Abstract
Objectives:
India has a high incidence of oral cancer due to multifarious tobacco use. The objective of this study was to assess the status of tobacco-related oral lesions over 16 years, in a screen-detected population.
Methods:
This cross-sectional study involved home visits of 2000 Delhi residents, previously screened for oral potentially malignant disorders/oral cancer and counseled for tobacco cessation. Their basic demographics and tobacco/alcohol history were noted followed by oral visual examination for any related mucosal abnormalities. The data thus obtained were statistically analyzed.
Results:
Two hundred and sixty-five individuals (13.2%) could be traced after 16 years. The status of oral lesions varied across the participants, mainly in terms of their location, type, number, and/or presence/absence; no oral malignancies were noted. Most individuals had either a decreased use (34%, p < 0.001) or had quit tobacco (25.7%, p < 0.001); 8.3% individuals from the former and 5.7% from the latter group showed complete lesion(s) regression. The overall change in the tobacco use and oral lesions showed a highly significant positive association (p < 0.05).
Conclusion:
A direct relationship exists between tobacco use and oral lesions. Repeated, tobacco cessation counseling provided by health-care professionals is effective. Oral screening of high-risk individuals, along with tobacco cessation, is thus essential.
Keywords
Introduction
Oral cancer is the 18th most common cancer occurring globally. It is eminently preventable, yet a major public health concern, especially in the Southeast Asia region. The mortality due to oral cancer in Southeast Asia has been reported to be 8542 cases in 2018. 1 Oral cancer incidence is also on a rise in India, which could be attributed to the widespread and multifarious tobacco use. It is the most common cancer among males and overall second most common cancer among both sexes in India. 1 Oral cancer constitutes about 15–20% of all the cancers as reported by surveys conducted in various cancer hospitals located across India. 2
Oral cancer can be detected at its early or potentially malignant stages, making it amenable to secondary prevention. Therefore, screening and early detection can help to markedly reduce the burden of oral cancer. 3 Screening for oral cancer implies looking for oral potentially malignant and cancerous lesions, in the asymptomatic phase. Early detection of oral cancer enables to reduce both the high mortality and morbidity associated with this condition and its treatment, such as severe loss of function, disfigurement, depression, and poor quality of life. 4 Oral visual examination (OVE), as a modality of screening for oral cancer, among the high-risk population residing in regions with high incidence, has proven useful in reducing the overall incidence and mortality rates. 5 Various studies have shown a strong association between the development of several oral mucosal lesions and tobacco consumption. A dose–response relationship between the amount of tobacco product consumed and the development of oral cancer has also been established. 2
With targeted measures toward tobacco control and increased awareness toward oral potentially malignant disorders (OPMDs) and oral cancer screening proving effective over the years, there is need for research focused on the early diagnosis of OPMDs and for identifying the current trends and diversity in tobacco usage, especially in regions showing high consumption. 6 Earlier, attempts have been made through cohort studies to estimate tobacco-related morbidity and mortality through cancer screening via OVE: by Gupta et al. among 36,471 tobacco users over 5 years in the rural areas of Ernakulam (Kerala), Srikakulam (Andhra Pradesh), and Bhavnagar (Gujarat) in India; by Sankaranarayanan et al. among 191,873 individuals in Kerala (India) with four screening rounds over 15 years; and by Chuang et al. involving biennial screening of 2,334,299 tobacco users in Taiwan. 5,7,8 A further cohort study in Mumbai was conducted by Gupta et al. on nearly 52,000 individuals over a period of 5–6 years with a view to assessing tobacco-related mortality in India. 9 A feasibility study conducted in Sri Lanka by Warnakulasuriya et al. in 1980s confirmed the reliability of utilizing primary health-care workers) for oral cancer screening. 10,11 The reproducibility of this model for oral cancer screening was also demonstrated by these authors. 12
The present study was undertaken to audit on the status of the tobacco-related oral lesions (majorly the OPMDs) over a period of 16 years, among a group of previously screened eligible participants (tobacco users), utilizing the simple, cost-effective method of OVE, the most feasible method for such low resource, large-scale population-based screening settings.
Methods
Baseline study/cross-sectional study
Previously, a cross-sectional study was carried out between the years 2002 and 2005 among 2000 individuals from the peri-urban population of New Delhi living within a 15-km radius of the All India Institute of Medical Sciences (AIIMS), New Delhi. The objectives were to analyze the tobacco habits and the prevalence of oral lesions, if any, and to determine the frequency of polymorphism in cancer predisposing genes involved in carcinogen metabolism—cytochrome P450 family 1 subfamily A member 1 (CYP1A1) and glutathione-s-transferase mu 1 (GSTM1), in patients with OPMDs. Face-to-face interviews were done by trained social workers (field workers) by house-to-house visits followed by an OVE. Their work was monitored by frequent visits by a dental surgeon and a general surgeon, who further examined the oral lesions and addressed any problems faced by the field workers. Individuals older than 10 years were included in the study and they were subjected to OVE, the details of which were recorded in a pro forma. A 6-month follow-up of the individuals was carried out for a 3-year period till 2005. Blood samples were collected and biopsies were performed by the dental surgeon or general surgeon (to confirm the clinical diagnosis and to exclude malignancy) from the participants with OPMDs, after obtaining written consent. The tobacco use history of the participants (present and ever users i.e. for 3 or more months) was recorded. For those participants who were diagnosed with OPMDs or oral cancer after biopsy were treated appropriately and followed up regularly at 6-month intervals. Leukoplakia and erythroplakia, greater than 4 × 4 cm, were surgically incised. Individuals with oral lichen planus (OLP) were prescribed multivitamins, advised a balanced diet, along with counseling, yoga, and meditation for stress control. Oral submucous fibrosis (OSMF) lesions were subjected to wedge biopsy along with that in any ulcerated area or growth; trismus was relieved by division of fibrous bands at the palatoglossal arch and pterygomandibular raphe to achieve an inter-incisor distance of 3–4 cm, along with administration of the trismus screw and advising of muscle relaxation exercises. Multivitamins and fibrinolysin combinations were also prescribed in OSMF, along with symptomatic treatment with infrared or ultraviolet rays.
Individuals with a history of tobacco consumption were informed about the ill effects of tobacco and counseled toward the cessation of the same at each visit every 6 months. Participants were also educated about the harmful effects of tobacco and the early signs and symptoms of oral cancer through different information education and communication material like short films (“AapMuskuraiye” developed by the Ministry of Health and Family Welfare) and pamphlets. Dental interventions, if required, were performed like grinding of sharp cusps and replacement of faulty dentures with well-fitting prosthesis. For the participants detected with oral cancer, radiotherapy or surgery was done based on the size (<2 cm diameter) with regular follow-ups every 6 months.
Current study/cross-sectional study
The same population was revisited after 16 years (2016–2017) by a team of dental surgeons and an assistant. Participants were briefed in detail regarding the study, written informed consent was obtained from those willing to participate, and the study was conducted according to the principles by the World Medical Association Declaration of Helsinki (version 2002). Information on basic demographics and detailed personal history (including tobacco and alcohol history) were obtained from the individuals by use of a household form and a tobacco history form (same as used in the baseline and previous screening rounds). Any change in the tobacco status, that is, increase, decrease, no change, quitting, or starting a new habit, was noted. The adverse effects of tobacco and alcohol use were explained to the current users and they were counseled to quit, while those who reported quitting these habits were encouraged not to restart. The oral cavity was subjected to OVE in its entirety and the presence of lesion(s) was noted in a case record form. The various structures of the oral cavity, such as the labial and buccal mucosae, retromolar areas, gingiva or alveolar ridges, vestibular mucosae, tongue (dorsoventral surface, lateral borders), the floor of mouth, and the hard and soft palate, were carefully inspected and palpated. The lesions were assessed for change in their status (location, type, number, and/or presence/absence) from the previous examination conducted more than a decade ago. In case of the presence of a lesion or suspicion of cancer, patients were requested to visit the oral health promotion clinic at Indian Council of Medical Research–National Institute of Cancer Prevention and Research (ICMR-NICPR), Noida, or the outpatient clinic at Centre for Dental Education and Research, AIIMS, New Delhi, for further evaluation and/or management. The frequency of polymorphism in the CYP1A1 and GSTM1 genes was not assessed in this visit.
Approval for the study protocol was obtained from the scientific and ethics review committee of the ICMR-NICPR (project ID: 5/13/3/TF/2001/NCD-III).
Definitions for the clinical diagnosis of oral lesions
Leukoplakia—a white plaque of questionable risk having excluded (other) known diseases or disorders that carry no increased risk for cancer. It has two main clinical types, namely, homogeneous and nonhomogeneous leukoplakia. Homogeneous lesions are uniformly white and flat with shallow surface cracks. Nonhomogeneous varieties include speckled—mixed, white (predominant) and red; nodular—small polypoid outgrowths, rounded red or white excrescences; verrucous—wrinkled or corrugated surface appearance; and proliferative verrucous leukoplakia—multiple, simultaneous lesions. 13
Erythroplakia—a fiery red patch that cannot be characterized clinically or pathologically as any other definable diseases. 13
OSMF—The clinical signs and symptoms depend on the stage of the condition. Stage 1 involves stomatitis with erythematous mucosa, vesicles, ulcers, melanotic pigmentation, and petechiae. Stage 2 includes fibrosis which is the hallmark of the disease. Early lesions show blanching of the oral mucosa with the development of vertical palpable fibrous bands in the buccal mucosa and circular bands around the oral commissure later. These features result in a mottled marble-like appearance of the mucosa. Other features include trismus, reduced mobility and size of the tongue, blanching floor of the mouth and palate, fibrotic and depigmented gingiva, shrunken or everted uvula, sunken cheeks, and so on. Stage 3 may present with additional lesions like leukoplakia, or speech and hearing deficit because of involvement of the tongue and the eustachian tube. 14
OLP—OLP may present with both red and white elements the most common being the reticular type, that is, fine white lines or striae, in a network or annular pattern, called Wickham’s striae. Other clinical varieties are papular (small white dots), plaque-like (homogeneous well-demarcated white plaque), bullous, erythematous/atrophic (homogeneous erythematous area), and ulcerative (fibrin-coated ulcers surrounded by an erythematous zone). The erythematous and ulcerative types are usually symptomatic. However, the reticular striae must be present along with these types to establish a clinical diagnosis of OLP. 15
Oral lichenoid lesion (OLL)—OLLs share the clinical features of OLP; however, the former are usually unilateral and have a topographical association with the causative agent. 16
Smoker’s palate and smoker’s melanosis—The most common oral mucosal clinical effects of smoking are dark brown mucosal pigmentations (smoker’s melanosis) and white leathery lesions of the palate (smoker’s palate). An initial erythema is followed by grayish white palatal mucosa, along with multiple red dots representing orifices of the minor salivary glands. 15
Tobacco pouch keratosis—This is characterized by a thick, corrugated/wrinkled, and white mucosal lesion in the area of tobacco contact, usually in smokeless tobacco (SLT) chewers. 17
Candidiasis—The pseudomembranous type typically presents with loosely attached membranes (pseudomembranes), which leave an inflamed, sometimes bleeding area if removed; the erythematous type presents as an erythematous area with diffuse borders; the chronic plaque type is characterized by a homogenous white plaque. Denture stomatitis is characterized by erythema in the denture-bearing mucosa, with granularity later. Angular cheilitis involves fissuring of the lip commissures, surrounded by erythema. Median rhomboid glossitis is clinically characterized by a central, erythematous area in the posterior part of the tongue dorsum, with sometimes an erythematous lesion in the corresponding palatal mucosa (kissing lesions). 15
Statistical analysis
The collected data were checked and validated for consistency and completeness and entered into MS Excel and exported to SPSS version 21. All the data were analyzed using SPSS version 21 and OpenEpi open-source software. A descriptive analysis was done for categorical variables and results were shown as proportions/percentages and continuous variables were summarized using mean ± standard deviation (SD). The McNemar test was employed to observe the change between past and present tobacco and alcohol habits, and χ 2 and Fisher’s exact tests were employed to determine the association between the oral lesion and tobacco habit status; a p value of <0.05 was considered significant.
Results
Among the 2000 individuals examined in the first visit (between the years 2002 and 2005), only 265 individuals (13.2%) were available during the visit in 2016–2017. The attrition rate of 86.8% was mainly due to participants belonging to the lowest socioeconomic strata, who were unavailable during the home visit due to various reasons such as migration to a different location within or outside the city (for a new job, moving out of rented accommodation), working hours, locked residence, and demolishment of the former residence or death. The mean age (±SD) was 48.7 ± 13.5 years, with 38.5% of individuals between 30 years and 40 years; most of the participants were males (83.8%) and married (97.4%). All the participants were from the urban slums/peri-urban New Delhi and from the upper lower class (modified Kuppuswamy scale; 68.7%; Table 1). Among the 265 participants, 37% were smokers, 48.3% used SLT, areca nut products (with or without tobacco; 9.1%), and 11% used both forms of tobacco; 22.6% also reported alcohol consumption. Of the total participants, 69.4% had one or more tobacco-related oral lesions.
Demographic details of the participants.

NA: not applicable.
The most commonly used smoked form of tobacco was bidi (31.3%); almost 30% of the bidi smokers and 5% of the cigarette smokers used the same brand as earlier. A variety of SLT products were used by the current study participants, the majority being khaini users (27.9%); around 18% gutkha chewers and 23% khaini users reported consumption of the same brand as earlier. The use of SLT products was widespread among both men and women. A significant change was observed between the present and past tobacco (cigarette, gutkha, supari) and alcohol (whisky, tharra) habit, including for those consuming both smoked and SLT (Table 2). Also, a statistically significant association (p < 0.001) was observed between smoked tobacco, SLT, alcohol, and their status of consumption (Table 3).
Change in the past and present prevalence of tobacco and alcohol use (n = 265).
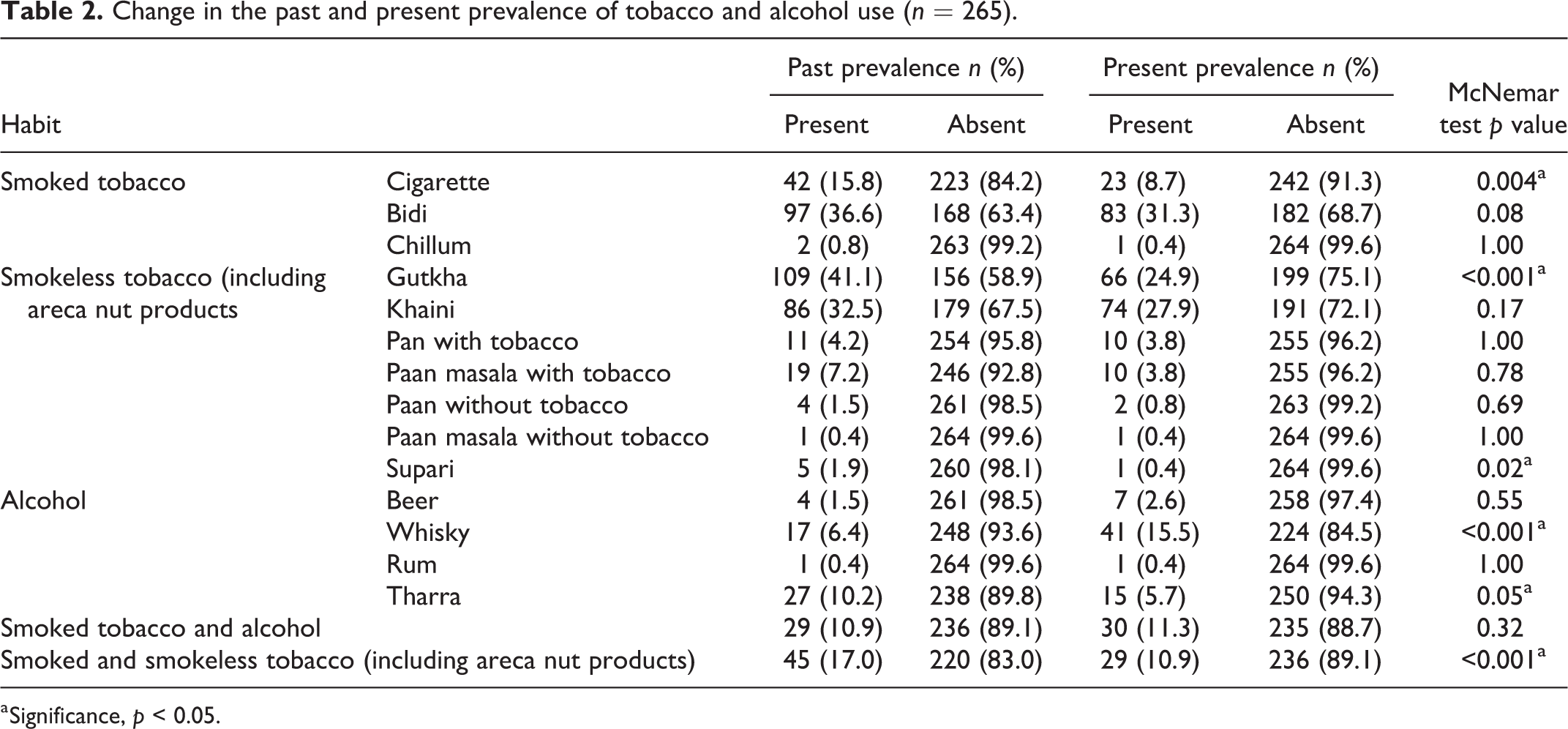
a Significance, p < 0.05.
Association between the various tobacco and alcohol products and their status of consumption (n = 265).

a Significance, p < 0.001.
Tobacco-related oral lesions were identified by the OVE method. Among participants presenting with lesions (69.4%), leukoplakia (34.7%) was the most common tobacco-induced lesion. In the two suspected cases of oral malignancy, punch biopsy for diagnostic confirmation could be performed in only one individual; however, he did not show malignant changes on histopathological evaluation. Due to poor general health and uncontrolled diabetes, a biopsy could not be performed for the other participant. The final diagnosis made was of nonhomogeneous leukoplakia for both cases. Hence, no cases of oral malignancy were found. One deceased patient’s relative reported (verbal autopsy) the cause of death as laryngeal cancer. Individuals untraceable on this screening round were searched for in the Delhi Cancer Registry at AIIMS; however, no cases of oral malignancies were reported even in this group. Candidiasis (2.6%) and other conditions such as chemical burn (induced by lime consumed with tobacco), tobacco-induced melanosis, and lichenoid reaction (4.9%) were among the other types of oral mucosal lesions seen (Table 4).
Tobacco-related oral lesions among the participants (n = 265).

NA: not applicable; OLP: oral lichen planus; OSMF: oral submucosa fibrosis.
Among the total tobacco users (n = 265), the majority reported either a reduced tobacco intake (34%) or quitting the tobacco habit (25.7%; Table 5).
Association between the status of the tobacco habit(s) and related oral mucosal lesion(s) (n = 265).

a Significance, p < 0.05.
In terms of change in the status of the lesion(s), 4.9% lesions were present at a different location from before, while 22.3% showed no change; 8.3% of individuals showed a change in the type of lesion present but without a change in its location, while 11.3% of individuals showed both changes in the type and location of the lesions. Around 6.0% individuals showed a change in the number (increase; along with the type) of lesion present, that is, an additional, new type of lesion(s) at a new site. Among those individuals with no lesions previously (n = 72), 16.6% (n = 44) presented with lesions currently, while 10.9% (n = 29) still had none. Around 19.6% (n = 52) individuals who had lesions earlier presented with none currently. The buccal mucosa was the most common site for the tobacco-related lesions to occur followed by the palate, labial mucosa, alveolar ridge or gingiva, buccal vestibule, and dorsum of the tongue. Overall, the change in the tobacco habit and the status of the tobacco-related oral mucosal lesions showed a highly significant association (p < 0.05). A statistically significant association was also observed between the change in the tobacco habit status in individuals with no current or previous lesions, those with no current lesion but who had lesions previously, and in those showing a different lesion at a different site (p < 0.05); the other categories of lesion status showed no association with tobacco habit status (p > 0.05). Interestingly, among those participants who previously had lesions but none currently (n = 52), 42.3% (22/52) had quit their tobacco habit, while only 15% of those participants (21/140), who still had lesions at the current study (“same lesion at same site,” “same lesion at different site,” “different lesion at same site,” “additional lesion at a new site,” and “different lesion at different site”), reported stopping of tobacco habits (Table 5).
Discussion
The current study was conducted in the urban slums/peri-urban areas of New Delhi. Among the large sample of 2000 individuals examined in the first visit, 13.2% individuals were available during the later visit in 2016 and 2017, resulting in a high attrition rate of 86.8%. In the previous large population-based study conducted in Taiwan, 23,34,299 tobacco users were screened as part of the National Nutrition and Health Survey; at 2 years’ follow-up, 5,99,103 individuals were found and a high attrition rate of 74% was reported, as in the present study. 8 Sankaranarayanan et al. also reported high attrition at the end of 2 years, that is, 67% overall and 71% in the tobacco and/or alcohol consumers screened in Kerala (India). 5 However, at the end of 5 years, the loss to follow-up reported was from 1% to 2.6% in the intervention group and between 2.4% and 13.9% in the control group, among the total population of 36,471 screened at Ernakulam (Kerala, India), Bhavnagar (Gujarat, India), and Srikakulam (Andhra Pradesh, India) by Gupta et al. 7 The majority of the tobacco users were found to be males between the third and fifth decade of life in the current study, similar to those screened in Taiwan, that is, males between 30 and 49 years of age. 8
The pattern of tobacco use varies in different parts of the world. In India, smokeless forms like gutkha, khaini, betel quid (paan), mawa, nass (Naswar), mishri, raw tobacco (sadapatta), and gudakhu are used. 6,18 Our study participants gave a history of using both smoked and smokeless forms of tobacco. Bidi was the most common smoked tobacco product used and khaini and gutkha were the common SLT forms consumed by this sample. Individuals in Ernakulam and Bhavnagar also showed maximum consumption of bidi in the smoked tobacco category, which was similar to our participants, while the population of Srikakulam smoked reverse chutta the most. 7 In contrast to the common SLT habit of khaini among our participants, betel quid with tobacco was consumed in Ernakulam, tobacco with lime in Bhavnagar, and plain tobacco in Srikakulam. 7 However, the Taiwanese population screened showed habits of betel quid and cigarette consumption, which was different from our participants. 8 Hence, an inter-regional and intraregional tobacco consumption variability is noted, owing to the geographic diversity in the available tobacco products.
The 16-year follow-up results here show a significant decrease (34%, p < 0.001) in the tobacco habit by the participants, with a significant percentage of tobacco quitters (25.7%, p < 0.001), owing majorly due to the cessation counseling provided by the health-care professional on the previous visits; at the previous visits (between 2002 and 2005), the tobacco quit rate noted was 8.1% at 6 months and 0.3% at 1 year. In the study by Gupta et al., in the intervention group who were provided health education for tobacco cessation, a remarkable progress was noticed in the percentage of tobacco quitters, that is, from 1.9% to 9%, from 4.9% to 17%, and from 2.1% to 13%, through years 1–5, in Ernakulam, Srikakulam, and Bhavnagar, respectively; the control group showed a reverse pattern, that is, from 5% to 3%, and from 9% to 4% in years 5 and 10, in the three districts, respectively. 7
The OVE is an established screening modality to detect OPMDs or very early stages of oral cancer. It involves systematic visual inspection of the different areas of the oral cavity by trained health-care professionals, under adequate light, using disposable instruments. 19 The Cochrane Review of 2013 reported on studies demonstrating a reduction in the mortality rate and improvement in survival as a result of OVE employed for population-based oral cancer screening. 20 A decrease by 24% in the oral cancer mortality in high-risk individuals screened four times in Kerala (India) and 26% among those who underwent biennial screening in Taiwan has been documented in the literature. 5,8 Population-based and targeted high-risk group (tobacco and alcohol users) screening can thus utilize OVE as a simple and cost-effective method in resource-deficient settings, to reduce the oral cancer mortality and incidence; it has been recommended once every 3 years among high-risk adults aged >35 years. 19
Among all the participants, the majority (69.4%) presented with OPMDs; however, no case of oral malignancy was observed. Among the former, leukoplakia was seen in majority (34.7%) of the individuals which is in line with the findings of Sankaranarayanan et al., Chuang et al., and Gupta et al. (for the Ernakulam and Bhavnagar populates), whereas in the reverse chutta users of Srikakulam, palatal lesions were more commonly seen. 5,7,8 The absence of oral malignant lesions in the current study, even after a long period of 16 years, may possibly be attributed to repeated, effective counseling to the tobacco and alcohol users regarding cessation of the same, during the previous screening visits by the health-care professionals. In addition to this, the reported reduction in tobacco use may also be attributed to the considerable changes in public health policy in India since. Even most of the 87% unavailable individuals, searched for in the Delhi Cancer Registry, may not have developed oral cancer; however, some may not have been registered in the same, while others may not have reported to the concerned hospitals.
Majority of the lesions (39.1%) were found in SLT users as compared to smokers (31.5%) and dual users (14.1%) in the present study; Chuang et al. also reported a higher percentage of OPMDs among consumers of betel nut alone as compared to smokers. 8 Gupta et al. also reported a higher number of tobacco chewers in the intervention group of Ernakulam presenting with OPMDs, unlike the smokers of the Bhavnagar screening population who showed a relatively higher percentage of these lesions. 7
In the study by Gupta et al., tobacco habit reduction, in turn, showed a decreased incidence of leukoplakia in Ernakulam and that of palatal lesions in the Srikakulam population. 7 A significant observation made in our study was that 8.3 and 5.7% of the individuals (p < 0.001) who showed completed regression of lesions also reported quitting and reducing tobacco use, respectively. This study thus demonstrates a link between the change in the overall lesion status and tobacco habits. Also, the presence of a higher percentage of tobacco quitters among the group showing no current lesions as compared to those with lesions at the current visit demonstrates the importance of stopping the tobacco habit in preventing the new incidence of lesions. Alcohol and tobacco have been shown to possess a synergistic effect in OPMDs and oral cancer development. 21,22 Various studies have demonstrated this synergistic effect of tobacco and alcohol, wherein the population attributable risk reported was 4, 33, and 35%, due to alcohol alone, tobacco alone, and due to the combination of tobacco and alcohol, respectively. 21 Alcohol also showed to increase the OPMD risk in tobacco users (past and current) in the study conducted by Maserejian et al. 23 In the current study, 23% of the study participants consumed alcohol and about 19% of them had OPMDs, the presence of which may be attributable to the additive effect of these risk factors.
The strength of this study is that the details regarding the tobacco habits were collected through a personal, face-to-face interview with the users during the house visits.
The limitations of the current study include identity of individuals after a lapse of 16 years, recall bias, and high attrition rate (due to majorly migrant population) resulting in a loss to follow-up. There are possibilities of underreporting of habits such as smoking and alcohol use by women, as these are still not as socially acceptable in India as the use of SLT products.
The study provides support to the conclusion that cessation/reduction of tobacco and alcohol use can prevent the progression/development of OPMDs including malignant transformation, as seen by the absence of development of such cases in our sample, even after a long period of 16 years. Preventive health programs should thus incorporate tobacco cessation as an important component along with oral cancer screening through OVE of high-risk populations, which could help in early detection of OPMDs and prevent malignant transformation of the same. Effective interventions and policies to assist cessation of the use of tobacco are mandatory to reduce the oral cancer-related mortality and morbidity.
Footnotes
Acknowledgments
The authors would like to thank Dr PC Gupta, Director, Healis Sekhsaria Institute for Public Health, Mumbai, Dr Anil Chaturvedi, NCI-NIH, and Dr R Sankaranarayanan, Visiting Scientific Advisor, IARC, for their valuable suggestions. The authors would also like to acknowledge the other members of the house-visit team—Mr. Santosh, Ms. Sheetal, and Mr. Kumar Chandan, National Institute of Cancer Prevention and Research, Noida, Dr Deepika Mishra, Department of Oral Pathology and Microbiology, CDER, New Delhi, for helping with the oral examination form, and the faculty at the Delhi Cancer Registry, AIIMS, New Delhi—Dr N Manoharan and Dr Rajiv Malhotra, for assistance in tracing the unavailable individuals in their database.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by ICMR-NICPR, Noida (India).
