Abstract
This study was aimed to investigate the protective effects of caffeic acid phenyl esther (CAPE) and Intralipid (IL) against hepatotoxicity and nephrotoxicity caused by acute intoxication of glyphosate (N-phosphonomethyl)glycine) (GI) in rats. Forty-nine Wistar Albino rats were randomly divided into seven groups as: I, Control; II, Intralipid (IL) (18.6 mL/kg, orally); III, CAPE (10 µmol/kg, intraperitoneally); IV, GI (4 mg/kg/day, intraperitoneally); V, GI + IL; VI, GI+CAPE; and VII, GI + IL + CAPE. Total antioxidant status (TAS) and total oxidant status (TOS) levels were measured in serum samples. Tissues were analyzed with hematoxylin and eosin (H&E) staining protocol. Bcl-2, Bax, and caspase-3 were evaluated by immunohistochemical method. The results revealed that, in hepatic tissues, the TAS levels were lower and the TOS levels were higher in the GI group compared to other groups. In renal tissues, the TAS levels were significantly lower in the GI group than in the control, IL, CAPE, and GI + IL + CAPE groups. The TOS levels were significantly higher in the GI group than in the control group. Moreover, histopathological analysis revealed severe hepatotoxicity in the GI group. In the GI + CAPE + IL group, hepatotoxicity recovered significantly. Nephrotoxicity was also observed in the GI group and moderately reduced in the GI + CAPE group. Biochemical results were confirmed by histopathologic examination. The results also revealed that CAPE and IL, due to their antioxidant effects, have a decreasing effect against both hepatotoxicity and nephrotoxicity caused by GI. Therefore, CAPE and IL may function as potential agents for supportive therapy since they decrease organ damage, or may facilitate the therapeutic effects of the routine treatment of patients with GI poisoning.
Keywords
Introduction
Glyphosate (N-phosphonomethyl)glycine) (GI), the most common organophosphorus compound used as herbicide, is widely used to kill unwanted plants both in agriculture and in non-agricultural landscapes. GI is the active ingredient in glyphosate-surfactant herbicides which are extensively used as non-selective herbicides. GI usually consists of glyphosate salts (e.g. isopropylamine/IPA, potassium, and di-ammonium salt), surfactants, anti-foaming agents, color agents, biocides, and inorganic ions for pH adjustment. 1
Glyphosate’s inhibition of cytochrome P450 (CYP) enzymes is an overlooked component of its toxicity to mammals. CYP enzymes play crucial roles in biology, one of which is to detoxify xenobiotics. Thus, GI enhances the damaging effects of other food-borne chemical residues and environmental toxins. Negative impact on the body is insidious and manifests slowly over time as inflammation damages cellular systems throughout the body. Some studies involving lifelong exposure in rodents have demonstrated liver and kidney dysfunction and a greatly increased risk of cancer with shortened lifespan. We evaluated the effects of GI against hepatotoxicity, nephrotoxicity, and oxidative stress.2,3
The clinical picture of severe GI poisoning may result in some clinical complications such as gastroenteritis, respiratory diseases, alterations in mental status, nephrotoxicity, acute renal failure, shock, neurological sequel, and death.4,5
Caffeic acid phenethyl ester (CAPE) is a compound that is structurally similar to the flavonoid found in bee propolis. It is an active component of propolis extract which has preventive effects on the reactive oxygen products and it occurs as a result of oxidative stress in toxic failure and ischemia-reperfusion injury and has a tissue protective effect. 6 Recent studies have shown that CAPE has anti-inflammatory, antioxidant, immunomodulatory, antimycotic, and anticarcinogenic effects.7–13
Lipid solutions (lipid molecules, Intralipid [IL] 20%, Liposyn 20%) have recently been reported to restrain the lipid-soluble toxic substances in the poisonings caused by lipid-soluble drugs (e.g. clomipramine, propranolol, bupropion, haloperidol, organophosphates). Nevertheless, these solutions have not been employed in the management of poisonings since their effects have not been studied. 14 We investigated the effectiveness of IL emulsion, considering that it may inhibit the effects of GI and reduce its toxic effects.
To date, no supportive therapy reducing organ damage caused by GI poisoning has been defined in the literature.
In this study, we aimed to investigate the protective effects of CAPE and IL against hepatotoxicity and nephrotoxicity caused by GI.
Materials and methods
Animals, care, and nutrition
A total of 49 female Wistar-Albino rats weighing 200–250 g were randomly divided into seven groups. Appropriate laboratory conditions along with a 12-h light/dark cycle and a room temperature of 21 ± 3°C were provided for all the rats. The animals were obtained from the Experimental Research Center of Mustafa Kemal University at Hatay. Ethical approval was obtained from the Experimental Animals Ethics Committee at Necmettin Erbakan University Experimental Medical Research Center, Konya, Turkey.
Animals and treatment
The 49 rats were classified into seven groups as control (n = 7), 20% IL (n = 7, 18.6 mL/kg, orally) (Intralipid; Fresenius Kabi AG, Turkey), 15 CAPE (CAPE Sigma C8221, Sigma-Aldrich Chemie Gmbh, Steinheim, Germany) (n = 7) (10 µmol/kg, intraperitoneally), 6 GI (n = 7) (4 mg/kg/day, orally), GI + IL (n = 7), GI + CAPE (n = 7), and GI + IL + CAPE (n = 7). CAPE and IL were given to the rats immediately after GI administration. The rats were sacrificed using ketamine (50 mg/kg; intraperitoneal [i.p.]; Ketalar; Parke Davis, Eczacibasi, Istanbul, Turkey) and xylazine (5 mg/kg; i.p.; Rompun; Bayer AG, Leverkusen, Germany) immediately after the administration of the drugs. The liver and kidney tissues were removed and longitudinally divided into two pieces. One piece was used for biochemical assays and the other piece was used for histopathological evaluations. Blood samples were drawn directly by cardiac puncture and the supernatants were removed after centrifugation, and stored at −80°C. Total oxidant status (TOS) and total antioxidant status (TAS) levels were measured in the serum samples.
Biochemical analysis
Serum TAS levels were measured by a novel, automatic, and colorimetric method developed by Erel. 16 This method provides perfect precision values lower than 3%. The TAS results were stated as nmol Trolox equivalent/mg protein and the TOS levels were expressed as nmol H2O2 equivalent/mg protein.
Immunohistochemistry
Immunohistochemical analysis was conducted using a Leica Bond-Max automated IHC/ISH platform (Leica Microsystems Inc., Buffalo Grove, IL, USA). Four micrometer paraffin sections were dewaxed in a Bond Dewax solution and rehydrated in alcohol and Bond Wash solution (Leica Microsystems). The retrieval of antigens was conducted using a high-pH (ER2) retrieval solution for a period of 15 min, which was followed by endogenous peroxidase blocking for 5 min on the device. The applications of anti-mouse monoclonal antibody Bcl-2 (C-2: sc-7382, Santa Cruz Biotechnology, Inc., in dilution 1:200), anti-mouse monoclonal antibody Bax (B-9: sc-7480, Santa Cruz Biotechnology, Inc., in dilution 1:100) anti-mouse caspase-3 (CPP32), and monoclonal antibody (clone JHM62, Leica Biosystems Ltd., Newcastle, UK) were performed at the rate of 1:50 dilution for 60 min at room temperature. A Bond Polymer Refine Red Detection system (Leica Microsystems) was used for detection with a 15-min post-primary step followed by the incubation with alkaline phosphatase-linked polymers for 25 min. Afterwards, the sections were counterstained with hematoxylin, dehydrated in alcohols, and mounted with the mounting medium (Sakura Finetek USA Inc., Torrance, CA, USA). The preparation stage of the tissues was monitored by two histopathologists blinded to the experimental groups. For counting the apoptotic cells, 10 randomly selected microscope fields were analyzed under a 400× magnification in a blind fashion.
Histopathologic analysis
Hematoxylin and eosin procedure
The tissues were first submerged into a solution containing 10% formaldehyde and then embedded into paraffin blocks. Four-micron sections were cut from the blocks by using a microtome. All the tissues were stained with H&E by using standard protocols. All the slides were analyzed by an Olympus BX51 microscope (Tokyo, Japan).
Histopathologic examination of the liver
The severity of hepatic injury was assessed using an ordinal grading system as follows: Grade 0, minimal or no sign of injury; Grade 1, mild injury accompanied by cytoplasmic vacuolation and focal nuclear pyknosis; Grade 2, moderate injury with enlarged nuclear pyknosis, cystoplasmic hypereosinophilia, and loss of intercellular borders; and Grade 3, severe necrosis with disintegration of hepatic cords, hemorrhage, and neutrophil infiltration. 17
Histopathologic examination of the kidneys
Renal injury was graded as follows: Grade 0, no diagnostic change; Grade 1, tubular cell swelling, loss of brush borders, nuclear reduction with up to one-third of tubular profile; Grade 2, in addition to Grade 1, greater nuclear reduction with up to two-thirds of tubular profile; and Grade 3, greater nuclear reduction with more than two-thirds of tubular profile.
Statistical evaluation
Data were expressed as mean ± standard error of mean (SEM). Statistical evaluations were performed using SPSS 11.5 for Windows (SPSS, Chicago, IL, USA). Normal distributions were determined by one-way ANOVA test, followed by Tukey’s post-hoc test. A P value of <0.05 was considered significant.
Results
Total antioxidant status (TAS) and total oxidant status (TOS) levels
The results revealed that, in hepatic tissues, the TAS levels were significantly lower in the GI group compared to other groups and the TOS levels were higher in the GI group compared to other groups (Table 1). In renal tissues, the TAS levels were significantly lower in the GI group than in the control, IL, CAPE, and GI + IL + CAPE groups. The TOS levels were higher in the GI group compared to other groups (Table 2).
Comparison of TAS and TOS levels in hepatic tissues.
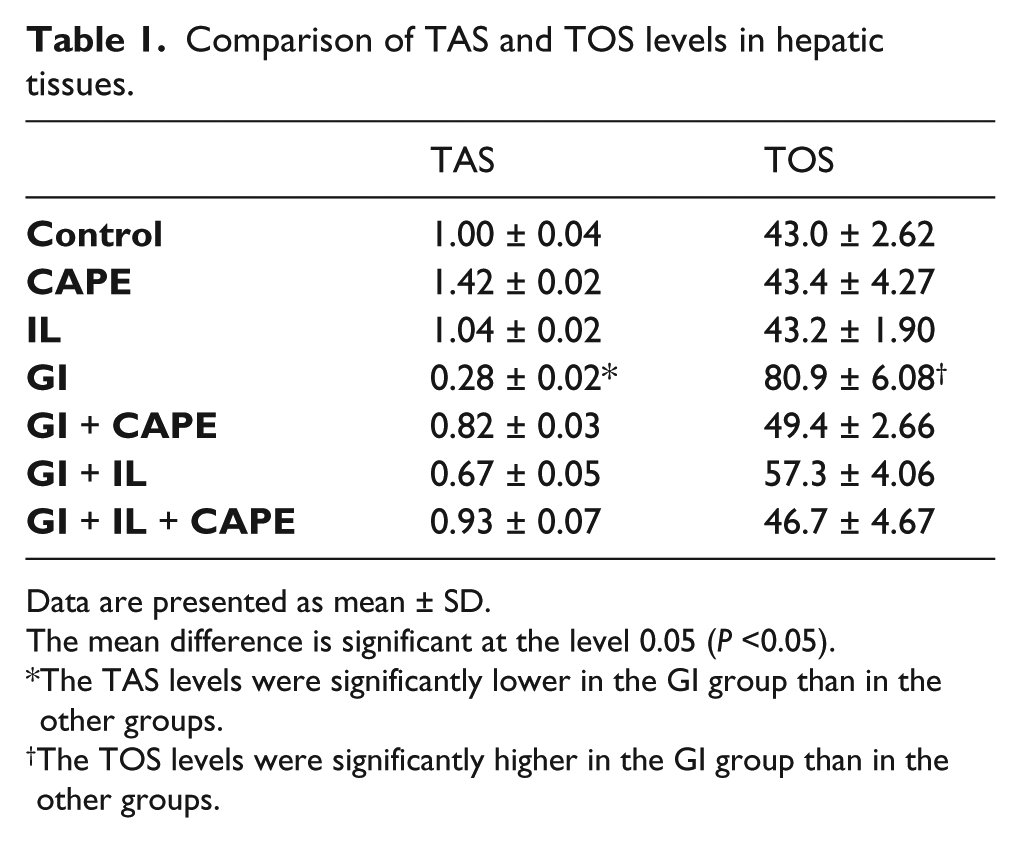
Data are presented as mean ± SD.
The mean difference is significant at the level 0.05 (P <0.05).
The TAS levels were significantly lower in the GI group than in the other groups.
The TOS levels were significantly higher in the GI group than in the other groups.
Comparison of TAS and TOS levels in renal tissues.

Data are presented as mean ± SD.
The mean difference is significant at the level 0.05 (P <0.05).
The TAS levels were significantly lower in the GI group than in the Control, CAPE, GI + CAPE + IL groups.
The TOS levels were significantly higher in the GI group than in the Control group.
Histopathologic results
Liver
H&E
Normal liver histomorphology was observed in the control group (Figure 1a). In the GI group, moderate toxicity, cytoplasmic hypereosinophilia or vacuolization, loss of intercellular borders, and inflammation or necrosis were detected (Figure 1b). In the GI + CAPE + IL group, a process of regressive regeneration that included regenerative changes suggestive of normal liver histology and focal cytoplasmic hypereosinophilia were observed (Figure 1c).

H&E method: (a) Normal liver histology (control group), (b) Severe liver damage and necrosis (GI), (c) Regressive regeneration including regenerative changes suggestive of normal liver histology and focal cytoplasmic hypereosinophilia (GI + CAPE + IL), (H&E, ×200). Immunohistochemical method (IHK): Apoptosis resulting from Bcl-2, Bax and caspase-3; (d) Immunoreactivity as opposed to the control group (IHK, ×200), (e) Immunoreactivity suggesting increased apoptosis in the liver tissues (GI) (IHK, ×400), (f) Apoptosis level in the GI + CAPE + IL group was only lower than the one in the GI group (IHK, ×400).
Immunohistochemical results
No immunoreactivity was observed in the control group (Figure 1d). In the GI group, signs of increased immunoreactivity were detected in the liver tissues (Figure 1e). We considered that the GI group was the only group in which the apoptosis significantly increased compared to the control group. In the GI + CAPE group, the apoptotic cell count was found lower compared to the GI group (Figure 1f).
Kidney tissues
H&E
Normal kidney morphology was observed in the control group (Figure 2a). In the GI group, mild tubular cell swelling, nuclear reduction, loss of brush borders, and severe renal tubular toxicity were observed (Figure 2b). Normal kidney morphology was observed in the GI + CAPE group (Figure 2c).
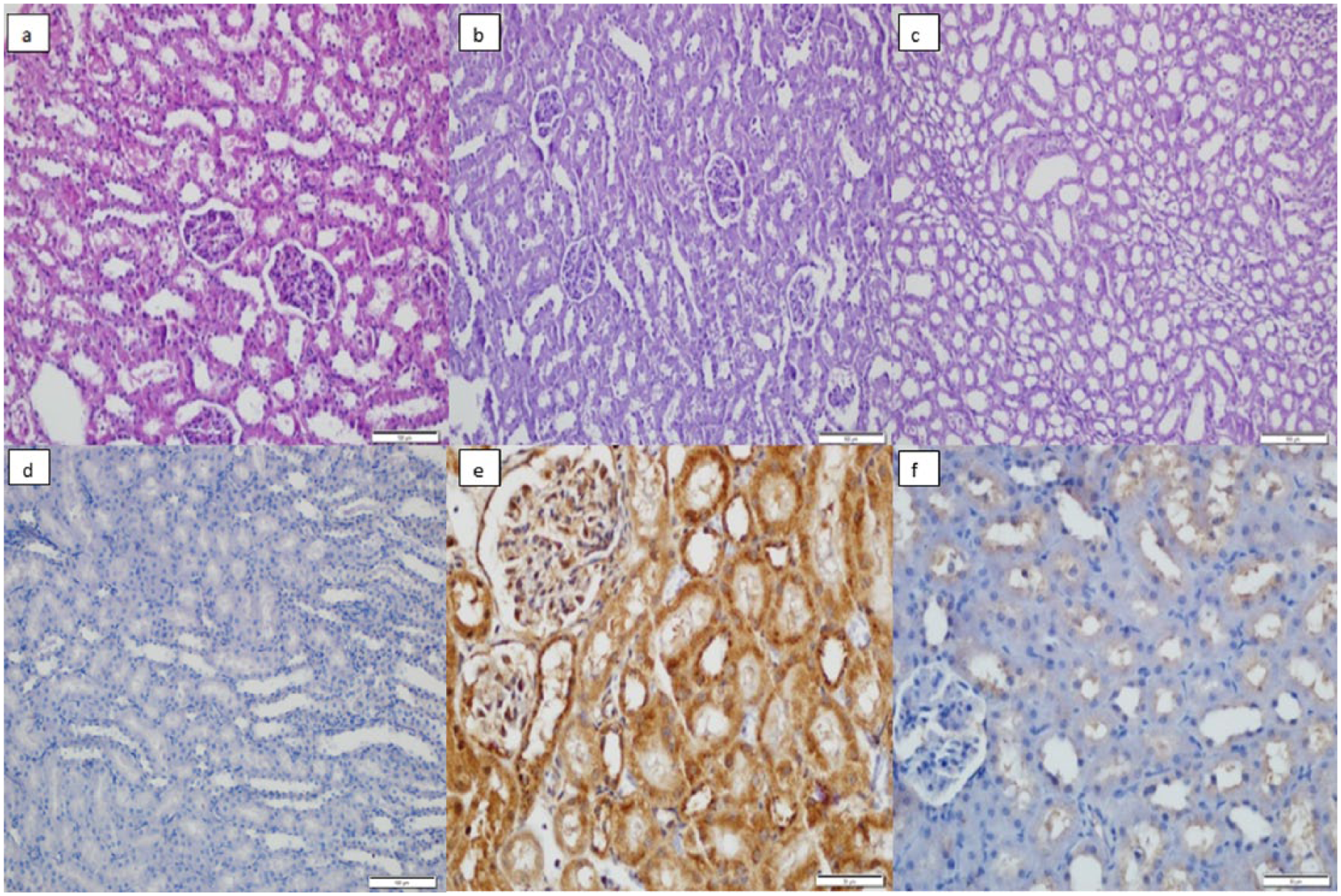
H&E method: (a) Normal kidney histology, (b) Severe renal tubular toxicity (GI), swelling of the tubular cells with brush border loss, degeneration, and many nuclear loss of the tubular cells, (c) Recovery in the kidney tissues (GI + CAPE). Immunohistochemical method: (d) Normal kidney histology, (e) Kidney damage (GI), swelling of the tubular cells with brush border loss, degeneration, and many nuclear loss of the tubular cells, (f) Recovery in the kidney tissues (GI + CAPE).
Immunohistochemical results
Normal kidney immunohistochemical results were found in the control group (Figure 2d). The histopathologic changes in the kidney tissues were examined by the apoptotic indicators including Caspase-3, bcl-2, and Bax. It was also revealed that GI led to increased apoptosis (Figure 2e) and GI + CAPE caused moderate apoptosis (Figure 2f).
Discussion
Acute kidney injury is the most common clinical presentation following GI intoxication. The kidney is an organ for excretion of GI components. For these reasons, early evidence of kidney injury could be used to predict the risk of a fatal outcome in GI intoxication. 1 CAPE has anti-inflammatory, antioxidant, and immunomodulatory properties. It also has protective effects against nephrotoxicity.7–13,18 Therefore, we aimed to investigate the effects of CAPE and IL against hepatotoxicity and nephrotoxicity caused by GI intoxication and thus we measured TAS and TOS parameters in liver and kidney tissues and we also evaluated apoptotic indicators including Caspase-3, bcl-2, and Bax by immunohistochemical analysis.
It is well established that glyphosate, a member of the general class of organophosphates, inhibits the enzyme 5-enolpyruvylshikimic acid-3-phosphate synthase (EPSP synthase), which is the rate-limiting step in the synthesis of aromatic amino acids in the shikimate pathway in plants. This pathway, while not present in mammals, is present in algae, Archaea, bacteria, fungi, prokaryotes, and unicellular eukaryotic organisms.2,19,20 Therefore, GI is a more popular agent than organophosphate pesticide (OP) in controlling insect pests of agriculture. Moreover, since OP inhibits acetyl cholinesterase in mammals, OP causes more clinic toxic effects compared to GI.
Studies reporting on GI poisoning have revealed that glyphosate-induced oxidative damage leads to a significant decrease in GSH levels and a significant increase in the MDA levels of the liver and kidney tissues. It has also been reported that treatment with G. biloba produces amelioration in the indices of hepatotoxicity and nephrotoxicity. In addition, in vivo results have shown that G. biloba extract is a potent protector against glyphosate-induced toxicity and its protective role is dose-dependent. 21
Although there are numerous studies reporting on oxidative stress induced by OP, there are very few studies investigating GI poisoning. In this study, the TOS and TAS levels in the liver and kidney tissues were measured as indicators of oxidative balance. The results revealed that GI administration led to significantly increased TOS and decreased TAS levels in both hepatic and kidney tissues compared to the control and other groups (Tables 1 and 2). However, there was no significant difference between the control and IL, CAPE, and GI + IL + CAPE groups. These findings indicate that GI has deteriorating effects on oxidative balance in both hepatic and renal tissues. Moreover, they also indicate that a combined therapy with CAPE and IL may reduce the toxic effects caused by GI.
A recent study suggested that IL administration seems to be an effective treatment modality in patients who ingested sufficient amount of glyphosate herbicide that is expected to bring about significant toxicity. 22 Another study reported that dichlorvos, which is an organophosphate, is able to induce apoptosis. 23 In our study, both the histopathological analysis and the biochemical results showed obvious morphological changes in the liver tissues in the GI group. Moreover, they also detected cytoplasmic hypereosinophilia or vacuolization, loss of intercellular borders, and inflammation or necrosis in the hepatocytes. These results were consistent with the signs of moderate toxicity. We also found that a process of regressive regeneration including regenerative changes considered as normal liver histology and focal cytoplasmic hypereosinophilia were observed in the GI + CAPE + IL group (Figure 1). In previous studies, GI is reported to cause acute tubular and glomerular necrosis and apoptosis, leading to impairment of kidney functions. 1 In the present study, however, histopathological evaluation revealed that the kidney tissues in the GI group presented with mild tubular cell swelling, nuclear reduction, and loss of brush borders due to the severe renal tubular toxicity caused by GI. These effects were moderately improved by GI plus CAPE (Figure 2).
CAPE has been reported to restore the liver functions impaired by Paraquat, which is one of the most widely used herbicides. 6 Similarly, we also observed immunoreactive changes and increased apoptosis in the immunohistochemical analysis of the liver tissues in the GI group. However, a significant increase in the count of apoptotic cells in liver tissues was only observed in the GI group compared to the control group (Figure 1). In addition, severe damage was detected in the kidney tissues of GI group and a significant improvement was observed in the kidney tissues in the GI + CAPE group (Figure 2).
In light of these findings, we conclude that CAPE and IL are capable of decreasing the hepatotoxicity and nephrotoxicity caused by GI by means of their antioxidant effects. Therefore, it is likely that CAPE and IL can be used as a supportive therapy since they decrease organ damage and can also facilitate the therapeutic effects of the routine treatment in patients with GI intoxication.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by Mustafa Kemal University Scientific Research Project Coordination (ID no. 1206M0127).
