Abstract
There is legitimate interest in the effects of recombinant human activated protein C (rhAPC) on various organs and individual patients, but the specific effects on organ tissues during early sepsis remain unknown. Differences in the levels of organ damage may influence responses to drug therapy. We aimed to investigate whether rhAPC induces organ-specific effects on inflammation and apoptosis using randomized, experimental trials with male NMRI mice. Animals underwent caecal ligation and puncture, and after 12 h, sepsis inflammation and apoptosis were assessed by plasma cytokines, gene expression ratios and immunohistochemistry (IHC). RhAPC-treated animals exhibited increased physical activity and decreased cytokine release compared to untreated animals (interleukin-6 reduction 58%, P < 0.001). CD14 expression was higher in the heart and liver and decreased upon rhAPC application in the heart (−35%), liver and kidney (both −60%). Macrophage inflammatory protein 2 (MIP2) expression decreased in the heart (−58%) but not in the liver or kidney. IHC revealed decreased cleaved caspase-3 in the heart and kidney due to rhAPC intervention. Preservation of the endothelial PC receptor was significant only in the heart during sepsis (P = 0.007). In early polymicrobial sepsis, inflammation was more pronounced in the heart and liver compared to the kidney. RhAPC exhibited protective effects, especially in the heart tissue, and led to reduced plasma levels of pro-inflammatory cytokines and improved physical activity.
Introduction
The protein C pathway plays a major role in the pathogenesis of sepsis. Recombinant human activated protein C (rhAPC) was introduced to treat sepsis in critically ill patients to prevent evolving organ dysfunction but was withdrawn after a clinical trial failed to show improvement over placebo. 1 Many concerns were raised about the design of the study, and questions remain concerning the effects of rhAPC in individual organs and patients. 2
There is legitimate interest in the effects of rhAPC on various organs and individual patients to find a treatment for inflammation-induced multi-organ dysfunction (MOD) 3 and other fields for the use of rhAPC.4,5
The key mechanisms of action of RhAPC are thought to be associated with anti-coagulative and anti-inflammatory effects. 6 It was previously shown that murine-activated protein C reduces the injury area in murine ischaemic stroke. 7 Currently, the molecular mechanisms of this neuroprotective effect remain unclear, although apoptosis has been blocked by activated protein C (APC) in the brain endothelium after ischaemic injury depending on endothelial protein C receptor (EPCR) and protease-activated receptor-1 (PAR-1). 8 The EPCR itself was identified as crucial for many of the therapeutic applications associated with recombinant APC, 9 and elevated levels in the plasma of critically ill patients have been associated with a higher risk of infection. 10 APC down-regulates the pro-apoptotic factor Bax and, conversely, up-regulates the anti-apoptotic factor B-cell lymphoma 2 (Bcl-2). 8
Gupta et al. 11 suggested that APC induces decreased inflammatory and apoptotic insults in sepsis-induced acute renal failure. In addition, Mosnier et al. 12 demonstrated variants of APC with reduced activity in the coagulation system that preserved anti-apoptotic activity, implicating at least two independent mechanisms of action. The anti-apoptotic effects of APC were shown on circulating mononuclear cells of septic shock patients as late as 24 h after the onset of septic shock. 13 Ekaney et al. 14 demonstrated a mitigating effect of APC on sepsis-related expression of messenger RNA (mRNA) encoding and ADAMTS13. They concluded that there may be a beneficial effect of APC at the molecular level.
The response to sepsis induced by caecal ligation and puncture (CLP) elicits organ-specific gene expression in a variety of organs. This gene expression response may be common to more than one organ or may even exert opposing effects in different organs. For example, interleukin (IL)-6 gene is up-regulated in the spleen and lung but is more lowly expressed in liver tissue. 15 Thus, the body’s response to sepsis is more diversely regulated among organs than initially suspected, which may lead to variations in the success of drug interventions.
RhAPC effectively decreases systemic pro-inflammatory cytokines, but data clarifying the organ-specific effects of rhAPC during sepsis are lacking. To the best of our knowledge, there are no data clarifying the organ-specific effects of rhAPC during early sepsis. We conducted this study to investigate whether rhAPC provides organ-specific effects on inflammation and apoptosis in the heart, liver and kidney in a murine CLP model.
Methods
Sepsis induction
The study was approved by the appropriate governmental institution and the local animal protection authorities (Thüringer Landesamt für Lebensmittelsicherheit und Verbraucherschutz (TLLV)). All experiments were performed in the laboratories of the Jena University Hospital in accordance with the principles for care and use of animals based on the Helsinki Declaration.
All mice (male NMRI mice; n = 30, body weight 30 ± 3 g) underwent a catheterization procedure. Spontaneously breathing animals under general anaesthesia (desflurane 8%–10% in oxygen/air mix with an FiO2 of 0.3) were fixed in the prone position. The right neck vessels were exposed under sterile conditions after local anaesthesia with 0.2 mL of 2% lidocaine (AstraZeneca, Wedel, Germany), and a central vein catheter (CVC; homemade from a sterilized polyethylene tube with an outer diameter of 0.61 mm) was implanted 1 cm deep into the jugular vein, enabling continuous intravenous application of fluids and drugs throughout the experiment. The CVC was tunnelled to the back of each mouse and guided through a flexible plastic tube (Drainobag 40; B.Braun, Melsungen, Germany) to prevent bite damage. A 27G cannula was inserted into the CVC to connect it to the syringe pump. Furthermore, an arterial transmitter (PA-C10; Data Sciences International, New Brighton, MN, USA) was implanted to measure heart rate (HR) and mean arterial pressure (MAP). The catheter consisted of a transmitter unit and a gel-filled tube, which was introduced and fixed in the right carotid artery. The transmitter unit was implanted subcutaneously in the abdominal region. Subsequently, after further local infiltration with lidocaine, the neck was closed by single sutures, and the mouse was re-transferred into the cage to rest for 48 h prior to sepsis by CLP. To prevent hypothermia, all animals were kept on a heating pad throughout the surgical procedure.
The CLP was performed under sterile conditions; a midline 1-cm laparotomy was performed. The caecum was identified, ligated approximately 1 cm from its base and then triple-perforated with a 21G needle. Faeces were protruded to ensure the perforations were opened. Then, the caecum was replaced and the abdominal cavity was closed with single sutures. The animal was transferred to its cage and reconnected to an intravenous line and syringe pump at a rate of 100 µL/h until the end of the study. All animals were sacrificed after 12 h.
The animals were randomly assigned to one of the three groups:
Sepsis induction and vehicle (0.9% NaCl infusion; sepsis);
CLP sepsis with rhAPC (drotrecogin alfa (activated); Lilly Inc., Bad Homburg, Germany) infusion (24 µg/kg/h rhAPC in 0.9% NaCl; rhAPC-sepsis);
Sham operation and vehicle infusion (Sham).
All animals received a bolus of 200 µL 0.9% NaCl when transferred to the cage.
Vital data measurements
The average of the last five measurements within 1 h of HR and the MAP using an arterial transmitter were recorded after 12 h of sepsis. To evaluate the clinical condition of the animals, the physical activity of each mouse was independently recorded by two investigators who were blinded to the treatment according to a predefined scoring system ranging from 1 (healthy) to 5 (in agony). 16
Sampling
After 12 h of sepsis, the animals were anaesthetized and placed in a prone position, and the abdomen and the thorax were opened. Blood samples were taken using pre-citrated syringes, and after centrifugation, the plasma samples were frozen according to the manufacturer’s guidelines until measurement. Each kidney was split lengthwise, and the left ventricle of the heart was used. Two portions of the kidney and the heart were frozen in liquid nitrogen and stored at −80°C, and one portion of the tissue samples was stored in 2% formalin for subsequent embedding into paraffin. The right lobe of the liver was also divided and stored in formalin as well as snap-frozen. RNA sampling and cryo-sectioning were performed in every animal.
Laboratory measurements
Plasma cytokines
Determination of IL-6, IL-10, monocyte chemoattractant protein-1 (MCP-1), interferon-γ (IFN-γ), tumour necrosis factor-α (TNF-α) and IL-12p70 levels was performed in duplicate using a cytometric bead array (CBA; BD Biosciences, Heidelberg, Germany). The CBA combines six bead populations with distinct fluorescence intensities with specific capture antibodies. These bead populations were quantitatively measured using a flow cytometer.
Immunohistochemistry
Cleaved caspase-3
Analysis of the 4-µm paraffin sections was performed using the avidin–biotin–peroxidase complex (ABC) method. 17 For the cleaved caspase-3 analysis, a rabbit polyclonal anti-cleaved caspase-3 antibody (CPP32; Zytomed, Berlin, Germany) was used (dilution 1:100) as the primary antibody after antigen retrieval (in 0.01 M citrate buffer, pH 6.0 in a microwave oven, 640 W for 15 min). A biotinylated goat anti-rabbit antibody (Vector Laboratories, Burlingame, CA, USA) was used as the secondary antibody. The peroxidase substrate was diaminobenzidine, and Harris haematoxylin was the counterstain.
EPCR
Cryo-sections of 8 µm were fixed in ice-cold acetone for 10 min and dried. After blocking with rabbit serum, the sections were incubated with a monoclonal rat anti-mouse CD201 (EPCR) antibody (Clone e-bio 1560; eBioscience, San Diego, CA, USA). Rabbit anti-rat biotinylated antibody (Vector Laboratories) was used as a secondary antibody for the ABC method. For the liver and kidney sections, an avidin–biotin blocking (Vector Laboratories) kit was applied before the primary incubation.
To quantify the amount of EPCR-positive or cleaved caspase-3-positive cells, respectively, 10 randomly chosen, adjoining fields in knight’s moves (no edge region) were analysed by two independent and blinded observers via light microscopy (original magnification, 400×) of each heart, liver and kidney, and the average was recorded. For the quantification of EPCR-positive cells, only sections with clearly identifiable vessels (specific brown endothelial staining) have been selected. For the quantification of cleaved caspase-3, only cells with a clearly identifiable positive (brown) staining have been considered as cleaved caspase-3-positive cells.
Gene expression
Polymerase chain reaction
RNA was extracted from the frozen tissue (RNeasy; Qiagen, Hilden, Germany), reverse-transcribed to complementary DNA (cDNA) (Quanti Tec; Qiagen) and analysed via real-time polymerase chain reaction (PCR) (RT2 real-time SYBR green/rROX PCR master Mix, PA-012; Bio-Rad, Hercules, CA, USA) with relative quantification using the housekeeper gene β-actin for analysis. Mouse-specific primers were used for Bax, Bcl-2, CD14 and macrophage inflammatory protein 2 (MIP2) (all from Biomers, Ulm, Germany) using an iCycler (Bio-Rad).
PCR was performed using 20 ng of cDNA and standard PCR protocols in 96-well PCR plates (ABgene, Epsom, UK) containing 25 µL reaction mixture/well with ReadyToGo PCR beads (Amersham, Braunschweig, Germany). The PCR primers were as follows: Bax: 5′-GCT GGA CAC TGG ACT TCC TC-3′ (forward), 5′-CTC AGC CCA TCT TCC AG-3′ (reverse); Bcl-2: 5′-CCT GTG GAT GAC TGA GTA CCT G-3′ (forward), 5′-AGA CAG CCA GGA GAA ATC AAA C-3′ (reverse); CD14: 5′-CAG AAT CTA CCG ACC ATG GAG-3′ (forward), 5′-GGA ACA ACT TTC CTC GTC TAG C-3′ (reverse); and MIP2: 5′-ACC AAC CAC CAG GCT ACA GG-3′ (forward), 5′-CCC TTG AGA GTG GCT ATG ACT T-3′ (reverse).
The PCR products were precipitated by adding 85 µL of 96% ethanol. The DNA was pelleted by centrifuging for 60 min at 3500 r/min and at 4°C using an Eppendorf 5810R centrifuge and then washed with 70% ethanol. The pellets were dried by evaporation using a Savant SpeedVac Plus SC210A and dissolved in 15 µL of water.
The sequencing primers were reverse primers for every product. The correct PCR products were verified by Sanger sequencing of the PCR products using Dye Terminator chemistry (v3.1; Applied BiosystemsTM, Thermo Fisher Scientific, Waltham, MA, USA) and PCR primers according to the manufacturer’s instructions. The PCR was run twice in triplicate.
Statistics
After verifying the data fit of a normal distribution (skewness <1.5), 18 differences between groups were evaluated by a one-way analysis of variance (ANOVA) and a post hoc Dunnett test. A pair-wise fixed reallocation randomization test was run for PCR diagnostics. 19 All data are reported as the mean ± SD unless otherwise indicated. P < 0.05 was considered significant.
Results
Activity and vital parameters
After 12 h of CLP, the physical activity of the rhAPC-sepsis group was significantly higher (P = 0.015; Table 1) compared to the sepsis group. There were no significant differences in HR between the groups (Table 1). The MAP was significantly higher in the sham group compared to the sepsis group (P = 0.031; Table 1).
Physical activity index, mean arterial pressure and heart rate after 12 h of sepsis.

RhAPC: recombinant human activated protein C; Sham: sham operated; sepsis: sepsis without treatment; rhAPC-sepsis: sepsis with treatment of rhAPC.
The physical activity index ranged from 1 (best activity) to 5 (agony).
P < 0.05 vs sepsis; †P < 0.01 vs sepsis.
Plasma cytokines
IL-6 was significantly increased in the sepsis group and decreased in the rhAPC-sepsis group, whereas IL-10 was not significantly decreased in the rhAPC-sepsis group (Table 2). MCP-1 and IFN-γ levels were significantly reduced after intervention in the rhAPC-sepsis group compared to the sepsis group (Table 2). TNF-α was not significantly lower in the rhAPC-treated mice compared to sepsis group, and IL-12p70 was not reduced due to rhAPC intervention (Table 2).
Serum cytokines after 12 h of sepsis.

RhAPC: recombinant human activated protein C; Sham: sham operated; sepsis: sepsis without treatment; rhAPC-sepsis = sepsis with treatment of rhAPC; IL: interleukin; MCP-1: monocyte chemoattractant protein-1; IFN-γ: interferon-γ; TNF-α: tumour necrosis factor-α.
P < 0.05 vs sepsis; †P < 0.01 vs sepsis.
Immunohistochemistry
Cleaved caspase-3
Heart
In paraffin-embedded heart tissue, the number of positive cells per visual field was 2.1 ± 0.6 in the sham-treated animals and 9.6 ± 4.0 in the sepsis group (P = 0.001). In the rhAPC-sepsis group, there was a reduction in the number of positive cells to 4.9 ± 2.7 (P = 0.010 vs sepsis; Figure 1(a) and (b)).

(a) Cleaved caspase-3 in heart, liver and kidney tissue after 12 h of sepsis. Therapy with rhAPC resulted in a decrease in the number of cleaved caspase-3-positive cells in the heart and kidney but not in the liver. Pc/lf = positive cells per visual field; black column = sham-operated group; white column = sepsis group (sepsis without treatment); red column = rhAPC-sepsis group (sepsis with treatment of rhAPC). The data are reported as mean ± SD. *P < 0.05 vs sepsis; †P < 0.01 vs sepsis. (b) Representative sections of heart tissue; arrows: cleaved caspase-3-positive cells. (c) Representative sections of liver tissue; arrows: cleaved caspase-3-positive cells. (d) Representative sections of kidney tissue; arrows: cleaved caspase-3-positive cells.
Liver
The liver tissue sections showed 10.8 ± 8.2 cleaved caspase-3-positive cells in the sham group and 16.2 ± 4.5 positive cells in the sepsis group (n.s.). Liver tissue in the rhAPC-sepsis group demonstrated 12.4 ± 6.2 active caspase-3-positive cells (n.s.; Figure 1(a) and (c)).
Kidney
The kidneys of the sham-treated mice had 1.4 ± 1.1 cleaved caspase-3-positive cells versus 5.0 ± 1.8 in the sepsis group (P = 0.03). RhAPC treatment resulted in a reduction to 1.6 ± 2.3 positive cells (P = 0.01 vs sepsis; Figure 1(a) and (d)).
EPCR
Heart
The number of cells positive for EPCR was 6.3 ± 1.0 in the sham-operated mice and was significantly reduced to 1.8 ± 1.3 positive cells in the sepsis group (P < 0.001). The treatment group showed less of a reduction upon administration of rhAPC (4.3 ± 1.4, P = 0.007 vs sepsis; Figure 2(a) and (b)).

(a) Endothelial protein C receptor in the heart, liver and kidney tissue after 12 h of sepsis. Therapy with rhAPC resulted in a reduced loss of anti-ePCR-positive cells in the heart. A similar finding was observed in the kidney tissue, although the result was not significant. There was no effect on ePCRs in liver tissue due to rhAPC treatment. Pc/lf = positive cells per visual field; black column = sham-operated group; white column = sepsis group (sepsis without rhAPC treatment); red column = rhAPC-sepsis group (sepsis with treatment of 24 µg/kg/h rhAPC). The data are reported as mean ± SD. *P < 0.05 vs sepsis; †P < 0.01 vs sepsis. (b) Representative sections of heart tissue; arrow: endothelial protein C receptor–positive cell. (c) Representative sections of liver tissue; arrow: endothelial protein C receptor–positive cell. (d) Representative sections of kidney tissue; arrow: endothelial protein C receptor–positive cell.
Liver
The liver tissue revealed comparable positive cell counts for all groups, with no significant differences (sham: 8.0 ± 2.3; sepsis: 6.3 ± 5.1; rhAPC-sepsis: 7.2 ± 3.8; Figure 2(a) and (c)).
Kidney
In the kidney, staining for ePCR revealed the highest cell counts in the sham-treated mice (9.5 ± 4.0; P = 0.036 vs sepsis) and a decrease to 3.8 ± 4.3 cells in the sepsis group. The rhAPC-sepsis group exhibited no differences in the number of ePCR-positive cells (6.8 ± 2.5; Figure 2(a) and (d)).
Gene expression
Bax
The normalized gene regulation of Bax in heart tissue was not significantly elevated in either the sepsis or the rhAPC-sepsis group (mean ± SEM-fold: 3.3 ± 2.0 (P = 0.119) and 2.8 ± 2.3 (0.259), respectively; Figure 3). Bax expression in the liver tissue of the sepsis group was 0.6 ± 0.3 (P = 0.303). RhAPC-treated mice showed Bax expression of 0.3 ± 0.3 (P = 0.439; Figure 3). In kidney tissue, the expression of Bax mRNA was 1.1 ± 0.7 (P = 0.917). The rhAPC-sepsis group exhibited a Bax expression of 1.4 ± 0.9 (P = 0.676; Figure 3).

Expression ratio of Bax in the heart, liver and kidney tissue after 12 h of sepsis compared to sham-treated animals and normalized to β-actin. Bax was non-significantly elevated in all investigated tissues. Black column = sepsis group (sepsis without treatment); white column = rhAPC-sepsis group (sepsis with treatment of 24 µg/kg/h rhAPC). The data are reported as mean ± SEM.
Bcl-2
In heart tissue, we found Bcl-2 expression of 1.3 ± 0.8-fold in the sepsis group (P = 0.699) and 1.1 ± 0.9-fold in the rhAPC-sepsis group (P = 0.942; Figure 4). Bcl-2 was expressed in the liver by 1.4 ± 0.9-fold in the sepsis group (P = 0.544) and 4.1 ± 3.8-fold in the rhAPC-sepsis group (P = 0.178; Figure 4). In the kidney, Bcl-2 expression was 0.8 ± 0.4 in the sepsis group (P = 0.668) and 0.8 ± 0.5 in the rhAPC-sepsis group (P = 0.821; Figure 4).
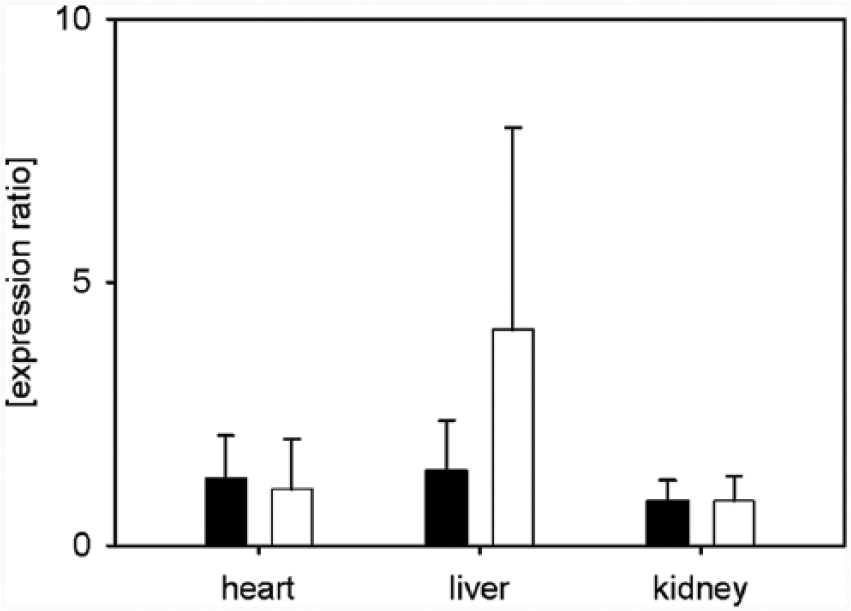
Expression ratio of Bcl-2 in the heart, liver and kidney tissue after 12 h of sepsis compared to sham-treated animals and normalized to β-actin. Bcl-2 was elevated in all tissues, although the increase was not significant in the liver tissue of the rhAPC-sepsis animals. Black column = sepsis group (sepsis without treatment); white column = rhAPC-sepsis group (sepsis with treatment of 24 µg/kg/h rhAPC). The data are reported as mean ± SEM.
MIP2
Cardiac MIP2 was increased by 16.6 ± 14.6-fold in the sepsis group (P = 0.029) and by 7.0 ± 7.8-fold in the rhAPC-sepsis group (P = 0.128; Figure 6). MIP2 was up-regulated in the liver by 39.2 ± 33.5-fold in the sepsis group (P = 0.007) and by 57.7 ± 60.5-fold in the rhAPC-sepsis group (P = 0.008; Figure 5). In kidney tissue, MIP2 was up-regulated by 8.0 ± 5.7-fold in the sepsis group (P = 0.040) and by 7.5 ± 5.1-fold in the rhAPC-sepsis group (P = 0.036; Figure 5).

Expression ratio of MIP2 in the heart, liver and kidney tissue after 12 h of sepsis compared to sham-treated animals and normalized to β-actin. The rhAPC therapy resulted in non-significantly decreased up-regulation in the heart, whereas the liver and kidney tissues were unaffected. Black column = sepsis group (sepsis without treatment); white column = rhAPC-sepsis group (sepsis with treatment of 24 µg/kg/h rhAPC). The data are reported as mean ± SEM. *P < 0.05; †P < 0.01.
CD14
The expression of heart tissue–derived CD14 was 37.3 ± 27.0-fold higher in the sepsis group compared to the sham group (P = 0.007). CLP in the rhAPC-sepsis group resulted in 24.7 ± 23.5-fold higher gene expression (P = 0.007; Figure 6). The up-regulation of CD14 in liver tissue was 111.1 ± 80.6 in the sepsis group compared to the sham-treated group (P = 0.006). In the rhAPC-sepsis group, the expression was 43.8 ± 41.6-fold higher (P = 0.004; Figure 6). Kidney-derived CD14 was up-regulated 6.7 ± 3.3-fold in the sepsis group compared to the sham group (P = 0.015). In the rhAPC-sepsis group, there was an up-regulation of 2.7 ± 1.7-fold, which was not significant (P = 0.223; Figure 6).

Expression ratio of CD14 in the heart, liver and kidney tissue after 12 h of sepsis compared to sham-treated animals and normalized to β-actin. Up-regulation of CD14 was attenuated by rhAPC therapy in all tissues but was more pronounced in the liver and kidney. In the kidney tissue, rhAPC administration resulted in non-significant regulation of CD14 expression. Black column = sepsis group (sepsis without treatment); white column = rhAPC-sepsis group (sepsis with treatment of 24 µg/kg/h rhAPC). The data are reported as mean ± SEM. *P < 0.05; †P < 0.01.
Discussion
In a model of early murine sepsis, we demonstrate organ-specific effects of rhAPC, especially in the heart, resulting in reduced pro-inflammatory gene expression at the mRNA level, less activation of caspase-3 and a reduction in the loss of EPCR.
Variations in responses to sepsis stimuli in different organs may reflect the possible heritable susceptibility to infectious diseases. Our findings regarding organ-specific inflammation are in line with demonstrated differential expression of pro-inflammatory proteins released by microparticles depending on the target tissue. 20
The early diagnosis of sepsis and therapy lead to significantly reduced mortality rates.21,22 Moreover, early administration of rhAPC within 24 h of sepsis results in superior survival rates compared with delayed initiation of therapy. 23 APC decreases pro-inflammatory effects and apoptosis, thereby modulating cell survival, which has been demonstrated in cell culture. 24 Furthermore, IL-6 was significantly reduced in rhAPC-treated patients. 25
rhAPC resulted in beneficial effects in treated mice compared to untreated sepsis mice with respect to physical activity (Table 1). Accordingly, this higher physical activity was reflected by anti-inflammatory effects of rhAPC infusion on plasma levels of IL-6, TNF-α and MCP-1, but not IL-12p70 (Table 2), similar to those observed in humans. 6 These effects were measured, although we used human APC instead of mouse-derived APC, resulting in anti-inflammatory effects rather than anti-coagulation effects for similar doses in mice. Therefore, we may have detected restricted results comparable to those observed when genetic variants of activated protein are used. 12 The lack of IL-12p70 up-regulation in our sepsis group compared to the rhAPC-sepsis group may be explained by the fact that TNF suppresses IL-12 production. 26 In addition, the ability of MCP-1 to suppress IL-12 production by macrophages may have contributed to this finding. 27
Murine (m) EPCRs are analogous to human (h) EPCRs with respect to ligand binding, and hAPC binds to mEPCRs with comparable affinity as to hEPCRs. 28 Cytoprotective effects (e.g. down-regulation of inflammatory mediators) are mediated by the EPCR. 29 In particular, cytoprotection is mediated by PAR-1 through its interaction with ePCR. 30 A certain pattern of ePCR distribution, with microvascular staining in most organs with higher levels, for example, in the liver and heart, could explain the differences in anti-inflammatory effects.
Gu et al. 31 demonstrated a rise in soluble ePCRs in serum, with a peak after 6 h accompanied by increased shedding of ePCRs upon exposure to endotoxin and thrombin and up-regulated ePCR mRNA. This shedding was mirrored by a decrease in ePCR-positive cells in the murine heart, liver and kidney in our sepsis group, although we only observed significantly higher levels of anti-ePCR-positive cells in heart after rhAPC treatment compared to the sepsis group (Figure 2). This finding is in line with the observed prevention of ePCR down-regulation by blocking nuclear factor (NF)-κB and the detection of organ-specific differences in ePCR down-regulation. 32
The decreased lower systemic inflammation in rhAPC-treated animals may have been due to a higher content of ePCRs contributing to anti-inflammatory activity in the investigated organs, 33 although the sepsis focus in our model was comparable in both the sepsis and the rhAPC-sepsis groups. Furthermore, the binding of rhAPC to ePCRs activates protease-activated receptors (PARs), 34 which could be related to the prevention of apoptosis by rhAPC.
There are two mechanisms by which apoptosis is stimulated after activation via the death receptor. The first is activation of caspase-3 directly by activation of caspase-8. The second involves members of the Bcl-2 family as an amplification loop to activate effector caspases. 35 We demonstrate increased cleaved caspase-3-positive cells in all investigated organs and a decrease upon treatment with rhAPC in the heart and kidney but not in the liver. Other authors obtained comparable results in wild-type mice for cleaved caspase-3 in heart sections, with a peak 8 h after lipopolysaccharide (LPS) challenge. 36 It was demonstrated in rats that, during experimental sepsis, rhAPC also improves cardiac dysfunction by down-regulating nitric oxide synthase pathways. 37
The measurements of cleaved caspase-3 first appeared to contradict our results for Bax and Bcl-2 gene expression. However, it was previously demonstrated that peaks in active caspase-3 and Bcl-2 may not be elevated simultaneously in sepsis and septic shock. 38 Because our model reflects the very early phase of sepsis, a possible explanation may be the activation of an amplification loop at later stages. At later stages of sepsis, our measurements may have revealed different results.
Further studies, however, are warranted to investigate the activation of additional (early and late) apoptotic markers to gain a better understanding of the organ-protective effects of rhAPC.
Apoptosis is regulated by two primary pathways, an extrinsic death receptor–mediated pathway and an intrinsic mitochondria-mediated pathway, by members of Bcl-2 families. The pro-apoptotic protein Bax is known to be stimulated by hypoxia, 39 which may occur during bacterial infection at the tissue level. In circulating mononuclear cells of patients suffering from septic shock, the Bax expression ratio was shown to be unaffected by sepsis and therapy with rhAPC, 13 in contrast to an earlier study by the same authors. 40 Although inflammation and apoptosis were investigated in circulating blood cells, data regarding these parameters in organ tissue are rare. In our study, we demonstrated a non-significant increase in Bax expression ratios in the heart and kidney and a decreased expression ratio in liver tissue. Treatment with rhAPC left the expression ratio unaffected, although the decrease in the liver was more pronounced after treatment (Figure 3).
In transgenic mice over-expressing Bcl-2 in T cells, decreased T-lymphocyte apoptosis in thymus and spleen and improved survival during sepsis were demonstrated. 41 The Bcl-2 expression ratios in our study were unaffected for all the investigated organs, although we detected a non-significant increase in the liver tissue upon treatment with rhAPC (Figure 4). This may have been due to an increased number of Bcl-2 family members in single organs, whereas other organs were unaffected. 42
MIP2 is a chemokine that attracts neutrophils and is secreted by various cells after pro-inflammatory stimulation. Murine heart cells synthesize and release MIP2 upon stimulation with pro-inflammatory cytokines. 43 MIP2 expression is induced within a short period after LPS injection or by pro-inflammatory cytokines and peaks after 4 h. There are organ-specific differences with respect to its peak expression; decreased MIP2 after a single shot of LPS and long-lasting increased expression are observed in liver tissue compared to heart tissue. 44 On one hand, the decrease in MIP2 during rhAPC therapy in the heart but not liver or kidney tissue in our study (Figure 6) may be explained by improved recovery of ePCR in rhAPC-treated mice compared to the sepsis group. On the other hand, the decrease in MIP2 may also be explained by the absence of down-regulation, indicating successful systemic anti-inflammatory treatment. MIP2 was elevated in rat kidneys with low levels of protein C 22 h after CLP, whereas normal protein C was accompanied by low MIP2. Furthermore, the receptor for APC (ePCR) was up-regulated in rats with low protein C levels treated with rat-specific APC. 11 Our data provide contradictory results, showing no decrease in MIP2 in animals treated with rhAPC (Figure 6). The differences in MIP2 suppression by rhAPC in mice suggest that species-specific APC has advantages in regulating lymphocyte migration into tissues. One limitation of this study was that we determined only the mRNA levels and not protein levels.
Furthermore, we detected inhibiting effects of rhAPC on the mRNA expression of CD14 in the heart, liver and kidney. The expression levels of CD14 in the heart and liver were up to 18-fold higher than those in kidney tissue, although the decrease due to treatment was more pronounced in the liver and kidney (−60%) than in the heart samples (−35%; Figure 7). These results reflect the decrease in pro-inflammatory plasma cytokines in rhAPC-treated mice, as CD14 inhibition may reduce the up-regulation of cytokines. 45 The induction of pro-inflammatory signalling is not necessarily mediated via CD14 in the heart, but CD14 plays an important role in augmenting inflammation. 46 However, liver microcirculation presented a CD14-independent LPS recognition and subsequent neutrophil attraction. 47 Thus, our results of MIP2 and CD14 are not contradictory, reflecting the anti-inflammatory effects of rhAPC and a CD14-independent attraction of white blood cells.
Variations in responses to the sepsis stimulus in different organ tissues may reflect the possible heritable susceptibility to infectious diseases. There are potential associations among pro-inflammatory cytokine gene polymorphisms, immunity pathway gene polymorphisms and the clinical outcomes of severe sepsis and septic shock. The inflammatory alterations in early sepsis may result in the up-regulation of apoptotic genes in subsequent stages of sepsis.
In our study, we demonstrated that inflammation and apoptosis developed to different extents at certain time points in the heart, liver and kidney tissue during early polymicrobial sepsis.
Organ-specific effects of rhAPC reduce the inflammatory response, especially in the heart tissue compared to the liver and kidney, resulting in reduced pro-inflammatory gene expression at the mRNA level, less activation of caspase-3 and a reduction in the loss of EPCR. These findings are consistent with decreased plasma pro-inflammatory cytokines and increased physical activity.
Thus, early administration of anti-inflammatory agents, such as rhAPC, may contribute to the reversal of sepsis-induced organ dysfunction, especially in the heart.
Footnotes
Acknowledgements
The authors thank Mrs Edith Wagner for performing the PCR.
Declaration of conflicting interests
G.M. received honoraria for consulting and research grants from B. Braun Melsungen GmbH and Adrenomed and lecture fees from B. Braun Melsungen GmbH. T.-P.S. received research grants and honoraria for lecture from B. Braun Melsungen. T.S. received honoraria for board membership from Astellas Pharma GmbH; lecture fees from Astellas Pharma, Bayer Health Care and AstraZeneca; and research grants from Bayer Health Care and Astellas Pharma GmbH. All the other authors declare that they have no competing interests.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
