Abstract
Background:
Oral cancer is a major public health problem in many parts of South Asia and ranking third among the cancer types in India. Numerous diagnostic, prognostic and metastatic biomarkers have been employed till date to assess various carcinomas. Stearoyl coenzyme A desaturase (SCD) is one such recently introduced biomarker that regulates key cell cycle events in normal cells and during carcinogenesis in neoplastic cells. Although SCD has been used to analyze the prognosis of breast and renal cell carcinomas, its role in oral epithelial dysplasia (OED) and oral squamous cell carcinoma (OSCC) remains unexplored. Therefore, there is a need to analyze the role of SCD in OED and OSCC to predict their biologic behaviour.
Objectives:
To assess the expression levels of SCD in OED and OSCC samples and correlate them with normal oral mucosa (NOM) to determine their prognostic potential.
Methodology:
Fifty five tissue samples of OED (20), OSCC (20) and NOM (15) was subjected to immunohistochemistry using SCD. Clinical details and follow-up data (3 years) were recorded. Clinical and histopathological parameters were statistically analyzed using descriptive statistics, univariate and multivariate analyses, Kruskal–Wallis analysis of variance test and Mann–Whitney
Results:
Descriptive analysis on OED and NOM showed a statistically significant difference in age between OED and NOM (
Conclusion:
The expression of SCD in OED was higher than NOM suggesting changes in cell cycle control in OED. An enhanced expression of SCD was identified in OSCC when compared to OED. Correlating the SCD expression with survival rate in OSCC patients revealed an increased SCD expression with diminished survival rate, indicating that SCD may be used to predict prognosis. However, the role of SCD in assessing the prognostic potential within the grades of OED and OSCC requires future research.
Introduction
The global epidemiology of oral squamous cell carcinoma (OSCC) indicates that its incidence is high in several south Asian countries including India. Majority of OSCCs in these populations arise from oral potentially malignant disorders (OPMDs) particularly among users of tobacco, alcohol and betel quid. 1 –5
The genesis of cancer is a very complex process that commences with phenotypic and genotypic changes.
6
The homeostatic equilibrium between cell growth and apoptosis is disturbed in carcinogenesis resulting in uncontrolled tumour cell proliferation. The cancer cells favour activation of lipid synthesis and signalling network during proliferation, which is mediated by metabolic events that facilitate activation of the fatty acid biosynthetic pathway. Saturated fatty acids (SFAs) and monounsaturated fatty acids (MUFAs) are the by-products that maintain the increasing demands of the rapidly dividing cells regulated by the cell cycle. The cyclic activity of fatty acid biosynthetic enzymes, namely, the adenosine triphosphate citrate lyase, acetyl-CoA carboxylase and fatty acid synthase causes increased production of SFA and its conversion into MUFA by way of stearoyl coenzyme A desaturase (SCD). SCD is an endoplasmic reticulum enzyme that catalyzes the synthesis of MUFA from SFA derived from the diet or
In normal cells, SCD is the principal target of growth factors and hormones that regulate key cell cycle events. SCD expression is proportionate to synthesis of phospholipids and cholesterol that facilitates MUFA in the genesis of cell membrane. In the absence of SCD, the cells favour increased accumulation of palmitic acid, a toxic product to the mitochondria and endoplasmic reticulum which leads to apoptosis. 8 In neoplastic cells, SCD overexpression is the cause of marked enrichment of phospholipids with MUFA. SCD has the ability to convert exogenously and endogenously synthesized SFA to MUFA, with the formation of SFA within cancer cells. 8
It has been observed that OKF6 and TERT1 cell lines (derived from head and neck SCC) showed an increased expression of SCD when compared to normal oral keratinocytes. Cells chronically exposed to tobacco have shown an overexpression of SCD by 2.6 folds that suggests its potential use as a diagnostic marker in head and neck epithelial malignancies. 9 Though elevated levels of MUFA have been observed in cancer cells in earlier studies, the mechanism of SCD in carcinogenesis is minimally explored. Despite research pertaining to SCD expression in malignancies of breast, kidney, liver, lungs, thyroid and pancreas, where increased levels of SCD have corresponded to poor prognosis, very few studies are available in the literature that explains the relationship of SCD expression with oral cancer. 10 –20 Also, evaluation of SCD expression in oral epithelial dysplasia (OED) may help in the prediction of the malignant potential of an OPMD at an earlier stage.
Our aim was to evaluate and compare the expression of SCD in formalin-fixed paraffin-embedded (FFPE) tissues of OED, OSCC and normal oral mucosa (NOM) specimens using immunohistochemistry to assess the prognostic potential of this biomarker.
Methodology
Sample collection and study setting
A retrospective study was conducted on FFPE tissue samples of OED, OSCC and NOM from the archives of Department of Oral Pathology, Faculty of Dental Sciences, Ramaiah University of Applied Sciences, Bengaluru, Karnataka, India.
Inclusion criteria
Histopathologically confirmed cases of OED, primary OSCC of patients who were chronic tobacco users and NOM of healthy subjects with and without the habit of using tobacco were included in the study.
Exclusion criteria
Recurrent cases of OSCC and subjects with metabolic and other systemic disorders were excluded.
The study was conducted from January 2016 to October 2017. The ethical clearance was obtained from institutional Ethical Clearance Board (FDS/EC/2014-16/PGST/29).
Immunohistochemical analysis
Streptavidin–biotin technique was used for immunohistochemistry. Tissue sections of 4-µ thickness were deparaffinized in xylene and rehydrated in graded alcohol. Antigen retrieval was performed with Tris ethylenediaminetetraacetic acid (EDTA) solution (pH 6.0), by treating the sections at 90–95°C for 15 min in a pressure cooker and then cooling to room temperature naturally. The slides were rinsed with phosphate buffered saline (PBS) thrice, each rinse lasting for 5 min. The sections were incubated with blocking solution of 3% bovine serum albumin at 37°C for 1 h to block the non-specific antigens. This was then followed by overnight incubation at 4°C using primary antibody, rabbit polyclonal SCD, unconjugated in liquid form (supplied in 1 × PBS buffer with 0.09% (w/v) sodium azide; Biorbyt Ltd, UK). The secondary antibody was conjugated by incubating at 37°C for 1 h and rinsing with PBS thrice for 10 min. Prior to microscopic examination, the sections were developed in diaminobenzidine (DAB) chromogen and counterstained in haematoxylin. Immunohistochemical results were semiquantitatively evaluated in a blinded pattern by two observers. This was observed at a magnification of ×400 using a compound light microscope. Brown staining identified at the perinuclear region of the cells was considered as positive as suggested by Wang et al. 15
A four-point scoring system was used to evaluate the distribution of cells stained per high power field as 0–25% = 1, 26–50% = 2, 51–75% = 3 and >75% = 4. The staining intensity was scored as negative = 0, weakly positive = 1, moderately positive = 2 and strongly positive = 3. A comprehensive score was obtained that was the product of staining intensity and the percentage of cells stained. The immunoscore of SCD was graded as low (<6) and high (≥6) expressions. 15
Statistical analysis
The results of SCD expression were evaluated in relation to different clinicopathological parameters and patients’ survival. Follow-up time for each patient was calculated in months for a period of 3 years. Cumulative survival was measured from the date of diagnosis until death by any cause or until the date of last patient contact. Patients who were alive at the time of last contact were considered as censored observation in cumulative survival analysis. Survival time was considered as censored if there was a time period for follow-up, the event (either death or recurrence) had not occurred. Event time distributions for these endpoints were estimated using Kaplan–Meier method. Cross tabulations were analysed by using the
Results
SCD expression and clinical parameters in OED and NOM
A descriptive analysis was performed to compare the clinical parameters of OED and NOM subjects. The clinical parameters considered were habits, gender and age (Table 1).
Descriptive analysis and comparison of clinical parameters in OED and NOM for SCD.

NOM: normal oral mucosa; OED: oral epithelial dysplasia; SCD: stearoyl coenzyme A desaturase.
Among the 15 NOM cases, 6(40%) reported risk habits, while 9(60%) were without any habit association. Eight (40%) subjects predominantly had smokeless tobacco habit in OED (
Considering the gender distribution within the subgroups, it was observed that within the 15 subjects of NOM group, 8(53.3%) were females and 7(46.6%) were males. Among the 20 OED cases, there was a male predilection (
In the NOM and OED cases, the age range identified was from 18 years to 80 years with a mean age of 49 years. The mean age within the NOM group was 39.7 years, whereas mild, moderate and severe OED cases presented with mean ages of 50.4, 56 and 68.2 years, respectively. A statistical significance with a
Correlation of SCD expression with clinicopathologic parameters in OED, NOM and overall comparison in OSCC.

NOM: normal oral mucosa; OED: oral epithelial dysplasia; OSCC: oral squamous cell carcinoma; SCD: stearoyl coenzyme A desaturase.
Cumulative survival and clinical parameters in OSCC
Based on TNM staging, 20 OSCC cases comprised of 9, 7, 3 and 1 cases each of stages I, II, III and IV, respectively.
Comparison of age, gender, site, habits and grade with cumulative survival was done using univariate analysis (Table 3).
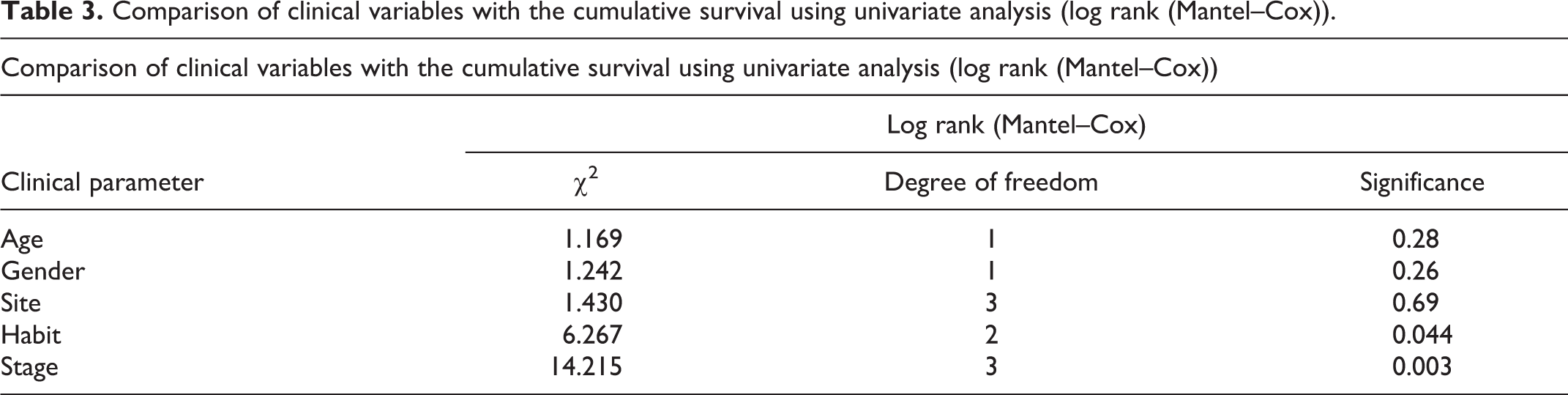
Comparison of clinical variables with the cumulative survival using univariate analysis (log rank (Mantel–Cox)).
The age range of the OSCC cases was 25–80 years, with 52.5 as the mean age. By Kaplan–Meier plot, it was observed that for the subjects above 50 years, the survival rate depreciated by 60–100% in a period of 10–40 months and for those below 50 years, the survival rate diminished to 80–100% in 20–35 months (Figure 1(a)). The events that occurred within the intervals were greater in subjects above 50 years compared to the subjects below 50 years. A statistically significant result was not attained (log-rank (Mantle score) test,
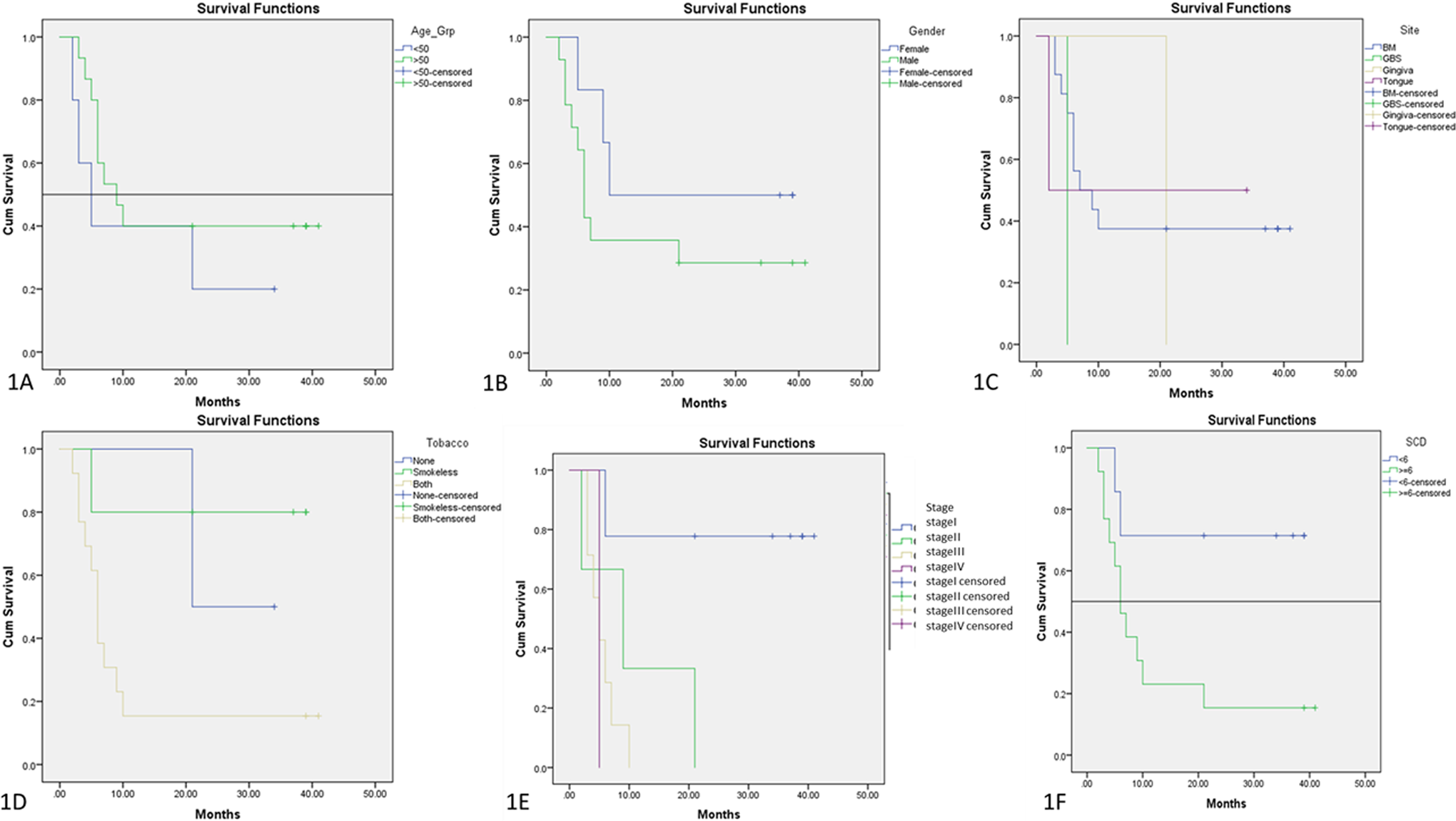
Kaplan–Meier plot comparing the clinicopathological parameters with cumulative survival. (a) Age, (b) gender, (c) site, (d) tobacco use, (e) stage and (f) SCD expression. SCD: stearoyl coenzyme A desaturase.
While considering the survival function by Kaplan–Meier analysis with gender, there was a decreased survival rate by 50% at 10 months in females and by 65% at 7 months in males (Figure 1(b)). This was not statistically significant (log-rank (Mantle score) test,
OSCC cases predominantly presented with tumours on the buccal mucosa, tongue and gingivobuccal sulcus. It was observed that survival rate of subjects with cancers on the tongue diminished to 50% in less than 5 months, whereas in buccal mucosa survival decreased by 65% in 10 months and 100% in cases of gingivobuccal sulcus location in about 5 months. However, due to limited number of samples (approximately 1) from gingiva, their survival rate could not be estimated (Figure 1(c)). The values obtained did not reveal statistically significant results (
There was a decrease in survival rate of non-habit-associated OSCC patients by 50% in 20 months. Subjects who consumed smokeless tobacco showed a decrease in survival rate by 20% in 5 months and those with both habits had a decreased survival rate by 85% in 10 months with increased incidents within the interval (Figure 1(d)). Log-rank (Mantle score) test showed a statistically significant difference with a
Stages IV, III and II OSCC patients showed a diminished survival rate of ≥95% in less than 5, 10 and 22 months, respectively, whereas stage I patients had a survival rate of 22% at 5–20 months (Figure 1(e)). Log-rank (mantle score) analysis gave a statistically significant difference between the OSCC stage and survival rate with a
Kaplan–Meier analysis comparing SCD expression with survival rate in OSCC cases showed diminished survival by 80% in 10 months in cases with IHC score >6, (increased SCD expression) and reduced survival rate by 25% in 5–20 months in cases with expression of SCD <6. The cut-off score was considered as >6 (Figure 1(f)). A statistically significant difference between IHC (SCD expression) scores >6 and <6 with a
Multivariate analysis (omnibus tests of model coefficients) was used to assess the cumulative survival of OSCC patients with the parameters of gender, age, site, habit, grade and IHC score. The analysis revealed an insignificant result (
Multivariate analysis of clinical parameters in OSCC with omnibus tests of model coefficients.

OSCC: oral squamous cell carcinoma; SCD: stearoyl coenzyme A desaturase; both: smokeless and smoked; nil: no habits; WDSCC: well-differentiated squamous cell carcinoma; MDSCC: moderate differentiated squamous cell carcinoma; PDSCC: poorly differentiated squamous cell carcinoma.
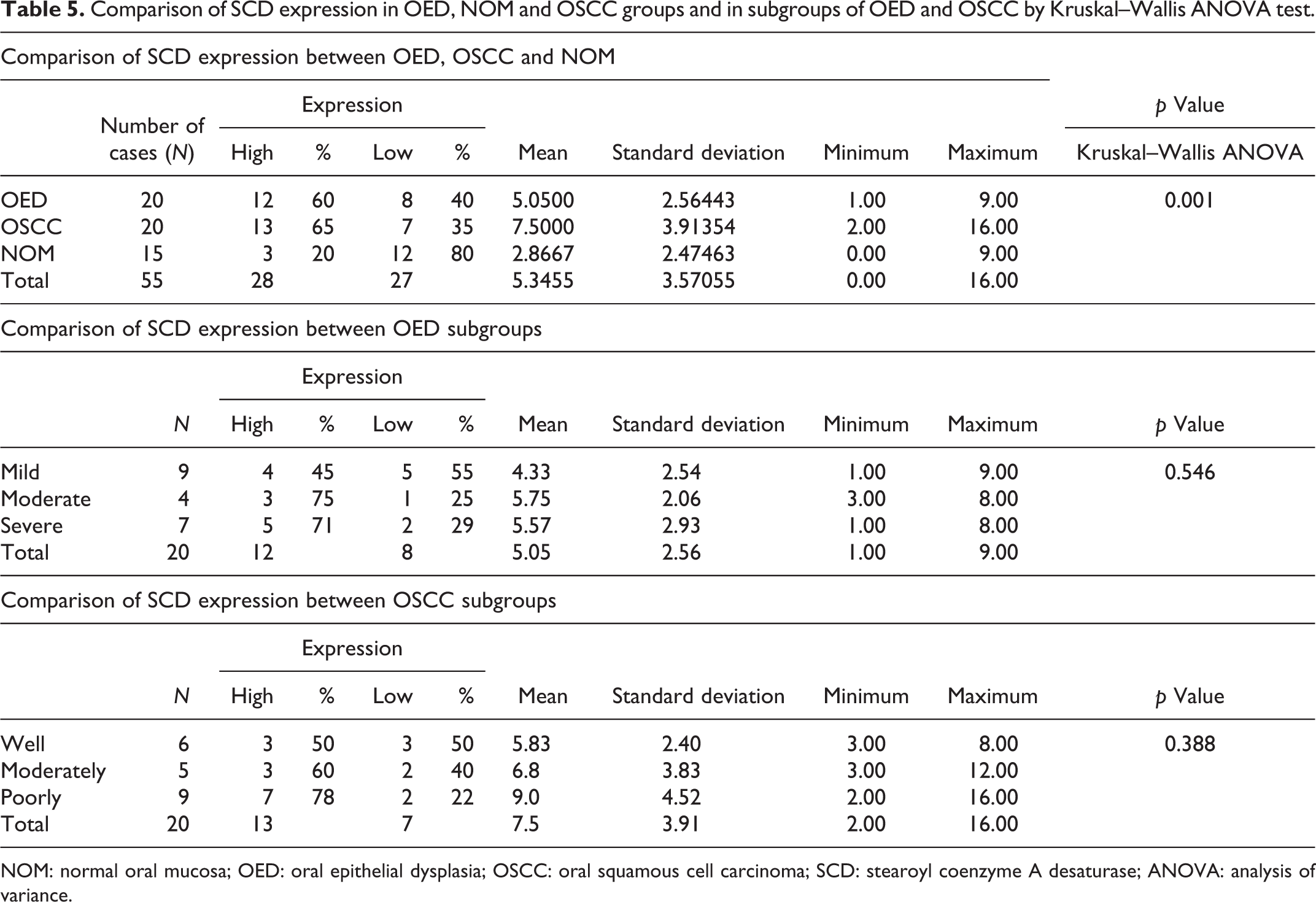
Among the 20 OED cases, 12 (60%) cases showed high expression of SCD in contrast to 8 cases (40%) with low expression. Similarly in OSCC, 13(65%) and 7(35%) cases showed high and low SCD expressions, respectively. Of 15 NOM cases, 12(80%) cases showed low expression and 3 (20%) cases displayed high SCD expression. Comparison of SCD expression between the study groups using Kruskal–Wallis ANOVA test showed a statistically significant difference with a
Comparison of SCD expression in OED, NOM and OSCC groups and in subgroups of OED and OSCC by Kruskal–Wallis ANOVA test.
NOM: normal oral mucosa; OED: oral epithelial dysplasia; OSCC: oral squamous cell carcinoma; SCD: stearoyl coenzyme A desaturase; ANOVA: analysis of variance.

Photomicrographs showing (H&E ×100) of NOM (A), OED (D) and OSCC (G). Photomicrographs of immunostained sections showing perinuclear staining of SCD immunoexpression in NOM (B) and (C), OED (E) and (F) and OSCC (H) and (I). H&E: hematoxylin and eosin; NOM: normal oral mucosa; OED: oral epithelial dysplasia; OSCC: oral squamous cell carcinoma; SCD: stearoyl coenzyme A desaturase.
Comparison between OED and OSCC, Mann–Whitney
Multiple comparison by Mann–Whitney

NOM: normal oral mucosa; OED: oral epithelial dysplasia; OSCC: oral squamous cell carcinoma; WDSCC: well-differentiated squamous cell carcinoma; MDSCC: moderate differentiated squamous cell carcinoma; PDSCC: poorly differentiated squamous cell carcinoma.

Photomicrographs showing (H&E ×100) different grades of OED; mild OED (A), moderate OED (D) and severe OED (G). Photomicrographs showing perinuclear staining of SCD immunoexpression in mild OED (B) (×100) and (C) (×400); moderate OED (E) (×100) and (F) (×400); severe OED (H) (×100) and (I) (×400). H&E: hematoxylin and eosin; OED: oral epithelial dysplasia; SCD: stearoyl coenzyme A desaturase.

Photomicrographs showing (H&E ×100) of WDSCC (A), MDSCC (D) and PDSCC (G). Photomicrographs of immunostained sections showing perinuclear staining of SCD immunoexpression in WDSCC (B) (×100) and (C) (×400), MDSCC (E) (×100) and (F) (×400) and PDSCC (H) (×100) and (I) (×400). H&E: hematoxylin and eosin; WDSCC: well-differentiated squamous cell carcinoma; MDSCC: moderate differentiated squamous cell carcinoma; PDSCC: poorly differentiated squamous cell carcinoma; SCD: stearoyl coenzyme A desaturase.
Among the 20 cases of OED, 9 cases belong to mild OED, while 4 cases belong to moderate OED and 7 cases to severe OED subgroups. In mild OED, five (55%) cases exhibited low expression of SCD and four (45%) cases exhibited high expression. Likewise, in moderate and severe OED, three (75%) and five (71%) cases, respectively, showed high expression, whereas one (25%) and two (29%) cases showed low expression when stained immunohistochemically with SCD. (Table 5 and Figure 3).
An increased expression of SCD was appreciated in higher grades of OED (Figure 3). However, a statistically insignificant difference with a
The OSCC group comprised of 20 cases of which 6 were of well-differentiated squamous cell carcinoma (WDSCC) grade, while 5 and 9 cases were moderate (MDSCC) and poorly differentiated squamous cell carcinoma (PDSCC) grades, respectively. In WDSCC, there were equal number of three cases (50%) showing high and low SCD expression. In MDSCC, three (60%) and two (40%) cases, respectively, showed high and low SCD expression. In PDSCC, seven (78%) cases presented with high expression, whereas two (22%) cases showed low SCD expression. It was observed that PDSCC showed greatest SCD expression when compared to the WDSCC and MDSCC counterparts but there was no statistically significant difference by Kruskal–Wallis ANOVA test (
Moreover, comparison of SCD expression between WDSCC and MDSCC, WDSCC and PDSCC and MDSCC and PDSCC by Mann–Whitney
Discussion
SCD is an iron-containing enzyme involved in mediating the biosynthesis of unsaturated fatty acids. SCD catalyses the -9-cis desaturation of palmitic acid and stearic acid to MUFAs, namely, the palmitoleic acid and oleic acid, respectively. These are principle components of membrane phospholipids, triglycerides and cholesterol esters. 8,21,22 The normal ratio of saturated to unsaturated fatty acids is essential in the regulation of cell growth and differentiation through effects on the physical properties of cell membrane, fluidity and signal transduction. Minute changes in this ratio, and in the amount of available unsaturated fatty acids, can influence the ability of the cell to respond to external stimuli. Variation in cellular fatty acid composition has been associated with a range of pathological conditions including neurological and vascular diseases, diabetes, obesity, hypertension and cancer. 21,22 SCD expression is observed in liver tissue and heart in response to high carbohydrate diet. Also, altered SCD expression and MUFA biosynthesis are observed in metabolic diseases like diabetes, obesity and atherosclerosis. In case of obesity and diabetes targeted cells like adipocytes and hepatocytes, muscle cells exhibit abnormally elevated lipid biosynthetic activity with a consequent overproduction of triacylglycerols and cholesteryl esters. 7 Therefore, elderly subjects and those exhibiting metabolic disorders like diabetes mellitus and cardiovascular disorders were excluded from the study.
In the current study, levels of SCD expression were analysed within varying grades of dysplasia, OSCC and NOM. Studies conducted on renal tissues, in carcinoma of breast, thyroid, pancreas, liver, and so on, have shown that SCD expression can be correlated with tumour aggressiveness and may serve as a prognostic marker. 10,13,15,16,19,20,23
In the current study, it was found that the elderly subjects and those who had habit association were significantly affected with OED when compared to those who were young with no habits. The persistent contact of the oral mucosa with tobacco for a longer time period could initiate and increase the grade of dysplasia. 24
There was a male predilection observed in the present study in OED group as they are highly exposed to deleterious habits in contrast to female gender. However, it was not of statistical significance probably due to limited sample size. This finding was in accordance with Gopinath et al. 25 on prevalence and risk factors for epithelial dysplasia and carcinoma within leukoplakic lesions.
Correlating the clinicopathologic parameters in OSCC in the current study, subjects who were habit associated with both smoked and smokeless tobacco had a significantly poor survival rate when compared to those using a single tobacco product or with no habits.
With respect to stage of OSCC, a significant association was found which deteriorated prognosis in advanced stages. However, subjects with stage I showed comparatively a good prognosis. The stages in OSCC advance with increase in tumour size, with or without infiltration of tumour cells into lymph node/nodes and may or may not be associated with metastasis. With increase in tumour size, the tumour cells tend to infiltrate subjacently, which could be the suggestive reason for reduced survival rate in stages III and IV in OSCC subjects. This finding was inconsistent with the data reported by Tirelli et al. on prognosis of oral cancer and its comparison with AJCC staging. 26
Poor prognosis was noted in OSCC of tongue followed by gingivobuccal sulcus and buccal mucosa, probably due to pooling of saliva containing carcinogens that may enhance the risk of these sites. This was, in accordance with Lee et al. 27 and Gopinath et al., 25 on carcinoma in dysplasia in Taiwan and south Indian populations, respectively. Male patients and those of >50 years of age had diminished survival rates when compared to females and subjects below 50 years age group, respectively. However, the latter two observations did not show significant results. Sharp cusps due to attrition that can cause trauma and chronic inflammation at the local region along with lower degree of keratinization and higher permeability of mucosa to carcinogens in these regions could be the suggestive reasons for their diminished survival.
An increased expression of SCD was noted in OED when compared to NOM; however, a statistical significance was not noted. Correlating the SCD expression with survival rate in OSCC patients revealed an increased SCD expression with diminished survival rate, thereby predicting a poor prognosis. This finding was in accordance with study conducted by Wang et al. on the association of high expression of SCD with poor prognosis in patients with clear cell renal cell carcinoma. 15
Univariate analysis was applied in the present study to compare clinical parameters with the survival rate of OSCC.
The present study showed significantly high expression of SCD in OED and OSCC when compared to that of NOM. This was in accordance with Roongta et al., where expression of SCD1 (an isoform of SCD) was relatively low in normal tissues in comparison to tumour tissues of colon, breast, lymphoid, prostate, gastric, ovarian, brain, kidney, liver, lung and skin. 14 Studies conducted by Belkaid et al. showed an enhanced expression of SCD in breast and prostate cancer tissues when compared to normal tissues. The authors have inferred that desaturated fatty acids are necessary for the survival of tumour cells and SCD can be a viable target for the development of novel agents for cancer therapy. 11
In case of OED, an increased expression of SCD was noted in higher grades of OED. Assay by Guo et al. 10 showed that chronic tobacco chewing induces molecular changes in a cellular system that causes dysplasia and eventually malignancy with increase in SCD expression. This observation was in accordance with the current study results where there was an increased SCD expression with the increase in grades of dysplasia.
Evaluation of OSCC cases exhibited higher expression of SCD with increase in the grades of carcinoma. PDSCC showed greatest SCD expression when compared to the WDSCC and MDSCC but there was no statistically significant difference. Similar results were observed in the studies done by Guo et al., 10 Wang et al., 15 Kim et al., 28 Noto et al., 29 Chen et al., 30 Noto et al., 31 Falvella et al., 32 Nie et al., 33 Walz et al. 34 and Hess et al., 35 where poor prognosis was noted with high SCD expression in cancers of breast, colon, liver, thyroid, kidney, oesophagus, prostate, lung adenocarcinoma, ovarian tumour and so on.
Unsaturated fatty acids are important precursors for various products in the cell, like phospholipids in the cell membrane, triglycerides for energy storage and diacylglycerols in signalling. However, contrasting results have been observed by Moore et al. on prostate carcinoma tissues and have stated that SCD catalyses the desaturation of palmitate and stearate to palmitolate and oleate. Alterations in the fatty acid composition could bring about an alteration in cell function and compromise the survival. 21,22
In the present study, we show a positive correlation of SCD expression with increased grades of OED and OSCC. The recognition of high SCD expression may alert the surgeon about the aggressive nature of potentially malignant and malignant oral lesions.
However, the limitations of the study are lack of objective method for assessing the expression levels and smaller sample size among the subgroups. Further research on specific mechanisms of SCD with follow-up cases, using larger samples with objectivity in assessment, is essential to determine its prognostic significance. SCD activity may be targeted using genetic and molecular methods to inhibit cell growth and induction of apoptosis. Therefore, SCD may act as a novel molecular target for the treatment of advanced stages of OED and OSCC patients.
Conclusion
In the present study, expression levels of SCD were compared among three independent groups, namely, NOM, oral potentially malignant and OSCC lesions. Cases diagnosed with OSCC and OED showed a higher SCD expression when compared to NOM, suggesting its clinical application. Among the OSCC and OED groups, a higher expression was seen in OSCC; within the subgroups, the SCD expression increased with increase in grades of dysplasia and by level of differentiation of OSCC which indicates that protein expression is correlated with the severity of the disease. Clinical parameters of tobacco usage and staging of OSCC showed a significantly positive correlation with the cumulative survival of patients. Also, the higher expression of SCD was associated with diminished survival that may alert the surgeon in predicting the clinical behaviour and prognosis of OED and OSCC patients, thereby enhancing the role of SCD as an independent prognostic biomarker.
Footnotes
Acknowledgements
The authors would like to thank Dr Puneet Gupta for the statistical analysis and also Mr Sriranga Raju and Mr Chetan Kumar for the technical support.
Authors’ contribution
Study design by RSR, SSV, VCH and SGP; literature review by MA, VCH, SGP and DA; manuscript editing by RSR, SGP, DA and SSV; manuscript review by RSR, DA, SN and SGP.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
