506 with concentrations of 0.02%–0.1% in ointment form has successfully been used for treatment of atopic keratoconjunctivitis (AKC)37,38 and giant papillary conjunctivitis. 39 Besides, topical 0.005% FK506 eye drop is a safe and effective treatment for patients with refractory vernal keratoconjunctivitis (VKC). 40 As FK506 may inhibit the inflammation at an early stage of fungal keratitis, 14 the antifungal drugs combined with FK506 may be the probable future of FK506 in the clinical scenario.
Conclusion
In conclusion, our study indicates that FK506 combined with natamycin alleviate fungi-induced corneal damage by suppressing NLRP3 inflammasome activation. In addition, the combination treatment with FK506 and natamycin may also be a potential therapeutic candidate for fungal keratitis.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
