Abstract
Introduction
Nucleotide-binding and oligomerization domain like receptors protein 3 (NLRP3) inflammasome-mediated interleukin (IL)-1β secretion plays an important role in the progression of Alzheimer’s disease (AD). Curcumin has been shown to improve cognitive impairment and learning ability of AD mice by reducing IL-1β secretion. However, its exact mechanism of action remains unclear. In the present study, we explored the relationship between the neuroprotective effect of curcumin and activation of the NLRP3 inflammasome pathway.
Methods
BV2 cells were primed with 500 ng/mL lipopolysaccharide (LPS) for 4 h and subsequently treated with 50 μM Aβ25-35 for 24 h or pretreated with 2.5–10 μM curcumin for 4 h and exposed to 50 μM Aβ25-35 for 24 h. The effects of curcumin and Aβ25-35 were assessed by the CCK8 assay. ELISA was used for the detection of IL-1β, IL-6, and tumor necrosis factor (TNF)-α levels in the supernatant of the cell culture medium. The viability of SH-SY5Y cells, which were incubated with conditioned medium (CM) was assessed using the CCK8 assay. The percentage of apoptotic SH-SY5Y cells incubated with CM was assessed using Annexin V-FITC/PI staining flow cytometry analysis. The expression levels of NLRP3, caspase-1 and IL-1β were observed by western blot and immunofluorescence staining analyses; the mRNA levels of
Results
Low (2.5 μM), medium (5 μM), and high (10 μM) concentrations of curcumin and 50 μM Aβ25-35 were used to perform the experiments in the present study. Curcumin attenuated the IL-1β, IL-6, and TNF-α release and increased SH-SY5Y cell activity, while decreasing the apoptotic percentage of SH-SY5Y cells using Aβ25-35 for cell stimulation (
Conclusion
Overall, the data indicated that curcumin is a promising neuroprotective agent for suppressing neuroinflammation by inhibiting the NLRP3 inflammasome pathway.
Introduction
Alzheimer’s disease (AD) is the most common cause of dementia, which has a high incidence rate among the elderly. Although multiple gene mutations have been proven to be associated with the risk of AD, 1 aging is still the main risk factor. It is widely recognized that the AD pathological features are characterized by neurofibrillary tangles (NFTs), which include intracellular hyperphosphorylated tau and senile plaques (SP) composed of extracellular accumulated amyloid-β (Aβ) peptides. In the early stage of AD, a high number of activated microglia accumulate around the SP, 2 whereas microglia that exhibit the M2 phenotype participate in phagocytosis and clearance of Aβ, which is considered the “main force” of Aβ clearance in the early stage of AD. 3 However, the persistent endogenous stimuli, including mutant superoxide dismutase (SOD), Aβ and hyperphosphorylated tau, can promote microglia to transform into the M1 phenotype. This phenotype secretes a large number of pro-inflammatory cytokines, which can in turn aggravate the deposition of Aβ or the abnormal hyperphosphorylation of tau and gradually damage the neurons and synapses. 4
Nucleotide-binding and oligomerization domain like receptors protein 3 (NLRP3) is a member of the pattern recognition receptor (PRR) NOD-like receptor family, which participates in the innate immune response. 5 The activation of the NLRP3 inflammasome can be divided into the two following steps: Priming and activation. In step 1, Toll-like receptors (TLR) on the cell membrane perceive the pathological stimuli, such as lipopolysaccharide (LPS) and subsequently cause an upregulation of the transcriptional levels of NLRP3, while activating NLRP3 through phosphorylation and deubiquitination. 6 In step 2, oligomerization of NLRP3 occurs combined with recruitment of apoptosis associated speck-like protein containing a card (ASC) and pro-caspase-1 by effective stimulation, such as urate crystals, aluminum salt, ATP, Aβ, and K+ efflux in order to complete the assembly of the inflammasome.6,7 Subsequently, caspase-1 is self-cleaved into activated caspase-1, which can mediate the secretion of pro-inflammatory cytokines, including interleukin (IL)-1β and IL-18. 8 The activation of the NLRP3 inflammasome and the increase of IL-1β in microglia have been observed in AD mouse models and brain tissues of patients with AD.9,10 In the AD transgenic mouse model, the spatial memory impairment of mice can be improved by knocking out the NLRP3 gene or using NLRP3 inhibitors (JC-124 or MCC950).10–14 The activation of the NLRP3 inflammasome further induces the pathogenesis of tauopathies, 15 which is another hypothesis of the development of AD. Therefore, this evidence suggests that the pathogenesis of AD is closely associated with the activation of the NLRP3 inflammasome mediated by microglia.
Curcumin, a polyphenolic compound of plant origin, has been used as an anti-inflammatory drug to treat certain diseases in Asia and other regions worldwide. Recently, it has been found that curcumin exhibits several pharmacological effects, such as antioxidation, scavenging of free radicals, anti-inflammatory, and antitumor activities.
16
Curcumin reduces the secretion of IL-1β by promoting the transformation of microglia to the M2 phenotype
Materials
Immortalized BV2 cells were obtained from the Cell Bank of the Chinese Academy of Science (Wuhan, China), whereas immortalized SH-SY5Y cells were obtained from the Beina Chuanglian Biotechnology Research Institute (Beijing, China). Dulbecco’s Modified Eagle Medium (DMEM), fetal bovine serum (FBS), penicillin-streptomycin and trypsin-EDTA (0.25%), and phenol red were purchased from GIBCO (Grand Island, NY, USA). Curcumin and Aβ25-35 were purchased from MedChem Express (Monmouth Junction, NJ, USA). LPS was purchased from Sigma-Aldrich (St. Louis, MO, USA). Mouse monoclonal antibody β-actin, goat serum, Cy3-labeled goat anti-rabbit IgG, HRP-labeled goat anti-mouse secondary antibody, and HRP-labeled goat anti-rabbit secondary antibody were purchased from Boster Biological Technology Co., Ltd. (Wuhan, Hubei, China). NLRP3 rabbit polyclonal antibody (118 kd) was purchased from affinity Biosciences (Cincinnati, OH, USA); mouse anti-caspase-1 monoclonal antibody (10 kD, 45 kd) was purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA) and mouse anti-IL-1β polyclonal antibody (17, 31 kd) from Cell Signaling Technology (Danvers, MA, USA). SYBR Green master mix was purchased from Vazyme Biotech Co., Ltd (Nanjing, Jiangsu, China).
Methods
Aβ25-35 preparation
As described previously, Aβ25-35 was diluted to a final concentration of 1 mM in serum-free DMEM and subsequently incubated for 2 weeks at 37°C to induce the aggregation of Aβ25-35. During its use, it was further diluted to 50 μM in medium. 19
Immortalized BV2 cell culture
Immortalized BV2 cells were cultured in DMEM containing 10% heat-inactivated FBS, 100 U/ml streptomycin, and 100 U/ml penicillin in a humidified 5% CO2/95% air incubator. The medium was replaced every 3 days. According to the experimental design, BV2 cells in the Aβ and curcumin intervention groups were primed with 500 ng/mL LPS for 4 h and subsequently exposed to different treatments. BV2 cells were cultured in DMEM as a control group.
Dose-dependency studies
A dose-dependency study was performed with increasing concentrations of curcumin [0, 2.5, 5, 10, 20, 40, and 80 μM] and Aβ25-35 [0, 15, 25, 50, and 75 μM] to determine the sublethal concentration of BV2 cells. The CCK8 assay was used to assess cell viability following 4 h incubation.
Preparation of conditioned medium
Components of CM.

Immortalized SH-SY5Y cell culture
Immortalized SH-SY5Y cell were cultured in DMEM containing 10% heat-inactivated FBS, 100 U/mL streptomycin and 100 U/mL penicillin in a humidified 5% CO2/95% air incubator. The medium was replaced every 2 days. According to the experimental design, SH-SY5Y cells were incubated with CM for 24 h. SH-SY5Y cells cultured in DMEM were used as a control group.
Detection of pro-inflammatory cytokines in the supernatant
Pro-inflammatory cytokines, including IL-1β, IL-6, and TNF-α were quantified using ELISA kits according to the manufacturer’s instructions. The assays were performed in triplicate.
CCK8 assay
SH-SY5Y cell viability was assessed using the CCK8 assay as previously described. 20 SH-SY5Y cells were seeded in a 96-well plate treated with CM for 24 h. The CCK8 reagent (100 μL) was added to each well. Following 4 h incubation at 37°C, the absorbance was recorded at 450 nm using a microplate reader.
Flow cytometry
SH-SY5Y cells were incubated in CM for 24 h and the total cell lysate was collected. The samples were centrifuged and the Annexin V-FITC/PI reagent was added. Subsequently, flow cytometry was used to detect the number of apoptotic cells.
Western blot assay
The western blot assay was performed as previously described. 21 Exposure of BV2 cells to different experimental conditions for 24 h was performed according to the study design. The determination of the total protein concentration was performed by the BCA protein assay kit following extraction of the total protein. The proteins’ concentration loaded was 40 μg per lane on a 12% SDS-PAGE gel and the proteins were electrotransferred to PVDF membranes, which were subsequently blocked with TBST buffer containing 5% non-fat milk and finally incubated with primary antibodies (diluted anti-NLRP3, caspase-1, and IL-1β 1:1,000, diluted β-actin 1:500) on a shaking platform at 4°C overnight. Following washing, the membranes were subsequently incubated with secondary antibodies, which were labeled with HRP (goat anti-mouse and goat anti-rabbit; 1:50,000) for an additional 2 h at room temperature. All blots were examined using ECL detection reagents and the scanned images were quantified by ImageJ software (USA). β-actin was used as a loading control.
Immunofluorescence staining assay
Immunofluorescence staining was performed as previously described. 22 Briefly, BV2 cells were fixed with 4% paraformaldehyde for 15 min, permeabilized with 0.5% Triton X-100 in PBS for 20 min and blocked with goat serum albumin in PBS for an additional 30 min. Following blocking, the cells were incubated with anti-NLRP3 primary antibody (1:50) at 4°C overnight. On the next day, the cells were washed with PBS and incubated with a secondary antibody (Cy3-labeled goat anti-rabbit IgG) at 1:100 dilution for 2 h. The nuclei were subsequently stained with DAPI for 5 min. Fluorescence images were obtained with OLYMPUS BX53. Fluorescence intensity was measured using Image J (USA).
qRT-PCR
PCR primer sequences.

Statistical analysis
All experiments were performed at least three times. All data are presented as arithmetic mean ± standard deviation. The statistical significance of the differences between the compared groups was determined using one-way analysis of variance (ANOVA) followed by Bonferroni post hoc test for multiple comparisons. SPSS (version 26.0) software (SPSS Inc., Chicago, IL, USA) was used. A
Results
Estimation of optimal curcumin concentration
Although curcumin exerts a neuroprotective effect, large doses are cytotoxic. BV2 cell viability was assessed by the CCK8 assay to estimate the optimal concentration of curcumin to be used in subsequent experiments. BV2 cell viability was not affected at concentrations of curcumin ≤10 μM. By contrast, very high concentrations of curcumin (>20 μM) significantly reduced the viability of BV2 cells. In order to clarify the protective effect of different doses of curcumin, low (2.5 μM), medium (5 μM), and high (10 μM) concentrations of curcumin were used to perform the following experiments (Figure 1(a)). BV2 cells were treated with various concentration of curcumin (a) and Aβ25-35 (b). Cell viability was assessed by the CCK8 assay. The data are shown as mean±standard deviation for three independent experiments; *
Aβ25-35 exhibits a dose-dependent cytotoxic effect on BV2 cells
BV2 cells were incubated with Aβ25-35 for 4 h to assess its cytotoxic effect on BV2 cells. A concentration range of frequently used doses was selected between 15 and 75 μM and the CCK8 assay was used to detect cell viability. The concentration of Aβ25-35 required to induce 50% loss of cell viability (IC50) was measured by the CCK8 assay and compared with the cell viability of the 0 μM group. The results indicated that Aβ25-35 induced a concentration-dependent cytotoxic effect on BV2 cells. Cell viability was decreased by 50.1% at a concentration of 50 μM Aβ25-35. Therefore, this concentration was selected as the IC50 of Aβ25-35 (Figure 1(b)).
Effects of curcumin on the secretion of the pro-inflammatory cytokines
Excessive pro-inflammatory cytokine generation is related to various inflammatory diseases. Therefore, ELISA was used to detect the levels of inflammatory cytokines in LPS-primed BV2 cells following stimulation with Aβ25-35. The secretion of the inflammatory cytokines (IL-1β, IL-6, and TNF-α) in the Aβ25-35-stimulated group was significantly higher than that of the control cells. The cells were pretreated with curcumin (2.5–10 μM), which decreased IL-1β, TNF-α, and IL-6 production compared with that noted in the Aβ25-35-stimulated group (Figure 2). Effects of curcumin on the expression levels of inflammatory cytokines in LPS-primed BV2 cells following stimulation with Aβ25-35. LPS-primed BV2 cells were treated with 50 μM Aβ25-35 for 24 h or pretreated with curcumin for 4 h and subsequently exposed to 50 μM Aβ25-35 for 24 h. The expression levels of IL-1β (a), IL-6 (b) and TNF-α (c) were evaluated using ELISA kits. The data are shown as mean ± standard deviation for three independent experiments; *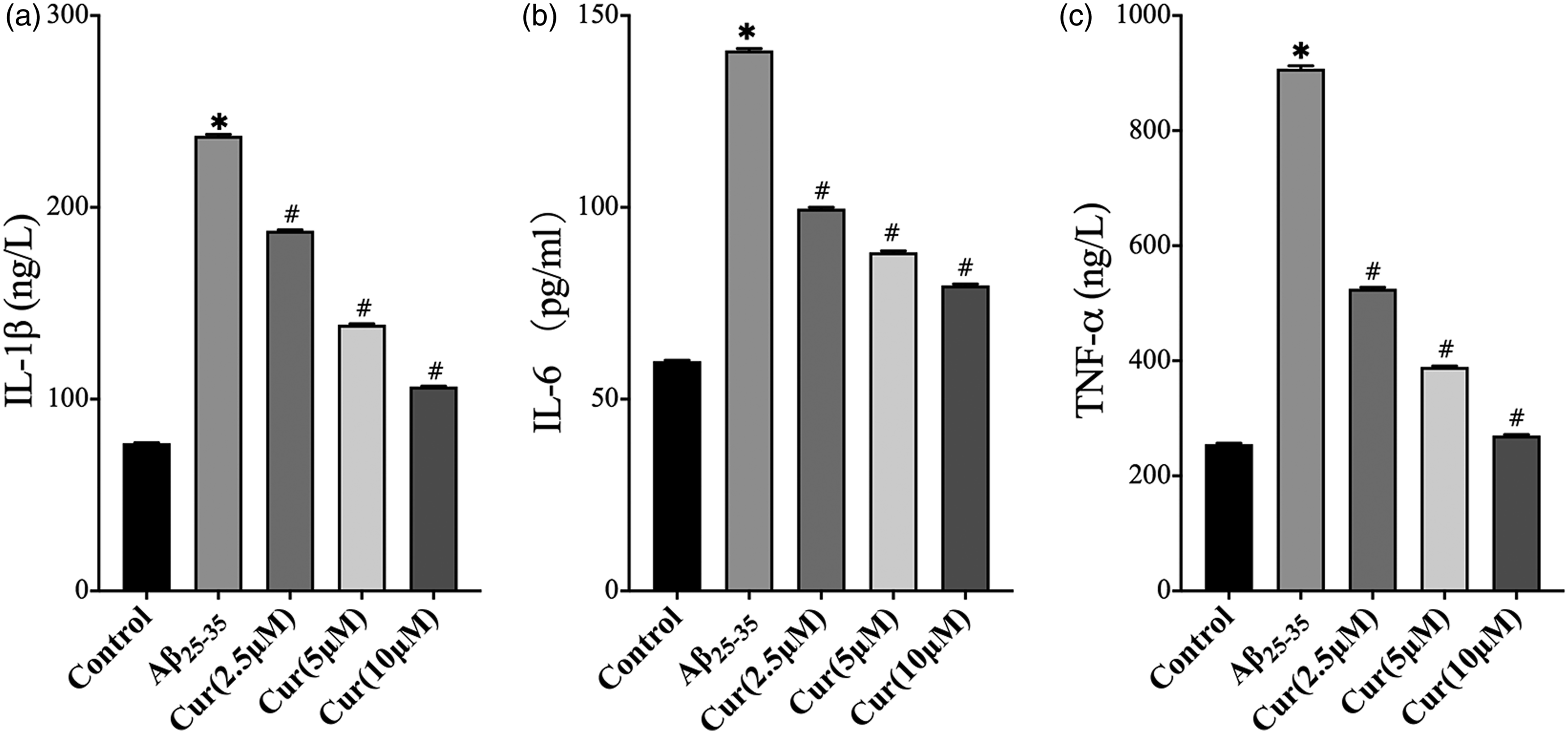
Effects of CM curcumin pretreatment on SH-SY5Y cell viability and induction of apoptosis
To evaluate neuronal apoptosis and viability induced by the inflammatory microenvironment, flow cytometry (Figure 3(a) and (b)), and the CCK8 assay (Figure 3(c)) were used. The apoptotic rate and viability of SH-SY5Y cells were increased in the CM-BV2/Aβ25-35 group compared with those of the control group. The apoptotic rate was reduced following incubation with curcumin (2.5–10 μM) compared with that of the CM-BV2/Aβ25-35 group, while, the viability of SH-SY5Y cells was increased in cells pretreated with curcumin (5–10 μM). Effects of curcumin on SH-SY5Y cell apoptosis and viability in CM. SH-SY5Y cells were incubated with CM for 24 h. The induction of apoptosis (a and b) of SH-SY5Y cells was detected by flow cytometry, whereas their cell viability (c) was measured by the CCK8 assay. The results shown are representative of three independent experiments, *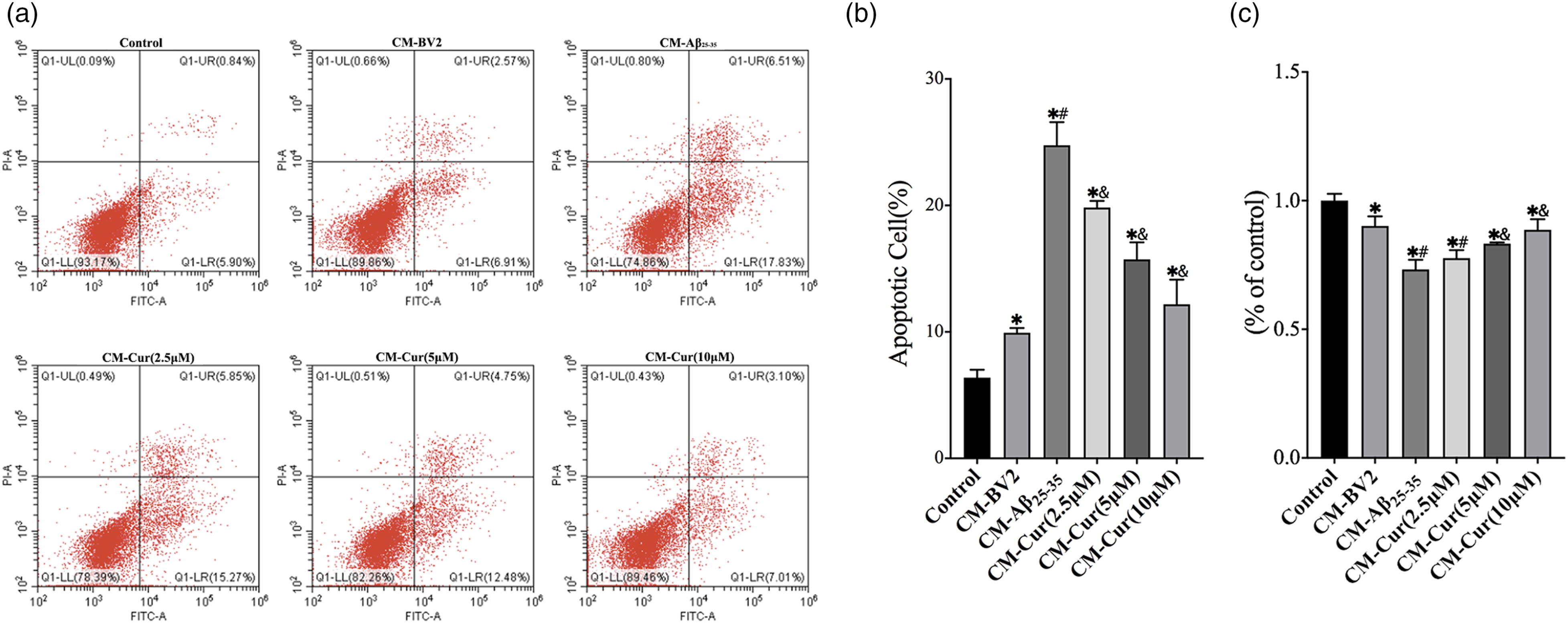
Effect of curcumin on the mRNA expression levels of the NLRP3 inflammation pathway-related genes
The qRT-PCR was used to detect the expression levels of the NLRP3 inflammation pathway-related genes. The results indicated that the mRNA levels of Effects of curcumin on NLRP3 pathway-related mRNA expression levels in LPS-primed BV2 cells following stimulation with Aβ25-35. LPS-primed BV2 cells were treated with 50 μM Aβ25-35 for 24 h or pretreated with curcumin for 4 h and subsequently exposed to 50 μM Aβ25-35 for 24 h. The mRNA levels of NLRP3 (a) caspase-1 (b) and IL-1β (c) were detected using qRT-PCR. The data are shown as mean ± standard deviation for three independent experiments; *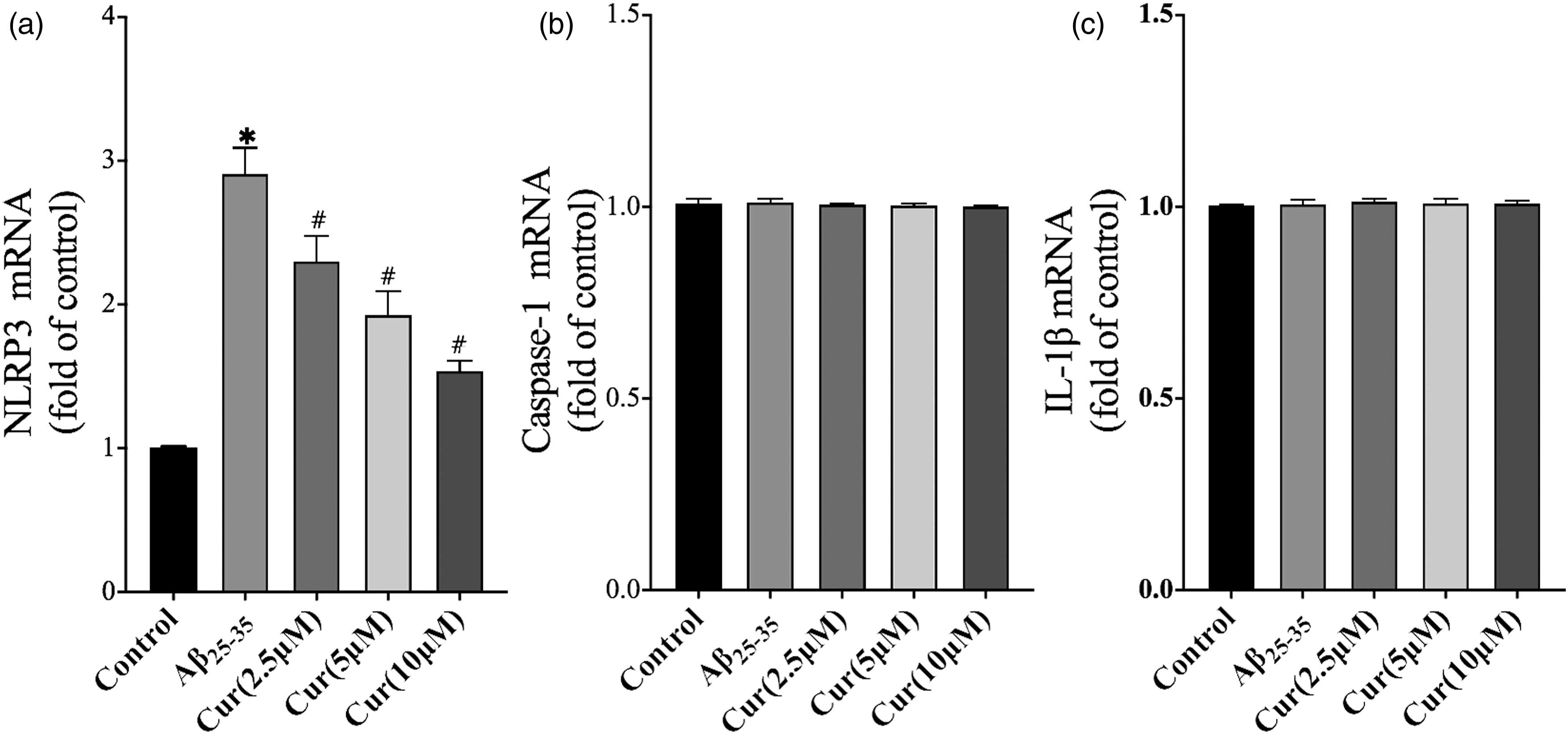
Effects of curcumin on NLRP3 protein expression
The NLRP3 protein plays a crucial role in the composition of the NLRP3 inflammasome, which is involved in the regulation of the central nervous system (CNS)-associated inflammation. Western blot analysis revealed that the expression levels of the NLRP3 protein in the Aβ25-35 group were upregulated, which were significantly different from the control group. Curcumin (2.5–10 μM) treatment significantly reversed this effect (Figure 5(a) and (c)). Immunofluorescence staining was used to further evaluate NLRP3 expression (Figure 5(b) and (d)). Red fluorescence indicates the expression of the NLRP3 protein. The data indicated that red fluorescence intensity was significantly elevated following stimulation of the cells with Aβ25-35. It was suggested that the production of the NLRP3 protein was increased. However, red fluorescence intensity was reduced following curcumin treatment. The results were consistent with those of the western blot assay. Effects of curcumin on the expression levels of the NLRP3 protein in LPS-primed BV2 cells following stimulation with Aβ25-35. LPS-primed BV2 cells were treated with 50 μM Aβ25-35 for 24 h or pretreated with curcumin for 4 h and subsequently exposed to 50 μM Aβ25-35 for 24 h. The expression levels of the NLRP3 protein were evaluated using immunofluorescence staining (a) and western blot analysis (b). The band density values of NLRP3 were normalized to those of β-actin and expressed as a ratio to the control group (c). The fluorescence intensity values of NLRP3 were normalized and expressed as a ratio to the control group (d). The data are shown as mean ± standard deviation for three independent experiments; *
Expression levels of cleaved caspase-1 and secretion of IL-1β
Caspase-1 and IL-1β exist in microglia as pro-active molecules. Caspase-1 is cleaved into the p10/p20 dimer. This process depends on the assembly and activation of the NLRP3 inflammasome that leads to the secretion of IL-1β. The expression levels of cleaved caspase-1 and the secretion of IL-1β were assessed using western blot analysis. The results indicated that the expression levels of cleaved caspase-1 (p10, 10 kd) and the secretion of IL-1β were increased in the Aβ25-35 group compared with those of the DMEM groups. BV2 cells were pretreated with curcumin and the expression levels of caspase-1 and IL-1β were decreased compared with those of the Aβ25-35 group. However, the expression levels of pro-caspase-1 and pro-IL-1β were not significantly different between the groups investigated (Figure 6). Effects of curcumin on caspase-1 and IL-1β expressions in LPS-primed BV2 cells following stimulation withAβ25-35. LPS-primed BV2 cells were treated with 50 μM Aβ25-35 for 24 h or pretreated with curcumin for 4 h and subsequently exposed to 50 μM Aβ25-35 for 24 h. The expression levels of caspase-1 and IL-1β were evaluated using western blot analysis (a). The band density values of caspase-1 (b and c) and IL-1β (d,e) were normalized to those of β-actin and expressed as a ratio relative to the control group. The data are shown as mean ± standard deviation for three independent experiments; *
Discussion
It has been recently accepted that neuroinflammation is a characteristic factor involved in the pathological process of AD. 23 Microglia are resident macrophages that participate in the immune regulation of the CNS. Aβ is generated by cleavage of the amyloid precursor protein (APP) through β-secretase and γ-secretase. 23 In the presence of Aβ, microglia can be activated causing the release of IL-1β via the activation of the NLRP3 inflammasome. Overexposure of neuronal cells to IL-1β causes neuronal damage through aberrant release and accumulation of glutamate. 24 Several studies have shown that microglia is a new target for the treatment of neuroinflammation.
In the present study, the expression levels of the inflammatory cytokines were examined in the supernatant of the cells using ELISA in order to observe the changes in the inflammatory microenvironment of the neurons following stimulation of microglia with Aβ25-35. The aforementioned results implied that the expression levels of IL-1β, TNF-α and IL-6 in the supernatant were significantly increased following exposure of the cells to Aβ25-35, which was in agreement with the findings reported by Heneka et al. in the frontal cortex and hippocampus of APP/PS1 mice. 11 In addition, SH-SY5Y cells were incubated with the CM, which contained pro-inflammatory cytokines in order to mimic the inflammatory microenvironment of AD in the brain. The results of the CCK8 assay revealed that CM exposure to Aβ25-35 decreased the activity of the neurons. Flow cytometry analysis indicated that neuronal apoptosis was increased. The findings revealed that the increase in the levels of the pro-inflammatory cytokines could lead to the decrease of neuronal activity and to the increase of neuronal cell apoptosis, which indicated Aβ25-35-mediated neurotoxicity.
The NLRP3 inflammasome is a multiple protein complex composed of NLRP3, ASC, and pro-caspase-1. Following stimulation of TLR by LPS in the microglia membrane, the expression levels of NLRP3 and pro-IL-1β are upregulated. Aβ can be identified by PRR and promotes NLRP3 inflammasome assembly by recruiting ASC and pro-caspase-1. Aβ is considered an intrinsic driving force for the activation of the NLRP3 inflammasome. The latter mediates the self-cleavage of pro-caspase-1 into the p20/p10 dimer, which is its active form that triggers the maturation and secretion of IL-1β.
The expression levels of the NLRP3/caspase-1/IL-1β pathway-related proteins were evaluated in microglia by western blot analysis following their stimulation with Aβ25-35. The results revealed that the expression levels of NLRP3 were increased. This finding was confirmed by immunofluorescence staining, indicating similar results, which were determined by the increase of red fluorescence in microglia. Subsequently, the expression levels of the related proteins, which acted downstream of the NLRP3 pathway, were assessed. The expression levels of the activated form of the NLRP3 inflammasome-dependent caspase-1, p10, and of the activated form of IL-1β, p17, were upregulated in microglia. Activation of caspase-1 (p20/p10 dimer) depends on the specific cleavage of pro-caspase-1 by the NLRP3 inflammasome, whereas activated IL-1β requires caspase-1 to cleave pro-IL-1β. Therefore, NLRP3 inflammasome-dependent caspase-1 (p10) and IL-1β (p17) protein expression levels were upregulated, indicating that Aβ induced the activation of the NLRP3 inflammasome in microglia. This finding is consistent with the results reported in previous studies.11,27 Stimulation of the cells with Aβ25-35 further increased the mRNA levels of
However, limited evidence is available on the regulation of the NLRP3 inflammasome in AD by curcumin. He et al. suggested that curcumin effectively inhibited the neuroinflammation induced by Aβ25-35 in microglia, in part by inhibiting the expression levels of HMGB1, TLR4, and RAGE.
28
The data reported in the present study indicated that curcumin dose-dependently inhibited the overexpression of IL-1β, IL-6 and TNF-α following stimulation of the cells with Aβ25-35. Moreover, it reversed neuronal activity and reduced neuronal apoptosis in the inflammatory microenvironment. It is suggested that curcumin can protect neuronal activity following Aβ injury. Concomitantly, curcumin significantly reduced the expression levels of NLRP3, caspase-1, and IL-1β in neuroinflammatory cell models and inhibited the upregulation of
The present study has certain limitations. First, the neuroprotective effect of curcumin was only assessed in
Conclusion
In summary, the mechanism by which curcumin suppresses neuroinflammation is still unclear and evidence concerning the interrelationship between curcumin and neuroinflammation is insufficient. Our results not only emphasize on the importance of microglia in the neuroinflammatory process of AD, but also point out that curcumin acts as an effective antagonist of microglia-dependent inflammatory signal transduction, which is mainly attributed to the regulation of the NLRP3/caspase-1/IL-1β inflammasome signaling pathway. Therefore, it is suggested that curcumin may be a potential drug candidate for the treatment of AD.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Education Department of Shaanxi Provincial Government (19JK0972), the Major projects of Yan'an University affiliated Hospital (2021ZD-03) and the National Natural Science Foundation of China (81760235).
Data availability statement
The authors state that all information provided in this article can be obtained from the author on request.
