Abstract
Aim:
We investigated the human papillomavirus (HPV) DNA distribution in oral lichen planus (OLP) samples using non-invasive methods, with the potential for much broader population screening.
Methods:
Three different non-invasive collection methods (dry storage, lysis buffer preservation, and use of a commercial tool to rake epithelial cells and immersion of the collection device into the provided rinse solution) were evaluated. HPV DNA was detected in brushed or scraped samples of the lesion site using a commercial HPV DNA polymerase chain reaction kit.
Results:
In the group of patients with OLP lesions, HPV was detected significantly more frequently than in the healthy control group (lysis buffer for control = 2.1%, p = 0.046; DNA•SAL™ rinse solution for control = 4.2%, p = 0.0003; dry storage for control = 6.3%, p = 0.0001) regardless of the different collection and preservation methods. HPV DNA was detected in 12.85% of the samples preserved in lysis buffer. HPV DNA was detected in 34.28% (p = 0.0048 vs. lysis buffer) of samples preserved in the DNA•SAL rinse solution. In the case of dry storage, HPV DNA was detected in 38.57% (p = 0.0008 vs. lysis buffer and p = 0.726 vs. DNA•SAL) of samples.
Conclusion:
The results suggest that the most effective sample preservation methods are provided by dry storage or DNA•SAL collection compared to lysis buffer. Our findings indicated that HPV DNA detection in superficial OLP scrapings has potential as a screening tool and has important applications for both research and clinical practice.
Keywords
Introduction
The presence of human papillomaviruses (HPVs) in the oral cavity of healthy individuals has been well documented. The normal oral mucosa may act as a supply for new HPV infections and/or as a source of recurring HPV-associated lesions. The prevalence of HPV in normal oral mucosa ranges(including both low- and high-risk types) from 0.6% to 81%. 1,2 HPV infection in the oral mucosa appears to be closely related with a range of benign papillomatous lesions, 3 whereas increasing amounts of data indicated a potentially important causative association of HPV infection in oropharynx cancer. 4-6
Furthermore, there is strong evidence available which suggests that a significant number of oral cancers are preceded by visible clinical changes that occur in the oral mucosa in the form of chronic white or red patches. 7 A World Health Organization-sponsored international workshop has recommended that the term “oral potentially malignant disorders” (OPMDs) be used, so as to include all the oral mucosal disorders that carry a risk of malignant transformation. 8,9 A recent systematic review recorded a strong association between HPV detection and OPMDs, when the same sampling technique was used for both cases and controls. 10 In addition, the prevalence rates of HPV association with OPMDs were reported to range from 0% to 85%. 6
Oral lichen planus (OLP) is a chronic inflammatory disorder with immune-mediated pathogenesis which is characterized by a variety of clinical subtypes. OLP is considered as an OPMD, in which HPV DNA is significantly more likely to be detected than in normal oral mucosa. 10 HPV DNA has been found in OLP lesions, with most of the studies conducted using oral biopsies, limiting the broad applicability for screening purposes. Questions still remain regarding the performance and quality of human DNA collected from oral mucosal epithelial cells using different collection and preservation methods. 11 We investigated the HPV DNA distribution in OLP samples with no visual lysis of epithelial mucosa, using different non-invasive collection methods, with the potential for much broader population screening.
Materials and methods
Seventy patients (54 women, age range 23–79 years; 16 men, age range 25–79 years), and 48 individuals (36 women, age range 21–75 years; 12 men, age range 23–76 years) used as age-matched control group, gave written informed consent to participate in the study. The patients’ rights have been fully protected by the institutional Bio-Ethical Committee of the Dental School of Aristotle University of Thessaloniki. Furthermore, the study was specifically conducted with complete anonymity of participants. All individuals who were included in the control group had healthy oral mucosa and cases with signs of oral infection or mucosal lesions as well as persons vaccinated against HPV were excluded. All patients suffering from any systemic disease, and those taking any medication, were excluded from this study. The inclusion criteria for the OLP patients were that they be newly diagnosed cases and not undergoing biopsy intervention for their lesion before sampling. At the first visit, all lesions were clinically typical reticular or plaque-type OLP with no visual lysis of epithelial mucosa, fulfilling the WHO criteria, with the diagnosis confirmed (later) by biopsy and histological examination. Prior to the sampling procedure, all the participants performed a mouthwash with sterile saline under strict supervision. Three different noninvasive collection methods were evaluated and samples were obtained by brushing or scraping the lesion site. All samples were preserved according to three different methods: dry storage (without the use of a buffer or stabilizer), in lysis buffer, and the case of a commercial tool used to scrape epithelial cells (DNA•SAL™, Oasis Diagnostics, Vancouver, WA 98686, USA) immersion of the collection device into the provided “rinse solution”. HPV DNA was detected in brushed or scraped samples using a commercial HPV DNA polymerase chain reaction (PCR) kit (AiD, Germany) and samples were differentiated into high risk (HR) and low risk (LR) genotypes. This kit enables detection of HPV and differentiation of genotypes by PCR and subsequent reverse hybridization with sequence-specific oligonucleotide probes (SSOPs).
Initially, two PCRs were performed, with DNA isolated from biopsy or scrape material. These reactions (referred as PN-1 and PN-2) were performed using the HPV detection kit, where a fragment of the HPV genome was amplified with specific biotin-labeled primers. The amplified gene fragments (amplicons) were characterized by a hybridization reaction with SSOPs, representing particular HPV genotypes and were immobilized on a nitrocellulose membrane. The DNA sequence which was used for the HR and LR HPV detection amplification was previously described. 12
The amplicons of PCR with PN I and PN II were mixed, denatured, and brought into contact with nitrocellulose strips in the wells of the incubation tray provided. The nitrocellulose strips were precoated with lines of immobilized gene probes for different genotypes of HPV, as well as control zones.
During hybridization, the denatured amplified DNA binds to the gene probes attached to the strips. A highly specific washing procedure ensures that only the hybrids survive if the probe’s sequence is 100% complementary to that of the amplified DNA. Streptavidin-coupled alkaline phosphatase binds to the hybrids of the gene probe and biotin-labeled, amplified DNA (1 ml of diluted conjugate to each strip, incubation for 30 min.). This complex was detected by a color reaction of substrate (1 ml to each strip, warmed to room temperature, and incubated for 10 min) to the alkaline phosphatase. Finally, the band pattern was analyzed using the specific evaluation sheet.
Additional HPV DNA typing was performed in the majority of the samples (56 cases) in order to confirm the credibility of the screening methods.
Interpretation of the results
A total of seven reaction zones were defined as capable of development on the nitrocellulose strip (Figure 1). The conjugate control reaction zone indicates the efficiency of the conjugate binding and was included at all times. The amplification control reaction zone contains a probe for the housekeeping gene Glyceraldehyde 3-phosphate dehydrogenase, which can be found in each human genomic DNA sample, and was used as a control for correct DNA isolation and amplification. Furthermore, there are five reaction zones which define the type of HPV in the specific specimen. The HPV HR reaction zone is developed if any of the following HR genotypes of HPV are present: 31, 33, 35, 39, 45, 51, 52, 53, 56, 58, 66, 68, 73, and 82. The HPV-16 reaction zone is developed if there is an HPV-16 genotype present. HPV-18 reaction zone is developed if there is an HPV-18 genotype present. HPV LR reaction zone is developed if any of the following LR genotypes of HPV are present: 6, 11, 40, 42, 43, and 44. The HPV-poly reaction zone is developed if any of the listed genotypes of HPV are present in the DNA sample.

A total of seven reaction zones are defined and are capable of development on the nitrocellulose strip. In this strip, there are positive reactions for HPV-poly, HPV HR, and HPV-18. HPV: human papillomavirus; HR: high risk.
The proportion and positive 95% confidence interval (CI) are presented for the frequency of HPV DNA. The proportion of HPV DNA was compared between the control and the patient’s group, as well as among the three sampling methods using Fisher’s exact test (Stata v11.0).
Results
In the healthy control group, HPV DNA was detected in 2.08% (one case with HPV LR band) of the samples preserved in lysis buffer, in 4.16% (one case of HPV HR band and one case with HPV LR band) of samples preserved in the DNA•SAL rinse solution, and in 6.25% (two cases of HPV LR band and one case with HPV HR band) of samples with dry storage. Regarding the group of patients with OLP lesions, HPV DNA was detected in 12.85% (6.9%, 22.6%) of the samples preserved in lysis buffer (three cases with HPV LR band, two cases with HPV-16, two cases with HPV HR band, one case with HPV-18, and one case with HPV-poly band). HPV DNA was detected in 34.28% (24.3%, 46%) of samples preserved in the DNA•SAL rinse solution (seven cases with HPV-16, six cases with HPV-18, five cases with HPV HR band, five cases with HPV LR band, and one case with HPV-poly band). In the case of dry storage, HPV DNA was detected in 38.57% (28.1%, 50.2%) of samples (eight cases with HPV-16, seven cases with HPV-18, six cases with HPV LR band, five cases with HPV HR band, and one case with HPV-poly band; p = 0.0048 DNA•SAL vs. lysis buffer, p = 0.0008 dry storage vs. lysis buffer, and p = 0.726 dry storage vs. DNA•SAL). The gender and mean ages of the HPV-positive patients with OLP lesions are presented in Table 1. The HPV DNA positivity in OLP lesions according to the collection methods is summarized in Table 2, and Table 3 presents HPV DNA positivity in OLP lesions according to their anatomical location of sample collection. In the group of patients with OLP lesions, HPV was detected significantly more frequently than in the healthy control group (lysis buffer for control = 2.1% (CI 0.4%, 10.9%), p = 0.046; DNA•SAL rinse solution for control = 4.2% (CI 1.2%, 14%), p = 0.0003; dry storage for control = 6.3% (CI 2.2%, 16.8%), p = 0.0001) regardless of the different collection or the preservation methods.
Demographics of HPV DNA positive cases in patients with OLP lesions.
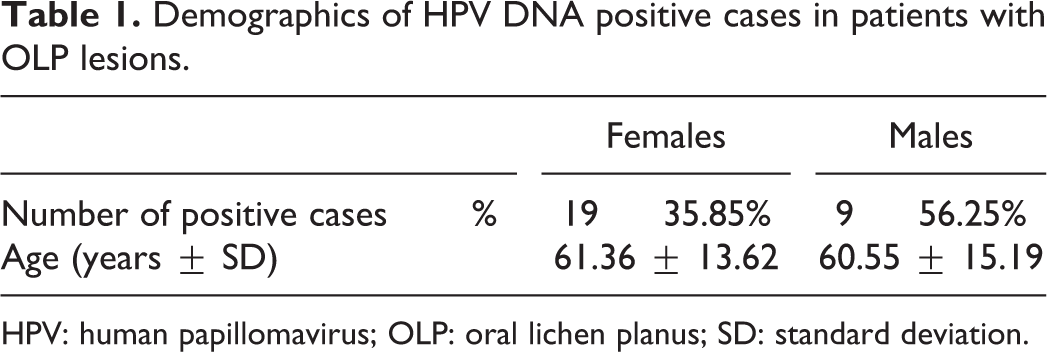
HPV: human papillomavirus; OLP: oral lichen planus; SD: standard deviation.
HPV DNA positivity cases in OLP lesions according to the collection methods.

HPV: human papillomavirus; OLP: oral lichen planus; LR: low risk; HR: high risk.
HPV DNA positivity in OLP lesions according to their anatomical location (dry storage of samples).

HPV: human papillomavirus; OLP: oral lichen planus; LR: low risk; HR: high risk.
Discussion
Detection of HPV infection is conducted routinely by morphological examination of cells from abnormal skin regions or cervical scrapes. HPV infection is suspected if the cytology identifies the existence of the typical morphological cellular feature of koilocytosis, with perinuclear halo formation and a thick cytoplasmic border. 13 However, with cytological diagnosis, it is highly possible to acquire false negative results. Furthermore, there is no other sensitive bioassay for diagnosis, since HPV is not easily cultivated in an in vitro cell culture system. The sensitive detection and differentiation of genotypes of HPV is of great clinical importance, especially for cancer prevention and diagnosis. Available assays based on molecular genetic methods are both costly and time-consuming. The reverse hybridization technique accomplished by this assay is both a time- and cost-sensitive method, which is able to detect and differentiate infections with HPV genotypes. 14 The assay reaches a sensitivity of at least 103 copies, which is the lowest concentration with clinical relevance. PCR is highly sensitive, widely used, and it is considered as the gold standard in a previous HPV meta-analysis. 10 However, PCR detection is prone to false positive results due to contamination which may arise during sampling, conditions of storage, processing, and due to any variation of PCR protocols. 10
In the scientific research papers published prior to 1998, 107 OLP samples were investigated for the presence of HPV DNA with either in situ hybridization or dot blot hybridization and 23% of the samples were reported positive. The most prevalent types detected were HPV-6 and HPV-11, followed by HPV-16. Since that time, 1929 samples (either scrapings or biopsies), from normal oral mucosa, have also been studied for HPV DNA and 11% of them were positive. 15 Additional studies have detected the presence of HPV in OLP, 16,17 concluding that in comparison with normal oral mucosa, the HPV detection rate in OLP was twice as high. The highest prevalence of HPV has been discovered in atrophic variants of OLP in comparison to other OLP forms. In addition, a systematic case control-based meta-analysis was conducted on the influence of HPV in oral cancers and OPMDs, including OLP. The results demonstrated that the HPV detection rate in OLP is considerably higher than that in normal oral mucosa, suggesting a specific role of HPV in this disease (odds ratio (OR) = 3.57; 95% CI: 1.16–10.97). This was also true for the presence of HPV-16 (OR = 7.23; 95% CI: 3.12–16.75). 10 Furthermore, in a recent study that investigated HPV in oral atrophic lichen planus lesions, HPV DNA was isolated in 15.9% of the samples, 18 which was similar or slightly less than reported in previous literature. 17,19,20 Both the common mucosal LR and HR HPVs were detected. Furthermore, it has been reported that none of the five atrophic OLP lesions, which progressed to invasive carcinoma, demonstrated positive for HR HPV, but two of those tested positive for HPV-6 and HPV-11 did transform. 18 The significance of this finding was interesting, because HPV-6 and HPV-11 are considered the two most common LR HPV types. However, earlier literature has reported that benign papillomas/condylomas, associated with either HPV-6 or HPV-11, have progressed to carcinoma when treated by irradiation. 21 In addition, an interesting finding was reported among head and neck cancers, where the outcome was worst for patients with HPV-6- and HPV-11-positive cancers (with or without linked HPV-16 infection). 22 Still any association of HPV infection with other cofactors and their impact (e.g. other infections, X-ray examinations, or carcinogens) in oral carcinogenesis is not known. Therefore, it is suggested that HR and LR HPVs may present a different behavior in oral mucosa than in the genital tract, because these two mucosal sites have a separate exposure to tobacco carcinogens and also have a different microbial milieu. 18
There is currently no validated screening method for HPV-related oropharynx cancer that has been shown to be effective. 23 There is an obvious difficulty getting scrapes out of tonsil or oropharynx and additionally HPV may lie in deeper crypts. HPV DNA has been detected in oral rinses and is associated with increased odds of HPV-related oropharynx cancer in case control studies. 10 This differs from the cervical cancer model, where persistent HPV infection is an established risk factor for developing high-grade cervical intraepithelial neoplasia and cervical HPV detection is being integrated into cervical cancer screening algorithms. 24
Our results suggest that the most effective sample preservation methods are provided by dry storage and/or DNA•SAL collection compared to lysis buffer. The dry storage method may be preferable when samples are to be processed immediately. The DNA•SAL collection and rinse solution offers sample preservation which is almost equally effective, when compared with dry storage, and may be preferable when adequate stabilization of samples is required for later or downstream testing.
It is not easy to explain why a chronically inflamed epithelium such as in OLP, thought to be infiltrated by T-cells, should be prone to supporting HPV infection. A possible explanation could be that mucosal alterations such as ulceration, that are frequently present in OLP, make it more susceptible to HPV infection 18 as the virus can enter the deeper layers of the mucosa due to breaks in the surface of an ulcerated OLP lesion. Another potential explanation could be the chronic use of steroids which may induce immune suppression, leading to upregulated HPV replication. 16
It is also noteworthy that although all our cases were clinically typical reticular or plaque type OLP, in one female patient with OLP lesions in the mandibular vestibule, HPV-16, HPV-18, HR positivity was detected and that patient was eventually diagnosed, by biopsy, with squamous cell carcinoma in the following year. Furthermore, a male patient with OLP reticular lesions in the palate, where HPV LR positivity was detected, has had reportly a previous history of genital condylomas.
The above-mentioned findings underline the fact that although previous reports suggested that higher HPV carriage was detected more frequently in erosive and atrophic types of OLP lesions, we cannot ignore the possibility of the potential malignant transformation in the non-erosive and non-atrophic types of OLP. Furthermore, it is important to point that as for the anatomical locations of HPV DNA positive lesions of OLP (the majority was detected in buccal mucosa and lateral border of the tongue, mainly to the anterior and to the border between anterior and posterior part of the tongue), these presented the same consistency with the OLP lesions without HPV DNA positivity.
It has been suggested that the progressive increase in HPV prevalence in lesions that have low potential to manifest oral malignant disease may be explained in two possible ways. 21 First, HPV plays a supplemental role in generating the progression of potentially malignant lesions, and secondly, lesions contribute an environment that is increasingly favorable for HPVs as their malignant potential increases. In both cases, the presence of HPV may have prognostic significance.
In addition, this study recorded an increased percentage of HPV positivity in men. Although the sample was small, it may be explained by the profound differences in exposure to risk factors (smoking, alcohol consumption, and dietary habits), which were not evaluated in the present project. In a previous study, it was reported that HPV was more prevalent in men and among smokers. Those findings were statistically relevant. That study also reported a 6.9% prevalence of HPV in the general US population, when they assessed a cohort of 5579 individuals without prior selection. 25
In a recent meta-analysis which studied the immensity of the relation between HPV and OLP showed that OLP patients have significantly higher HPV prevalence (OR: 6.83; 95% CI: 4.15–11.27) than healthy controls. 26 Furthermore, it was observed that in the subgroup surveys, the association of HPV and OLP varied significantly according to the geographic region and population, among the 12 countries, in different continents. In the above-mentioned study, the ORs of HPV/OLP association were reported to range from 1.00 to 138.05. 26 The same meta-analysis investigated the association between HPV and the clinical forms of OLP (erosive-atrophic (EA) and non-EA) which was comparable and more intense in the EA OLP lesions (OR: 9.34) than in the non-EA OLP lesions (OR: 4.32). 26 In addition, in a previous study, the risk of progression to oral malignancy for non-EA OLP lesions was approximately 0.5%, whereas in the EA OLP, lesions increased at least to 3.5–4.00% during similar follow-up periods. 20 The above-mentioned data lead to a hypothesis that differences in the HPV prevalence may have an effect on OLP malignant potential and amplify the necessity for HPV screening not only in the OLP patients but also in all the OPMD lesions.
In conclusion, our findings indicated that HPV DNA detection in superficial OLP scrapings has potential as a screening tool, with important applications for both research and clinical practice. From a technical standpoint, a custom designed oral scraper could be helpful in improving the sampling procedure and may allow more accurate diagnosis of HPV infection. It may also represent a reliable method to investigate the biological characteristics of HPV infection in OLP.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
