Abstract
There are similarities between the clinical conditions associated with genital and oral mucosal human papillomavirus (HPV). The role of HPV infection in oral carcinogenesis is currently controversial. Also, factors underlying this infection are still unclear. The frequent location of HPV lesions in areas of trauma indicates the role of mechanical irritation as a necessary event for the penetration and/or reactivation of the virus into the epithelium. Oral mucosa is frequently exposed to mechanical irritation, as it is in constant and intimate association with hard tissues. During the oral HPV infection, favorable conditions must be generated for the infection to be successful. Some of them, associated with proliferation of basal keratinocytes, are related to the presence of external stimuli, such as chronic mechanical irritation (CMI). The objective of this work is to demonstrate, through the presentation of three clinical cases, the possible role of CMI in the pathophysiology of HPV lesions of the oral mucosa.
Introduction
In 1977, Zur Hausen suggested human papillomavirus (HPV)’s carcinogenic probability on genital mucosa. 1 Following this, more knowledge was gained on HPV and its interactions with host cells, tissues, and the immune system. In 1983, Syrjänen et al., based on consistent morphological and immunohistochemical studies, proposed that HPV might be involved in oral carcinogenesis. 2 In 2011, the International Cancer Research Agency indicated the existence of a causal association between HPV and oral cancer, proposing it as an independent risk factor. 3,4 Today, more than 200 types of HPV genotypes have been characterized, and more than 40 of these genotypes can infect the female genital tract. 3 The role of HPV in cervical cancer is indisputable. However, some factors underlying this infection are still unclear. 5 HPV lesions are usually located at sites exposed to mechanical irritation, such as fingers, elbows, knees, anus, and perineum. 6 The frequent location of HPV lesions in areas of trauma indicates the role of mechanical irritation as a necessary event for the penetration of the virus into the epithelium.
There are similarities between the clinical conditions associated with genital and oral mucosal HPV. The role of HPV infection in oral carcinogenesis is currently controversial, as the detection rates using molecular biology techniques are widely variable (from 0% to 100%). 7 One way of HPV transmission to the oral mucosa is through sexual contact. HPV infection may be due to orogenital sex and risky sexual behaviors. People who practice oral sex have a 50% more chance of acquiring the virus. Tongue, lips, palate, and floor of mouth are the most frequent places where infection begins. 8 Also, the virus may be transmitted by mouth-to-mouth contact, autoinoculation, or vertical transmission in the birth canal. 9
Successful infection with HPV occurs when the virus manages to enter and bind to basal keratinocytes. Some authors indicate that some membrane receptors are involved in this process. These cell receptors are expressed during cellular migration and proliferation. In a study on rabbits that addresses the relationship between microerosions and viral infection, the wound created 3 days prior to the inoculation significantly increased the efficiency of the infection, allowing us to understand the importance of the inflammatory microenvironment created around the wound. 10
Oral mucosa is frequently exposed to mechanical irritation, as it is in constant and intimate association with hard tissues such as teeth, dentures, and so on. In turn, when there is an injurious agent (ill-fitting dentures, sharp cusps, damaged dental restorations, parafunctional habits etc.) 11 which generates chronic mechanical irritation (CMI), basal keratinocytes overexpress syndecan 1 and thus may strongly upregulate the ability to attach and internalize HPV. The permissiveness for HPV is an important event for establishing a persistent infection and a biological requirement for the viral propagation and carcinogenesis. 12 Unlike in other mucosal sites, the relationship between CMI and oral mucosa is an important consideration to be taken into account in the oral HPV infection.
There are many studies in the current literature addressing the causal relationship between HPV and oral cancer. However, there are no studies where CMI is registered as a cofactor in association with HPV. The objective of this work is to demonstrate, through the presentation of three clinical cases, the possible role of CMI in the pathophysiology of HPV lesions of the oral mucosa. This work was approved by the Health Science Ethical Committee, Dental College, Universidad Nacional de Córdoba, Argentina, in accordance with the Helsinki Declaration.
Case 1 involves a 25-year-old young adult male who attended with 1-month evolution of a lesion located on the lingual frenulum. An elevated, pedunculated lesion was observed, with multiple digitiform elements. In addition, the patient had both lower central incisors with enamel angle fractures, with a sharp surface. Among his sexual habits, cunnilingus was a frequent sexual behavior (Figure 1). The lesion was surgically removed. The histopathological diagnosis was condyloma acuminatum, and HPV 6 was found in the excised tissue examined by polymerase chain reaction (PCR) technique. Mechanical irritation caused by the sharp lower incisors generated during oral sex around the lingual frenulum could have allowed the virus to access the epithelium and its subsequent infection and development of the lesion.

Wart-like lesion (condylomata acuminatum) on the lingual frenulum and related to sharp incisal edges of lower teeth.
Case 2 involved a 74-year-old adult female who attended with multiple white lesions. The most significant lesion was an asymptomatic slightly raised white plaque on the lower right residual ridge. The lesion was located on the denture bearing area, where one of the attachments supporting the denture was fractured a year ago, generating continuous friction between the contacts of a rough denture surface with the alveolar ridge mucosa (Figure 2(b)). HPV16 was detected by PCR and the lesion was surgically removed. The histopathological findings of the hematoxylin–eosin-stained tissue revealed a mucosa fragment with an acanthotic-stratified squamous epithelium exhibiting parakeratosis, koylocitosis, and HPV-associated moderate dysplasia (Figure 2(a)). The epithelial–connective tissue junction was completely preserved. These HPV-associated histopathological features were described as koilocytic dysplasia by Fornatora et al. 13 The persistent mechanical irritation of a rough surface on the probably infected mucosa could have reactivated the virus and contributed to the progression of the lesion.
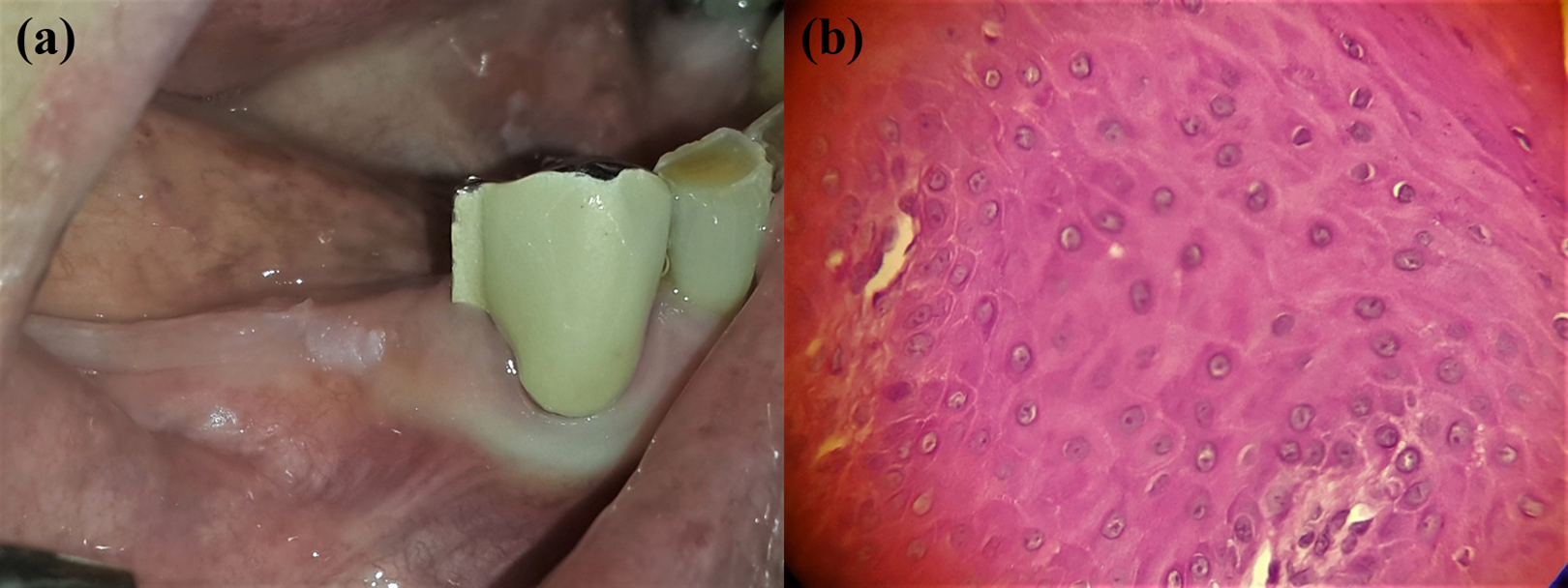
(a) Circumscribed hyperkeratotic plaque, close to fractured prosthetic attachment. (b) Basal and intermediate squamous cells with large and irregular perinuclear halos (koilocytes).
Case 3 involves a 55-year-old adult female who attended the dental office with a lesion located on the lateral border of the tongue. During oral examination an erythroleukoplakia with an indurated area was observed close to the lower left bicuspids that were lingually inclined. A metal component of a previous prosthesis was fixed to the alveolus devoid of its denture. As the patient did not use her lower denture, the lingual border of fixed metal object impacted against the tongue during swallowing. (Figure 3 (a)). The patient neither smoked nor drunk alcohol. Toluidine blue–guided incisional biopsy was performed (Figure 3(b)).The histopathological diagnosis was a microinvasive squamous cell carcinoma (Figure 3(c) and (d)), and HPV16 was detected by PCR. The dental and prosthetic factors and functional CMI could have facilitated and induced the progression of the viral cycle.

(a) Speckled leukoplakia-like lesion located on the left tongue border. (b) Toluidine blue staining allowed to recognize dysplastic suspicious zones. (c) A microinvasive squamous cell carcinoma. (d) Epithelium with evident dysplastic changes and koilocytes affecting all the epithelial thickness.
Methods of HPV detection
In all cases, for HPV detection, lesional cells were obtained by brushing with a sterile endocervical cytobrush. Viral DNA was extracted corresponding to the L1 region of the viral genome. It was amplified by PCR with MY09 and MY11 primers. Further, HPV-DNA-positive samples were typed by restriction fragment length polymorphism and mixed with restriction enzymes. Finally, the obtained pattern was compared with the published data.
Discussion
Epithelial infection with HPV begins when the host cell expresses membrane receptors that allow binding to the virus and its subsequent entry. The first basal and parabasal keratinocytes receptors involved in this process are alpha 6 beta 4 integrins and syndecan 1. The overexpression of these receptors after superficial injuries in the epithelium 14 shows that microabrasions could exist prior to the infection and the presence of HPV-associated lesions in areas affected by CMI. This is seen in case 1 where there is an irritative factor at the site of the lesion in a young male adept at oral sex practices. Figure 4 (a) to (d) illustrates the possible role of CMI in oral mucosal HPV infection.

(a) and (b) A traumatic condition begins to irritate healthy epithelium of the oral mucosa. (c) A traumatic microerosion allows the exposure of the basal keratinocytes to the oral microenvironment. Basal cells express membrane-specific receptors such as syndecan 1. (d) Successful infection with HPV occurs when the virus manages to bind to specific receptors. (e) Viral latency is restricted to the basal layer of the epithelium. (f) to (h) If the latently infected mucosa is associated with a CMI condition, microerosions are generated again and stimulate basal cells proliferation. (i) and (j) The virus reactivates due to a mechanical stimulus and begins to develop an HPV-related lesion. Modified from Gravitt. 15 CMI: chronic mechanical irritation.
HPV infection may be acute, chronic, or latent, but unlike other viruses, HPV latency’s mechanism is not exactly defined. The latency of HPV can be modified by various epithelial or microenvironmental conditions, leading to the recrudescence of the disease. 14 The persistence of the virus in the absence of a clinical lesion does not necessarily exclude viral activity because during latency there are low levels of genomic replication and production of viral transcription proteins. 16 During the latency phase, HPV can be removed (clearance) from the infected epithelium through a process mediated by Langerhans cells 17 or begin to synthesize new viral particles leading to its reactivation. 16
Evidence of HPV latency in human infections is based on clinical observations but is always anecdotal evidence because of ethical concerns. These observations led researchers to develop experimental animal models with compelling results that provide scientific evidence on HPV reactivation caused by CMI. Those animal models included cattle bladder cancer associated with bovine papillomavirus (BVP4), equine skin tumors associated with BVP 1 and 2, rabbit oral papillomavirus, canine oral papillomavirus, and cotton tail rabbit papillomavirus (CRPV). Tumors associated with BPV were found in places of mechanical irritation subsequent to infection, suggesting the possible reactivation of the latent virus. A study of
It is unethical to follow the role of CMI as a relevant ancillary event for oral HPV infection. Oral HPV lesions and HPV-associated potentially malignant disorders must be treated and the treatment should involve the control of local CMI factors. In these three cases, there was no proven HPV-PCR diagnosis before CMI events, but the evolution of these lesions (especially in cases 2 and 3) before the CMI events was carefully recorded by anamnesis. Due to this ethical unfeasibility, the available evidence is limited to different papillomavirus experimental animal models. However, in vivo research could be applied to clinical situations, allowing us to confirm the role of CMI in the oral HPV infection and reactivation.
CMI experimentally generates not only a hyperproliferative epithelial state but also an inflammatory microenvironment that acts as a promoter of cell growth. The mechanism by which CMI contributes to oral carcinogenesis is not currently identified; however, an epithelial wound would facilitate the absorption of other carcinogens, promoting increased mitosis of the germ strata in response to an attempt to repair tissue damage. 20,21 In addition, Tezal described a possible association of chronic inflammation and viral replication, taking into account the basal cell proliferation generated by epithelial microabrasions. 22 The presence of HPV in CMI areas, as in cases 2 and 3, could be a risky association, because the CMI would facilitate the progression of viral infection. Such a link could have been a key factor in the development of dysplasia (case 2) and progression of carcinoma (case 3).
Gravitt proposed that cervical HPV latency could be reactivated by the mechanical irritation generated during sexual intercourse. The mechanical stimulus on the infected basal cells would stimulate the cellular machinery and the transcription of viral proteins. Likewise, infected basal cells begin to differentiate, promoting the healing of such wounds and, thus, reactivate HPV infection as described above. 15 This proposed mechanism could be easily reproduced in the oral cavity, due to the irritative effect exerted by defective teeth and ill-fitting dentures on the mucosal surface (Figure 4(f) to (j)).
In this work, we have presented some clinical cases in which after the CMI events, an HPV-associated lesion appeared, proving its presence with molecular biology techniques. Our data corroborate with the findings of Perry et al. who observed different lesions caused by HPV that developed in CMI areas, highlighting the role of the virus in oropharyngeal carcinogenesis, even in those patients who do not have traditional risk factors such as tobacco and alcohol consumption. 23
Conclusion
CMI could generate microenvironmental conditions that allow the penetration and reactivation of HPV in the oral mucosa. Therefore, CMI should be considered as a cofactor in the process of oral carcinogenesis. In turn, this suggests that CMI must be recorded in studies that address HPV infection in oral mucosa, related to or not related to oral cancer. Further, due to the limitation of this few cases presented in this review, more studies with a strong evidence-based study design are needed to elucidate the relationship between CMI and oral HPV infection.
In the prevention of CMI of the oral mucosa, dentists could play a key role, since they are capable of the detection and treatment of CMI factors. It would be very important to consider CMI as a conditioning element of HPV infectious cycle in the prevention and early detection of oral cancer.
Footnotes
Acknowledgements
The authors would like to thank Dr Ruth Ferreyra, Oral Pathology Department, Dentistry College, Universidad Nacional de Córdoba, for her support with the histopathological figures. This case report and review of the literature was carried out in the framework of the doctoral thesis titled “Differentiation and Proliferation Cell Biomarkers in Chronic Mechanical Irritation of the oral mucosa and its relationship with biological factors of carcinogenic risk.”
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
