Abstract
Keywords
Introduction
Coronavirus disease 2019 (COVID-19) is a highly infective disease caused by a novel coronavirus which was named severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Lungs and airways are believed to be the main targeting organs of SARS-CoV-2. However, accumulating evidence supported that SARS-CoV-2 infection not only resulted in damage to lungs but also other critical organs, which significantly increased their mortality.1,2
Association of SARS-CoV-2 infection and kidney injury has been noted in previous studies.1,3,4 Kidney injury was reported to be common in COVID-19, especially in critically ill patients.4,5 Acute kidney injury (AKI), an adverse clinical predictor, was considered to be associated with increased mortality of COVID-19.1,6 Several pathogenic mechanisms including direct virulence of SARS-CoV-2, cytokine storms, microcirculatory dysfunction, and hypoxia have been proposed to explain the etiology of kidney injury in COVID-19 patients.7,8 Cytokine storms, resulting from SARS-CoV-2 infection, are believed to be a key event in the development of kidney injury in COVID-19. However, the speculations are mainly based on observations of severe sepsis and other coronavirus infection. To date, limited study evaluated a relationship between dysregulation of cytokines and kidney injury in COVID-19.
The primary goal of the present retrospective study was to evaluate whether dysregulation of cytokines was involved in the etiology of kidney injury in COVID-19 patients.
Methods
Study design and participants
This was a retrospective observational study carried out from 28 January 2020 to 30 March 2020 in Optics Valley Branch of Tongji Hospital. All consecutive patients who tested positive by polymerase chain reaction testing of a nasopharyngeal/oropharyngeal sample for SARS-CoV-2 were enrolled in the study. These patients were treated by Fujian Medical Team aiding Hubei province. Exclusion criteria were as follows: patients younger than 18; patients with incomplete laboratory data of kidney function; patients with a known history of chronic kidney disease; patients with autoimmune diseases or were on drugs affecting serum cytokines. This study was approved by the Ethics Committee of Zhongshan Hospital, Xiamen University (approval no. 2020–012). Written informed consent was obtained from all subjects. All methods are carried out in accordance with relevant guidelines and regulations.
Data collection
We extracted the demographic data, days from illness onset to admission, laboratory data, clinical symptoms, and signs from electronic medical records. The laboratory data consisted of renal function tests, complete blood count, hemostasis parameters, inflammatory markers, and serum cytokines. All data were reviewed by a team of trained physicians.
Definition
Based on the guidelines for diagnosis and management of COVID-19 (seventh edition, in Chinese), 9 COVID-19 patients were divided into four categories: mild, moderate, severe, and critical cases. Mild cases: the clinical symptoms are mild and no pneumonia manifestation can be found in imaging. Moderate cases: patients have symptoms such as fever and respiratory tract symptoms, etc., and pneumonia manifestation can be seen in imaging. Severe cases: adults who meet any of the following criteria: respiratory rate ≥ 30 breaths/min; SpO2 ≤ 93% at rest; PaO2/FiO2 ≤300. Patients with greater than 50% lesion progression within 24–48 h in pulmonary imaging were also defined as severe cases. Critical cases: patients who meet any of the following criteria: occurrence of respiratory failure requiring mechanical ventilation; presence of shock; other organ failure that requires monitoring and treatment in the ICU. Since only four patients met the criteria of critical cases, severe and critical cases were combined into one group. Therefore, there were three groups finally: mild, moderate, and severe to critical group.
Calculation of glomerular filtration rate
Estimated glomerular filtration rate (eGFR) was calculated using the CKD-EPI equation: eGFR = 141×(minimum of standardized serum creatinine [mg/dL]/κ or 1) α ×(maximum of standardized serum creatinine [mg/dL]/κ or 1)−1.209 × 0.993age×(1.018 if female) × (1.159 if black), where κ is 0.7 if female and 0.9 if male and α is −0.329 if female and −0.411 if male. 10
Cytokine measurement
Blood samples were collected from the patients on admission or the second day after admission. Chemiluminescent immunoassay by Siemens Immulite 1000 analyzer was used to measure serum cytokines including interleukin (IL)-1β, IL-2R, IL-6, IL-8, IL-10, and tumor necrosis factor (TNF)-α according to the manufacturer’s instructions.
Statistical analysis
All continuous variables were presented as mean ± standard deviation (SD) or median (percentile 25, percentile 75) depending on whether they were normally distributed or not. Categorical variables were presented as number (percentage). Normality was checked with a QQ-plot and Shapiro-Wilk normality test. 11 One-way ANOVA was used to compare the baseline data between groups stratified by COVID-19 severity when the data were normally distributed; otherwise, the Kruskal–Wallis H (K) test was used. 11 Categorical variables were compared by the Chi-square test or Fisher’s exact test. 11 Descriptive data not in normal distribution were log-transformed before analysis. Correlation analyses were evaluated by the spearman rank correlation test. Multiple linear regression was performed to determine the independent predictors of eGFR and cystatin C. p < .05 was considered statistically significant. Statistical analyses were performed using SPSS 22.0 (SPSS Inc, Chicago, IL). GraphPad Prism 8 (GraphPad Software, USA) was used to draw the graphs.
Results
Baseline characteristic of COVID-19 patients according to disease severity
The baseline demographic and clinical data in different groups of COVID-19.
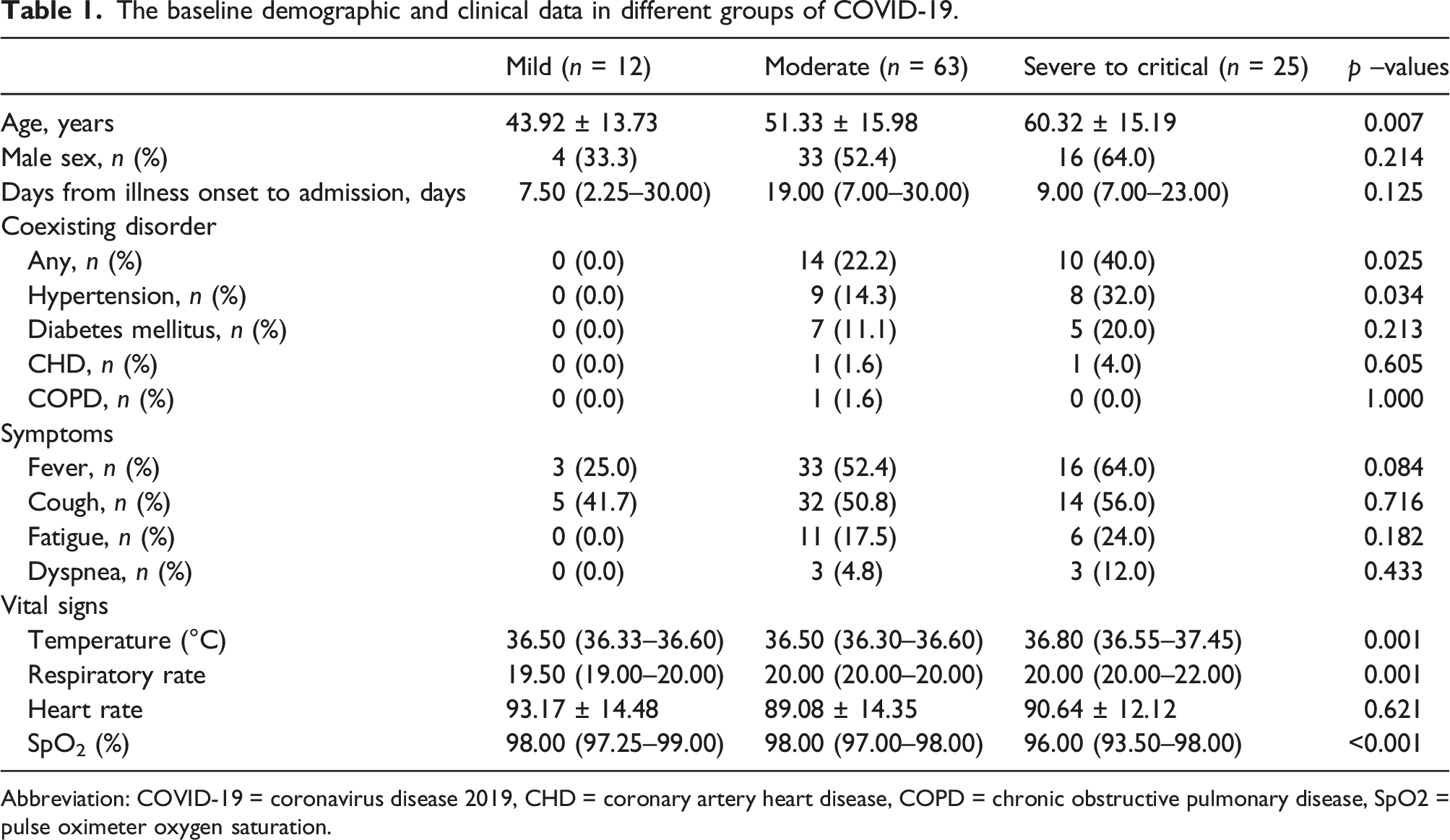
Abbreviation: COVID-19 = coronavirus disease 2019, CHD = coronary artery heart disease, COPD = chronic obstructive pulmonary disease, SpO2 = pulse oximeter oxygen saturation.
Laboratory results of COVID-19 patients according to disease severity
The laboratory data in different groups of COVID-19.

Abbreviation: IL = interleukin, TNF = tumor necrosis factor, Hs-CRP = high-sensitivity C-reactive protein, PCT = procalcitonin, COVID-19 = coronavirus disease 2019, PLT = platelet, CK = creatine kinase, LDH = lactic dehydrogenase, APTT = activated partial thromboplastin time, PT = prothrombin time, FIB = fibrinogen, IL = interleukin, TNF = tumor necrosis factor.

Comparisons of kidney injury markers among different groups stratified by COVID-19 severity (a). blood urea nitrogen; (b). serum creatinine; (c). eGFR; (d). cystatin C).
Factors associated with kidney injury in COVID-19
Spearman rank correlation coefficients between kidney injury and demographic data, laboratory data, and cytokines.

Abbreviation: IL = interleukin, TNF = tumor necrosis factor, Hs-CRP = high-sensitivity C-reactive protein, PCT = procalcitonin, eGFR = estimated glomerular filtration rate.

Correlations between cytokines and eGFR (a). IL-1β; (b). IL-2 R; (c). IL-6; (d). IL-8; (e). IL-10; (f). TNF-α).

Correlations between cytokines and cystatin C (a). IL-1β; (b). IL-2 R; (c). IL-6; (d). IL-8; (e). IL-10; (f). TNF-α).
Role of cytokines in kidney injury in COVID-19
Stepwise multiple linear regression was used to determine the independent predictors of kidney injury. The factors significantly associated with kidney injury were included as independent variables. The results showed that age (β = −0.565, p < .001), log TNF-α (β = −0.230, p = .002), log IL-6 (β = −0.212, p = .007) were independently related to elevated eGFR. Age is one of the factors used to calculate eGRF. When age was excluded from independent variable, the results indicated that any coexisting disorder (β = −0.188, p = .041), log TNF-α (β = −0.316, p = .001), log IL-6 (β = −0.331, p = .001) were independently related to elevated eGFR. Using cystatin C as dependent variable, multiple linear regression showed that log TNF-α (β = 0.534, p < .001) and log IL-6 (β = 0.405, p < .001) were independent predictors of elevated cystatin C.
Discussion
Our results showed that kidney injury was significantly correlated with the severity of COVID-19. Cytokine IL-6 and TNF-α were independent predictors of elevated eGFR and cystatin C in COVID-19 patients. Our findings provided a direct evidence to support an important role of cytokines in kidney injury in COVID-19.
Kidney injury in COVID-19 patients has been reported in several previous studies.1,3,4 It is estimated that AKI occurs in 3%–29% of COVID-19 patients. 12 A study analyzing 701 COVID-19 patients found that the prevalence of proteinuria and hematuria were 43.9% and 26.7%, respectively. 14.4%, 13.1% and 13.1% of COVID-19 patients had elevated serum creatinine, elevated blood urea nitrogen and eGFR under 60 mL/min/1.73 m2, respectively. 5.1% patients suffered from AKI during follow-up in this study. 1 Another study focusing on critically ill adult patients showed that 29% of the 52 patients had AKI. 3 A postmortem analysis of 26 COVID-19 patients showed evidence of acute proximal tubule injury manifested as the loss of brush border, vacuolar degeneration, dilatation of the tubular lumen with cellular debris. 8 COVID-19 is a systemic illness affecting multiple organs. The above evidence indicated that kidney injury should not be neglected when treating this disease.
In the present study, the disease severity was significantly associated with kidney injury. Our findings indicated that it is necessary to have a close monitoring and evaluation of kidney injury in severe to critical COVID-19 patients. Based on the results of our study, we could also conclude that SARS-CoV-2 infection was a risk factor of kidney injury. The underlying mechanisms are multifactorial. Cytokine storms are believed to be one of the most important mechanisms explaining kidney injury in CVOVID-19 patients. 13 Previous evidence have shown a role of cytokines in the development of AKI after cardiac surgery or during sepsis.14,15 A clinical study focusing on patients with severe sepsis showed that increased log IL-6 and APACHE II score were significant risk factors of AKI in patients with severe sepsis. 14 Zhang et al. 15 included 960 patients from six academic centers and revealed that elevated first postoperative IL-6 and IL-10 concentration were significantly associated with higher risk of AKI in patients after cardiac surgery. Furthermore, Previous studies have evaluated the relationship between cytokines including IL-8, TNF-α, IL-2R, IL-10, IL-6 and COVID-19 disease severity and confirmed a strong relationship.16,17 However, limited study focused on association between kidney injury and cytokine storms in COVID-19.
This study sought to identify factors associated with kidney injury in patients with COVID-19. Our study provided a direct evidence of cytokines involvement in the kidney injury in COVID-19. A better understanding of the pathogenesis of kidney injury in COVID-19 will help to promote novel therapies for this condition. It has been reported that tocilizumab therapy, an IL-6 inhibitor, can effectively decrease inflammatory markers, improve radiological imaging, and reduce the demands of ventilatory support. 18 Another study also showed the benefits of tocilizumab treatment in improving symptoms and imaging findings in COVID-19. 19 Our results suggested that log IL-6 was a major independent predictor of kidney injury. Therefore, it will be interesting to explore whether tocilizumab treatment can alleviate the kidney injury in COVID-19 in the future. In addition, TNF-α was also found to be an independent predictor of kidney injury in our study, which highlighted the important role of TNF-α in kidney injury in COVID-19. Feldmann et al. 20 proposed that anti-TNF therapy should be evaluated in patients with COVID-19 on hospital admission to prevent progression after summarizing the current evidence. Our results supported this proposal by providing the direct evidence.
The CKD-EPI equation was reported to be as accurate as MDRD in the patients with eGFR < 60 mL/min/1.73 m2 and substantially more accurate in the patients with eGFR > 60 mL/min/1.73 m2. 21 So it is a reliable marker assessing kidney function. Cystatin C was considered as a reliable marker of kidney function and could be useful for early diagnosis of kidney injury. 22 The result of multivariate analysis of eGFR was similar to that of cystatin C, which confirmed the reliability of these two cytokines in predicting kidney injury.
There were some limitations associated with this study that must be noted. Firstly, the retrospective design allowed us to support a correlation but not a causative relationship. It was also associated with a higher susceptibility to errors caused by bias and confounding. Secondly, no calculation of sample size was carried out prior to this study, and the sample size of the present study might be relatively small, raising the possibility of a type 2 error. A study with a larger sample size could result in more robust conclusions. Thirdly, other data affecting kidney injury such as risky occupation for kidney injury, body weight, and nutritional status were not collected. Finally, the prevalence of AKI could not be evaluated due to lack of longitudinal data of serum creatinine.
Conclusion
Taking together, the present study suggested that cytokines IL-6 and TNF-α were independently related to kidney injury in COVID-19 patients, which suggested that elevated serum cytokines might be involved in the development and progression of this disorder in COVID-19. This result provided a therapeutic target for the treatment of COVID-19 related kidney injury. Animal experiments and clinical studies with a larger sample size are warranted for a better understanding of the mechanisms.
Footnotes
Acknowledgements
We sincerely appreciate all front-line medical staff for their hard work and sacrifice.
Authors’ contributions
LDC, LH, and XBZ conceived and designed the study, analyzed the data and drafted this manuscript. YS, YPH, SJY, and JY contributed to analysis of the data, and revising of the manuscript. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China [82170103], Young people training project from Fujian Province Health Bureau [2020GGB057] and Xiamen Medical and Health Guidance Project [3502Z20214ZD1043].
Ethics approval
This study was approved by the Ethics Committee of Zhongshan Hospital, Xiamen University (approval no. 2020–012).
Written informed consent
Written informed consent was obtained from all subjects.
Availability of data and material
The data that support the findings of this study are available from the corresponding author upon reasonable request.
