Abstract
Variants of the interleukin-1β (IL-1β) gene have been associated with type 2 diabetes (T2D) and coronary artery diseases (CAD). However, association of IL-1β polymorphisms with diabetic patients having CAD clinical manifestation has not been studied yet. In this study, we aim to decipher the role of IL-1β common promoter variants with susceptibility/resistance to development of CAD in T2D patients. T2D patients with (n = 134) or without CAD (n = 533) were enrolled. A total of 513 essentially healthy individuals from the same population were included in this study as control. Plasma levels of IL-1β were quantified by enzyme-linked immunosorbent assay (ELISA) kit as per instructions from the manufacturer. IL-1β promoter variants (T-31C and C-511T) were genotyped by polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP). T2D patients displayed higher level of plasma IL-1β in comparison to healthy controls. Prevalence of variants for IL-1β (C-511T) polymorphism was higher in diabetic patients compared to controls (CT: P < 0.0001, OR = 3.01; TT: P < 0.0001, OR = 2.45). IL-1β (C-511T) polymorphism was linked with plasma IL-1β levels. Interestingly, heterozygous mutants (CT) were most prevalent in T2D individuals with CAD compared to those without CAD (P = 0.03, OR = 1.82). Furthermore, low-density lipoprotein (LDL) and triglycerides were elevated in T2D patients with CAD than in patients without heart-related disorder. No significant association of other polymorphism (T-31C) was noticed with susceptibility to T2D or diabetic patients with heart disorders. IL-1β (C-511T) variants are associated with elevated plasma IL-1β levels. Mutation at the IL-1β promoter region (C-511T) predisposed subjects to the development of T2D and CAD manifestation in diabetic patients.
Introduction
Diabetes is a major health-related complication in humans which results when the pancreas cannot produce sufficient insulin (type 1 diabetes) or when the human is unable to utilize insulin efficiently (type 2 diabetes; T2D; World Health Organization, 2016). Despite more than 350 million subjects with diabetes worldwide, associated with the socioeconomic burden, the pathogenesis of T2D is poorly known. The most vital physiological feature of T2D patients is insulin resistance characterized by improper response of tissue to insulin. Primarily depletion of β-cell mass and dysfunctional β cells majorly contribute towards the T2D pathogenesis. 1 Progression of T2D can be classified into two distinct stages: pre-diabetes and overt diabetes.2,3 In the pre-diabetes stage, insulin resistance is minimized by increased secretion capacity of β-cell, whereas overt T2D is characterized by failure of the compensatory mechanism and subsequent degradation of functional β-cells, 4 causing aggravated hyperglycemia.
Management of diabetes is very much crucial and an important clinical practice for diabetes is the aggressive management of various risk factors that lead to clinical complications. Among several such risk factors, cardiovascular disease (CVD) remains a major cause of mortality in T2D patients. CVD in T2D is often found to be more severe and result in a higher rate of complications than those who are not diabetic, 5 and about one-quarter of mortality in T2D subjects is contributed by CVD. 6 A population-based multinational study across 28 different countries suggested that the overall prevalence of CVD in T2D patients ranged from 21.6% to 34.2%. 7 Among different CVDs, the incidence of coronary artery disease (CAD) increases along with age and occurs in the younger age population with diabetes.8,9 An estimate indicates that about 75% of subjects with T2D died as a consequence of several heart-related diseases, including CAD. 10
T2D is characterized as a low-grade chronic inflammation condition often demonstrated with hyper secretion of pro-inflammatory cytokines. Emerging evidence suggests that interleukin-β (IL-1β), a major pro-inflammatory cytokine playing a wide role in auto-inflammatory diseases, acts as a key promoter of systemic and tissue inflammation in T2D.11,12 An enhanced IL-1β expression has been marked in a high-glucose milieu in human monocytes and macrophages.13,14 Furthermore, elevated IL-1β has been reported to contribute to β-cell failure disabling insulin production.15,16 Moreover, upregulated levels of IL-1β have been observed in the heart and retinal vessels of diabetic rats. 17
The importance of inflammation on pathogenesis of CAD is well investigated. 18 IL-1 has been shown as a crucial molecule for induction of post-infarction inflammatory response and is associated with pathogenesis of cardiac remodelling. Therefore IL-1 signalling cascade has been targeted by various studies to find out the promising therapeutic target for myocardial infarction patients. 19 In mice, elevated IL-1β gene expression has been reported after myocardial infarction. 20 Furthermore, higher levels of IL-1β mRNA have been documented in CAD patients, 21 indicating an imperative role of IL-1β in progression of CAD.
Differential mRNA expression or plasma levels of IL-1β have been attributed to various factors, and polymorphisms in promoter region of IL-1β gene is one among them. 22 Various single-nucleotide polymorphisms (SNPs) have been reported at the promoter region, but T-31C (rs1143627) and C-511T (rs16944) are two common function polymorphisms widely investigated (https://www.ncbi.nlm.nih.gov/gene/3553). Candidate gene-based association study, including these two common SNPs (T-31C and C-511T) with T2D23–26 and CAD,27–34 has been carried out independently in different populations, which yielded contradictory results. However, reports on the role of IL-1β variants on predisposition to CAD in T2D patients are lacking.
As IL-1β plays an important role in both T2D and CAD, we hypothesized that common variants in the promoter region of IL-1β gene would be associated with susceptibility to development of T2D and diabetes with coronary artery disease. To test this, we performed a hospital-based case-control study in the Chinese population and investigated for possible genetic association for development of T2D with or without CAD.
Materials and methods
Study subjects
In this observational study, T2D patients who attended the outpatient department (OPD) of the Department of Endocrinology, The First People’s Hospital of Yunnan, during the period of November 2015–May 2018, were enrolled. Diabetes was diagnosed by measurements of various biomarkers such as fasting blood sugar (FBS), postprandial blood sugar (PPBS) and glycosylated haemoglobin (HbA1c) in plasma as prescribed by the American Diabetic Association (FBS > 150 mg/dL, PPBS > 200 mg/dL, HbA1c > 7mmol/mol). 35 For all patients, standard care was followed for anti-diabetic treatment such as appropriate diet, oral hypoglycemic drugs with or without insulin and only insulin treatment. The diabetic patients were further grouped into patients with or without CAD. Therapeutic intervention for CAD was made using lipid lowering drugs such as statins and fibrates. Patients having significant renal impairment, planned coronary intervention or planned surgical intervention, acute coronary syndrome, history of lactic acidosis, type I diabetes, or any significant hepatic, renal or cardiovascular dysfunctions were excluded from the present investigation. Healthy subjects from similar population were enrolled as controls. All controls were healthy and with no history of any heart-related anomalies or diabetes. Different biochemical parameters (risk factors) such as triglycerides, total cholesterol, low-density lipoprotein (LDL) and high-density lipoprotein (HDL) of patients and healthy controls were also measured in the laboratory facility available in the hospital. Written informed consent was collected from patients and controls and the study protocol was approved by the Institutional Human Ethical Committee of The First People’s Hospital of Yunnan. A post hoc power analysis was performed by G*Power 3.1.9.4 software with a smaller effect size (0.01), α error of probability (0.05) and observed 79.48% power to detect genetic association for the enrolled sample size.
Determination of plasma IL-1β levels
About 5 mL of heparinized blood was collected intravenously from each patient immediately before coronary angiography. Plasma was separated after centrifuging blood at 2000 r/min for 5 min and stored at −20°C till further use. Plasma IL-1β levels were measured using a highly sensitive immunoassay kit (Human IL-1β Quantikine High Sensitivity ELISA (enzyme-linked immunosorbent assay); R&D systems, Minneapolis, MN, USA) according to manufacturer’s instructions.
Genomic DNA isolation and genotyping of IL-1β polymorphisms
Whole blood of 200 µL was used for extraction of genomic DNA by kit according to manufacturer’s instructions (QIAGEN DNA blood mini kit). Extracted DNA was quantified and stored at −80°C until use. Two functional SNPs (T-31C and C-511T) in the promoter region were genotyped by polymerase chain reaction (PCR) followed by restriction fragment length polymorphism methods (RFLP), as described by our group earlier. 36 Briefly, for IL-1β T-31C polymorphism, the following primer set was used [5′-agaagcttccaccaatactc-3′ and 5′-agcacctagttgtaaggaag-3′] to obtain 239 bp amplicon. The amplified product was digested (AluI) and based on differential fragments genotype of the subject was determined as follows: TT = 137 + 102 bp, TC = 239 + 137 + 102 bp and CC = 239 bp.
Primers 5′-tggcattgatctggttcatc-3′ and 5′-gtttaggaatcttcccactt-3′ were employed for amplification of IL-1β gene flanking C-511T promoter polymorphism. The amplified product size was 304 bp and after digestion with restriction enzyme (AvaI) based on restriction fragment length pattern, the individuals were genotyped. Homozygous mutant (TT) lost the restriction site for AvaI and thus remained undigested (304 bp). Wild type (CC) has one restriction site for the restriction enzyme and produces 190 and 114 bp size fragments. Heterozygous mutant (CT) had three fragments: 304, 190 and 114 bp.
Statistical analysis
Genotype and allele frequencies of IL-1β polymorphisms (C-511T and T-31C) in healthy controls and patients were calculated by counting in Microsoft Excel sheet. The distribution of genotypes was tested for Hardy–Weinberg equilibrium by in-house formulated Microsoft Excel sheet. All statistical investigations were performed by GraphPad Prism 6.01. Mean plasma levels of IL-1β in healthy controls and T2D patients were compared by Student’s t-test. Furthermore, mean IL-1β quantity in different clinical manifestations of T2D patients and controls were investigated by analysis of variance (ANOVA) followed by Tukey’s post-test. A P value less than 0.05 was considered as significant. Prevalence of IL-1β promoter polymorphisms (C-511T and T-31C) in controls and T2D patients was analysed by Fisher’s exact test. For genotypes and allele comparison, a probability value of <0.02 (0.05/2) was taken as significant after the Bonferroni correction for two SNPs under investigation.
Results
Baseline characteristics
The distribution of sociodemographic characteristics in patients and healthy controls is shown in Table 1. A total of 667 subjects diagnosed with T2D were included in the study, of which 134 patients (20%) were found to be diagnosed with CAD. Participants were divided into three groups: healthy controls (Group 1), subjects with T2D without CAD (Group 2) and subjects with T2D with CAD (Group 3). The mean age of participants in the three study groups was comparable and gender disparity among the study groups was absent. No significant difference was observed in the levels of FBS, PPBS and HBA1c between T2D patients with or without CAD. Although no significant difference was observed in the total cholesterol and HDL levels among the three study groups, T2D patients irrespective of CAD manifestation displayed significantly higher levels of LDL compared with healthy controls. In addition, diabetic patients with CAD had a significantly upregulated LDL and triglyceride levels in comparison to those without CAD manifestation. Prevalence of hypertension was more frequent in diabetic patients (both groups) compared to healthy controls. Particularly, diabetic patients with CAD had a higher prevalence of hypertension compared with patients without heart dysfunction. However, prevalence of alcohol intake practice and smoking habits of participants were comparable among all the three groups (Table 1).
Baseline characteristics of patients and controls.

CAD: coronary artery diseases; T2D: type 2 diabetes; OHA: oral hypoglycemic drugs; NA: not applicable; HDL: high-density lipoprotein; LDL: low-density lipoprotein; SE: standard error.
Data are in mean ± SE or number (%) as applicable.
T2D patients displayed higher plasma IL-1β
The distribution of plasma IL-1β concentrations in three different study groups is shown in Figure 1. The mean levels of IL-1β were found to be 3.61 ± 0.92, 8.04 ± 1.06 and 8.53 ± 0.87 pg/mL in healthy controls, T2D patients without CAD and with CAD clinical manifestation, respectively. Overall, the results showed significantly higher IL-1β levels in T2D patients with or without CAD compared to healthy controls. However, diabetic patients with CAD clinical manifestation showed a marginal increase in IL-1β levels compared to those without CAD, but this difference could not reach the significance level.

Plasma levels of IL-1β in healthy controls and different clinical categories of type 2 diabetes (T2D). Plasma levels of IL-1β in healthy controls (n = 513) and T2D patients (n = 667) were quantified in all participants by ELISA as per manufacturer’s instructions. T2D patients were further categorized into patients with (n = 134) or without (n = 533) coronary artery diseases (CAD). Mean IL-1β levels in different clinical categories were compared by analysis of variance (ANOVA) followed by Tukey’s post-test. A P value <0.05 was considered as significant.
Distribution of IL-1β promoter polymorphisms in healthy Chinese
Polymorphisms (T-31C and C-511T) in IL-1β promoter region were genotyped by PCR-RFLP in 513 healthy Chinese and the prevalence of genotypes is shown in Table 2. For IL-1β T-31C polymorphism, heterozygous mutant (TC: 52%) were more frequent compared to homozygous mutant (CC: 26%) and wild type (TT: 22%). In contrast, for other polymorphisms (C-511T), CC genotypes were most frequent (58%) compared to heterozygous mutant (CT: 35%) and homozygous mutant (TT: 7%). Distribution of genotype among healthy Chinese were in Hardy–Weinberg equilibrium (T-31C: χ2 = 0.92, P = 0.33; C-511T: χ2 = 1.60, P = 0.20).
Prevalence of IL-1β promoter polymorphisms (-511C > T and -31T > C) in controls and type 2 diabetes patients.
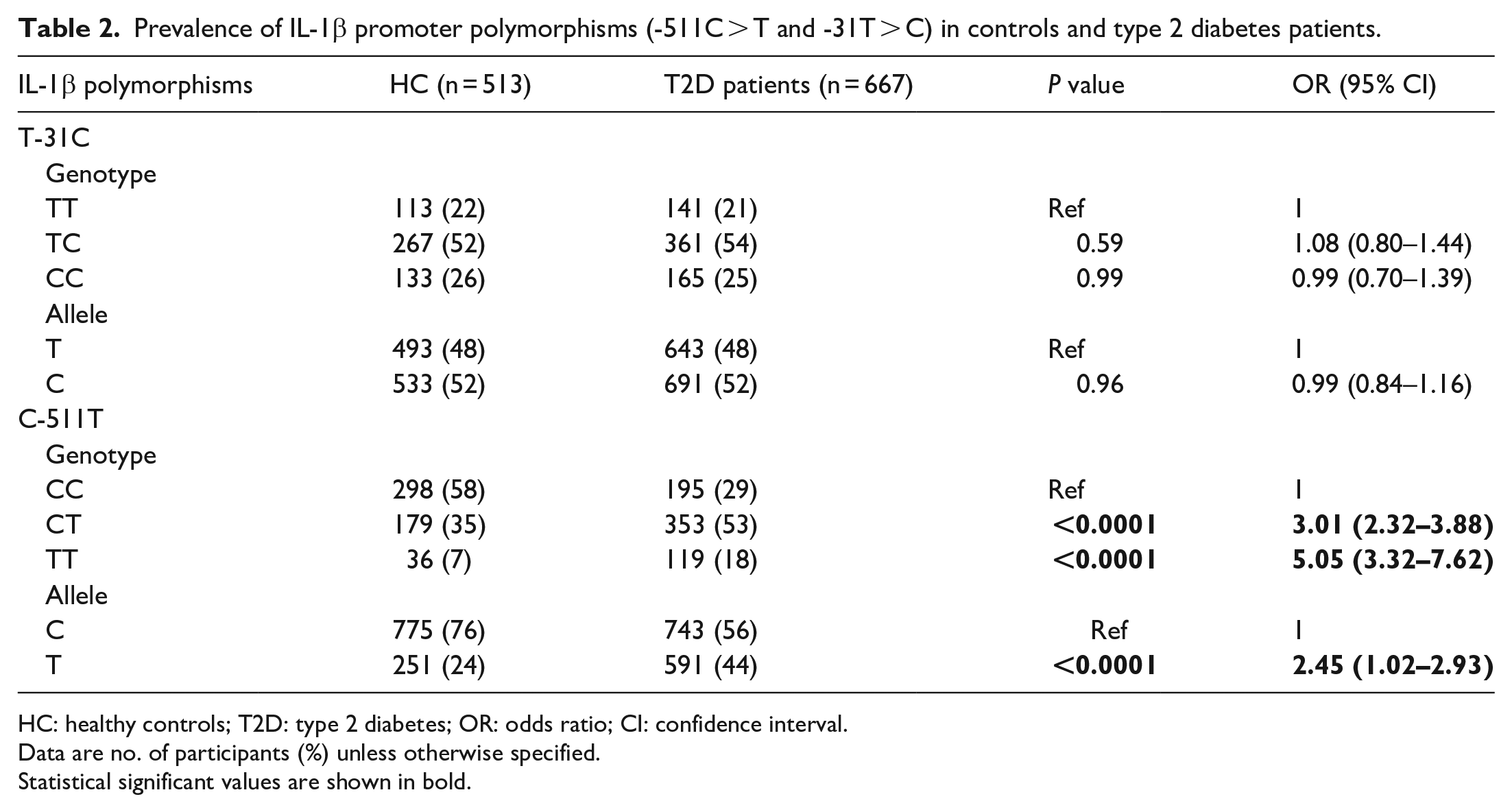
HC: healthy controls; T2D: type 2 diabetes; OR: odds ratio; CI: confidence interval.
Data are no. of participants (%) unless otherwise specified.
Statistical significant values are shown in bold.
Association of IL-1β (C-511T) variants with T2D
To investigate the possible association of IL-1β promoter polymorphisms (T-31C and C-511T) with susceptibility to development of T2D, we compared the distribution genotypes among healthy controls and T2D patients. As shown in Table 2, heterozygous (CT) and homozygous mutants (TT) for IL-1β (C-511T) polymorphism were more frequent in T2D patients compared to healthy controls (CT: P < 0.0001, OR = 3.01; TT: P < 0.0001, OR = 5.05). Similarly, the prevalence of minor allele (T) was also significantly higher in T2D patients compared with controls (P < 0.0001, OR = 2.45). However, the distribution of genotypes for IL-1β (T-31C) polymorphism was comparable among controls and patients (Table 2).
TT genotype of IL-1β (C-511T) polymorphism predispose to CAD in diabetic patients
The role of IL-1β variants in predisposition to CAD has been contradictory.28–34 In the present investigation, we observed a significant association of IL-1β (C-511T) polymorphism with predisposition to T2D development in our population; furthermore, we hypothesized their possible role in the development of CAD in T2D patients. The prevalence of IL-1β variants in controls, T2D patients with or without CAD was compared by Fisher’s exact test and is shown in Table 3. Like previous observations (Table 2), the heterozygous (CT) and homozygous mutants (TT) for IL-1β were more frequent in T2D patients with or without CAD compared with healthy controls (HC vs T2D with CAD (CT: P < 0.0001, OR = 3.09; TT: P < 0.0001, OR = 8.04); HC vs T2D without CAD (CT: P < 0.0001, OR = 2.99; TT: P < 0.0001, OR = 4.39)). In the same line, minor allele (T) was also more prevalent in T2D patients (both with and without CAD) than healthy controls (HC vs T2D with CAD (P < 0.0001, OR = 2.32); HC vs T2D without CAD (P < 0.0001, OR = 3.04)). Most importantly, when distribution of IL-1β (C-511T) polymorphism was compared among different clinical categories with respect to CAD, homozygous mutant (TT) was more frequent in T2D patients with CAD than those without CAD. However, the distribution was unable to reach significance levels after the Bonferroni correction (P = 0.03, OR = 1.82). Prevalence of other IL-1β (T-31C) polymorphism was comparable among healthy controls and different clinical categories of T2D patients with reference to CAD (Table 3).
Distribution of IL-1β (-511C > T and -31T > C) polymorphisms in healthy controls and type 2 diabetes patients with or without coronary artery diseases.

HC: healthy controls; T2D: type 2 diabetes; CAD: coronary artery diseases; OR: odds ratio; CI: confidence interval.
Data are no. of participants (%) unless otherwise specified.
Genotype–phenotype relationship of IL-1β promoter polymorphisms.
Statistical significant values are shown in bold.
Although various studies have focused on the genetic association of IL-1β promoter polymorphisms (T-31C and C-511T) with a wide range of diseases in humans, the functional relevance of these variants is underexplored.36–39 In this study, we investigated the association of IL-1β polymorphisms (T-31C and C-511T) with alteration of plasma IL-1β. As shown in Figure 2(a), subject to TT genotype for IL-1β C-511T polymorphism displayed higher levels of plasma IL-1β compared to CC (P < 0.0001) and CT (P < 0.0001). However, there was no such association among genotypes for IL-1β (T-31C) polymorphisms (Figure 2(b)).

Association of IL-1β (T-31C and C-511T) polymorphisms with plasma levels of IL-1β. Plasma levels of IL-1β in T2D patients and controls were quantified by ELISA and mean levels were compared in different genotypes for (a) IL-1β (T-31C) and (b) (C-511T) polymorphisms by ANOVA and Tukey’s post-test. A P value <0.05 was taken as significant.
Discussion
In China, there has been a rapid increase in the prevalence of T2D in subjects since the past three decades.40,41 A national survey conducted during 2007–2008 in the general Chinese population showed that the prevalence of coronary heart disease (CHD), stroke and combination of both was 0.6%, 0.8% and 1.37%, respectively, in China mainland. 42 Furthermore, prevalence of CAD in the Chinese population with T2D has also been reported. For example, a study in Taiwan reported that the prevalence of stroke was 2.3% in T2D subjects, 43 whereas a study in Beijing suggested an incidence of stroke to be 5.5% in patients with T2D. 44 T2D with CAD manifestation significantly increases the mortality rate of diabetic patients. Recently, a review highlighted the prevalence of CAD in T2D patients at around 23.6% in a total patient size of 44,602, including different regions of the western Pacific. 45 In accordance with this observation, we observed an overall prevalence of 20.0% (134 out of a total of 667 T2D subjects). Although crude prevalence of CAD in the Chinese population with T2D is substantially presented, no approach has been made on the incidence of CAD among T2D subjects in the Chinese population; our study tries to fulfil this gap.
It is well known that the major basis of morbidity and mortality in diabetes is CVD, which is exacerbated by hypertension. Furthermore, hypertension is an important risk factor for diabetes-related vascular complications. Hypertension substantially increases the risks of CHD, stroke and nephropathy in diabetic patients. Earlier reports have demonstrated that with the coexistence of hypertension with diabetes, the risk of CVD increased by 75% when compared to overall morbidity and mortality. 46 In line with earlier observations, this study also showed a significantly higher prevalence of T2D subjects with CAD having hypertension compared to those without CAD.
We investigated the significance of various risk factors in T2D and CVD by examining various factors such as LDL, HDL, total cholesterol, triglycerides, hypertension, FBS, PPBS, HbA1C, smoking habits and alcohol intake. Significantly elevated level of LDL was observed in Group 3 subjects compared to that of Group 2. Previously, the importance of LDL in CVD manifestation has been documented and an enhanced risk of 12% has been shown in patients with LDL level more than 10 mg/dL. 47 Thus, possibly high levels of LDL in T2D patients lead to develop CAD in them. In contrast, no remarkable difference was observed in the levels of total cholesterol and HDL among the study groups. Interestingly, we also observed higher levels of triglycerides in Group 3 patients compared to those in Group 2. Enhanced titre of triglycerides has been considered as an independent predictor for CVD. A fasting triglyceride level of more than 150 mg/dL has been considered as a definitive risk factor for CVD. 48 However, the role of triglycerides as a risk factor for developing CAD in T2D is still a debatable issue. 49 Our data show elevated triglycerides level in T2D subjects with CAD supporting possible role of this factor on onset of CAD. Other baseline parameters were comparable among T2D patients with or without CAD, indicating no possible role in the pathogenesis of CAD in T2D patients. In an earlier report, 50 comparable levels of FPBS and HbA1C (%) in T2D patients with and without CHD have been documented in both men and women. The result of this study is consistent with previous reports. In addition, levels of PPBS were also comparable in two clinical categories of T2D patients. However, in an independent report on Chinese patients, 51 PPBS levels were significantly higher in T2D subjects with cardiovascular complications compared to those without the disease.
Previous studies have indicated an important role of IL-1β in the outcome and pathogenesis of T2D. IL-1β has been shown to contribute to β-cell failure leading to insufficient production of insulin.16,52 Significantly higher IL-1β mRNA levels were found in T2D subjects compared to healthy controls. 1 Our results showing higher levels of IL-1β in T2D subjects (with and without CAD) compared to healthy controls matched with earlier reports. Furthermore, the role of IL-1β in CAD has been well documented. Elevated levels of IL-1β have been reported in CAD patients in comparison to healthy controls, suggesting an important role in the pathogenesis of CAD. However, in this study, plasma levels of IL-1β between T2D patients with or without CAD were comparable.
Attempts have been made to decipher the functional relevance of IL-1β promoter polymorphisms (T-31C and C-511T) by various groups; however, these yielded contradictory results. For IL-1β (T-31C) polymorphism, transfectional model showed association of TT genotype with higher expression level compared to CC genotype.37,38 It is believed that mutation at the -31 promoter region alters binding of TATA box protein and possibly enhances the transcription rate. 38 However, similar association of IL-1β variant (T-31C) was not observed in our studied cohort. Similarly, earlier reports also failed to demonstrate the significant genotype–phenotype association of IL-1β (T-31C) with plasma levels of protein.36,39 These discrepancies could be attributed to the different models used for investigation. In line with an earlier investigation on the Chinese population, 36 in this study the other IL-1β (C-511T) variant was associated with plasma levels of IL-1β. TT genotype was related to higher, heterozygous with intermediate and CC genotype was linked with the lowest plasma levels of IL-1β. However, an earlier report failed to demonstrate such association. The exact mechanism of how C-511T variants associated with plasma levels of protein is not known. Possibly, variation at the -511 region of IL-1β gene facilitates binding of certain transcription factors and that ultimately enhances the transcription rate of IL-1β gene.
The present candidate gene approach association study revealed the significant role of IL-1β -511 variants with susceptibility to T2D development. Furthermore, we observed association of higher plasma IL-1β with these mutants (CT and TT genotypes). Possibly, subjects harbouring these variants produced higher plasma IL-1β which may lead to β-cell failure and disabling of insulin production.15,16 Variants of IL-1β polymorphism (rs1143634) are also associated with T2D susceptibility in a Finland cohort. However, an earlier investigation in Chile with T2D patients failed to demonstrate such association with IL-1β promoter -511 mutants. 24
This study is the first of its kind to investigate the possible association of IL-1β promoter polymorphism with susceptibility to CAD manifestation in T2D patients. We observed a higher frequency of TT genotype of IL-1β C-511T polymorphism in T2D patients with CAD than those without CAD (P = 0.03), indicating a possible predisposing factor for CAD development. Previous genetic association studies in different populations such as African-Brazilian, 32 Polish 31 and a recent meta-analysis 34 have demonstrated a significant association of IL-1β (-511) with susceptibility to CAD. However, the mechanism of this polymorphism (IL-1β C-511T) is not known. Similarly, reports also demonstrate the association of IL-1 β (C-511T) polymorphism with susceptibility to different clinical manifestations of T2D such as diabetic nephropathy23,26 and periodontitis. 24
This study has several advantages over earlier reports. Most importantly, to the best of our knowledge, this is the first report to investigate the role of IL-1β in T2D patients with CAD clinical manifestation. Second, we have included a larger number of patients and healthy controls for this investigation. Third, this is a genetic association study comprising both genetic and phenotypic association of IL-1β with predisposition to development of T2D and diabetic patients with CAD clinical manifestation. However, this study has several limitations too and they need to be addressed. In this study, we have included only two promoter polymorphisms (T-31C and C-511T). Other SNPs in the promoter region and coding region in IL-1β, those believed to alter the expression and plasma levels of Il-1β, were not included. Furthermore, other clinical manifestations such as diabetic nephropathy, retinopathy and neuropathy were also not analysed in the present report.
Conclusion
T2D patients displayed higher levels of IL-1β compared to healthy controls. However, plasma levels of IL-1β were comparable among patients with or without CAD. Furthermore, variants of IL-1β (C-511T) were associated with predisposition to development of T2D and diabetes with CAD manifestation. However, studies in different population are required to validate our findings.
Footnotes
Acknowledgements
The authors thank all the participants of the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from the Institutional Human Ethical Committee of The First People’s Hospital of Yunnan (IHEC/FPHY/462).
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Informed consent
Written informed consent was obtained from all subjects before the study.
