Abstract
Background and objective
Cisplatin, an effective drug against cancer, commonly induces nephrotoxicity; limiting its therapeutic efficacy and application. In this study, Cisplatin NanoComposite (Cis NC) was formulated successfully from irradiated chitosan coated Cisplatin and MgO nanoparticles (CHIT/Cis/MgO NPs) to promote cisplatin release in a more sustained manner to improve therapeutic efficacy via the reduction of its nephrotoxicity. To compare the relative induced renal toxicity of cisplatin with Cisplatin NanoComposite, histological and biochemical mechanisms underlying nephrotoxicity were investigated.
Methods
Thirty rats were equally separated to three groups, first group received saline injections and adjusted as the control group, the second group was injected intra-peritoneal with cisplatin 0.64 mg/kg b. wt./day for 6 weeks, the third group was injected intra-peritoneal with Cis NC 5.75 mg/kg b. wt. daily for 6 weeks.
Results
Cisplatin-induced renal functional impairment and histopathological damages in the kidney; also, cisplatin disrupted the balance of the redox system in renal tissue, stimulated the inflammatory reactions in the kidney via triggering signal transducer and activator of transcription-1 (STAT1) dependent pathways. Moreover, Cisplatin-induced activation of mammalian target of rapamycin mTOR and inactivation of AMPK/PI3K/Akt signal pathway, and was coupled with induction of p53 activity and the executioner caspase3 to induce apoptotic renal cell death. On the other hand, Cis NC exerted a minimal stimulatory effect on apoptotic and inflammatory signal cascade with negligible renal functional and morphological alterations.
Conclusion
We postulated that Cis NC may be a valued possible drug to decrease the cytotoxicity of cisplatin thus reserves the renal function and structure.
Introduction
Cisplatin is commonly applied in the treatment of a diversity of solid tumors in medical practice due to its simple administration and great efficacy. 1 Cisplatin efficiency is proportionate to its dose. Nevertheless, cisplatin has a relatively limited safety margin and exhibits significant dose-limiting side effects, such as vomiting, nausea, ototoxicity, neurotoxicity, and particularly nephrotoxicity. 2 Hence, there is an urgent need to minimize renal damage in patients treated with cisplatin, also the cisplatin pathogenesis must be explained to create a novel medicine to mitigate cisplatin induced nephrotoxicity. 3
Platinum composite loaded polymeric carrier has been formulated for passive and active targeting. 4 To improve and manipulate limitations of traditional chemotherapies, nanoparticles have been designed and formulated with optimum size and surface characteristics to be loaded with active anticancer drugs, chitosan is a flexible transporter for gene and drug delivery. This is accredited to the reactive amino group of glucosamine in the C2 position, which could easily be used for chemical alteration, binding with medications and cell or tissue targeting moieties, and forming complexes with DNA, short interfering RNA, and oligonucleotides. 5
In recent years, the pathophysiological basis and the molecular mechanism underlying cisplatin mediated kidney damage have been elucidated. A previous study 6 illustrated that cytotoxicity endpoint is indicated by toxicity markers which include the formation of reactive oxygen species, depletion of antioxidant enzymes, lysosomal and mitochondrial membrane damage, stimulation of apoptosis-related gene expression, and finally cell death. There is a confirmed indication that inflammation, oxidative stress, apoptosis, and necrosis can play critical roles in cisplatin provoked renal toxicity. 7
Current evidence proposes that cisplatin penetrates cells causing mitochondrial dysfunction and lipid peroxidation yields accumulation in renal tissues, leading to fast production of reactive oxygen species (ROS) moreover activating the oxidative metabolic system. Accumulation of ROS above inadequate antioxidant mechanism consequences in the diminution of glutathione (GSH), superoxide dismutase (SOD), and a rise in MDA level in addition to the enhancements of inflammatory factors including iNOS, NF-κB, Tumor Necrosis Factor-alpha, and interleukin-1β.
8
The pro-inflammatory cytokines production, mitochondrial dysfunction also defects in the PI3K/Akt cellular pathway, collectively trigger the apoptosis signaling pathway.
9
Previous studies indicated that cisplatin induced excessive renal cell death and kidney damage which can be efficiently improved via regulating AMP-activated protein kinase (AMPK) also its downstream PI3K/Akt as well as p53 signal pathways.10,11 AMPK is a main metabolic switch, which is essential for cells to preserve the regular energy metabolic rate and redox equilibrium.
12
Therefore, AMPK activation may ameliorate cisplatin induced kidney damage.13,14 PI3K, the main downstream target of AMPK, regulates cellular characteristics such as proliferation, survival, and apoptosis15,16
The cytoplasmic transcription factor STAT1 participates in the signal cascade triggered via cellular stress and cytokines. Those signals facilitate its phosphorylation and translocation toward the nuclei, where it controls genes associated with inflammation, for instance; inducible nitric oxide synthase, 19 TNF-α, 20 also genes involved in apoptosis, for example, Fas, caspases, and TNF-related apoptosis-inducing ligand (TRAIL). 21 STAT1 also induces apoptosis in association with the cell cycle regulator p53. 22 Interestingly, STAT1 knockdown by Short interfering RNA (siRNA) against STAT1 alleviated cisplatin-mediated activation of p53, indicating that p53 activity is regulated by STAT1. 23
In this study, the treatment with Cisplatin NanoComposite (Cis NC) relatively reduced the toxic effect of free cisplatin on renal tissue. It was revealed that treatment with Cis NC limited the inflammation, apoptosis, and oxidative stress by regulating AMPK/PI3K/Akt - mTOR as well as STAT1/p53 signal pathways thus reserved renal tissue from damage.
Materials and methods
Chemicals
Chitosan (CHIT) with an average Mwt (100,000–300,000) was supplied by Sigma Co. USA. Magnesium nitrate hexahydrate (MgNO3.6H2O), NaOH, and other reagents were graded analytically and they were used as it is without any treatment, provided by Acmatic Co. Egypt. Urethane was obtained from Sigma Aldrich Corporation (USA). Cisplatin (cisPtCl2 (NH3)2) was acquired from Oncotec Pharma company.
Steps of preparation of Cisplatin NanoComposite (Cis NC)
Preparation of Magnesium Oxide NPs coated with CHIT
First, to prepare a 1% CHIT solution, 1gm of chitosan was dissolved in a 1% acetic acid solution (100 mL) and vigorously stirred with a magnetic stirrer till completely homogenate. Next, thoroughly combine equal volumes of CHIT and magnesium nitrate solution (1 Radiation facility
The MgO/CHIT/Cis milky white dispersion then exposed to 10 kGy dose of γ-radiation using the 137Cesium source (gamma cell-40) available at The National Center for Radiation Research and Technology (NCRRT), Egyptian Atomic Energy Authority (Cairo, Egypt), manufactured by Canadian Atomic Energy Co., Ltd (Ontario, Canada). Gamma radiation exposure will break down the CHIT molecular chains to a very low molecular weight, ensuring CHIT coating of Cisplatin and Magnesium Oxide NC. The formed precipitate after irradiation is washed several times by decantation for 2 days to ensure the removal of excess Na OH, the suspended mixture of MgO/CHIT/Cis was placed in a clean container and cautiously sealed till used.
Characterizations of MgO/CHIT and MgO/CHIT/Cis nanocomposite
The IR study of MgO/CHIT and MgO/CHIT/Cis nanocomposite samples were verified by Fourier-Transform Infrared Spectroscopy-Vertex 70 spectrophotometer (Bruker, Germany). Transmission Electron Microscopy (TEM) was applied to evaluate the bulk and shape of the prepared MgO/CHIT NPs. The formed nanoparticles powder was suspended in doubly distilled water then dispersions were dropped on an ultrathin carbon-reinforced copper grid and dehydrated in an oven overnight. TEM JEOL: JEM-100cx was applied for this determination. The X-ray diffractogram for Magnesium Oxide NPs was obtained by Rigaku 2550D/max VB/PC X-ray diffractometer.
Animals
Male Wister rats (of approximate weight 110–120 g) were acquired from the Nile Pharmaceutical Company, Cairo, Egypt. The rats were kept in the animal facility of the National Center for Radiation Research and Technology, Cairo, Egypt. Before beginning the experiment the rats were kept to adapt for a week. They were kept under standard laboratory conditions of light/dark cycle (12/12 h) at a temperature of 25 ± 2°C and humidity of 60 ± 5%. The rats were accommodated in cages with free access to drinking water and food. Rats were provided with suitable standard laboratory food (pellets). All animal experiments were carried out in accordance with the National Institutes of Health guide for the Care and Use of Laboratory Animals (serial:18–2019) in accordance with international ethical considerations.
Experimental design
Thirty rats were randomly separated to three main equal groups, 10 rats each, kept in separate cages and categorized as follow: a. Group 1: Normal control group
Rats received normal saline, designated as an untreated control group. b. Group 2: Cisplatin intoxicated group
Rats were treated 0.64 mg/kg b. wt. daily for 6 weeks, the choice of the dose was according to 1/10 LD50 injected intra-peritoneal c. Group 3: Cis NC treated group
Rats were treated 5.75 mg/kg b. wt. daily for 6 weeks the choice of the dose was according to 1/10 LD50 injected intra-peritoneal
By the end of the experiment, rats were anesthetized by ether to withdraw blood samples from the heart of each rat. Blood samples were permitted to clot and formerly were centrifuged for 15 min at 3000 r/min. Then, rats were sacrificed via cervical dislocation; part of the kidneys were quickly dissected, washed with ice-cold saline, dried, and weighed for biochemical analysis. Another part of the kidney was separated and was reserved for histopathological investigation.
Determination of kidney function
Serum urea and creatinine were measured by bio-diagnostic kits (Cairo, Egypt)
Determination of oxidative stress markers
Lipid peroxidation end products in kidney tissue homogenate were estimated via the thiobarbituric acid (TBA) test for malondialdehyde (MDA), following the method described by Yoshioka et al. 24 Reduced glutathione (GSH) contents were measured as defined by Beutler et al. 25
Determination of 8-OHdG, iNOS, and NADPH oxidase
ELISA kits (BioSource Int., Camarillo, California, United States) were used to determine levels of 8-OHdG, iNOS, and NADPH oxidase on kidney tissue homogenate consistent with the manufacturer’s protocol.
Determination of caspase3, interleukin-1β, and Tumor Necrosis Factor-alpha
The collected serum was used to determine concentrations of IL-1β, and TNF-alpha in addition to tissue caspase3 using ELISA technique (BioSource Int., Camarillo, California, United States) as described by the protocol supplied via the manufacturer.
Estimation of Nuclear factor kappa B, STAT1, and P53 gene expression in kidney tissue homogenate via quantitative real-time polymerase chain reaction (qPCR)
Extraction of RNA and synthesis of cDNA
To examine the alterations in mRNA expression for NFkB, STAT1, and P53, TRIzol reagent (Life Technologies, USA) was used to isolate total RNA from 50 mg kidney tissue. Reverse transcriptase (Invitrogen) was used to synthesize complementary DNA (cDNA) first strand using 1 mg RNA template.
Reverse transcription-polymerase chain reaction (RT-PCR)
Primers list used in polymerase chain reaction.

Western immunoblotting
TRIzol reagent (Invitrogen) was used to extract proteins from kidney tissue in accordance with the protocol supplied via the manufacturer (ThermoFisher). Each lane was loaded with 20 μg of protein and separated by 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) then transferred onto polyvinylidene difluoride (PVDF) membrane. Membranes were blocked with blocking solution containing 5% non-fat milk for 2h at room temperature then were washed with Tris-buffered saline with Tween 20 (TBS-T), and then incubated overnight at 4°C with primary antibodies toward AMPK, p-AMPK, PI3K, p-PI3K, Akt, p-Akt, mTOR, p-mTOR, and β-actin was used as a loading control. The membranes were washed several times with TBS-T before being incubated with horseradish peroxidase-conjugated secondary monoclonal antibodies for 2h at room temperature then were washed several times with TBS-T. Using Amersham detection kit to develop and visualize membranes by chemiluminescence in accordance with the manufacturer’s instructions before being exposed to X-ray film. Primary and secondary antibodies were acquired from Cell Signaling Technology (USA). Densitometric analysis of the autoradiograms was used to quantify AMPK, p-AMPK, PI3K, p-PI3K, Akt, p-Akt, and mTOR, p-mTOR proteins, using a scanning laser densitometer (Biomed Instrument Inc., USA). All values were normalized to β-actin and results were expressed as arbitrary units.
Histopathological examination
A histological examination was performed on renal tissue specimens from each study group to identify and compare the histological alterations that resulted from Cisplatin and Cis NC treatment. Tissue samples were fixed with 10% buffered formalin, dehydrated with a gradient concentration of alcohol, and immersed in xylene, then embedded in paraffin. A microtome (Leica, Germany) was used to take 5-μm thick sections of each tissue sample on glass slides. After clearing paraffin, the sections were stained with hematoxylin then counterstained with Eosin to be analyzed microscopically below the light microscope at original magnification of _x400, and the images were obtained by a digital camera (Nikon, ECLIPSE, TS100, Japan).
Statistical analysis
The data analysis was performed using SPSS (version 20) to analyze the data by one-way analysis of variance (ANOVA). After that a post hoc test (LSD) for multiple comparisons was performed. The obtained data were expressed as mean ± standard error (SE). Considering
Results
Chemical evaluations
Characterization of MgO/CHIT and MgO/CHIT/cisplatin nanocomposite
IR spectrum of cisplatin and MgO/CIS coated with chitosan
The characteristic IR bands for the MgO NPs, Cisplatin (Cis), and MgO/Cis coated with chitosan are illustrated in Figure 1. From infrared analysis of the Cis sample, the chief bands appeared due to extending vibrations of the OH group at 3326.5 cm-1 because of moisture content, and the stretching vibration at a range of 582.8 cm-1 to 603.9 due to the Pt-O-Pt linkage. The IR of MgO nano dispersion demonstrates a stretching vibration manner that appears in the range of 587–633 cm-1 was attributed to the Mg–O–Mg interactions. Two separate bands are noticed around 968.8–1636 cm−1, which are associated to the bending vibration of hydroxyl group provided by absorbed water molecules on the surface of the prepared particle samples. The stretching vibration of the OH group appeared at 3289.4 cm−1.
28
The appearance of the OH bands was owing to the adsorbed moisture on the MgO surface. IR spectroscopy of MgO NPs, Cisplatin, and Magnesium Oxide/Cisplatin coated with chitosan.
IR spectrum of magnesium oxide/Cisplatin coated with chitosan (MgO/Cis/CHIT) as shown in Figure 1 represents a combination of all constituents of the prepared nanocomposite sample, where the characteristic bands of CHIT were observed at: 3360 cm-1 (for the OH groups), 2972 cm-1 (for C–H group), 1777 cm-1 (for the amide carbonyl groups), 1424 cm-1 (for CH2 bending vibration), and 1059 cm-1 (for C=O group), (Figure 1). The appearance of these bands confirms the preservation of the CHIT characteristic structural features after the embedding of the MgO/Cis NPs within the polymeric matrix. Furthermore, a noticeable shift in the IR bands of CHIT-MgO NC resulted from the influence of embedded magnesium oxide NPs due to their interaction with CHIT chains. These observed shifts are especially because of the interaction between the NH and OH groups, confirming the H-bonding interaction of these groups with magnesium oxide molecules.
X-ray diffraction
The X-ray diffractogram of prepared MgO nanoparticles is illustrated in (Figure 2) which confirms that the synthesized MgO is of hexagonal structure
29
detected with no impurities. The average size of the prepared nanoparticles was estimated to be 4.3 nm using Scherer formula: Crystallite average size D = k λ/β (cos θ) XRD pattern of pure MgO NPs.
Where D is the crystallite size in nm, λ is the radiation wavelength (0.154 nm for Cu K), β is the bandwidth at half-height and θ is the diffraction peak angle and k is a constant (0.9). After a correction for the instrumental broadening, the average value of the magnesium oxide NPs size was found to be 4.26 nm.
Transmission electron microscopy of MgO NPs
Transmission electron microscopy (TEM) is the best applicable technique used to evaluate and investigate the morphology of materials, and to conclude the magnitude of molecular spots at the nanoscale. MgO samples were shown by TEM images in (Figure 3). The TEM images of MgO nanoparticles (Figure 3) show a systematic structure of MgO nanoparticles and also demonstrate that the MgO nanocrystallites have equivalent lamellar morphologies with a hexagonal shape and the sizes of the particles vary, giving an average size of nearly 10.32 nm. TEM photo of MgO NPs (dispersion).
Biochemical studies
The effect of Cis NC or cisplatin on kidney function
The effect of Cis NC or cisplatin on the renal function test of normal male rats.

Each value represents the mean ± standard error of 10 records.
asignificantly different from the control group at (
bsignificantly different from the cisplatin group at (
The effect of Cis NC or cisplatin on oxidative stress biomarkers in renal tissue
The effect of Cis NC or cisplatin on oxidative stress biomarkers in normal male rats.

Each value represents the mean ± standard error of 10 records.
MDA: malondialdehyde; 8-OHdG: 8-hydroxydeoxyguanosine; GSH: Reduced glutathione.
asignificantly different from the control group at (
bsignificantly different from the ciplatin group at (
The effect of Cis NC or cisplatin on the levels of NADPH oxidase and iNOS in renal tissue
To identify whether oxidative stress is associated with the progress of cisplatin induced kidney injury, we measured the levels of NADPH oxidase and iNOS in all groups. An apparent elevation in the content of NADPH oxidase and iNOS following cisplatin challenge or Cis NC administration was observed when compared with control. Also increases were more pronounced in the cisplatin intoxicated group compared with Cis NC administered rats. (Figure 4, Effects of Cis NC or cisplatin on the levels of NADPH oxidase, and INOs. All values were expressed as mean ± S.E. a 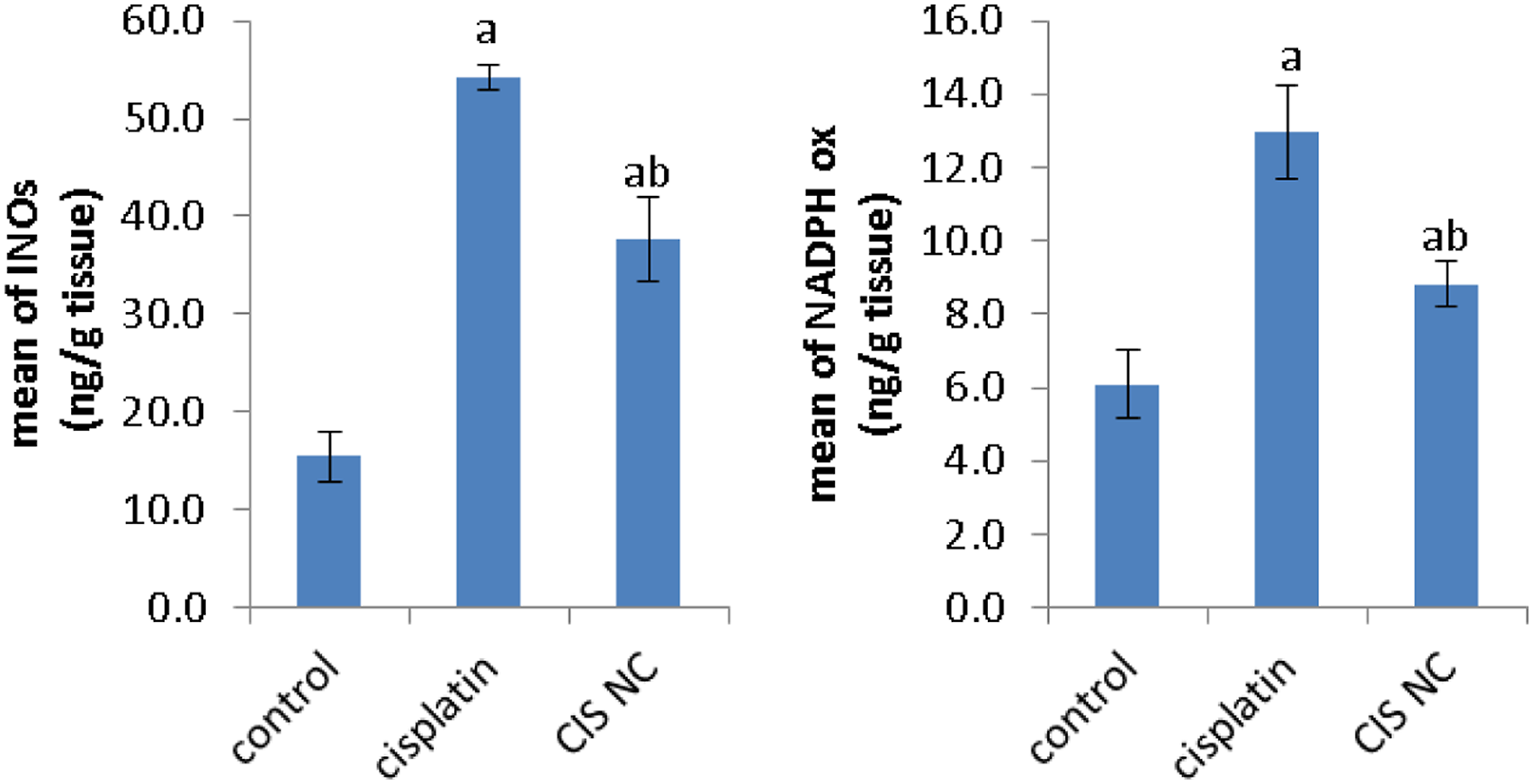
The effect of Cis NC or cisplatin on inflammatory cytokines
In comparison with normal group, the levels of TNF-α and IL-1β were significantly elevated in serum of cisplatin or Cis NC groups. However, the changes in the levels of inflammatory factors in the Cis NC treated group showed relatively reduced levels when compared to the cisplatin group (Figure 5, Effects of Cis NC or cisplatin on the content of inflammatory cytokines. All values were expressed as mean ± S.E. a 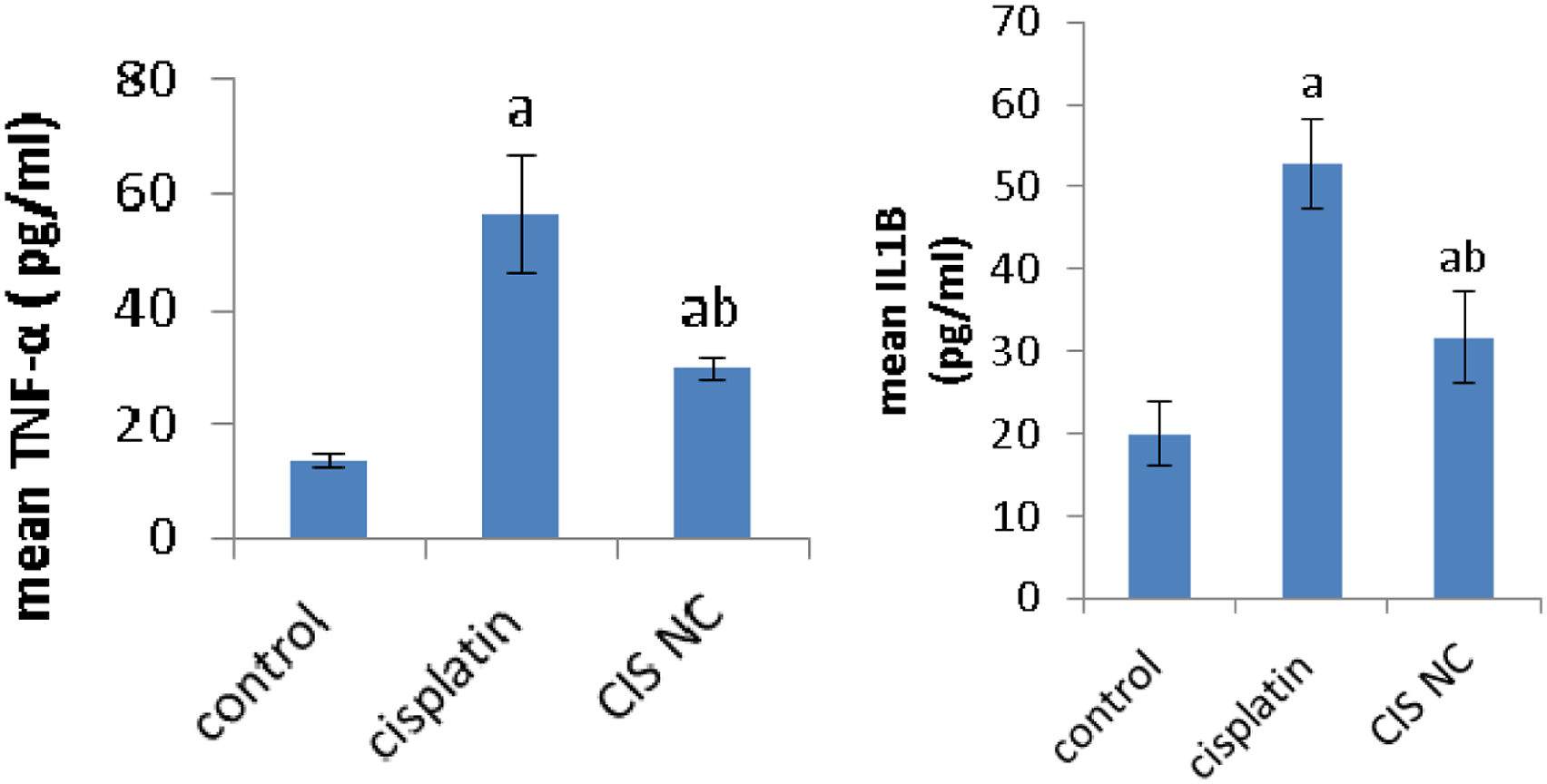
The effect of Cis NC or cisplatin on NF-κB gene expression
To confirm the involvement of cisplatin or Cis NC in the changes induced in inflammatory mediators, we further estimated NF-κB gene expression levels which increased significantly by cisplatin challenge or Cis NC administration above the levels observed in control. However, the level of NF-κB gene expression in the Cis NC group is less manifested compared with the Cisplatin group (Figure 6, Effect of Cis NC or cisplatin on NF-κB gene expression level in renal tissue All values were expressed as mean ± S.E. a 
Effect of Cis NC or cisplatin on STAT1 gene expression in renal tissue
STAT1 is an essential constituent of inflammatory and apoptotic signal pathways and shows a key role in regulating numerous cellular stresses. Cisplatin intensely augmented the expression of STAT1 above its detected levels in Cis NC or normal renal tissue (Figure 7). The effect of Cis NC or cisplatin on the gene expression levels of STAT1. All values were expressed as mean ± S.E. a 
Effect of Cis NC or cisplatin on p53 gene expression in renal tissue
The p53 tumor suppressor protein shows a key role in regulating numerous cellular stresses and cell apoptosis. Cisplatin caused a greater increase in the expression of p53, than in Cis NC when compared to control (Figure 8). The effect of Cis NC or cisplatin on the gene expression levels of p53. All values were expressed as mean ± S.E. a 
The effect of Cis NC or cisplatin on caspase 3 level in renal tissue
In order to study the apoptotic influence of cisplatin or Cis NC, we measured the content of executioner caspase-3 by ELISA in renal tissue, where cisplatin induced greater activation of the caspase-3 level compared to control or Cis NC treated groups (Figure 9). Effect of Cis NC or cisplatin on caspase 3 level in renal tissue. All values were expressed as mean ± S.E. a 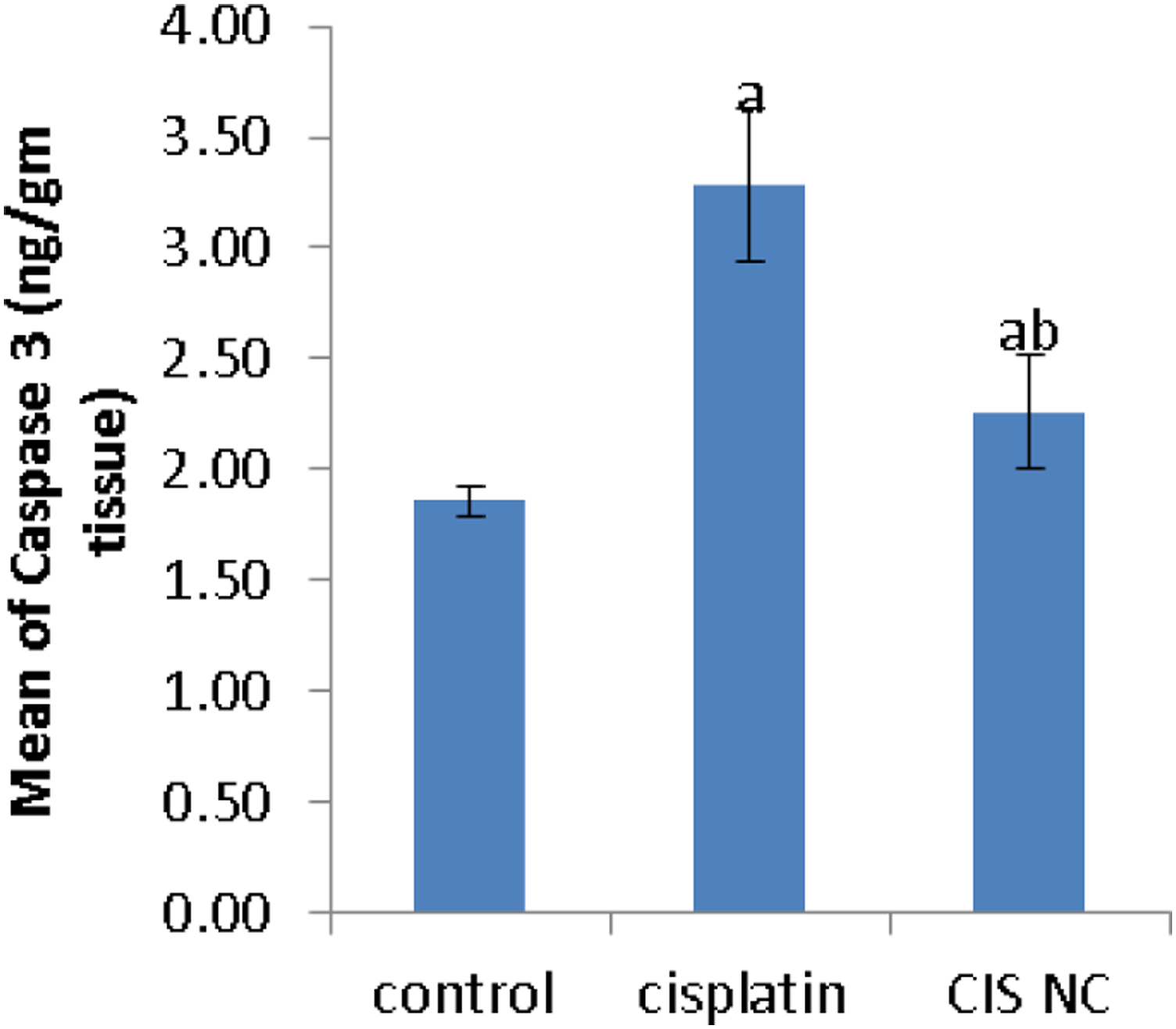
The effect of Cis NC or cisplatin on the AMPK/PI3K/Akt and mTOR signaling pathways in renal tissue
Figure 10(A) and (B) illustrated that the expression of p-AMPK, p-PI3K also p-Akt were reduced after cisplatin intoxication. Remarkably, Cis NC slightly affects the expression of p-AMPK, p-PI3K, and p-Akt when compared to the cisplatin intoxicated group. Furthermore, the phosphorylated form of mammalian target of rapamycin (mTOR) also increased in the renal tissue of cisplatin or Cis NC treated rats compared to control rats. However, the changes in Cis NC treated rats are less manifested as compared to Cisplatin intoxicated rats. (Figure 10(A) and (B)). A, B. Effects of Cis NC or cisplatin on the protein expression level of AMPK/PI3K/Akt/mTOR signaling pathway along with the used loading control β-actin. Protein expression was estimated via quantitative analysis of relative protein expression in the kidney of each group of rats. The column charts show the ratio between the phosphorylated and total of the respective protein. Values were expressed as mean ± S.E. 
Effect of Cis NC or cisplatin on renal histopathological changes
Histopathological alterations of the rat kidney were considered to evaluate the efficiency of Cis NC in protecting the kidney from the deleterious effects of free cisplatin. Examination with a light microscope of kidney tissues in a normal rat showed a typical glomerular structure and renal tubular interstitial without indication of inflammatory infiltration or cell necrosis (Figure 11 (a)). The rat intoxicated with cisplatin revealed massive inter-tubular hemorrhage replaced some tubules with degenerative changes in the tubules (Figure 11(b)). Moreover, the renal tubules in some cases are expanded, in others replaced by massive leukocytes, which may be progressive to necrosis. In other rat kidneys treated with cisplatin dilated renal tubules were seen besides leukocyte infiltration. In addition, the renal cortex presented glomerular atrophy, the remaining glomerular tuff was atrophied owing to minor fibrosis, Bowman’s space was widened, and the basement membrane was thickened (Figure 11(c), (d), and (e)). In comparison, Cis NC treatment revealed a normal structure devoid of degenerative modifications or necrosis. In some specimens, congested intertubular blood vessels with albuninous casts in renal tubules were shown (Figure 11(f) and (g)). a; The kidney of a control rat shows normal structure (H& E × 400), b; The kidney of a cisplatin rat presents massive intertubular hemorrhage replacing some tubules with degenerative changes in the tubules (H& E × 400), c; The kidney of a cisplatin administered rat revealed extended renal tubules and other tubules replaced by massive leukocytes (H& E × 400), d; The kidney of a cisplatin rat shows glomerular atrophy, widened capsular spaces, dilated renal tubules, and leukocyte infiltration (H& E × 400), e; The kidney of a cisplatin administered rat shows degenerative changes of renal epithelium which progressive to necrosis of tubules and other loss of its details (H& E × 400), f; kidney of the Cis NC rat with normal renal tissues (H& E × 400), and g; The kidney of the Cis NC rat shows normal renal tissues with congested intertubular blood vessels with albuminous casts in the renal tubules (H& E × 400).
Discussion
Several methods have been tried to minimize toxicity and enhance the efficacy of cisplatin, one of which is to apply nano-technology as a means of drug delivery method. Chitosan is one of the most plentiful natural polysaccharides, it has been considered comparatively safe because of its biodegradable and biocompatible properties, thus increasing its biomedical applications as drug delivery systems.30,31 In this study, we demonstrated that Cisplatin Nano Composite efficiently manifested lower side effects on renal tissue, it has less nephrotoxic impact compared with cisplatin. Cisplatin induced kidney damage by triggering inflammation, oxidative stress, and apoptosis and the fundamental molecular mechanisms can be via inhibition of AMPK/PI3K/Akt and stimulation of mTOR, STAT1/p53 signaling pathways.
Cisplatin nephrotoxicity is mediated by different cellular events, including DNA injury, reduced protein synthesis, mitochondrial dysfunction, and membrane peroxidation are due to failure to scavenge the produced free radical molecules in the body. 32 In accordance with the preceding study, 33 our findings revealed that the morphological changes and kidney dysfunction represented by elevated levels of biomarkers of renal function tests including urea and creatinine caused by cisplatin challenge were associated with enhanced lipid peroxidation, GSH depletion, in addition to an increased accumulation of oxidative damage marker 8-OHdG, which may be a consequence of cisplatin-mediated increase in free radical generation or decreased redox potential of the mitochondria. 34 Thus the current induced oxidative stress cascade caused by cisplatin intoxication contributes to renal tubular damage in rats. Previous reports have also clarified that accumulative cisplatin causes the generation of great quantities of ROS in cells, which leads to the progress of oxidative stress, which is a dominant pathogenic element in cisplatin induced kidney injury 35 However, we found a negligible increase in MDA levels, marginally decreased GSH levels, and a non-remarkable elevation in 8-OHdG in the Cis NC treated group as compared to the dramatic disturbance caused by cisplatin. It is obvious from our finding that Cis NC was not efficient in producing significant oxidative stress on normal renal cells, thus confirming its safety and biocompatibility. This finding was in agreement with Leekha et al. 36
As revealed during the present study, cisplatin increased renal NADPH oxidase in addition to iNOS protein expression. Current reports propose that, as well as ROS mediated through NADPH oxidase expression, nitrosative stress plays a significant part in cisplatin induced nephrotoxicity. 37 Our results revealed that Cis NC treatment did not result in demonstrable changes in oxidative/nitrosative stress in renal tissue as compared with cisplatin.
In addition, there is evidence that locally produced ROS triggered the production of TNF-α post-renal injury, which through p38 MAPK activate NF-κB.
7
Our data revealed activation of NF-κB associated with an elevated significant rise in the pro-inflammatory cytokines level including; IL-1β and TNF-α in the serum of animals intoxicated with cisplatin as associated with the relative control. Also, the results obtained in this research involve upregulation of STAT1 in cisplatin mediated nephrotoxicity. Cisplatin activated STAT1 through ROS generation by means of NADPH oxidase. A variety of genes involved in inflammation were regulated by STAT1, such as iNOS,
19
TNF-
Apoptosis is a biological mechanism triggered by multiple variables and is also coordinated by a number of cell death signaling pathways. 38 Apoptotic machinery activation is linked to mitochondria, where p53 activation promotes the transcription of pro-apoptotic genes such as Bax, resulting in enhanced mitochondrial permeabilization and release of cytochrome c, which stimulates caspase-3 and initiates apoptosis via an oxidative stress related mitochondrial pathway. 39 It was confirmed that cisplatin-mediated renal injury was associated with cell apoptosis. 9 we revealed that cisplatin induced activation of the executioner caspase-3 and p53, the upstream regulator of the apoptotic pathway, thus triggering cell apoptosis. Previous reports demonstrated that cisplatin challenge evoked apoptosis and inhibited the cell cycle in kidneys by suppressing the expression of Bcl-2 and increasing Bax and p53 expression; proposing that the inhibition of p53 activation may be a main objective for alleviating cisplatin nephrotoxicity. 40 Therefore, in this study, the gene expression of p53 was measured. The results indicated that the p53 protein expression level was significantly amplified after cisplatin intoxication, which was shown to a lesser extent in the group treated with Cis NC.
Earlier studies have also established that cisplatin induces apoptosis via AMPK-mediated signaling pathways, which is crucial in the enhancement of cisplatin mediated renal damage. 41 Therefore, cisplatin induced renal injury can be mitigated through AMPK stimulation.13,14 In addition, the downstream target of AMPK is the PI3K/Akt signal and is closely associated with survival and cell growth, The PI3K/Akt signaling pathway’s role in renal damage is debatable; several researchers have established that stimulation of PI3K/Akt was associated with renal damage and was correlated with the generation of abundant ROS as well as apoptosis following cisplatin administration,10,42 whereas others found that the PI3K/AKT signaling pathway was inhibited following cisplatin administration and was associated with mitochondrial dysfunction. 43 Our current study postulated that cisplatin treatment caused inhibition of AMPK/PI3K/Akt expression in renal tissue, which was reliable with earlier results. 13 Also, we confirmed that Cis NC treatment possesses a less inhibitory effect on the phosphorylation cascades of AMPK/PI3K/Akt when compared to cisplatin. As protein synthesis and cell growth are controlled by the essential regulator mTOR which is controlled via its upstream regulator PI3K/Akt. 40 We investigated the influence of cisplatin or Cis NC on mTOR stimulation; we found that Cis NC treatment showed no significant increased phosphorylation of mTOR compared to free cisplatin. PI3K/Akt can be positively activated by its direct regulator AMPK, the AMPK pathway can be suppressed by triggering ROS, causing inhibition of its downstream substrate PI3K/Akt phosphorylation, also AMPK is a negative regulator of mTOR, we demonstrated that cisplatin challenge caused inhibition of AMPK, resulting in mTOR over-activation. Researches on animal models demonstrated that the mTOR signaling pathway was activated in chronic kidney disease, where mTOR increases pro-inflammatory and pro-fibrotic cytokines expression in addition to the infiltration of inflammatory cells in the interstitium, leading to renal function damage. Obviously, activation of AMPK and inhibition of mTOR ameliorate the progression of renal injury induced upon cisplatin challenge in different models.14,44 Several studies have confirmed that PI3K/Akt regulates the Bcl-2 anti-apoptotic protein expression and is an upstream effector of p53.45,46 Therefore we postulated that induced kidney cell apoptosis by cisplatin may be via inhibition of AMPK activity and its downstream survival signal PI3K/Akt with triggered activation of p53 and the executioner caspase 3, this effect is less pronounced by Cis NC on renal cells.
Cisplatin intoxicated rats revealed condensation of kidney cell necrosis and protein cast, as well as vacuolization of the renal cortex, where Cis NC shows relatively normal architecture without degenerative changes or necrosis. All these collective results suggest that Cis NC exerted potential renal protection compared to free cisplatin. Previous studies have also examined the impact of cisplatin loaded nanocarriers on cells in comparison with cisplatin, and the findings demonstrated that cisplatin treated renal cells revealed a greater percentage of apoptotic and necrotic cells compared to the cisplatin loaded Nano-carriers, 5 which is in agreement with the current study.
The results of this work indicated that preparation of cisplatin Nano-composite may decrease cisplatin kidney injury; this is probably owing to the gradual release of cisplatin from the Nano-carriers and decreased cisplatin toxicity on renal cells, thus reserving the renal structure and function.
However, more research is needed to identify the drug toxicity, and effectiveness of the formulated Nano-composite on further in-vivo and large scale of cell lines, in addition to the need of combined treatment with novel designed Nano-antioxidants 47 to reduce oxidative stress related toxicity of cisplatin. Also, understanding of biological processes and available studies concerning the pharmacokinetics and bioavailability of newly designed Nano based drugs should be considered to develop an appropriate treatment strategy. Furthermore, recent advances in nanotechnology have resulted in the development of a Nano-carrier based cisplatin in a various designated forms such as polymeric NPs, lipid-based Nano formulations, inorganic NPs, and carbon-based NPs, under preclinical and clinical phase trials with innovative impact in cancer treatment targeting with increased safety margin and minimized toxicity to normal cells. 48
Conclusions
We can conclude that Cis NC may be a valued promising agent to minimize nephrotoxicity induced by cisplatin where it exerts a minimal disrupting effect on redox balance in renal tissue, and less inhibitory effect on AMPK activity, and its downstream survival signal PI3K/Akt with a low stimulatory effect on expression of mTOR and STAT1/P53 dependent apoptotic and inflammatory signaling pathways. However, future additional preclinical studies are needed to elucidate the beneficial effects of using Nano-based drugs to improve bioavailability and reduce drug toxicity.
Declarations
Footnotes
Acknowledgements
We wish to acknowledge the contribution of Dr Faten Ismail Abou El Fadl associate Prof. of chemistry,
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Consent for publication
Was provided to SAGE.
Availability of data and materials
All data generated or analyzed during this study is included in this article.
Ethics approval
All animal experiments were carried out in accordance with the National Institutes of Health guide for the Care and Use of Laboratory Animals (serial: 18–2019) in accordance with international ethical considerations.
Animal welfare
The present study followed international and national guidelines for animal treatment and complied with relevant legislation.
