Abstract
Objective:
To examine the immunohistochemical expression of four members of the type 1 growth factor receptor family in oral squamous cell carcinomas (OSCCs) and to correlate with clinical outcomes.
Materials and methods:
Sixty OSCCs from a patient cohort in Sri Lanka were included in the study. Five sections from each carcinoma were immunostained with antibodies to epidermal growth factor receptor (EGFR)/c-erbB-1, c-erbB-2/HER-2/neu, c-erbB-3/HER-3 and c-erbB-4/HER-4. Two clones were used to stain for c-erbB-2/HER-2/neu. Semiquantitative analysis of immunoreactivity was carried out by scoring the intensity of expression and proportions of positively stained cells. A logistic regression analysis was performed to examine positive expression against overall survival.
Results:
There was heterogenous expression of the four receptors, positivity ranging from 26% to 60%. Co-expression of all four markers was observed only in 1–3% of the tumours. Both membranous and cytoplasmic expressions were observed, EGFR showing predominantly membranous expression and c-erbB-2 showing only cytoplasmic staining. In logistic regression analysis, none of the growth factor receptors were significantly predictive of overall survival.
Conclusion:
Type 1 growth factor receptors are highly expressed in oral carcinomas, EGFR being the predominant marker.
Keywords
Introduction
In the past few decades, many studies have focused on identifying key genes and signalling pathways, which help us to understand the molecular mechanisms of oral carcinogenesis and development of the disease. The key objective of such studies has been to develop diagnostic and prognostic molecular biomarkers for oral squamous cell carcinoma (OSCC). One of the key molecules that has received much attention relates to the epidermal growth factor receptor (EGFR) that belongs to the type 1 family of growth factor receptors. EGFR is a member of this family composed of four different receptors – EGFR/c-erbB-1, c-erbB-2/HER-2/neu, c-erbB-3/HER-3 and c-erbB4/HER-4 – all of which are transmembrane proteins with tyrosine kinase activity. EGFR has an extracellular domain, which provides a ligand-binding site for multiple ligands, EGF, transforming growth factor alpha and amphiregulin which are specific ligands of the EGFR. Few others, such as β-cellulin, heparin-binding EGF and epiregulin, are less specific ligands that bind to EGFR and c-erbB4. Seven growth factors bind to EGFR/HER-1, none bind to c-erbB-2/HER-2, two bind to c-erbB3/HER-3 and seven ligands bind to c-erbB-4/HER-4. 1 Upon ligand fixation, EGFR homodimerization or heterodimerization with another HER receptor occurs, leading to the activation of the intracellular tyrosine kinase. This stimulates kinase signal transduction pathways involved in tumour proliferation, differentiation, cell migration and invasion.
The type 1 family of growth factors and their receptors play an important role in normal development, wound healing and cancer. 2 Role of EGFR and its ligands in the pathogenesis of OSCCs is confirmed by a number of studies that have shown overexpression of this protein in a majority of cancers at high frequency. On average, 50–80% of oral carcinomas have been found to overexpress EGFR. 3 -15 In contrast, studies that have examined the expression of other members of the family are generally more limited.
So far, few studies have investigated HER-2/neu in OSCCs. To the best of our knowledge, a Taiwan study has reported on HER-2/neu expression during the development of oral cancer 16 and more recently by few other authors in OSCC tissues. 17 -19 The roles of HER-3 and HER-4 have not been widely studied.
All four members of the receptor family were investigated by O-charoenrat et al. and Rautava et al. 20,21 A combined study of EGFR, HER-2/neu and HER-3 investigated a series of 111 patients with SCC by Xia et al. 22 Evidence suggests that cooperation of multiple ErbB receptors and cognate ligands is necessary to induce cell transformation. 23 For these reasons, we aimed to investigate all members of the ErbB (HER) tyrosine kinase receptor family in a single cohort of OSCCs.
Materials and methods
Sixty pathologically confirmed OSCCs of the oral cavity were included in the study. The samples were obtained during the course of diagnosis and treatment planning of patients attending two teaching hospitals in Central Sri Lanka.
The detail of the sampling and inclusions/exclusions are already described and published in an earlier publication. 24 Age and sex distributions of patients and anatomical subsites are shown in Table 1. Sample was dichotomized as tongue and other intra-OSCCs with reference to anatomical location, because there was no significant number of samples from other intra-oral sites within the selected cohort to consider them separately.
Characteristics of the sample.a

aFrom Dissanayake (2017).
Antibodies, pretreatments and dilutions used for immunolocalization in paraffin-embedded tissues are shown in Table 2. Five adjacent sections from each tumour were used for immunostaining performed with antibodies to EGFR, c-erbB-2, c-erbB-3 and c-erbB-4, which included two clones to c-erbB-2. Two clones against c-erbB-2 were used, with the aim of identifying the true pattern of expression, because variation in expression has been observed in different types of tissues including OSCCs. Immunostaining was carried out using the ABC staining kit (DAKO) according to the manufacturer’s instructions and recommended reagents with 3,30-diaminobenzidine (DAKO) as chromogen applied for 5 min. Counterstaining was with Harris haematoxylin for 2 min. Specificity of the immunoreaction was monitored by replacing primary antibodies with non-immune sera. Positive and negative control sections were included in each run. The positive controls included an oral epithelial cell carcinoma known to express EGFR (membranous expression) and two adenocarcinomas of the breast known to express c-erbB-2 (membranous expression) and c-erbB-3 and 4 (cytoplasmic expression).
Antibodies, their clones and concentrations used in the present study.

EGFR: epidermal growth factor receptor.
In OSSCs, gradient of intensity of staining was assessed as negative = 0, mild = 1 (+), moderate = 2 (++) and strong = 3 (+++). Tissues with ++ and +++ immunostaining were considered as demonstrating overexpression.
Semiquantitative analysis of immunoreactivity was jointly carried out by two observers blinded to clinicopathological characteristics, and for statistical purposes each case was classified as negative (0; 0–5% positivity) or positive (1; >5% of positivity).
Review of literature revealed, these scores used in the present study were the most commonly used, comparatively reliable and less expensive, evaluating criteria that have been employed to assess the overexpression of type 1 growth factor receptor proteins.
The associations between categorical variables were evaluated by
Results
The study group included 60 patients: four-fifth of the patients (
At the time of last follow-up examination, 25/60 (41.6%) patients were alive and did not show any evidence of recurrent disease. There were nine (15%) patients with residual disease who were under further treatment. Twenty-four (40%) of 60 had died due to the disease within this follow-up period and 2 patients had died due to other causes and 1 of them had residual disease at the time of death.
Epidermal growth factor receptor
The positive control used showed strong EGFR membranous positivity. The normal oral mucosal samples showed membranous pattern of expression with very low intensity. The distribution of the staining was mostly limited to the basal cell layer with few suprabasal layers stained in normal epithelium. In minor salivary glands, acinar cells and scattered ductal cells in the lamina propria also displayed immunoreactivity.
Expression of EGFR protein was found in 57% (34/60) OSCCs with monoclonal antibody EGFR 113. These 34 (57%) tumours showed EGFR immunoreactivity in more than 5% of neoplastic cells. Rest of the cases were EGFR negative or scored less than 5% of cells as positive (
c-erbB-2
Breast cancer positive controls showed strong membranous expression with little cytoplasmic positivity. The normal mucosal samples also showed cytoplasmic expression with mild intensity. Adjacent normal salivary tissues in tumour sections also showed moderate to strong cytoplasmic positivity. c-erbB-2 expression was strictly cytoplasmic with both clones used in the study sample and no membranous staining was found. The expression was limited to few suprabasal layers; however, basal layer was also positive in some tumours.
Staining with the two antibodies (CB11 and 3B5) demonstrated negative expression in 15 and 24% tumours (respectively), while 58 and 45% had positive expression in comparison with normal tissues. c-erbB-2 was strongly overexpressed (++, +++) in 16 (26.7%) tumours with monoclonal antibody CB and in 19 (31.7%) with clone 3B5.
C-erbB-3 and c-erbB-4
The breast cancer positive control samples used showed strong cytoplasmic expression. The normal mucosal samples showed cytoplasmic expression with mild intensity. The staining in the normal control epithelia extended to both suprabasal layers as well as basal layer. Salivary acini in the mucosal tissue adjacent to the tumours showed mild to moderate cytoplasmic expression. The expression/overexpression of c-erbB-3 and c-erbB-4 in the oral carcinomas was also cytoplasmic except six tumours which showed a mixed membrane/cytoplasmic staining for both markers.
Five cancers (8.4%) were c-erbB-3 negative and had no immune staining in the neoplastic cells; 15/60 (25%) tumours had expression similar to normal epithelium. Forty (66.6%) tumours overexpressed (++, +++) c-erbB-3 protein. Six (10%) tumours of these 40 positive cases showed membrane staining as well.
Three cancers (5%) were c-erbB-4 negative and had no immune staining in the neoplastic cells; 21/60 (35%) tumours had expression similar to normal epithelium. Thirty six (60%) tumours overexpressed (++, +++) c-erbB-4 protein. Six (10%) tumours of these 31 positive cases showed membrane staining as well. Tumours with c-erbB-4 protein overexpression had a tendency to be well differentiated (data not shown).
Table 3 gives the summary of the numbers and percentage of tumours, which were immunopositive and immunonegative for the expression/overexpression of all four growth factor receptor family members when reacted with five different antibodies. Figures 1 to 5 present immunoreactive patterns observed with five different antibodies.
Immunoreactivity of type 1 growth factor receptor family members in 60 oral carcinomas.
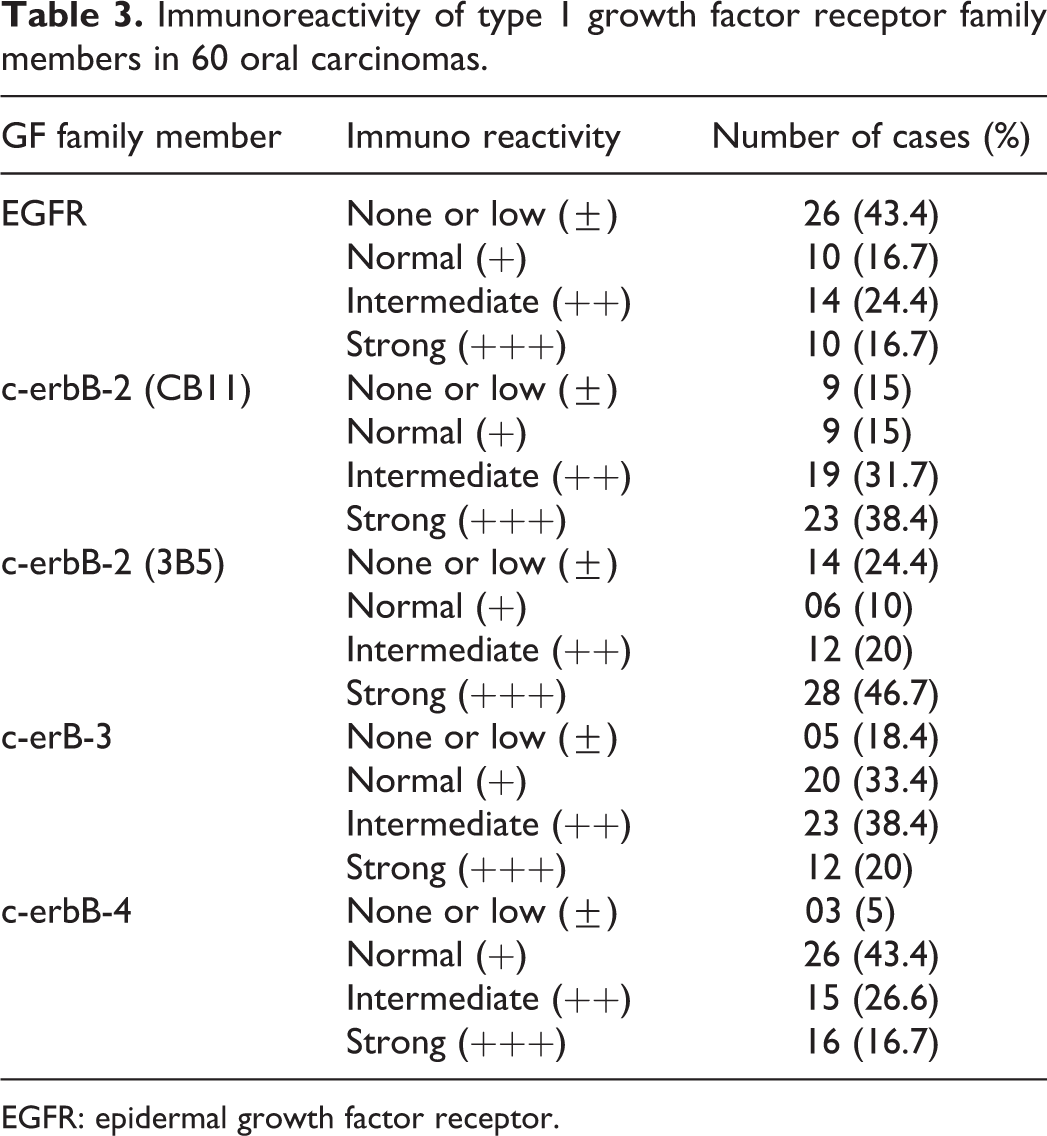
EGFR: epidermal growth factor receptor.

Immunostained section of an OSCC of the study sample. Overexpression of EGFR (with monoclonal antibody EGFR 113): membranous pattern (×200). OSCC: oral squamous cell carcinoma; EGFR: epidermal growth factor receptor.

Strong cytoplasmic staining of a section of OSCC in the study sample which overexpressed c-erbB-2 protein with antibody NCL CB11 (×100). OSCC: oral squamous cell carcinoma.

Strong cytoplasmic staining of a section of OSCC in the study sample which overexpresses c-erbB-2 protein with antibody 3B 5 (×100). OSCC: oral squamous cell carcinoma.

Overexpression of c-erbB-3 oncoprotein in tumour islands of the OSCC (mostly cytoplasmic) with monoclonal antibody RTJ-1 (×100). OSCC: oral squamous cell carcinoma.

Overexpression of c-erbB-4 oncoprotein in tumour islands of the OSCC (mostly cytoplasmic) with monoclonal antibody HRF-1 (×100). OSCC: oral squamous cell carcinoma.
Co-expression of all four markers was observed only in 1.6% of tumours, when CB11 monoclonal antibody was used to assess c-erbB-2 protein, whereas it was 3% with monoclonal antibody 3B5.
Table 4 gives the details about overexpression of each of the other markers in relation to EGFR overexpression. Two tumours of 60 overexpressed both EGFR and c-erbB-2 assessed with monoclonal antibody CB11. Both these markers were overexpressed by nine tumours when c-erbB-2 was assessed using monoclonal antibody 3B5. Simultaneous overexpression of EGFR and c-erbB-3 was observed in 13 of 60 tumours. Both EGFR and c-erbB-4 were overexpressed in 10 of 60 tumours.
Expression/overexpression of EGFR in relation to expression/overexpression of c-erbB-2, c-erbB-3 and c-erbB-4.a

EGFR: epidermal growth factor receptor.
aLevel of significant; with reference to each marker
No significant relationships were found between the EGFR score and clinicopathological factors. Overexpression of EGFR, c-erbB-2 CB11, c-erbB-2 3B5, c-erbB-3 and c-erbB-4 showed no significant prognostic value in terms of survival.
Discussion
The significance of EGFR protein in tumorogenesis came into much consideration after Cohen and Merlino et al. 25,26 Their in vitro studies demonstrated abnormally high levels of EGFR in A431 cell line established from a mucocutaneous carcinoma of the vulva −10 to 50 times more when compared with other tumour cell lines. Early studies confirmed that the expression of EGFR is a common phenomenon in most of the cell types but most abundantly in epithelial cells. 27
Our interest in examining EGFR expression in SCCs relates to translation of these findings to direct molecular-targeted therapy. There are several ways of blocking EGFR biological activities in head and neck cancer: either acting intracellulary by inhibiting EGFR phosphorylation or by targeting the extracellular part of the receptor and blocking dimerization and thus activation. The inhibition of phophorelation is targeted by the so-called small molecules such as gefitinib, erlotinib and lapatinib; the blocking of the extra cellular domain of the receptor is dealt by monoclonal antibodies such as cetuximab, panitumumab, matuzumab and nimotuzumab. 28
Immunohistochemical expression and overexpression of EGFR in oral and head and neck cancers have been shown in several key studies mentioned earlier. An Indian study on multiple molecular markers reported that EGFR was the most frequently expressed (150/178 or 84%) biomarker of their cases.
13
Some authors also report on the expression pEGFR – the active molecule.
29,30
Latter authors found that more than one-third (41%) of the cases expressed the activated form of this protein.
30
In some reported studies, the immune histochemical results are validated by EGFR gene copy number studies. These studies illustrate that EGFR plays an important role in oral and head and neck cancer development. Nakata et al demonstrated that EGFR copy number status is a more reliable indicator than EGFR protein overexpression in tongue cancers and that the copy number increase is not correlated with EGFR protein levels.
31
However, Ryott et al. reported that EGFR gene copy number was significantly associated with EGFR protein expression (
In the present study, we observed that 57% of OSCC expressed EGFR, that is, in line with other authors. Although most of the studies had reported the EGFR positive immunoexpression in the cell membrane, co-expression of EGFR both in the cell membrane and in the cytoplasm of the tumour cells has been reported by Monteiro et al. (2012). 10 We did not observe any cytoplasmic expression of EGFR in the present sample. However, presence of both membranous and cytoplasmic expression has been shown to affect the prognosis of oral cancer patients 10 and in other cancers. 35
Although some of the studies have correlated this marker with clinical and pathological variables, 4 -6,8,36 -40 our data are in agreement with reports of other authors that concluded that the study of EGFR expression does not give any additional information on the clinicopathological status of patients with oral cancer. 12,41,42 This may be because of the participation of this marker in both the early stages and more advanced cases of HNSCC. Moreover, some studies have identified a statistically significant correlation between ‘high’ EGFR expression with certain pathologic factorseg; tumour invasion. 29 This was a limitation in our study that we did not separately analyse high expression groups (++, +++) against tumour factors.
The influence of EGFR expression on poor prognosis of oral cancers has been highlighted in several studies. 3,5,6,22,43 Contradictory results with no association was reported by Monteiro et al., Solomon et al., Jonsson et al. and Galvis et al., though a trend was found in the latter study. 10,13 -15 In contrast to this, Maiorano et al. showed that the overexpression of EGFR is associated with a prolonged survival. 44 These contradictory results are discussed in a literature review that found five studies reporting EGFR expression having an influence on survival while two studies with negative findings. 45
Few studies have compared the utility of pEGFR against EGFR expression as prognostic markers. 30,46 In multivariable analysis, Monteiro et al. reported that only pEGFR expression (and tumour stage) proved to be of independent prognostic value on survival. 30 They concluded that pEGFR has more prognostic value than EGFR expression of a tumour. No correlation has been found between high EGFR expression and pEGFR expression levels 29 and for this reason it would be prudent to estimate pEGFR expression in future studies.
HER-2 gene is amplified or overexpressed in 10–34% of breast cancers and historically, HER-2-amplified metastatic breast cancer is an aggressive cancer. 47 In oral cancers, some studies reported the expression of c-erbB-2 as membranous 48 and the others as mixed, cytoplasmic and membranous 22 or even as cytoplasmic (only). The studies that examined HER-2 in OSCCs have reported divergent results with reference to pattern of expression. While cytoplasmic staining comparable to normal tissue was found in all but one tumour, 19 the authors found no overexpression of HER-2/neu in head and neck cancers (HNSCC). HER-2/neu overexpression was not identified in any of the cases examined by BShintani et al. 49 A further study reported HER-2 expression and gene amplification are rarely found in HNSCC and the subset of OSCC. 17 In 11 of 97 biopsies (11.3%), membranous overexpression (score 2+ and 3+) of HER-2/neu was shown by immunohistochemistry and the authors found no correlation between survival and HER-2/neu overexpression. 18 However, our data indicate the overexpression of HER-2 in oral cancers falls within the range of breast cancers (10–34%) irrespective of the antibody used. These data imply that HER-2 overexpression may have a role to play in oral cancer in a way similar to breast cancers. There is little published data on c-erbB-3 and c-erbB-4 alone on oral cancer to make any comparisons with our data.
Previous authors have investigated all type 1 growth factor receptors as we did in our study. Their observations suggested that the expression of all EGFR members was significantly associated with shortened patient survival, and the association was strongest for HER-2/neu overexpression. 22 However, only 47 of 111 cases entered to the study had all clinical data. In a further study conducted among the four family members, EGFR appears to be an independent prognostic marker. 20 Co-expression of all four markers was found to be limited in our study 1–3%. Overexpression of the four markers demonstrated a heterogeneous pattern and we did not find any correlation with expression patterns. However, co-expression of more than a single c-erbB receptor may play a significant role in the pathogenesis of HNSCC. Studies involving whole genome sequencing in head and neck cancer have stratified head and neck cancer to several subtypes based on high or low expression of EGFR in addition to few other markers. 50
One of the important clinical applications of assessing EGFR status of a tumour is the possibility of using it as a target for anticancer molecular therapy for tumours with EGFR expression. Numerous strategies for inhibiting EGFR are in current use, which include monoclonal antibodies (e.g. cetuximab) and tyrosine kinase inhibitors (e.g. gefitinib, erlotinib), which may be used in combination with other therapies. 51 With biologically guided tumour characterization, personalized treatment protocols can be designed for individual patients
Conclusion
A heterogeneous pattern of expression of the family of type 1 growth factor receptors is reported in the present study. These data suggest that when planning molecular-targeted therapy for OSCC, it is necessary to investigate the tumours for each member of the EGF receptor family. Further clinical trials are needed to test whether in addition to EGFR other members of the family may also serve as candidates for molecular-targeted therapies.
Footnotes
Acknowledgements
The author wish to thank Prof. Newell W. Johnson and Prof. Saman Warnakulasuriya for their encouragement and supervision of my laboratory studies. Prof. W. J. Gullick (University of Kent) kindly gifted two antibodies (CB11 and 3B5) for this study. Dr Derek Cooper PhD helped the author with statistical analysis of data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
