Abstract
The study aims at evaluating the protective effects of Ajwa dates extract against diclofenac sodium (DFS)-induced nephrotoxicity. A total of 30 male albino mice were divided into three groups. Group 1 received water per oral gavage for 14 days and saline per intra-peritoneal (IP) dose for the days 12–14. Group 2 received water like Group 1 and a daily IP dose of DFS for the days 12–14. Group 3 received 1 gm/kg Ajwa extract in water for 14 days and a daily IP dose for the days 12–14. Biochemical investigation revealed DFS-induced increase in serum urea and creatinine levels along with depleted antioxidant capacity, altered lipid profile, and hyperglycemia. Ajwa extract successfully protected against DFS-induced adverse effects, normalizing renal parameters biochemically and histologically, and prevented its hyperlipidemic and hyperglycemic effects. Ajwa protected against endogenous antioxidant capacity depletion in treated animals. Hydroalcoholic Ajwa extract is a promising candidate for protection against DFS-induced hyperlipidemia and nephrotoxicity.
Introduction
Natural products represent proper therapeutic solutions, as they are an inexpensive and easy source for a wide range of therapeutic applications. Some natural drugs, among them Ajwa, gained a significant value as dietary supplements and treatment of several types of diseases, as they are mentioned in Prophet Muhammad’s recitations (prophetic Hadith). Ajwa date fruits are commonly consumed in several parts of the world and are an inevitable part of the Arabian diet. 1
Ajwa is only cultivated in the Al-Madinah region of western Saudi Arabia and is known for its high nutritional properties. Ajwa date fruit is also rich in polyphenols, flavonoids, and iso-flavonoids which posses antioxidant and free radical–scavenging activities, and sterols and lignans which are important for lowering cholesterol level and thus reducing the risk of cardiovascular-related disorders and explaining their wide range of health benefits. 2
Date fruit extract has been reported to possess anti-inflammatory as well as an anti-cancer effects antibacterial activities against different bacterial strains in addition to hepatoprotective, neuroprotective, gastroprotective as well as nephroprotective effects. 2
Non-steroidal anti-inflammatory drugs (NSAIDs) like diclofenac sodium (DFS) represent a group of drugs that are utilized for several indications including pain analgesia as a main use. DFS is a benzene–acetic acid derivative that is completely absorbed after oral administration and eliminated as glucuronide and sulfate conjugates of the metabolites. 3
DFS has analgesic, anti-inflammatory, and antipyretic properties in addition to antibacterial effects. It is effectively indicated in ankylosing spondylitis, migraine, dysmenorrhea, osteoarthritis and mild to moderate pain, in addition to its off-labeled indication for juvenile idiopathic arthritis. In addition, it was also reported as a standard anti-inflammatory drug against several noxious stimulants like carrageenan-induced paw edema, hot plate, and acetic acid–induced writhing DFS effects are regarded mainly to its potent inhibitory effect on prostaglandins, which mediate the inflammatory responses among other functions via the competitive binding to cyclooxygenases (COX). 4
Despite these therapeutic actions of DFS, it has a list of reported adverse effects that include the increased risk of fatal or nonfatal stroke, coronary death, and nonfatal myocardial infarction. DFS was also linked to a significant increased risk of upper gastrointestinal complications in a dose-dependent manner, in addition to immune-mediated hepatotoxicity due to its metabolism and conjugation in the liver. DFS induces a state of oxidative stress within the kidney that elicits nephrotoxicity in response to its role in up-regulating proinflammatory cytokines’ expression. 3
Several studies aimed at investigating natural drugs ability to protect against DFS-induced toxicities. 5 Interestingly, until now, no studies have been done to investigate the possible effects of Ajwa fruit extract on DFS-induced nephrotoxicity. For this reason, we aimed in this research at investigating the protective and ameliorative effect of a hydroalcoholic extract of Ajwa on DFS-model of nephrotoxicity.
Materials and method
Chemicals
DFS ampoules for injection (75 mg/3 ml) were procured from Jamjoom Pharma, Jeddah, Saudi Arabia. All other chemicals were of analytical grade and purchased from local providers.
Extraction of Ajwa dates
Ajwa dates were purchased from local markets in Almadinah Almunawrah and dried after removal of pits. About 500 g of dried and ground date flesh was extracted by being macerated in 500 ml of hydroalcoholic solution for 1 week with frequent shaking and then the supernatant was filtered. The marc was repeatedly extracted two times in the same way, and the supernatants from the three extraction processes were pooled and dried until a semisolid residue is formed.
Antioxidant activity of Ajwa extract
DPPH (2,2-diphenyl-1-picrylhydrazyl) assay was used to assess the antioxidant capacity of hydroalcoholic Ajwa extract as mentioned earlier. 6 Briefly, serial dilutions of Ajwa extract were mixed with DPPH (8 mg/100 mL methanol) for 30 min then the decrease in the absorbance was assessed spectrophotometrically at 517 nm using ascorbic acid as a reference. Each measurement was repeated three times and the antioxidant activity was calculated using the following formula
% of inhibition = 100 × (1–(absorbance of extract/absorbance of ascorbic acid))
Determination of total phenolic and flavonoid contents of Ajwa extract
Total phenolic and total flavonoid contents of hydroalcoholic Ajwa extract were determined spectrophotometrically by Folin–Ciocalteu reagent method and aluminum chloride method, respectively, as previously described. Gallic acid and rutin were used to create calibration curves for total phenolics and flavoniods, respectively. 7 The content of phenols of flavonoids was calculated using the linear equations of the respective calibration curves. The total phenolic content was expressed as gallic acid equivalent (GAE) per gram of dry extract, and the total flavonoid compounds was expressed as rutin equivalent (RE) per gram of dry extract.
Experimental design and sample collection
A total of 30 adult male albino mice (weight range: ~30 g) were used for this study. Mice were randomly divided and kept in plastic cages and exposed to 12 h/12 h cycles of light and darkness. All animals had access to standard animal chow and water ad libitum and were maintained at a temperature of 23°C–25°C. Animals were allowed to accommodate for 1 week before starting the experimental procedures. Procedures involving animals and their care were conducted in accordance with international laws and policies. Animals were assigned to three groups as follow: Group 1: healthy control animals receiving water per oral gavage and the same volume of saline intra-peritoneal (IP) instead of DFS as used in Groups 2 and 3. Group 2: animals received water per oral gavage for 14 days and started to receive 50 mg/kg of DFS per IP from day 12 through day 14 along with water per oral gavage. Group 3: animals received 1 g/kg Ajwa extract per oral gavage for 14 days and started to receive 50 mg/kg of DFS (1.5 mg/30 g mouse) per IP from day 12 through day 14 along with Ajwa extract (30 mg/30 g mouse). Animals were sacrificed 24 h after the last DFS dose. Blood was collected by cardiac puncture in glass tubes and was allowed to clot at room temperature for 20–25 min before the serum was separated by centrifugation (3000 r/min, 20 min). Collected serum samples were kept at −20°C until further use. Kidneys were collected from animals of the different groups and either snap frozen or prepared for histological examination.
Assessment of biochemical parameters
Serum or tissue samples from the different groups were assessed for their content of glucose, creatinine, urea, triglycerides, total cholesterol, lipid peroxidation, total thiol, and catalase activity using commercially available kits according to the manufacturer’s instructions.
Histopathological analysis
Immediately after the mice were sacrificed, the kidneys were rinsed in normal saline then fixed in 10% formalin. After dehydration, the tissues were cleared in xylol and finally embedded in paraffin. Five-micrometer-thick sections were stained with hematoxylin/eosin to evaluate the morphological changes.
Statistical analysis
Values were represented as means ± standard error of means SEM (n = 10) using GraphPad® prism 6. Experimental results were statistically analyzed using the Student’s t-test for unpaired data, with a
Results
Free radical–scavenging activity of Ajwa extract
Assessment of the in vitro free radical–scavenging activity of natural products using DPPH is a widely used method. In this assay, the absorbance of DPPH negatively correlates with the antioxidant capacity of the tested material. The hydroalcoholic extract of Ajwa dates showed strong antioxidant capacity compared to ascorbic acid based on the calculated % of inhibition, which was calculated as 71% ± 4.2% at the concentration of 100 µg/mL compared to 88% ± 3.8% for ascorbic acid at the same concentration, and 82% ± 4.6% for Ajwa extract at 150 µg/mL compared to 92% ± 2.1% for ascorbic acid at the same concentration (values represent mean ± SD of triplicates).
Total phenolic and flavonoid content of Ajwa extract
Assessment of the total phenolic contents was performed using Folin–Ciocalteu method as GAE in mg/g of the extract and was found to be 311.5 GAE/g. It was calculated using the calibration curve equation Y = 0.0031X – 0.0027, where R2 = 0.998.
Total flavonoid content was assessed using aluminum chloride method as mentioned in the methods section and was expressed as REs in mg/g of the extract. Based on the standard curve equation Y = 0.0029X – 0.004, where R2 = 0.997. The flavonoid content of Ajwa extarct was calculated as 227.4 RE/g.
Effect on biochemical parameters
DFS administration induced a significant increase in blood glucose level when compared with saline-treated control mice. It also altered the lipid profile of test animals as observed in the form of a significantly increased serum triglyceride and total cholesterol levels when compared to control animals. Interestingly, a significant reduction in blood glucose level, as well as normalization of serum triglycerides and cholesterol, was observed in animals that were pre-treated with Ajwa extract (Table 1).
Effect of the different treatments on the listed biochemical parameters.

Nephrotoxicity is a possible side effect of DFS and is observed in the form of increased serum levels of urea and creatinine. In our results, using IP injections of DFS altered renal parameters as observed by a significant increase in serum urea as well as serum creatinine level when compared to healthy control animals (Table 1). Pretreatment with the Ajwa date extract resulted in a protective effect against DFS-induced nephrotoxicity as evidenced by the normalized serum urea and creatinine levels that were comparable to healthy controls.
Effect on catalase activity, total antioxidant capacity, and lipid peroxidation
Compared to control animals, DFS treatment resulted in a significant reduction in catalase activity as well as total antioxidant capacity, whereas a significant increase was observed in lipid peroxidation (Figure 1(a)–(c)). Interestingly, Ajwa treatment significantly restored catalase activity and the depleted antioxidant capacity induced by DFS. In addition, Ajwa treatment also induced a significant reduction in DFS-mediated increase in lipid peroxidation.
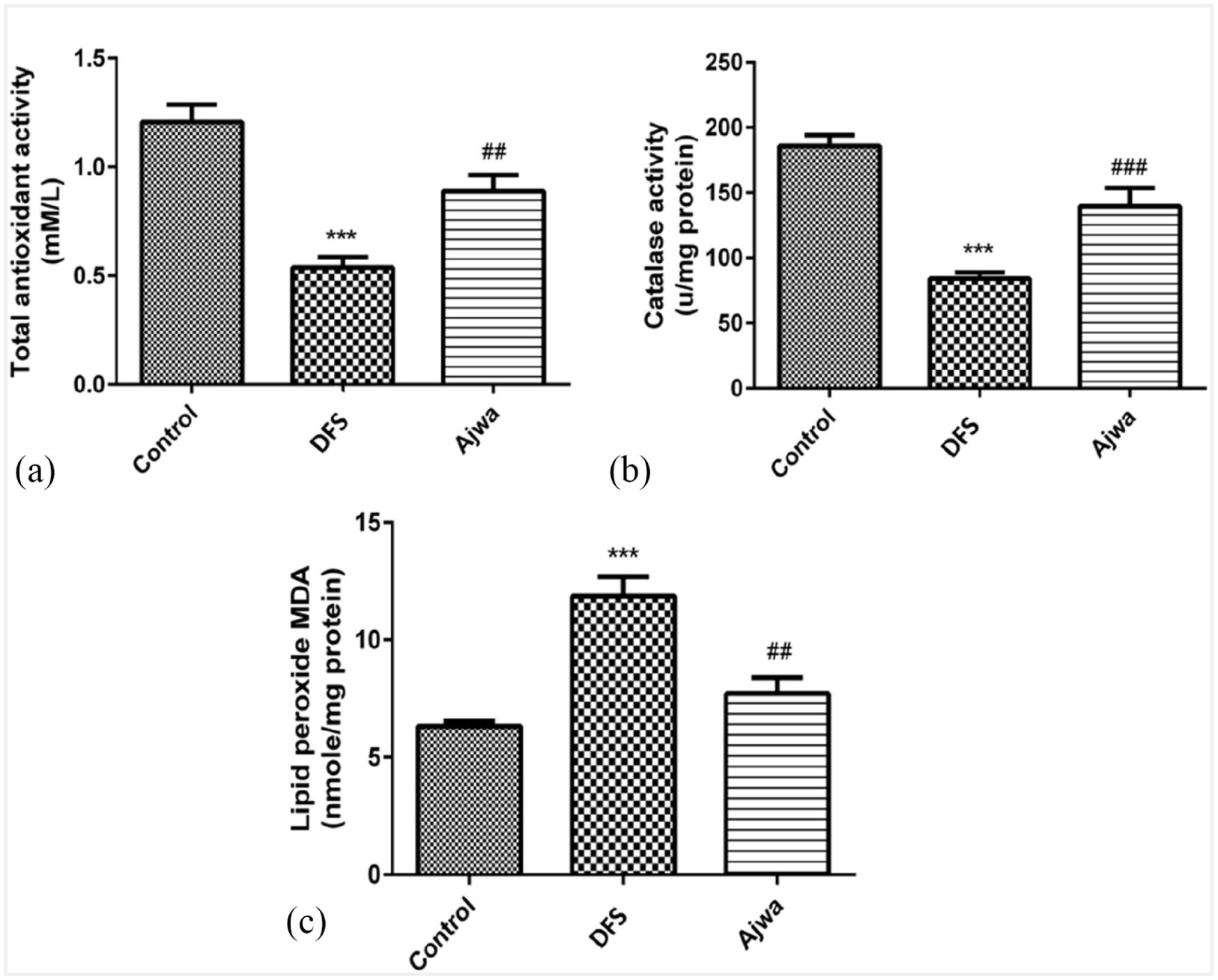
Effect of the different treatments on total antioxidant capacity (a), catalase activity (b), and lipid peroxidation (c).
Histopathology studies
In control animals, no pathological changes were detectable in the kidneys as seen in Figure 2(a). No hemorrhagic changes or necrosis were observed in the kidneys of all test groups; however, focal degenerative changes of the tubules presented in the form of cloudy swelling, congestion, inflammation as well as hypercellularity of glomerular tufts were detected in animals treated with DFS (Figure 2(b): arrows and arrowheads). Interestingly, treatment with Ajwa ameliorated these effects in the respective animals resulting in much less congestion, inflammation, and swelling as well (Figure 2(c) and (d)).

Histopathological examination of kidney tissues stained with hematoxylin/eosin: (a) control group shows no pathological changes; (b) DFS treated group where arrows in (b) shows congestion, and arrowheads show hypercellularity of glomerular tufts; (c) Ajwa /DFS treated group. The pretreatment with Ajwa protected against the effects of DFS on kidneys; and (d) scoring of the pathological changes in kidney tissue.
Discussion
DFS is an anti-inflammatory and analgesic that is mainly metabolized in the liver with about 65% of its metabolites excreted in urine and 35% in bile. Many reports recorded hepatotoxic and nephrotoxic effects as well as gastric ulceration and cardiovascular effects after DFS administration which can be attributed to the oxidative stress induced by DFS and its metabolites on the different tissues. 3 The oxidative stress–induced nephrotoxic effects of DFS impair glomerular filtration, resulting in abnormal renal parameters. 8
In this study, administration of DFS to adult male mice induced an increase in serum urea and creatinine levels, hyperglycemia, and alteration of the lipid profile compared to healthy control animals. Significant depletion of antioxidant capacity and catalase activity and induction of lipid peroxidation as well as histopathological changes were also reported in this study.
High doses of DFS induce hyperglycemia due to its cytotoxic effects on pancreas and insulin-producing cells, in addition to decreasing insulin sensitivity as reported earlier. 9 It is also lipolytic, as it modulates the production of prostaglandins, which regulate lipolysis; resulting in an increase in total cholesterol, triglycerides as well as free fatty acids. 10
Ajwa fruit was reported to possess protective properties in different pathological conditions, depending on its content of antioxidants (phenols, flavonoids, melatonin, carotenoids, and vitamins A, E, and C) and free radical–scavenging enzymes. 2 Several researchers reported positive effects of Ajwa date flesh extracts on nephrotoxicity as it normalized renal parameters and ameliorated the deterioration of the proximal tubular regions; which agrees with our findings that showed improvemet in the endogenous antioxidant capacity and preserved renal tissue against the detrimental effects of DFS. 2 After improving the level of antioxidants, Ajwa date extracts reduced lipid peroxidation in arthritic rats as well as protected against lead acetate–induced toxicity in rabbits as shown by others. 2
Hyperlipidemia together with hyperglycemia represent significant cardiovascular risk factors that contribute to the development of cardiovascular disorders in DFS-treated individuals. In our study, Ajwa ameliorated the hyperglycemic and hyperlipidemic effect of DFS, similar to previous reports. This can be regarded to its dietary fiber and phytochemical content, which modifies lipid and cholesterol metabolism and interferes with lipase activity.2,11 Studies have shown antihyperlipidemic and hence cardioprotective effects for B-sitosterol, anthocyanins, saponins, and selenium are found in Ajwa. 11 In addition, flavonoids and phenolic compounds protect the pancreatic functions, promoting the antidiabetic effect of Ajwa extract. 2 An earlier study on diabetic patients showed that Ajwa might help in controlling blood glucose and lipid profile due to its low glycemic index which is associated with its high fructose/glucose content. 12
In conclusion, this study represents the hydroalcoholic Ajwa extract as a protective drug against DFS-induced renal damage and cardiovascular risk factors, suggesting the routine consumption of Ajwa dates by patients receiving DFS for long periods to protect them against the possible DFS-induced damages.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
