Abstract
Occlusion of arteries in the brain is a common cause of cerebral infarction which induces inflammatory response and oxidative stress resulting in neuronal apoptosis and disruption of neurological function. The present study investigated the protective roles of an nicotinamide adenine dinucleotide phosphate oxidase inhibitor, apocynin, against cerebral infarction. Rat went through a surgery of middle cerebral artery occlusion and a subset of rats was treated with apocynin by intraperitoneal injection. The volume of cerebral infarction and water content were measured. Neuronal apoptosis, inflammatory response, and oxidative stress were assessed following middle cerebral artery occlusion and apocynin treatment. We found that apocynin significantly improved neurological function, increased forelimb placement test scores, and suppressed balance beam walk latency in rats with cerebral infarction. Histological and biochemistry analysis revealed that apocynin lead to a significant reduction in the volume of cerebral infarction as well as cerebral water content, suppressed neuronal apoptosis, oxidative stress, and inflammatory response induced by middle cerebral artery occlusion. Finally, we found that apocynin suppressed Tlr4/nuclear factor-k-gene binding signaling pathway that was upregulated in rats with cerebral infarction. Our results indicate that apocynin may represent a potent therapeutic strategy in alleviating neurological dysfunctions in patients with cerebral infarction.
Introduction
Cerebral infarction or stroke is a common disease associated with multiple neurological deficits including dementia, aphasia, and hemiplegia. 1 Minutes following the onset of stroke, reduced blood flow induces a complicated cascade of pathological processes including inflammatory response and oxidative stress. This further leads to massive cell death and subsequently brain damage. Blood flow blockage, which induces cerebral infarction, may result in different degrees of symptoms and outcomes depending on the size and region of the brain being impacted, such as loss of sensation or paralysis on the opposite side of the body of the affected brain side, problems in vision and language, loss of memory, or even death.
Nicotinamide adenine dinucleotide phosphate (NADPH) oxidases are important sources of reactive oxygen species (ROS), whose upregulation is frequently observed as a result of brain injury such as cerebral ischemia and reperfusion. 2 NADPH oxidases are implicated in multiple neurological disorders and highly relevant to neurological dysfunction, neuroinflammation as well as neural degeneration. NADPH oxidases are involved in mediating brain injury and blood–brain barrier (BBB) disruption in middle cerebral artery occlusion (MCAO) induced cerebral ischemia/reperfusion. 3 NADPH oxidase deletion or NADPH oxidase activity inhibition protects against ROS production and BBB disruption and attenuates brain lesion. 3
Apocynin is a selective NADPH oxidase inhibitor extracted from Picrorhiza kurroa roots and has been show to suppress superoxide production in various cell types, although it was also shown that apocynin did not inhibit vascular NADPH oxidases while acted as an antioxidant. 4 The neuroprotective role of apocynin has been reported in various scenarios. 5 Apocynin prevented ischemia/reperfusion-induced BBB damage. 3 In addition, it has been shown that apocynin attenuated cerebral infarction induced by transient focal ischemia manifested by reduced infarct size which is accompanied by suppression of NADPH oxidase activity and reduced production of superoxide. 6
The molecular mechanism underlying how apocynin treatment results in attenuation of cerebral infarction is elusive. Previously, it has been shown that Tlr4/ nuclear factor-k-gene binding (NF-κB) signaling pathway was enhanced as a result of cerebral ischemia/reperfusion and suppression of this pathway alleviated neurological dysfunction and brain damage. 7 In a rat model of traumatic brain injury, apocynin administration attenuated behavioral impairment and neuronal damage by suppressing inflammation mediated through Tlr4/NF-κB signaling pathway. 8
We hypothesized that MCAO induced impairment of neurological function could be attenuated by apocynin treatment through suppression of neuronal apoptosis and inflammation. In the current study, we tested this hypothesis by investigating the protective effects of apocynin against cerebral infarction induced neural functional disruption and the underlying molecular mechanisms. We identified multipotent effects of apocynin in alleviation brain artery occlusion induced cerebral infarction.
Materials and methods
Rat CI model
All animal experiments were conducted in accordance with The First Affiliated Hospital of Wenzhou Medical University’s Institutional guideline. Adult male Sprague–Dawley rats were purchased from SLAC (Shanghai, China) and maintained in our animal facility under standard environment with free access to water and food. A total of 44 rats were used in the study and divided into surgery and/or drug treatment groups: sham, cerebral infarction (CI), and CI + apocynin. CI was induced by MCAO according to a previously described procedure. 9 Briefly, rats were anesthetized with 20% urethane by intraperitoneal (i.p.) injection and fixed on an operation bench in the supine position. Following an incision at the cervical midline, common carotid artery (CCA), external carotid artery (ECA), and internal carotid artery (ICA) were exposed and isolated from surrounding tissue. Following ligations of CCA at the proximal end and ECA at the root, a nylon suture was inserted into CCA from ICA to the origin of CCA and fixed after meeting resistance. The incision was sutured. Rats were recovered and housed until further analysis. Rats in the sham group went through the same operations except the ligation. Apocynin was dissolved in 1% dimethyl sulfoxide (in saline) and i.p injected into rats immediately following MCAO surgery at a dose of 50 mg/kg according to previous studies.6,8 After the first injection, apocynin was injected daily until the rats were euthanized.
A subset of rats was subjected to neural function analysis for 28 days and this duration was chosen according to a previous study 8 : sham (n = 6), CI (n = 6), and CI + apocynin (n = 8). The rest of experimental rats were euthanized for histological and biochemical analysis at post operation day 5 according to a previous study 9 : sham (n = 8), CI (n = 8), and CI + apocynin (n = 8).
Forelimb placement test
At post operation days 1, 7, 21, and 28, rats were subjected to forelimb placement test as described previously. 10 Briefly, the subject was held by the torso with one forelimb fixed to a table and stimulated by brushing the vibrissae of the rat against the table edge. Rats with intact neurological function would place the free forelimb to the table upon vibrissae stimulation. A total of 10 stimulations were tested each forelimb and the number of times the free forelimb touched the table was recorded. Data were presented as the mean percent of times that forelimbs of each rate touched the table edge for each group.
Balance beam walking test
Balance beam walking test was performed to assess motor coordination according to a previous procedure. 8 Each rat was trained on the balance beam for 3 days prior to surgery and tested on post operation days 1, 7, 14, 21, and 28. The latency (s) for the rats to traverse the beam was measured. The maximum observation time is 60 s. Rats that did not cross the beam was placed back to the holding cage and the time was recorded as 60 s. Three trials were performed for each rat. The average time was calculated for each group from each test day.
Neurological score assessment
Neurological deficits of each rat were assessed according to a 5-point system with a higher score indicating more severe deficit based on previous studies.9,11 The scores were assigned to the rats based on following criteria: 0—no deficit, 1—mild deficit observed through tail suspension during which subject was unable to extend forelimb fully, 2—moderate deficit with rats walking in a circle to the side of cerebral infarction, 3—severe deficit with rats falling to the side of cerebral infarction, 4—very severe deficit with rats showing reduced conscious level and lacking of spontaneous walk.
Triphenyltetrazolium chloride staining
Triphenyltetrazolium chloride (TTC) staining was used to measure cerebral infarction volume as described previously. 9 Briefly, rat brain was placed at −20°C for 5 min. After sequential slicing every 3 mm, brain samples were treated with 2% TTC solution for 20 min at 37°C and 4% paraformaldehyde for 10 h. Brain slices were then imaged and analyzed using the BI-2000 Medical image analysis system (Olympus Optical Co., Ltd, Tokyo, Japan). Total infarction volume was calculated by adding all slices in each brain.
Cerebral water content measurement
Determination of cerebral water content was accomplished by the dry–wet method. 9 First, wet weight was acquired from freshly dissected brain. Dry weight was acquired following a drying step for 24 h at 100°C. Cerebral water content was represented as the percentage of water, which was the difference between dry weight and wet weight, over wet weight.
TUNEL staining
Neuronal apoptosis was assessed by terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick end labeling assay (TUNEL) staining. 9 Brain sections were incubated in 2% H2O2 for 10 min at room temperature and in 20 g/mL non-deoxyribonuclease (DNase) proteinase K (Merck Drugs & Biotechnology Inc., Darmstadt, Germany) at 37°C for 30 min. Following phosphate buffer saline (PBS) wash, brain slices were incubated with terminal deoxynucleotidyl transferase (TdT) (F. Hoffmann-La Roche & Co., California, USA) and biotin-dUTP (F. Hoffmann-La Roche & Co.) for 1 h at 37°C, washed with PBS, and incubated with a mixture of streptavidin–HRP enzyme (F. Hoffmann-La Roche & Co.) (10 μL) and Biotin-dUTP (490 μL) for 1 h at 37°C. Staining was developed by a 3′-diaminobenzidine (DAB) solution (F. Hoffmann-La Roche & Co.). Hematoxylin (Solarbio Science & Technology Co., Ltd., Beijing, China) was used for counter staining. Ratio of TUNEL positive cells over total cells (%) was calculated based on cells counts from 10 randomly acquired images.
Western blot analysis
Western blot analysis was performed to assess relevant protein levels according to a standard protocol. 12 Briefly, rat cerebral cortices were isolated and homogenized in lysis buffer. An equivalent amount of protein from each sample was subjected to blotting. The following antibodies were used: anti-cleaved Caspase 3, anti-Bcl-xL, anti-tumor necrosis factor (TNF)-α, anti-interleukin (IL)-1β, anti-Tlr4, anti-NF-κB-p65, anti-phosphorylated-NF-κB-p65, and anti-β-actin from Abcam, Cambridge, UK. Proteins were detected with ECL plus. Data were analyzed by ImageJ and normalized to β-actin. Protein expression relative to sham group was presented.
Oxidative stress assessment
Activity of superoxide dismutase (SOD), content of malondialdehyde (MDA) and catalase (CAT) activity were measured to assess oxidative stress as previously shown.9,13 Briefly, freshly isolated brains were dried and homogenized in saline at 1:9 (w:v). Supernatant was saved for further analysis after centrifuging at 3000 r/min for 15 min at 4°C. SOD activity was measured with xanthine oxidase approach and presented as unit/mg (U/mg) protein. MDA content was measured by its reaction with thiobarbituric acid and presented as µg/mg protein.
Real-time polymerase chain reaction
mRNA levels of relevant genes were assessed by real-time polymerase chain reaction (RT-PCR) as previously described. 7 Total RNA was extracted from rat brain tissue using TRIzol (Invitrogen, Carlsbad, CA, USA). RT-PCR was performed using SYBRGreen I kit (Takara, Otsu, Shiga, Japan) through ABI PRISM 7500 Real-Time PCR System (ABI, Carlsbad, CA, USA). β-actin was used as an internal control. Relative target gene expression was analyzed using the 2−ΔΔCt approach. Data in each group were first normalized to the internal control and then normalized to Sham group. Primers used in this current are Tlr4-forward 5′-GGACTCTGCCCTGCCACCATTTA-3′ and Tlr4-reverse 5′-CTTGTGCCCTGTGAGGTCGTTGA-3′; NF-κB-p65-forward 5′-GTGCAGAAAGAAGACATTGA-3′ and NF-κB-p65-reverse 5′-AGGCTAGGGTCAGCGTATGG-3′; Actb-forward 5′-ACTGGCATTGTGATGGACTC-3′ and Actb-reverse 5′-CAGCACTGTGTTGGCATAGA-3′.
Statistical analysis
Data were presented as mean ± standard deviation (SD). Statistical difference between indicated groups was determined by Student’s t test or analysis of variance (ANOVA) analysis. Difference is regarded significant with P value <0.05.
Results
Apocynin accelerates neural functional recovery in rats with CI
We investigated neural function in rats with CI for 4 weeks following induction of CI by MCAO surgery. We found that MCAO surgery drastically disrupted neural function on day 1 post-surgery followed by slow and incomplete recovery after 4 weeks (Figure 1). Administration of apocynin significantly accelerated neural functional recovery.
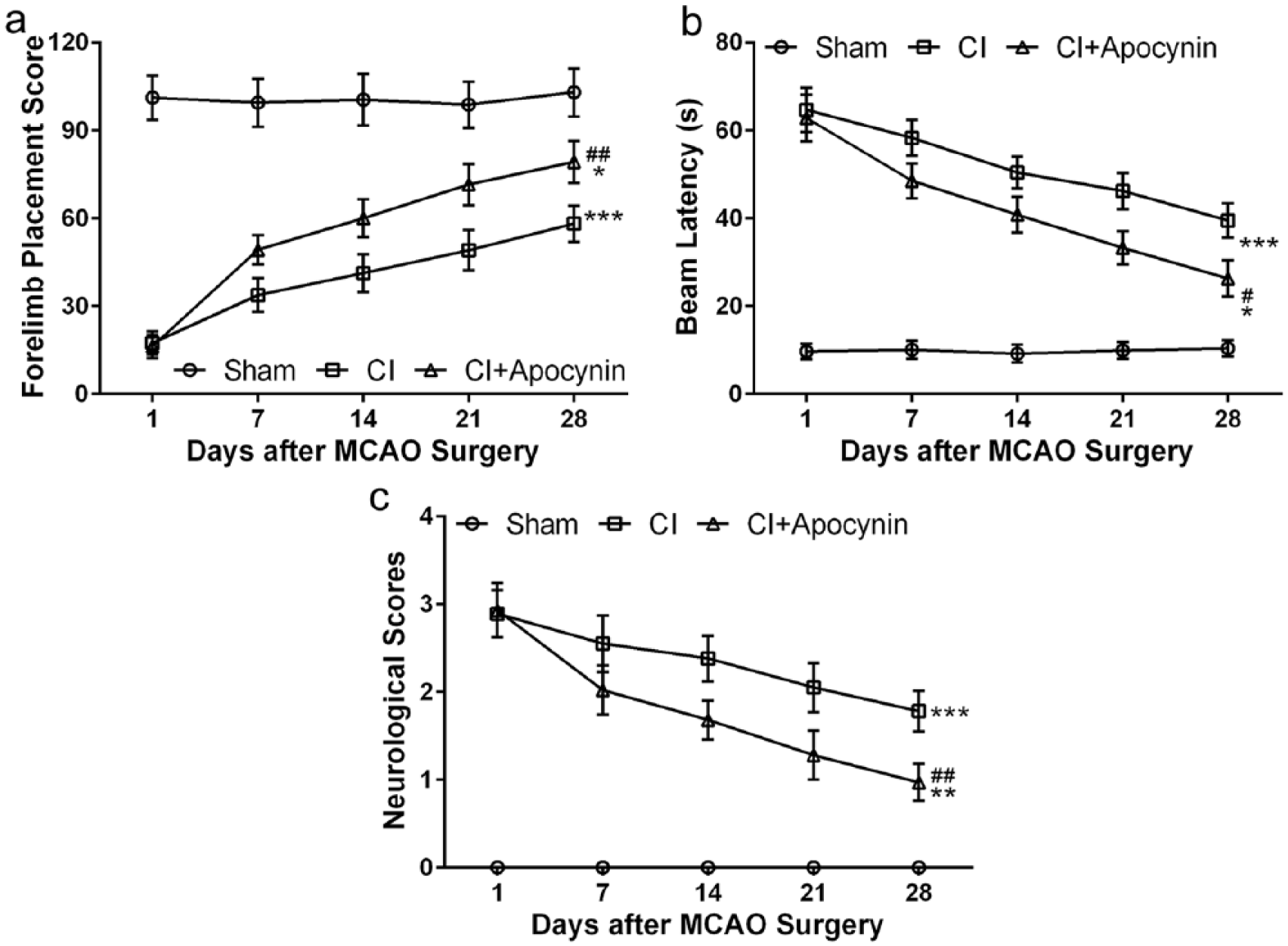
Apocynin accelerated neurological function recovery in rats with cerebral infarction during 4 weeks of tracking test. (a) Comparisons of scores of forelimb placement test of rats among three groups (%). (b) Comparisons of beam walk latencies during the balance beam walking test among three groups. (c) The neurological scores of rats in the three groups. Data are presented as mean ± SD. *P < 0.05, **P < 0.01, and ***P < 0.001 compared to sham group; #P < 0.05 and ##P < 0.01 compared to CI group.
In the forelimb placement test, average scores of sham-operated mice were around maximum in all trials from day 1 to day 28 post-surgery (Figure 1(a)). MCAO surgery lead to a drastic reduction in forelimb placement score on day 1 post-surgery and all the following trials till day 28. Apocynin treatment in CI rats significantly increased forelimb placement scores, although these scores were significantly lower than that of the sham group.
In the balance beam walking test, the latency for the rats to walk through the balance beam significantly increased in CI group compared with control group (Figure 1(b)). Apocynin treatment significantly reduced walking latency in CI rats and recovered faster.
In addition, we assessed the overall neural function and assigned each rat with a neurological score (Figure 1(c)), with a lower score (from 0 to 4) indicating better performance. We found that all score of the rats in the sham group is 0, suggesting that they all functioned normally after operation. An average of 3 points was assigned to rats of both CI and CI + apocynin groups 1-day post-surgery indicating the presence of severe neural deficits in these rats. The scores of rats from both CI and CI + apocynin groups reduced gradually from 1- to 28-day post-surgery with rats from CI + apocynin group showing significantly lower scores than that of the CI group.
Apocynin attenuates rat cerebral infarction induced by MCAO surgery
We examined cerebral infarction by TTC staining (Figure 2(a)). Quantification of CI volume revealed that MCAO surgery induced large area of CI which was significantly reduced as a result of apocynin treatment (Figure 2(b)). We then measured cerebral water content. The water content in the sham group was about 75%, while it increased to over 80% in the CI group (Figure 2(c)). Apocynin treatment significantly reduced water content in CI rats compared to those without treatment.

Apocynin meliorated cerebral infarction in rats 5 days after MCAO surgery. (a) Representative TTC staining of brain tissue among different groups. (b) Volume of cerebral infarction was quantified from TTC staining. (c) Cerebral water content was quantified from different groups. Data are presented as mean ± SD. *P < 0.05, **P < 0.01, and ***P < 0.001 compared to sham group; #P < 0.05 and ##P < 0.01 compared to CI group.
Apocynin reduces neuronal apoptosis in rats with CI
We found that MCAO surgery drastically increased the number of cells with positive TUNEL staining, indicating an increase of apoptotic cells in CI group when compared to sham operation (Figure 3(a) and Figure 3(b)). The number of cells undergoing apoptosis was significantly reduced by apocynin administration.

Apocynin suppressed neuronal apoptosis in rats with cerebral infarction. (a) Representative TUNEL staining of neuronal cells in the infarct area of the brain among different groups and quantification of cell apoptosis (b). Data are presented as mean ± SD. *P < 0.05 and ***P < 0.001 compared to sham group; #P < 0.05 compared to CI group.
We further confirmed this observation by examining two proteins involved in the apoptotic pathway, the anti-apoptotic Bcl-xL and pro-apoptotic Caspase-3. Western blot analysis (Figure 4(a)) showed that MCAO surgery significantly reduced Bcl-xL expression which was partially reversed by apocynin treatment (Figure 4(b)). On the other hand, Caspase-3 levels were significantly increased rats with CI which was also partially reversed by apocynin treatment (Figure 4(c)).

Apocynin altered Bcl-xL and caspase 3 expression in rats with cerebral infarction. (a) Bcl-xL and caspase 3 protein expressions in the infarct area of the brain were determined by Western blot analysis. (b) Quantification of Western blot analysis as normalized to sham group, the loading control. Data are presented as mean ± SD. *P < 0.05, **P < 0.01, and ***P < 0.001 compared to sham group, #P < 0.05 and ##P < 0.01 compared to CI group.
Apocynin suppresses oxidative stress and inflammation in rats with CI
We observed a significant increase of oxidative stress in rats following MCAO surgery as indicated by reduced SOD activity (Figure 5(a)), increased MDA content (Figure 5(b)), and reduced CAT activity (Figure 5(c)). Similarly, MCAO surgery also lead to an induction of inflammatory response manifested by increased levels of proinflammatory cytokines TNF-α and IL-1β Figure 5(d) and (e) . We found that strikingly, apocynin administration in rats with CI not only suppressed oxidative stress by increasing SOD and CAT activities and decreasing MDA content, but also reduced TNF-α and IL-1β levels.

Apocynin suppressed oxidative stress and inflammation in rats with cerebral infarction. SOD activity (a), MDA content (b), and the level of CAT (c) were measured from the infarct area of the brain in the three groups. (d) Western blot analysis of TNF-α and IL-1β expression in the infarct area of the brain. (e) Quantification of Western blot results as normalized to sham group. Data are presented as mean ± SD. *P < 0.05, **P < 0.01, and ***P < 0.001 compared to sham group; #P < 0.05 and ##P < 0.01 compared to CI group.
Apocynin suppresses Tlr4/NF-κB signaling in rats with CI
Finally, we examined Tlr4/NF-kB signaling pathway within cerebral cortex. We found that the mRNA levels of Tlr4 (Figure 6(a)) and NF-κB (Figure 6(b)) were significantly increased following MCAO surgery. Apocynin treatment suppressed both Tlr4 and NF-κB mRNA expression. Similarly, Western blot analysis (Figure 6(c)) revealed that MCAO surgery significantly enhanced Tlr4, phosphorylation of NF-κB protein, and NF-κB protein expression which was suppressed when combined with apocynin administration (Figure 6(d)).
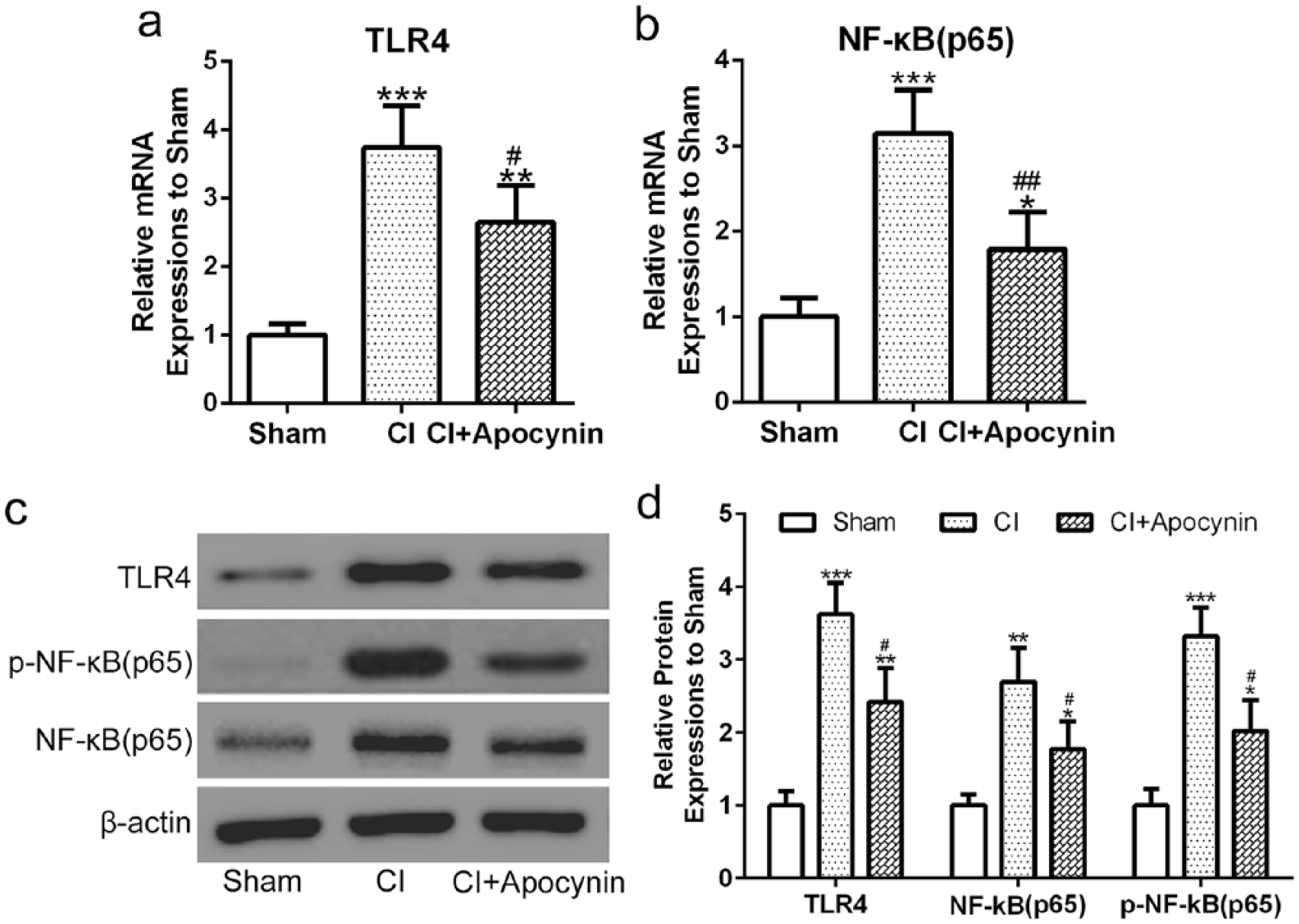
Apocynin inhibited Tlr4/NF-κB signaling pathway in rats with cerebral infarction. (a–b) mRNA expression of Tlr4 and NF-κB p65 in the infarct area of the brain was measured by real-time PCR. (c) Protein expression of Tlr4, phosphorylation of NF-κB p65 and NF-κB p65 in the infarct area of the brain was assessed by Western blot analysis. (d) Quantification of western blot results as normalized to sham group. Data were normalized to sham group. Data are presented as mean ± SD. *P < 0.05, **P < 0.01, and ***P < 0.001 compared to sham group, #P < 0.05 and ##P < 0.01 compared to CI group.
Discussion
Apocynin has been shown to suppress NADPH oxidase activity and alleviate MCAO induced cerebral infarction. The underlying molecular mechanism is unclear. In the current study, we investigated the effects of apocynin on neurological function of rats with cerebral infarction and explored the underlying mechanisms. We found that MCAO surgery lead to severe disruption of neurological functions followed by a slow and incomplete recovery. Apocynin administration significantly accelerated neurological function recovery. In addition, apocynin significantly reduced cerebral infarction volume and water content following MCAO surgery. Investigation of the underlying molecular mechanism revealed that apocynin suppressed MCAO-induced apoptosis, oxidative stress, and inflammatory response through Tlr4/NF-kB signaling pathway.
Cerebral infarction is the most common cause of adult neurological dysfunction and disabilities that are frequently long term or even lifelong. 14 In this study, we took advantage of the MCAO model to explore the beneficial effects of apocynin in relieving neurological dysfunction in rats with cerebral infarction. Previously, it has been shown that rats with diabetes induced by alloxan administration and cerebral infarction by MCAO showed drastic increase in neurological score, suggesting severe deficits of neurological functions in these rats. 9 Similar to this report, we also observed a high average score of nearly 3 points 1 day after MCAO surgery meaning that these rats were unable to maintain a standing position and they fell to the side of injury. Four weeks after surgery, these mice still showed substantial neurological impairments and abnormal walking pattern. The neurological scores gradually reduced to 2 points at this time point. In addition, rats with cerebral infarction also performed poorly in other behavioral assessments, including forelimb placement test and balance beam walk test. The majority of rats undergone MCAO surgery were unable to place the free limb to the table edge during the experiment after 1 day and even after 4 weeks of recovery, the average score was only about 50, while the average score of sham group remained about 100, indicating almost all control rats performed perfectly during this test. Similarly, majority of rats were unable to cross the balance beam within 60 s while the average time for control rats was less than 10 s. Four weeks after surgery, it still took an average of 40 s for rats to cross the balance beam, indicating the presence of long-term neurological deficits.
Our results with apocynin administration following MCAO surgery indicated that apocynin did not elicit an immediate response. Rats in the CI + apocynin exhibited similar severity in neurological function assessment 1-day post-surgery. However, we found that apocynin substantially accelerated the recovery process and neurological functions assessed at 4 weeks after surgery were much better than that of MCAO surgery alone. It is worth noting that the recovery is not complete in both CI and CI + apocynin groups, suggesting that CI may have induced some permanent damage to the brain which cannot be reversed by apocynin treatment alone.
Disruption of BBB integrity is frequently observed in animal models of cerebral infarction as well as other brain injury models such as traumatic brain injury, leading to increased BBB permeability.15–17 As a result of BBB disruption, brain edema is also induced by brain injury. Consistently, we observed a substantial increase in cerebral water content of rats with cerebral infarction. Along with this observation, previously studies suggested that apocynin protects against brain injury–induced BBB disruption. 18
Massive neuronal loss following stroke is a key cause of neurological dysfunction and suppressing neuronal death is among the most important focuses to promote recovery from therapeutic perspective.19,20 Cerebral ischemia induced by MCAO surgery in animal models is associated with massive cell death and neuronal loss. 21 In the present study, neuronal apoptosis was confirmed by TUNEL staining. We found that MCAO surgery lead to massive neuronal death, down regulation of anti-apoptotic Bcl-xL, and apoptotic marker active Caspase 3. Importantly, our results suggest that apocynin treatment significantly suppressed cell death and reversed Bcl-xL and Caspase 3 expression.
Two cellular processes that cause neuronal apoptosis are oxidative stress and inflammatory response following onset of cerebral infarction. 22 Increased oxidative stress is responsible for more severe cerebral infarction in rats with spontaneous hypertension. 23 Oxidative stress is caused by ROS over-production whose primary source is NADPH oxidases. 24 A previous study showed that cerebral infarction correlated with elevated activity of NADPH oxidase as well as superoxide level, both of which were suppressed by apocynin. 6 Apocynin is a naturally available antioxidant and an inhibitor of NADPH oxidase. Multiple disease models have suggested that apocynin relieves oxidative stress. Consistently, our observation of reduced SOD activity and elevated MDA contents suggests that oxidative stress is reduced by apocynin in rats with cerebral infarction. While conflicting roles of apocynin on the oxidative stress have been reported previous, our study confirmed its role as an antioxidant and suppressed oxidative stress induced by MCAO surgery in the rat cerebral infarction model. However, our study did not provide direct evidence showing that apocynin-induced alleviation in brain damage resulted from MCA surgery was through inhibition of NADPH oxidase activity.
Similar to oxidative stress, inflammatory response is induced as a result of brain injury. 25 We detected increased levels of TNF-α and IL-1β which were suppressed by apocynin treatment in rats with cerebral infarction. In fact, in addition to our study, apocynin has been shown to exert an anti-inflammatory role in various scenarios, including Parkinson’s disease and spinal cord injury.26,27 A further investigation on the underlying molecular mechanism revealed that Tlr4/NF-κB signaling pathway was abnormally upregulated in rats with cerebral infarction. A previous study showed that Tlr4 inhibition suppresses neuronal apoptosis and attenuates neurological deficits in rats with diabetes and cerebral infarction. 9 Our study together with this previous study suggests that TLR4/NF-κB is involved in apocynin-mediated improvement of neurological function and suppression of apoptosis.
It is interesting to note that apocynin partially suppressed all the adverse effects induced by the MCAO surgery. However, this may not be so surprising. Previous studies have shown that oxidative stress, inflammation, and apoptosis are connected to each other in various systems. For example, in an in vitro model of neuronal degeneration, a dopaminergic toxin–induced oxidative stress leads to neuronal apoptosis and inhibition of NADPH oxidase activity, while inhibition of NADPH oxidase not only suppressed oxidative stress but also rescues neuronal cells from apoptosis. 28 Similarly, oxidative stress has been shown to be linked to inflammation through Toll-like receptors. 29 Consistent with the previous reports, we found that MCAO surgery leads to a cascade of cellular events including increased oxidative stress, inflammatory response, and neuronal apoptosis. It is thus not surprising that apocynin suppressed all these adverse events resulting in improved behavioral and structural neuronal functions.
While it is striking to observe all these effects of apocynin in this CI model, one limitation of our study is that we only used one CI model. In the future, the function of apocynin should be characterized in more detail using different CI models.
Conclusion
In summary, our findings suggest that apocynin significantly improved neurological functions and accelerated recovery after induction of cerebral infarction probably by suppressing neuronal apoptosis, oxidative stress, and inflammatory response through Tlr4/NF-kB signaling pathway. These promising results warrant further investigation of the use of apocynin as a potential therapeutic strategy against cerebral infarction.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by Research and Demonstration Application Project of Social Development in Jiaxing, Zhejiang (2016AY23048).
