Abstract
Objective:
To determine the prevalence of human papillomavirus (HPV) DNA in oral squamous cell carcinoma (OSCC).
Methods:
A total of 88 OSCC specimens collected between 2006 and 2013 were available for the study. DNA was extracted using formalin-fixed, paraffin-embedded specimens and analysed for the presence of 18 HPV genotypes using a nested polymerase chain reaction using consensus forward primer (GP-E6-3F) and two consensus back primers (GP-E7-5B and GP-E7-6B). Plasmid DNA of HPV 16 and 18 was used as positive controls.
Results:
HPV DNA was detected in 3 of the 88 samples, a prevalence of 3.4%. Genotypes detected were 16, 18 and 52.
Conclusion:
The overall prevalence of HPV DNA was 3.4%. Only high-risk genotypes were detected. This low prevalence of high-risk types of HPV suggests that the HPV virus may not have a significant role in the development of oral cancers in Ghana, unlike higher rates described elsewhere in the world, especially in Western countries. Surveillance of future prevalence of HPV and attention to other major risk factors is warranted.
Introduction
In sub-Saharan Africa, cancers of the oral cavity and the oropharynx were found to be the most commonly reported malignancies among head and neck squamous cell carcinomas (HNSCCs) in a systematic review over the period 1990–2013. 1 The review included 7750 cases of which 90% were HNSCC. 1 These, combined sites, also constituted the third most common malignancy sites, after prostate and liver in males, and breast and cervix in females. 2
The aetiology of oral squamous cell carcinoma (OSCC) is multifactorial with alcohol and tobacco consumption considered to be the main risk factors. 3,4 There is considerable variation in incidence due to geographic location, ethnicity and culture and in the anatomic subsite/s affected. Tobacco, both smoked and smokeless, heavy alcohol consumption and areca nut, both alone and as a component of betel quid, are established causes of OSCC in Asia and Europe. 5,6
Data concerning the aetiology of HNSCC in sub-Saharan Africa are unreliable due to the absence of established cancer registries in most countries. Nevertheless, it is noteworthy that the review by Faggons et al. 1 found that 43% of patients used tobacco and 42% used alcohol; these factors being more strongly associated with laryngeal SCC than with oral cavity or oropharyngeal cancer.
Oncogenic human papillomavirus (HPV) infections have a well-established association with uterine cervical carcinoma, and the prevalence of cervical HPV infection in Africa is high. Indeed, HPV is the primary cause of cervical cancer, which is a leading cause of death by cancer in females across the continent. 7
Recently, the International Agency for Research on Cancer (IARC) has acknowledged oncogenic genotypes of HPV, also referred to as high risk or hrHPVs, as causing a subset of HNSCCs, particularly those that arise in the oropharynx, notably base of the tongue and tonsils. 8 Approximately, one-third of the total head and neck cases in the world have been shown to be associated with hrHPV infection, but wide geographic variation exists. 9 HPV infection and its association with oropharyngeal squamous cell carcinoma (OPSCC) and OSCC is supported by the fact that OPSCC and OSCC are occurring in people who have not been heavily exposed to alcohol or tobacco and by the detection of HPV DNA in tumour tissues. 10
There is much variation in HPV prevalence worldwide, and controversies on its role in oral cavity cancers have been debated. Data on HNSCC for Ghana and elsewhere in sub-Saharan Africa are sparse. We therefore investigated the prevalence of the HPV virus in histologically diagnosed cases of OSCC in a Ghanaian population.
Methods
A total of 88 formalin-fixed, paraffin-embedded (FFPE) archival blocks of incisional biopsy specimens with histological diagnosis of OSCC accessioned for the period January 2006 to December 2013 at the Oral Pathology Department, University of Ghana School of Medicine and Dentistry, College of Health Sciences, located at the Korle-Bu Teaching Hospital, were analysed. Clinico-pathological data of tumour sites, tumour differentiation and demography of patients were retrieved from the pathological records.
The study was approved by the Ethical and Review Committee of the School of Medicine and Dentistry, College of Health Sciences, University of Ghana, Legon.
Histological grading was determined according to the WHO classification as grades 1, 2 and 3 which referred to well, moderately and poorly differentiated cancers, respectively. 11
Preparation of genomic DNA
Genomic DNA was prepared using the method described by Dabić et al., 12 with slight modifications. The FFPE blocks were sectioned while maintaining utmost precautions to avoid interblock contamination of DNA. Five 10-µm thick sections were cut from each paraffin block into 2.0 ml sterile microcentrifuge tubes. A new blade was used for every block. Thorough cleaning of the microtome with single use of brush and forceps and a regular change of gloves in between blocks as well as changing the water bath for each block ensured that there was no interblock transfer of DNA.
These sections were incubated in 250 μl lysis buffer, (1 mg/ml proteinase K (Sigma, Sigma-Aldrich Corporation St Louis, Missouri, USA), in 50 mM Tris-hydrochloride pH 8, 1 mM ethylenediaminetetraacetic acid and 0.5% Tween-20 (Sigma, USA)) for 16 h at 56°C. Proteinase K was inactivated at 100°C for 5 min and the lysate centrifuged at 13,000 r/min for 5 min, 0.2–2 µl of the supernatant was then used for polymerase chain reaction (PCR) amplification and the rest of the DNA lysate stored at −20°C for future use.
Detection of HPV DNA by nested multiplex PCR
Primer sets (Eurogenerics Brussels, Belgium) and the method described by Sotlar et al. 13 were used. The HPV genome was amplified using consensus forward primer (GP-E6-3F) and two consensus back primers (GP-E7-5B and GP-E7-6B). The PCR reaction mix of 50 µl contained 10× PCR buffer (Biopioneers Co., USA), 2.5 mM magnesium chloride, 200 µM of each of the 4 oligonucleotide triphospates, 15 pmols of each E6/E7 consensus primer and 1.25 units of Taq polymerase enzyme (Biopioneers Co., USA). DNA lysate (0.2–2 µl) was used as template for the amplification reaction and sterile double distilled water was used to make up the volume to 25 µl.
The DNA amplification was carried out using a thermal cycler. The cycling parameters for the first round PCR with the E6/E7 consensus primers were 94°C for 4 min followed by 40 cycles of 94°C for 1 min, 40°C for 2 min, 72°C for 2 min and a single final elongation step of 72°C for 10 min. For each reaction, positive controls (HPV 16 and 18 plasmid DNA) and a negative control (double-distilled water) were included. In the second round nested PCR, 1 µl of first round PCR product and 15 pmols of forward and reverse primers for genotyping were used with the above-mentioned other parameters remaining the same. The genotyping cycling parameters were 94°C for 4 min followed by 35 cycles of 94°C for 30 s, 56°C for 30 s, 72°C for 45 s and a single final elongation step of 72°C for 4 min.
Primers for the identification of high-risk genotypes 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66 and 68 and low-risk genotypes 6/11, 42, 43 and 44 were used. The primers were in four cocktails, each containing four to five different primer pairs (Table 1). The PCR products were run on a 2% agarose gel and stained with 0.5 µg/ml ethidium bromide to detect the presence of amplified DNA fragments with a hundred base pair DNA molecular weight marker run alongside.
Sequences of synthetic oligonucleotide primers used for HPV genotyping.

HPV: human papillomavirus.
Statistical analysis
This was carried out using SPSS version 20 (IBM Corporation, Armonk, New York, USA). Categorical variables such as gender and age group were summarized as proportions and percentages. Prevalence of HPV positivity was compared among gender, age group and histological differentiation of OSCC using χ 2 test and significance set at p < 0.05.
Results
A total of 88 of 104 blocks provided clean data. Due to deficiencies in the records, some blocks had to be excluded. The mean age of patients was 57.3 years; 54 (61%) were men, giving a male to female ratio of 1.6:1. The greater proportion, 49% (n = 43), was moderately differentiated; followed by well 36% (n = 32) and poorly differentiated 15% (n = 13). PCR assays revealed the presence of HPV DNA in 3 of 88 (3.4%) tumour specimens (Figures 1 and 2). HPV genotypes detected in these three cases were 16, 18 and 52. Two of the positive results were found in males, and one in a female, all of whom were <60 years of age. All three positive OSCCs were of moderate differentiation. The associations between HPV positivity and clinicopathologic variables (gender, age and histological grading) are shown in Table 2. No significant associations were found between the presence of HPV DNA and gender (χ 2 = 0.037, df = 1 and p = 0.848), age (χ 2 = 2.588, df = 1 and p = 0.108) or histological grading (χ 2 = 3.25, df = 2 and p = 0.197).
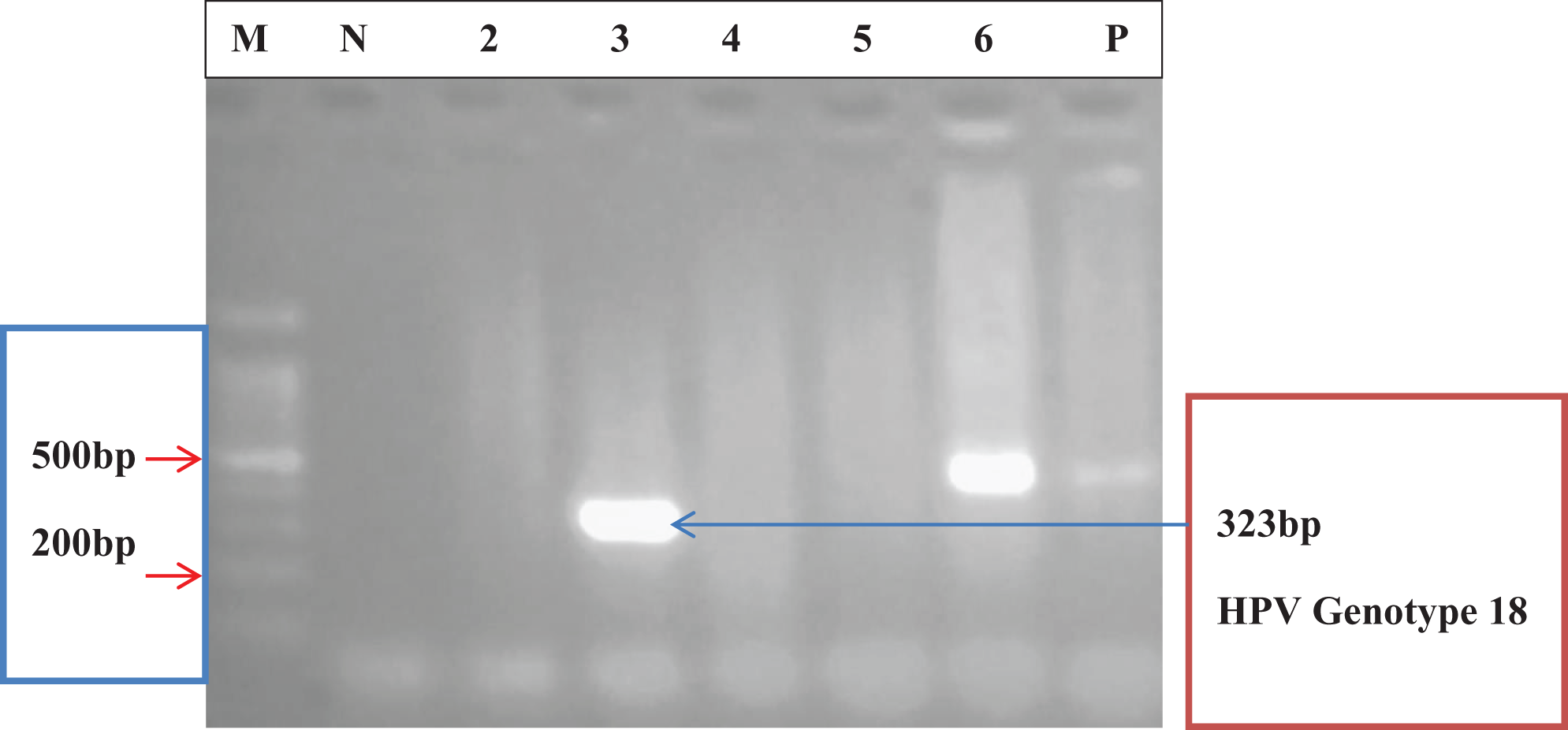
Agarose gel electropherogram of amplified DNA fragments for HPV genotypes 16 and 18. Ethidium bromide stained 2.0% agarose gel electropherogram of amplified DNA fragments from paraffin-embedded samples of patients with oral squamous cell carcinoma infected with HPV. From left to right: Lane M = 100 bp molecular weight marker; Lane N = negative control; Lanes 2, 4 and 5 = PCR negatives; Lane 3 = PCR positive (HPV genotype 18, 323 bp); Lane 6 = PCR positive (HPV genotype 16, 457 bp); and Lane P = positive control (HPV genotype 16, 457 bp). (Amplification was done with cocktail 1 primers from the work of Sotlar et al. 13 ). PCR: polymerase chain reaction; HPV: human papillomavirus.
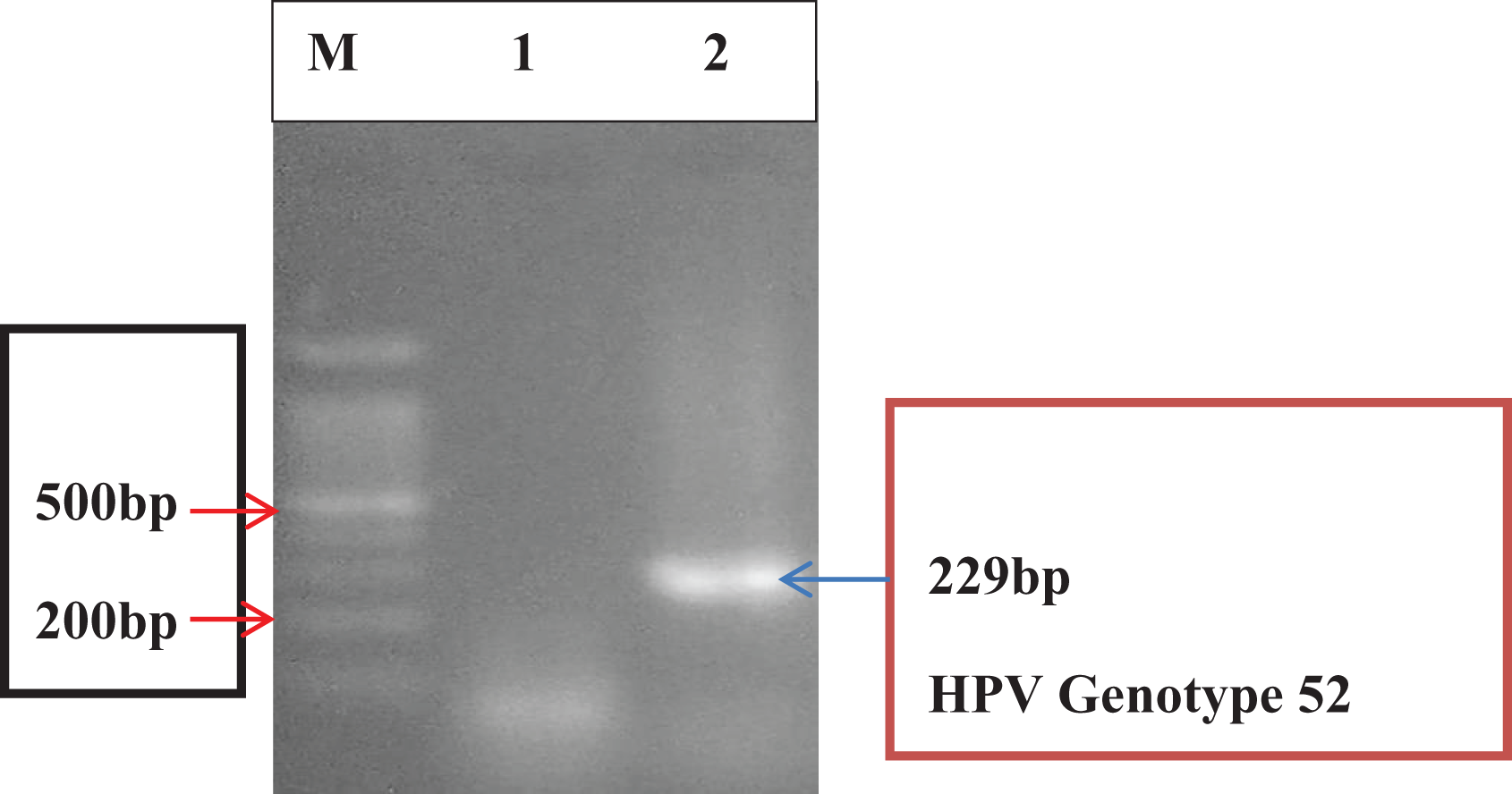
Agarose gel electropherogram of amplified DNA fragments for HPV genotype 52. Ethidium bromide stained 2.0% agarose gel electropherogram of amplified DNA fragments from paraffin-embedded samples of patients with oral squamous cell carcinoma infected with HPV. From left to right: Lane M: 100 bp molecular weight marker; Lane 1: PCR negative sample; and Lane 2: PCR positive (HPV genotype 52, 229 bp). (Amplification was done with cocktail 2 primers from Sotlar et al. 13 ). PCR: polymerase chain reaction; HPV: human papillomavirus.
HPV DNA distribution.

HPV: human papillomavirus.
Discussion
Approximately, one-third of the total head and neck cases in the world have been shown to be associated with high-risk HPV infection, but wide geographic variation exists. 9,14 HPV infection and its association with OPSCC and OSCC is supported by the fact that cases are occurring in people who have not been heavily exposed to alcohol or tobacco and by the detection of HPV DNA in OSCC tissues. 10 Our study did not include any analysis on lifestyle risk factors. HPV DNA was detected in 3.4% of cases in our study. Similar low HPV prevalence has been reported in Nigeria 15 (0%), Mozambique 16 (0%), South Africa 17,18 (0%) and (11.86%), respectively, Senegal 19 (2.44%) and Ghana 20 (13.79%) as presented in Table 3.
African studies reporting HPV prevalence in OSCC.

HPV: human papillomavirus; PCR: polymerase chain reaction; OSCC: oral squamous cell carcinoma; FFPE: formalin-fixed, paraffin-embedded; IHC: Immunohistochemistry; ISH: in situ hybridization.
Our findings are somewhat different from prevalences reported from other parts of the world, notably Asia, Europe and North America, which have recorded high prevalence rates. In a systematic review of published studies of HNSCC biopsies by Kreimer et al., 21 the highest HPV prevalence in OSCC, as analysed by study location, was recorded for Asia (33.3%), followed by North America (16.1%) and Europe (16%). Results from Central and South America, Australia and Africa, combined as ‘others’, gave a prevalence rate of 18.1%. Kreimer et al. review was conducted in 2005 and over the years sexual practices may vary leading to a rise or fall of these prevalence data.
Even within the same geographical location, ethnic disparities exist. African Americans have been shown to record lower HPV prevalence rates for OPSCC (4%) as compared to 34% in White patients, as reported by Settle et al. 22 Men, younger age-cohorts and Whites have higher exposures to the oral sexual behaviours associated with oral HPV16 infection and OSCC as reported by Herbenick et al. 23 : Africans and African Americans are less likely to have oral sex as compared to other races and this may explain the lower prevalence of HPV DNA in OSCC in our part of the globe. 24,25
Across the Asia–Pacific region, high HPV DNA prevalence in OSCC has been reported from South East Asia (48.6%), East Asia (42.8%) and South Asia (36.6%), with low prevalence in Australia (5.6%). 9 A global meta-analysis by Ndiaye et al. 26 reported the highest regions consistent with the Asia–Pacific study, with the lowest prevalences noted in Africa.
In a review of the literature by Miller and White, 27 HPV DNA was detected significantly more often (p < 0.001) in frozen OSCC (115 of 223; 51.6%) than paraffin-embedded tissue (136 of 628; 21.7%). Smeets et al. 28 also considered frozen biopsies as the gold standard for a meaningful HPV detection. Most studies encountered in our review of the literature employed FFPE specimens, with detection rates of HPV DNA for the oral cavity ranging from 0% to 37.5%. The highest figure was found in the South Asia region using PCR technique as published by Shaikh et al. 9 (37.5%) but lower figures were reported by Matzow et al. 29 (2.6%), Ndiaye et al. 26 (24.2%) and Blumberg et al. 16 (0%). FFPE was used in our study because frozen specimens were not available to us.
Several techniques have been employed for detection of HPV DNA. Assays considered to be of low sensitivity include immunoperoxidase assay, immunofluorescence and in situ hybridization (ISH). Moderate sensitivity assays are Southern blot hybridization (SBH), dot blot hybridization and reverse blot hybridization. An assay considered to be of high sensitivity is PCR. 27 HPV DNA was detected in OSCC significantly (p < 0.005) more often (122 of 329; 37.1%) in studies that used PCR than studies that used moderate sensitivity assays (84 of 334; 25.2%; e.g., SBH) and low sensitivity assays (61 of 362; 16.9%), for example, Immunohistochemistry (IHC), ISH as reviewed by Miller and White. 27
Recent studies have largely used PCR, which is sensitive and cost-effective. 26 In a limited resource setting such as ours, we found PCR to be quite practicable. However, PCR can be overly sensitive and may amplify contaminant HPV from the laboratory environment if stringent precautions are not taken. In an early retrospective review of the literature up to the mid-1990s, Miller and White 27 described appreciable prevalences from studies using paraffin-embedded specimens and PCR. We were careful to avoid contamination, and the prevalence rate was low. We are thus confident that our data are real, and the comparatively low prevalence is more likely to be explained by cultural and behavioural differences compared to societies with higher rates of transmission.
The use of early gene primers E6 and E7 in the PCR has been shown to be two to three times more efficient (p < 0.001) in the detection of HPV DNA in OSCC than the use of late gene primers. 27 We used early primers. The L1 gene is used as the amplification target in PCR in several laboratories as it is more stable in fixed tissues and its product is equivalent to the whole HPV genome. In a recently reported study from Cordoba, Argentina L1 gene was used for PCR amplification of saliva and tongue scrapings and reported a prevalence of HPV in 3% in their population. 30 It is important to note that be it L1 or the E6/E7 oncogenes, positive results merely indicate the presence of the virus: they show association, not causation. 28 We also take note of a recent study illustrating the importance of analyzing E2 expression as a marker for stably integrated HPV. 31
Ghana, with a population of 28 million, like most of West Africa, does not have an established population-based cancer registry making it impossible to assess the true incidence and prevalence of all cancers. One cannot, therefore, compute attributable risk for hrHPV infections and HNSCC in the nation, and the population burden this represents.
To our knowledge, there is only one previously published study from Ghana describing the prevalence of HPV in HNSCC, using conventional PCR methods. Kaba et al., 20 in a retrospective review of archival HNSCC specimens and patient demographic and clinical data between January, 2007 and December, 2009 in the Department of Pathology, Korle-Bu Teaching Hospital, Accra, reported HPV DNA in 4 out of 29 OSCC cases (13.8%) using multinested PCR. The present study in the same hospital, over 8 years (2006–2013), and employing the same techniques, found only 3/88 (3.4%) positive. In both studies, the number of cases is small, and it is possible that DNA quality may have deteriorated over time.
The HPVs isolated from OSCC around the world include HPVs 6, 11, 16, 18, 22, 31, 33, 35, 38, 58, 68 and 70. The most identified genotypes are 16 and 18. 14,21 HPV16 is described as present in 90% of cases of HPV+ HNSCCs worldwide. 21 HPV 16 was the most common type in all sites and regions with the exception of OSCC in Africa where HPV 18 was the most common type. 9 HPV 16 was also the most common genotype reported in OSCC in Asia–Pacific. 9 HPV 16 was also the predominant genotype in the work by Kaba et al. 20 here in Ghana. Our study does not present that picture. HPV genotypes identified in the positive specimens in this study were high-risk category types 16, 18 and 52. These genotypes, according to the IARC 2009, are most frequently found in oral and cervical cancers. 21 Notable here is the finding of genotype 52 which is more frequently a cervical genotype. These genotypes have also been found in cervical studies done in Ghana by Attoh et al. 32 In their study on HPV genotypes in cervical carcinoma, they detected 8 out of 18 different genotypes and all were high-risk types. Genotype 18 was predominant (84%) in cervical carcinoma tissue followed by 24% of genotype 16. Co-infections with 18 and 16 were seen in 18% of cases. Other genotypes seen in this study were 35, 52, 56 and 66 at 2% each. 32 Shaikh et al. 9 also found a few cases of HPV DNA genotype 52 in their meta-analysis of the Asia–Pacific Region.
Most cases with HPV DNA from this study were moderately differentiated. This is likely because the literature reports that ISH and PCR studies showed that HPV signals were often strongest in more differentiated regions and in more differentiated cells in OSCC. 27 In this study, however, moderately differentiated OSCC was of higher prevalence over well differentiated OSCC.
The three specimens that were found to have detectable HPV DNA were from males, aged 40–49 and 50–59 years and a female who was less than 40 years old. Although one cannot generalize from such low numbers, this is consistent with the world literature showing higher rates in young, male patients. 9 Likewise, although our cases were from floor of the mouth, hard palate and tongue, no conclusions can be drawn regarding oral site susceptibility.
Limitations of the present study included exclusion of a number of samples because blocks were not found or the records provided were inaccurate. This affected sample size. Aetiological factors such as alcohol and tobacco use could not be explored because such information was not provided on the patient biodata forms sent to our pathology laboratory. Our cases were derived from a single pathology laboratory, which may limit generalization.
Conclusion
In light of the presumed low prevalence of HPV DNA in OSCC in the present study, investigations of other risk factors such as alcohol and tobacco use, predisposing factors such as diet and nutrition, genetic predisposition, sexual and oral hygiene and the role of other microbes need to be explored. Multicentre studies using fresh, frozen specimens need to be carried out. It also necessary to conduct surveillance studies on HPV infection in oral carcinomas.
Footnotes
Acknowledgements
The authors acknowledge the technical support of Miss Linda Azuyie of the Oral Pathology Department and the editorial support of Prof Grace Evangeline Parkins of the Oral and Maxillofacial Department.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
