Abstract
Objective:
Currently, oral squamous cell carcinoma (OSCC) is one of the most fatal cancers of all head and neck malignancies. Despite advancements in therapy, the mortality and morbidity remain high. Hence, it is essential to identify useful prognostic markers for high-risk individuals with OSCC to decide on treatment protocols. Centrosomal protein 55 (
Materials and Methods:
Forty pathologically diagnosed cases of OSCC were included in the study: 20 each of early and advanced OSCC cases. Formalin-fixed paraffin-embedded archival samples were used. The sections were immunohistochemically stained with Cep55 antibody. The expression levels of Cep55 were correlated with clinical parameters and disease outcome.
Results:
A higher expression of Cep55 was observed in advanced stage compared to early stage of OSCC. The Cep55 expression showed no significant relation with respect to clinical staging, pathological grading and site, except for tongue. Cep55 overexpression is significantly associated with poor survival.
Conclusion:
The present study suggests that Cep55 could play an important role in determining the biological behaviour and survival of OSCC patients independent of tumour staging and pathological grading. Thus, assessment of Cep55 expression could navigate the surgeons to plan an appropriate treatment.
Introduction
Oral squamous cell carcinoma (OSCC) is the third most common cancer in India, 1,2 and the major risk factors are widely known. 3 –5 Even with the provision of advanced treatment modalities, the morbidity and mortality rates still remain high in India. OSCC patients have high potential for invasiveness through regional lymph node metastasis and recurrence. 6 –8 Exact reasons for recurrence and metastasis are yet unclear. Several factors may contribute to treatment failure, and our primary interest relates to understanding the biological behaviour of OSCC at molecular and cellular levels.
OSCC patients with similar clinical stages and/or pathological grades can often have different clinical outcomes. Tumor, Nodal and Metastasis (TNM) staging and pathology grading are insufficient to accurately predict prognosis and survival. Thus, it is important to investigate the biomarkers that are able to predict the biological behaviour and to assess their utility for determining the probability of survival of OSCC patients.
Centrosomal protein of 55 kDa (Cep55) is a protein that in humans is encoded by the
Cep55 is initially located at the centrosome in interphase but in anaphase of mitosis, Cep55 moves to the central spindle part of the cell. Later, it is recruited to the mid-body for cytokinesis for the regulation of normal cell division. If there is premature or prior recruitment of Cep55 at the mid-body, then the cell fails to divide into two daughter cells, resulting in cells with impaired mid-body release, which are more likely to undergo differentiation. Thus, appropriate localization of Cep55 to the mid-body is crucial to determine the fate of a daughter cell. 10,16,18 –21
Various studies have shown overexpression of Cep55 in lung, ovarian, breast, urinary bladder, colon cancers and in gastric carcinoma, hypothesizing its role as an oncoprotein, and studies have correlated the expression levels of Cep55 with stage of disease, metastasis and angiogenesis. 22 –29
However, the role of Cep55 in assessing the biological behaviour of OSCC has been less explored. Our aim was to correlate the expression levels of Cep55 in OSCC patients with the clinical and pathological parameters to predict survival.
Materials and methods
A retrospective study was undertaken on 40 cases of primary OSCC with complete clinical and follow-up records for the period of November 2014 to January 2017. The cases were grouped by the T stage as group 1 (T1 and T2 as early OSCC) and group 2 (T3 and T4 as advanced OSCC) based on the tumour size. All cases were confirmed histopathologically using World Health Organization (WHO) criteria, and clinical data were collected from the Department of Maxillofacial Surgery, Faculty of Dental Sciences (FDS), Ramaiah University of Applied Sciences (RUAS). The formalin-fixed paraffin-embedded tissue blocks were retrieved from the archives of the Department of Oral Pathology. The cases who had recurrence of OSCC and undergoing preoperative radiotherapy/chemotherapy were excluded from the study. The inclusion criteria were pathological confirmation of OSCC, availability of clinicopathological information and informed consent by the subjects. The study protocol was reviewed and approved by the Ethics Committee of FDS, RUAS (FDS/EC/2014-16/PG ST/28).
Immunohistochemistry
The standardization of Cep55 antibody for immunostaining was performed by diluting the antibody in phosphate-buffered saline (PBS) in graded concentrations of positive control tissue of breast carcinoma 30 as suggested by the manufacturer (Figure 1(a) and (b)).

Photomicrographs of breast cancers and different grades of OSCC under ×400 magnification in routine haematoxylin and eosin stain and immunoexpression with Cep55 marker. Positive control – breast carcinoma for Cep55 expression (a) and (b). Early stage: (c) and (e) haematoxylin and eosin–stained sections; (d) and (f) immunohistochemical expression of Cep55. Advanced stage: (G) and (I) haematoxylin and eosin–stained sections; (h) and (j) immunohistochemical expression of Cep55. OSCC: oral squamous cell carcinoma; Cep55: centrosomal protein 55.
A standard immunohistochemical staining protocol was performed. Tissue sections of 4 µm thickness were deparaffinized in xylene and rehydrated in graded alcohol. Antigen retrieval was performed with 10 mM of Ethylenediaminetetraacetic acid-8 (EDTA-8), by treating the sections at 90–95°C for 15 min in a pressure cooker and then cooling to room temperature naturally. The slides were rinsed with PBS thrice at an interval of 5 min. The sections were incubated with the blocking solution of 3% bovine serum albumin at 37°C for 1 h to block the non-specific antigens, followed by overnight incubation at 4° C with 1:400 anti Cep55 primary mouse monoclonal antibody (Abcam, Kolkata, India). The secondary rabbit polyclonal antibody (Mach-1; Biocare, Bengaluru, India) was conjugated by incubating at 37° C for 1 h and then rinsing it with PBS thrice for 10 min. The sections were treated with 3,3’-diaminobenzidine tetrahydrochloride (DAB) chromogen and counterstained with haematoxylin.
The immunohistochemical expression of Cep55 in stained sections was examined, and cytoplasmic staining was considered as positive for Cep55 expression. The intensity of the stain was graded (Table 1) according to the criteria given by Jiang et al. 22 Finally, the immunohistochemical score (IHS) was calculated as the product of the percentage of cells stained and their staining intensity. The Cep55 expression was evaluated from five randomly selected high-power fields for each section and was assessed for intensity and staining distribution by two observers to reduce the bias. The mean scores given by the two observers were used. The resultant value of IHS ≥4 was considered as positive and <4 as negative. 22
Scoring criteria used in reporting Cep55 expression.

Cep55: centrosomal protein 55.
Statistical analysis
Data analysis was performed using SPSS version 22. Correlations of Cep55 expression with clinical and pathological parameters were analyzed using the
The expression of Cep55 was classified as high at ≥4 and low at <4 using the Kaplan–Meier method. The cases with incomplete data were excluded. All statistical analyses were considered significant when the
Results
Initially 40 cases were considered; however, due to inadequate tissue, staining errors and nonspecific staining, eight cases were excluded from interpretation. Therefore, the data from 32 patients used in the statistical analysis are presented here. Of the 32 cases, 25 (78%) showed positive Cep55 expression, while 7 cases (22%) showed negative Cep55 expression (Table 2).
The distribution of
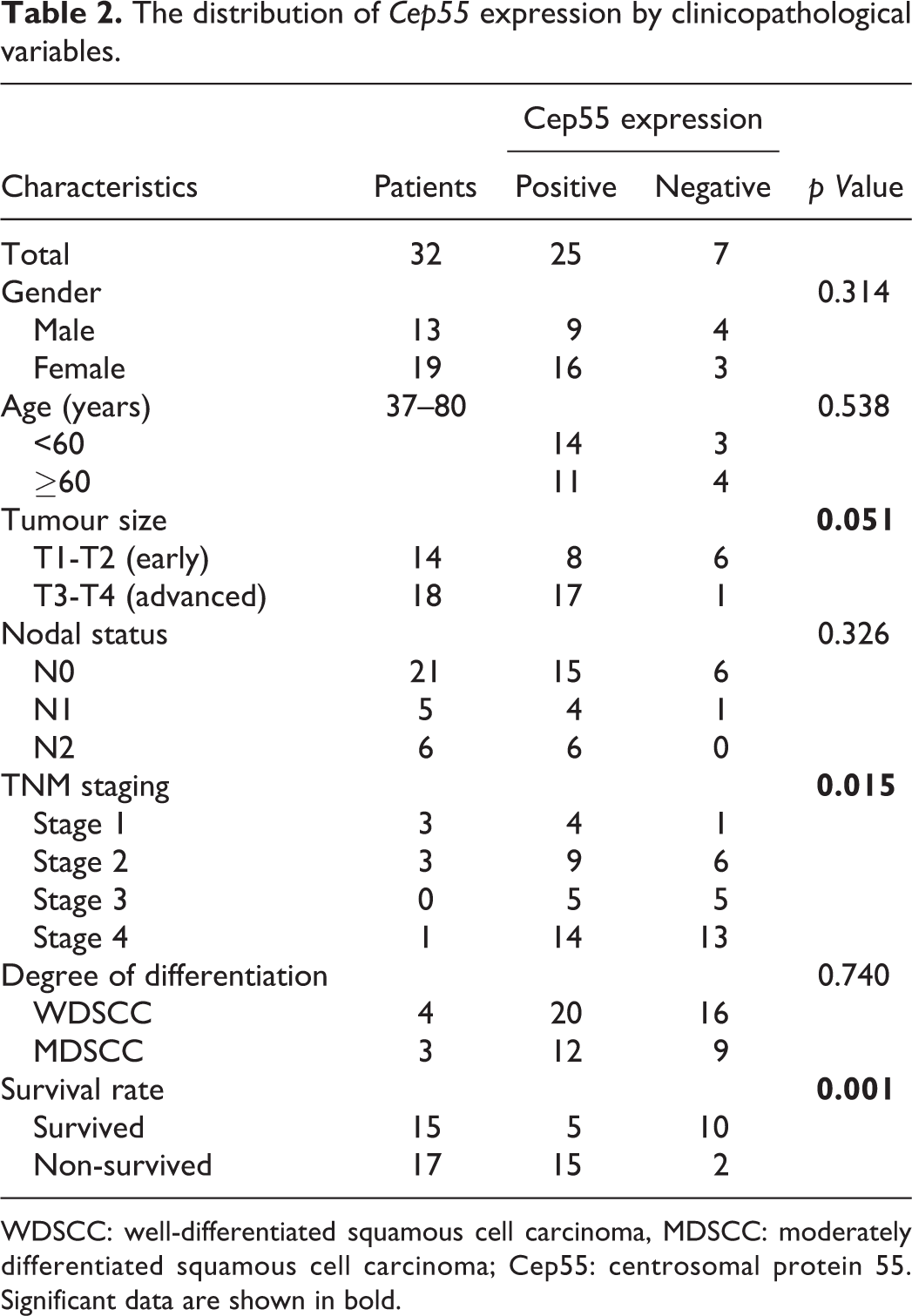
WDSCC: well-differentiated squamous cell carcinoma, MDSCC: moderately differentiated squamous cell carcinoma; Cep55: centrosomal protein 55. Significant data are shown in bold.
Correlation of Cep55 expression with clinicopathological parameters
Tumour site
A significant difference was found in the expression of Cep55 in tongue cancers compared with other sites (
Early versus advanced
We noted a statistically significant result (

Graphs illustrating the distribution of Cep55 expression in early versus advanced OSCC (a), by nodal status (b), by tumour stage (c), by degree of differentiation (d) and with respect to patient survival (e). OSCC: oral squamous cell carcinoma; Cep55: centrosomal protein 55.
Nodal status
Higher Cep55 expression was found in 15 cases (46%) of N0 compared with 4 (13%) and 5 (16%) cases of N1 and N2, respectively. These differences were statistically insignificant (
Clinical staging
Cep55 expression was higher in stage 4 (13/14 cases) followed by stage 3 (5/5 cases), stage 2 (5/9 cases) and stage 1 (1/4 case). An increased expression was observed as the tumour stage progressed with a statistically significant
Tumour grading
There were no statistically significant differences in Cep55 expression and histological degree of differentiation (
Survival analysis
By correlating the survival of the OSCC patients with Cep55 expression, it was observed that the expression levels were directly proportional to the survival rate. Of the 32 patients, 20 (63%) showed positive Cep55 expression and 12 (27%) showed negative expression. Among these 20 positively immunoexpressed patients, 15 (75%) died within 3 years of the follow-up, while 5 (25%) survived. Of the remaining 12 patients who showed negative Cep55 immunoexpression, 2 (16.6%) of them died and 10 (83.3%) subjects survived. These findings were statistically significant (
The Kaplan–Meier curves show that the survival of the OSCC patients was site-specific: Patients with tongue OSCC had a shortened survival period of <30 months (Figure 3), with a drop in the survival rate to 40% compared to other sites (gingivobuccal sulcus, retromolar trigone and buccal mucosa (BM)). Patients with cancers of BM had a better survival rate of 60–100% with >50 months of survival.

Kaplan–Meier curve depicting the patient survival rate in relation to tumour site.
OSCC patients with >4 immunohistochemical score had close to 20% survival rate by 24 months, as most of the cases with overexpression of Cep5 in our study died within a short span of <24 months (Figure 4).

Kaplan–Meier curve depicting the patient survival rate in correlation with the IHS of ≥4
According to the multivariate analysis of 32 OSCC cases, tumour sites, mainly tongue (
Multivariate analysis of clinicopathological parameters.

SE: standard error; CI: confidence interval.
Discussion
The biomarker Cep55 has recently been explored through cellular as well as molecular levels to study its role in the process of oncogenesis. 16
Cep55 is a centrosomal protein that controls the cell cycle during the mitosis phase mainly acting on the PI3K/Akt pathway. Any aberrant mutation of this gene (leading to overexpression) results in the dysregulation of the PI3K/Akt pathway. 16 Thus, Cep55 has been considered as an oncogene. The action of Cep55 through the PI3K/Akt pathway is in conjugation with VEGF, p16, p53, p85 and p110 by increasing the stability of the subunit of the PI3K/Akt complex, resulting in an increased activity of Akt for cell survival, cell growth and angiogenesis. 16,31
The role of Cep55 as a tumour maker has been studied in several human cancer types, for example, colon, lung, breast and ovarian cancers. 22,23,25,27,30 The expression levels of Cep55 have been correlated with varying parameters, for example, clinical staging, prognosis and metastasis. However, there is a dearth of similar studies on assessing the biological behaviour of OSCC. Hence, our aim was to compare the expression levels of Cep55 with various clinical and histological parameters in OSCC and survival.
Our study findings of Cep55 expression on the early (T1 and T2) and advanced (T3 and T4) OSCC tumours and their correlation with patient survival were in accordance with Chen et al. 7 on OSCC samples and Jiang et al. 22 on pulmonary adenocarcinoma. The data suggesting its role during tumour advancement in OSCC patients are given in Table 2.
Literature suggests that the higher expression of Cep55 might result from the cellular transformation of a normal cell to a tumour cell by the activation of the PI3K/Akt pathway, resulting in tumour cell proliferation. 32
Chen et al. 7 have suggested that the Cep55/FLJ10540 has a direct role in stimulating Matrix metalloproteinase (MMP2) and Forkhead box protein M1 (FOXM1) for tumour cell migration and metastasis, resulting in poor prognosis and decreased survival rate in patients with oral cavity cancer.
The current study did not indicate that high Cep55 expression was associated with advanced nodal disease. These observations corresponded with the data of Tao et al. who studied the nodal status (N0, N1 and N2) in cases of gastric carcinoma. 28 However, studies conducted by Jiang et al. 9 on oesophageal carcinoma and Chen et al. 33 on nasopharyngeal carcinoma showed contrasting results, indicating that high Cep55 expression was associated with poor prognosis in locally advanced cases.
The current study showed greater Cep55 expression in stage IV followed by stages III, II and I, with a statistically significant
There were statistically insignificant differences between Cep55 expression and pathological grading with increased expression noted in well-differentiated squamous cell carcinoma (WDSCC) compared to moderately differentiated squamous cell carcinoma (Table 2). It is known that histological grading and aggressive behaviour of OSCC are independent assessors. 34 So, this could be one of the reasons for higher Cep55 expression in WDSCC, albeit with aggressive behaviour. Other than the above-mentioned reason, subjective errors in histological grading and smaller sample size can also lead to insignificant results. Our results were in accordance with the results of Chen et al. who evaluated the histological grading in cases of OSCC and stated that it is an independent indicator of prognosis. 7 However, the contrasting results were published by Jiang et al. 9 on histological grading of oesophageal carcinoma and Tao et al. 28 on gastric carcinoma with poor prognosis.
Previous studies by various authors (Jiang et al., Wang et al. and Tao et al.) have stated that Cep55 has a definitive role in the development and progression of other cancers through cell cycle regulators and proteins controlling the PI3K/Akt pathway. They have shown that overexpression of Cep55 results in the activation of FOXM1, MMP2 and other oncogenes, enhancing the progression of a tumour. 7,22,28,35
In the current study, the Cep55 expression was found to be higher in patients who died of the disease compared to the surviving patients with a statistically significant
In our study, the multivariate analysis and Kaplan–Meier curves showed that the tumour aggressiveness and survival of OSCC patients were dependent on Cep55 immunoexpression, whereas clinical staging and pathological grading were independent survival predictors. Hence, our study results suggest that Cep55 could be a biomarker for the invasion of cancer cell and alter the biological behaviour in OSCC patients, resulting in a shortened survival rate. Therefore, Cep55 expression could be used as an early prognostic biomarker to decide on the plan of treatment to improve the quality of life in patients with OSCC. Limitations of the present study were the small sample size with the uneven distribution of clinical groups.
Conclusions and future directions
From the present study, it is noteworthy to conclude that the biomarker Cep55 would indicate the survival of patients based on the biological behaviour of OSCC, independent of clinical staging and pathological grading. To our knowledge, this is the first study to report on the correlation of Cep55 expression with survival on OSCC. Nevertheless, a study with a larger sample size and advanced molecular studies could help us to understand the exact role of Cep55 in their pathogenesis and predicting the behaviour of OSCC.
Footnotes
Acknowledgements
The authors would like to thank Mr Sridhar and Mrs Ashwini at Oncore Diagnostics, Dr Puneet Gupta statistician and Mr Chetan Kumar for their technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
