Abstract
Keywords
Introduction
Sepsis is the main matter of death in intensive care units because it subsequently leads to life-threatening organ impairment. 1 The liver is an important visceral organ involved in host defense against infections as well as in other vital functions. Importantly, liver injury and liver dysfunction are common clinical manifestations that eventually lead to poor outcomes in sepsis,2–4 and septic liver injury is deemed to be an independent factor of mortality. 5 Therefore, prevention of septic liver injury is essential for the control of this condition. 6
Immune-responsive gene 1 (Irg1), also referred to as aconitate decarboxylase 1, is an inflammation-induced mitochondrial enzyme that decarboxylates cis-aconitate, a TCA cycle intermediate, to produce the bio-active metabolite itaconate.7,8 Previous findings have found that Irg1/itaconate plays an essential role in oxidative stress,9,10 cellular metabolism as well as immune and inflammatory responses.11–13 Interestingly, the development of septic liver injury is closely associated with inflammation and oxidative stress.3,6 However, it is still unclear whether Irg1/itaconate also regulates the progression of septic liver injury.
The transcription factor nuclear factor erythroid 2-related factor 2 (Nrf2) acts as a pivotal function in defense response against oxidative stress 14 via motivating the transcription of antioxidative enzymes, such as heme oxygenase 1 (HO-1) and NADPH quinone oxidoreductase 1 (NQO1).5,14 Enormous references indicated antioxidative enzymes was decreased in Nrf2 knockout mice or siNrf2-treated cells.15,16 Moreover, the antioxidant enzymes have a shielded influence on hepatocytes from oxidative stress damage in the liver.11,17 Furthermore, the Nrf2 pathway has been broadly reported as an effective way to alleviate hepatic disease. 11 Previous studies have found that Nrf2 is one of major targets that responsible for the major bioactivities of Irg1/itaconate. 8 Then, we speculated that Irg1/itaconate might regard as a tutelar factor in septic liver injury.
In the study, the level of Irg1 was detected in mice with systemic exposure to lipopolysaccharide (LPS) in liver, the main toxic component of Gram-negative bacteria. 18 To explore the latent significance of Irg1, Irg1 knockout (KO) mice and their wild-type (WT) littermates were exposed to LPS and the grade of septic liver injury was assessed. To detect the therapeutic significance, a cell-permeable itaconate derivative called 4-octyl itaconate (4-OI),19–21 was administered and its potential effects on liver injury were tested.
Materials and methods
Study animals
Male C57BL/6J mice (8-week old; 18-22 g) were bought from the Experimental Animal Center of Chongqing Medical University, while Irg1 knockout (Irg1-/-) ones were obtained from Cyagen (Guangzhou, China). The mice were then given a traditional laboratory diet and free access to drinking water within a regulated environment (20-25°C, 50% ± 5% re-humidity, 12 h dark/light cycle). Furthermore, the animals were permitted to adapt to laboratory conditions for a minimum of 1 week prior to the experiments. All laboratory procedures concerning animals were ratified by the Ethics Committee of Chongqing Medical University (No. LL-202239).
Reagents
LPS (E. coli, 055: B5) was bought from Sigma (St Louis, MO, USA), while 4-OI (C13H22O4,242.3, Purity≥98%) was bought from Cayman Chemical (USA). Evans Blue was bought from Solarbio (Beijing, China). Biochemical assay kits for the detection of aspartate aminotransferase (AST, cat. No. C010-2-1), alanine aminotransferase (ALT, cat. No. C009-2-1), malondialdehyde (MDA, cat. No. A003-1-2) and superoxide dismutase (SOD, cat. No. A001-1-2) were bought from Nanjing Jincheng Bioengineering Institute (Nanjing, China), with enzyme-linked immunosorbent assay (ELISA) kits for tumor necrosis factor alpha (TNF-α, cat. No. EMC102a) and interleukin-6 (IL-6, cat. No. EMC004) obtained from Neobioscience (Shenzhen, China). Regarding antibodies, the rabbit anti-mouse IRG1 antibody (cat. no. 19857,1:1000), rabbit anti-mouse nuclear erythroid 2-related factor 2 (Nrf2) antibody (Cat No. 16396-1-AP, 1:1000), rabbit anti-mouse heme oxygenase 1 (HO-1) antibody (Cat No. ab189491, 1:2000) and rabbit anti-mouse β-actin antibody (Cat No. bs0061R, 1:5000) were acquired from Cell Signaling Technology (USA), Proteintech (Wuhan, China), Abcam (Cambridge, UK) and Bioss (Beijing, China), respectively. These rabbit anti-mouse antibodies, which are extracted from rabbits and are kinds of antibodies reacted with mice, are the primary antibodies. And the goat anti-rabbit IgG antibody (Cat No. bs-80295G-HRP, 1:5000) which is our secondary antibody were bought from Bioss (Beijing, China).
Experimental protocol
LPS (15 mg/kg, melted in saline) was injected intraperitoneally into the animals to create a model of septic liver injury. 22 The same volume of saline was injected intraperitoneally in control group. The total number of mice used in the study was 48. To explore the pathological outcome of Irg1, septic liver injury was also modeled in both Irg1-/- mice and wildtype (WT) littermate controls. The four groups of mice were separated (n = 4): WT control group, WT + LPS group, Irg1 -/- control group, Irg1 -/- +LPS group, with serum and liver samples subsequently harvested (18 h post LPS exposure) for further experiments. To investigate the exudation of Evans blue in the liver, 16 mice were randomized into four groups in WT mice (n = 4): control group, 4-OI group, LPS group, LPS+4-OI group. Because Evans blue is a kind of dye, it can affect other experiments. Hence, we added 16 mice treated as above described to explore the pharmacological significance, cell-permeable itaconate derivate 4-OI was administered intraperitoneally (50 mg/kg, melted in DMSO) before harvesting serum and liver samples 18 h after LPS exposure. 23 Mice were anesthetized intraperitoneally and sacrificed with 1% pentobarbital sodium at a dose of 0.01 mL/g.
Histopathology
Paraffinized sections of liver tissues were processed prior to hematoxylin and eosin (H&E) staining. The degree of liver injury was then assessed by scoring the H&E-stained sections under 200x magnification as reported previously: 11 level 0: no sign of injury; level 1: slight damage by cytoplasmic vacuole formation; level 2: middle to heavy damage with broad nuclear consolidation and absence of intercellular boundaries; level 3: heavy necrosis with bleeding and neutrophil infiltration.
Enzyme-linked immunosorbent assay
Liver tissues were homogenized and centrifuged at 4°C (12000×g, 10 min). For the resulting supernatant, the levels of IL-6 and TNF-α were measured using enzyme-linked immunosorbent assay (ELISA) as described by the manufacturer (Neobioscience).
Evans blue staining
After injecting 2% of Evans Blue solution through the tail vein, the animals were spread transcardially and bloodless by open-heart surgery with PBS (5 U/ml of heparin) 1 h post injection. Livers were subsequently homogenized in N.N-dimethylformamide (1 mL/100 g tissue), incubated at 60 C for 48 h and centrifuged at 5000×g for 15 min. The absorbance of the supernatant was then examined at 620 nm using a microplate reader, with the concentration of the Evans blue dye eventually determined based on a standard curve.
Biochemical analysis
To determine the grade of liver injury, the concentrations of ALT and AST in serum were measured using commercially available detecting kits as required by the manufacturer (Nanjing Jincheng Bioengineering Institute).
Oxidative stress indices assay
Liver tissues were homogenized and centrifuged at 4°C (12000×g, 10 min). To estimate antioxidant status, the levels of MDA and the activity of SOD were assessed using the kits according to the instruction of manufacturer in the supernatants of liver tissues.
Real-time quantitative polymerase chain reaction
Primer sequences used for real-time qPCR.

Irg1, Immune-responsive gene 1; HO-1, heme oxygenase 1.
Western blotting
Liver tissues were lysed in RIPA buffer to obtain proteins which were subsequently quantified using BCA kits. The total extracted proteins were then detached from SDS-PAGE and shifted to PVDF membranes. After closing the membranes with skim milk, overnight incubation was carried out at 4°C in the existence of the primary antibodies. Subsequently, the appropriate secondary antibody was incubated for 1h at room temperature. Finally, specific bands were visualized on the FUSION SOLOS Imaging System (VILBER BIO IMAGING, FRANCE) using enhanced chemiluminescence (ECL) method.
Statistical analysis
GraphPad Prism eight was utilized to examine the data which were then represented as the mean ± standard deviation (SD). One-way ANOVA with Tukey’s multiple comparisons test was subsequently applied for multi-group comparisons, 25 while t-test allowed comparisons between two groups. Results were defined as statistically significant at p < .05.
Results
LPS exposure induced the upregulation of Irg1 in the liver
It was previously discovered that, in experimental animal models with liver ischemia-reperfusion, colitis, acute lung injury and acute kidney injury, there was an increase in the expression of Irg1.19,23,26,27 Similarly, current research found that the mRNA level of Irg1 in the liver was notably higher in mice exposed to LPS compared to controls (Figure 1(a)). These results were also reflected in the western blot analysis where an elevation in the level of hepatic IRG1 was noted in LPS-challenged mice (Figure 1(b) and (c)). Lipopolysaccharide (LPS) exposure induced the upregulation of Irg1 in the liver. Septic liver injury in wild type (WT) and Irg1 knockout (Irg1-/-) mice was triggered by intraperitoneal injection of LPS. (a) mRNA levels of Irg1 in liver tissues were determined by RT-qPCR. (b, c) Levels of the IRG1 protein in liver tissues was also tested by Western blot (n = 4; mean ± SD; **p < .01; one-way ANOVA; ***p < .001; ****p < .0001).
Irg1 deficiency aggravated septic liver injury
To research the pathological implications of Irg1’s upregulation, Irg1 knockout mice (Irg1-/-) were generated and the absence of expression in the liver was verified by PCR and western blotting (Figure 1). In contrast to the wild-type (WT) mice, LPS induced higher levels of serum ALT and AST in Irg1-/- mice (Figure 2(a) and (b)). Similarly, histological analyses indicated that LPS-induced hepatic abnormalities, such as necrotic area (red arrows), in Irg1-/- mice were more severe than those in WT mice (Figure 2(c) and (d)). Irg1 deficiency aggravated septic liver injury which were triggered in wild type (WT) and Irg1 knockout (Irg1-/-) mice through intraperitoneal injection of lipopolysaccharide (LPS). (a, b) The serum levels of ALT and AST were detected by biochemical analysis. (c, d) Liver slices were stained with H&E (100× and 200× magnification) and the grade of liver damage was scored (red arrows represented necrotic areas) (n = 4; mean ± SD; t test; *p < .05; ****p < .0001).
Irg1 deficiency exacerbated inflammation and oxidative stress
Additionally, the levels of hepatic TNF-α and IL-6 as well as the degree of Evans blue exudation in liver tissues increased in LPS-treated Irg1-/- mice as opposed to the WT ones (Figure 3(a) to (c)). Meanwhile, the MDA production in the liver was dramatically accelerated in the LPS-exposed Irg1-/- mice compared to WT mice (Figure 3(d)). And the activity of antioxidant enzyme SOD was significantly reduced in LPS-challenged Irg1-/- mice (Figure 3(e)). Thus, deletion of Irg1 enhanced hepatic inflammatory responses and oxidative stress. Irg1 deficiency exacerbated inflammation. (a, b, c) The hepatic levels of TNF-α, IL-6 as well as the leakage of Evans blue in liver tissues were measured by ELISA. (d, e) The MDA content and SOD activity were detected by biochemical analysis. (n = 4; mean ± SD; t test; *p < .05; **p < .01; ***p < .001).
Irg1 modulated Nrf2 activation
The Nrf2/HO-1 antioxidant pathway is thought to be a central target related to the bioactivities of Irg1.
28
The results of this study proved that hepatic levels of HO-1 mRNA were notably lower in LPS-treated Irg1-/- mice contrasted with WT ones (Figure 4(a)). Additionally, the western blot experiments indicated that Nrf2 and HO-1 proteins in the liver of LPS-exposed Irg1-/- mice were less abundant than in WT mice exposed to the LPS (Figure 4(b), (c), (d)). Irg1 modulated Nrf2 activation. (a-d) Septic liver injury in wild type (WT) and Irg1 knockout (Irg1-/-) mice were triggered by intraperitoneal injection of lipopolysaccharide (LPS). mRNA levels of HO-1 (a) and levels of Nrf2 and HO-1 proteins (b to d) in hepatic tissues were determined by RT-qPCR and Western blot (n = 4; mean ± SD; t test; *p < .05; **p < .01).
Itaconate modulated Nrf2 activation
Itaconate is the enzymatic product of Irg1 which motivates the Nrf2/HO-1 pathway.19,29 To investigate the modulation of Irg1/itaconate on the hepatic Nrf2/HO-1 pathway, the cell-permeable derivate of itaconate, 4-OI, was administered and the results revealed that 4-OI raised the mRNA levels of HO-1 (Figure 5(a)). Furthermore, western blotting also showed that the amount Nrf2 and HO-1 proteins increased after 4-OI treatment (Figure 5(b) to (d)). Therefore, Irg1/itaconate activated the hepatic Nrf2/HO-1 pathway in mice with septic liver injury. Itaconate modulated Nrf2 activation. (a-d) Septic liver injury was triggered by intraperitoneal injection of lipopolysaccharide (LPS) and 4-octyl itaconate (4-OI) was then administered. mRNA levels of HO-1 (a) and levels of Nrf2 and HO-1 proteins (b to d) in hepatic tissues were determined by RT-qPCR and Western blot (n = 4; mean ± SD; one-way ANOVA; *p < .05; **p < .01; ***p < .001).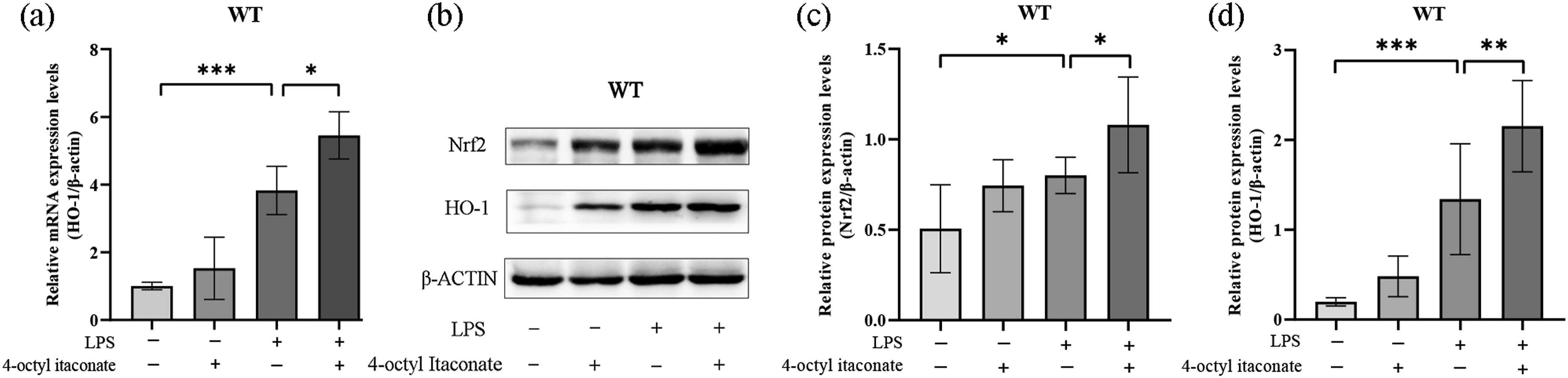
Supplementation with 4-OI alleviated septic liver injury
The current research also investigated the pharmacological significance of administering 4-OI on septic liver injury. In LPS-exposed mice which were given 4-OI, the concentrations of ALT and AST were lower contrasted with the LPS group (Figure 6(a) and (b)). HE staining also showed that providing 4-OI mitigated histological abnormalities, such as hemorrhage (red arrows) and inflammatory cell infiltration (yellow arrows) (Figure 6(c) and (d)). Recruitment with 4-OI relieved septic liver injury. Septic liver injury was triggered by intraperitoneal injection of lipopolysaccharide (LPS) and 4-OI was then administered. (a, b) The serum contents of ALT and AST were tested by biochemical analysis. (c, d) Liver slices were stained with H&E (100× and 200× magnification) and the grade of liver damage was scored (red arrows represented hemorrhage; yellow arrows represented inflammatory cell infiltration) (n = 4; mean ± SD; one-way ANOVA; *p < .05; **p < .01; ***p < .001; ****p < .0001).
Supplementation with 4-OI reduced inflammation and oxidative stress
Treatment with 4-OI also lessened the amount of TNF-α and IL-6 in septic liver, while suppressing the exudation of Evans blue in the liver (Figure 7(a) to (c)). Furthermore, we observed that pretreatment with 4-OI in WT significantly declined the amount of MDA and increased the activity of SOD compared to the LPS treated mice (Figure 7(d) and (e)). Thus, it seems that 4-OI could have prospective value as drug therapy against septic liver injury. Supplementation with 4-OI reduced inflammation. (a, b, c) The hepatic levels of TNF-α and IL-6 as well as the leakage of Evans blue in the liver were determined by ELISA. (d, e) The MDA content and SOD activity were assessed by biochemical analysis. (n = 4; mean ± SD; one-way ANOVA; *p < .05; **p < .01; ***p < .001; ****p < .0001).
Discussion
The present study revealed that systemic LPS exposure upregulated hepatic Irg1, while deletion of Irg1 exacerbated septic liver injury. As a result, serum contents of ALT and AST are significantly enhanced in the liver since they are important biochemical hallmarks for assessing liver function. 30 Moreover, the histological abnormalities in septic liver injury, such as necrotic area, hemorrhage and inflammatory cell infiltration, are more serious. Therefore, an upregulation of hepatic Irg1 might act as a phylactic part to limit the progression of septic liver injury. Additionally, increasing evidence suggests that some metabolic enzymes/metabolites take an active part in the management of signal transduction as well as in the progress of various disorders.31,32
In this context, LPS stimulation binds Toll-like receptor 4 (TLR4) to secrete proinflammatory cytokines. 1 TNF-α represents an important cytokine that is released during tissue damage and is involved in inflammatory responses. 33 Indeed, it triggers an inflammatory cascade to induce other proinflammatory cytokines and adhesion molecules, such as IL-6. 23 An uncontrolled inflammatory response may lead to the development of septic liver injury. 34 Current research showed that deletion of Irg1 resulted in higher concentrations of pro-inflammatory TNF-α and IL-6 in the liver, hence suggesting that the gene might serve as a passive modulator of hepatic inflammation in sepsis. Therefore, inhibition of TNF-α and other proinflammatory cytokines could be essential to attenuate septic liver injury. In fact, this is not surprising as the anti-inflammatory benefits of Irg1 have already been reported both in vivo and in vitro.9,11,35,36
In addition to inflammation, the development of liver injury is closely related to oxidative stress. 11 Evidence has proved that LPS exposure led to the alteration of antioxidant enzyme levels and excessive production of free radicals.23,31 The results of our study indicated absence of Irg1 resulted in higher MDA production and lower SOD activity in the liver. Therefore, control of hepatic inflammation and oxidative stress could be among the hepatoprotective activities of Irg1.
Nrf2 is one of the major targets through which Irg1 exerts its bioactivity as itaconate alkylates cysteine residues of Keap1, a cytoplasmic protein that can degrade Nrf2 by ubiquitination.19,29 More specifically, the alkylation of KEAP1 cysteine residues allows the separation of the keap1-NRF2 complex, resulting in the accumulation of the Nrf2 protein and its subsequent translocation to the nucleus.26,37 NRF2 then binds to antioxidant response elements (ARE) within the nucleus to exert protective effects and promote the transcription of its downstream antioxidant factors such as HO-1.38,39 Consistently, this study linked more severe liver injury with compromised activation of Nrf2 pathway, with previous findings also establishing Nrf2 as a protective factor in septic liver injury, while oxidative stress is linked to the development of septic liver injury.39–41 Thus, Nrf2 could be a key target leading to the beneficial effects of Irg1 in septic liver injury.
In addition to the suppression of oxidative stress-induced hepatocyte impairment, Nrf2 can also participate in the modulation of inflammatory responses.20,42,43 Indeed, previous results have shown that Nrf2 dampens inflammation by preventing the transcription of pro-inflammatory cytokine, activating the production of the anti-inflammatory cytokine as well as inhibiting the excitation of nucleotide-binding oligomerization domain-like receptor family pyrin domain-containing 3 (NLRP3) inflammasome.42–45 Therefore, modulation of redox-sensitive pro-inflammatory signaling might also contribute to the beneficial impact of Irg1 in septic liver injury.
Since itaconate is highly polar and difficult to permeate cell membranes, the present study used cell-permeable 4-OI to investigate potential pharmacological benefits of Irg1/itaconate in septic liver injury. 20 Previous study reported itaconate modulated macrophage metabolism and immunity and exerted protective effects against oxidative stress in hepatocytes.12,20 The results indicated that supplementing 4-OI stimulated the Nrf2-HO1 pathway, restrained oxidative stress and inflammatory response as well as alleviated liver injury in experimental animals. Accordingly, 4-OI might have underlying value as medication against septic liver injury.
However, our study has some limitations. Firstly, we didn’t use power analysis for sample size calculation. Second, although the Nrf2 pathway activated by itaconate has been reported in many studies, the mechanism of whether the inhibition of the Nrf2 pathway aggravated septic liver injury should be further investigated. Finally, we only validate the results in animal experiments. Cell experiments and clinical trials are absent. Further studies are warranted.
Conclusion
In conclusion, the current study figured that septic liver injury is linked with the upregulation of hepatic Irg1, genetic deletion of Irg1 exacerbated septic liver injury, which was concomitant with compromised excitation of Nrf2 antioxidative pathway. Interestingly, supplementation with the cell-permeable 4-OI stimulated Nrf2 pathway and alleviated septic liver injury. Although the mechanisms underlying the hepatoprotective benefits of Irg1/itaconate need to be further researched, the current study suggests that Irg1/itaconate might have latent significance for the pharmaceutical intervention of septic liver injury.
Footnotes
Author contributions
GL and LZ designed the experiments.TZ, KL and JQ acquired the data. TZ, KL, YQ, JL and QZ analyzed and interpreted the data. TZ wrote the manuscript. GL, LZ, XW and KL handled the funding and supervision. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Chongqing (grant no. cstc2020jcyj-msxmX0254; cstc2019jcyj-msxmX0294), the Graduate Innovative Special Fund Projects of Chongqing (grant no. CYS21248), the Chongqing Municipal Public Health Bureau of Chongqing People’s Municipal Government (grant no. 2022MSXM006), and the Science and Technology Research Program of Chongqing Municipal Education Commission (KJQN202000444).
Ethical statement
Data availability statement
The data that support the findings of this study are available from the corresponding author.
