Abstract
Objectives:
Curcumin has been shown to exhibit anti-neoplastic effects. However, due to its poor bioavailability, the use of curcumin as an anti-cancer drug is limited. Thus, it is necessary to identify molecules as an alternative to curcumin that could serve as anti-cancer targets. In this study, we attempted to understand the underlying curcumin-mediated signalling pathways contributing to anti-neoplastic effects of curcumin.
Methods:
We carried out mass spectrometry-based phosphoproteomic analysis of head and neck cancer cell line, CAL 27, treated with and without curcumin to identify curcumin-mediated signalling pathways. Serine/threonine kinases were enriched using titanium dioxide.
Results:
This resulted in the identification of 5921 phosphopeptides corresponding to 1878 proteins. Of these, 275 and 183 phosphopeptides corresponding to 335 and 242 proteins (≥2.0-fold) were found to be hyper- and hypo-phosphorylated, respectively, in response to curcumin treatment. Calcium/calmodulin-dependent protein kinase kinase 2 (CaMKK2), a serine/threonine kinase, and its downstream target protein kinase AMP-activated non-catalytic subunit beta 1 (PRKAB1) were found to be hypo-phosphorylated when treated with curcumin. Further, silencing or inhibiting CaMKK2 resulted in decreased invasion and colony forming ability of not only CAL 27 cells but also other head and neck squamous cell carcinoma (HNSCC) cell lines. Further, Western blot analysis showed that curcumin-mediated signalling is corroborated by CaMKK2.
Conclusions:
Taken together, our results suggest that CaMKK2 could be a novel therapeutic target in HNSCC and can serve as an alternative to curcumin.
Keywords
Introduction
Curcumin, chemically known as diferuloylmethane, is the active component derived from
Curcumin exerts its effects by targeting multiple proteins such as enzymes, receptors, proteins implicated in cell signalling, cell cycle, regulation, various chemokines, cytokines and cell adhesion. 7 However, the use of curcumin is limited due to its low solubility and bioavailability, faster metabolism and rapid excretion from the system. 8,9 However, several attempts are being made to overcome these problems that limit the utility of curcumin. One of the approaches that could be employed is to identify the downstream signalling pathways mediated by curcumin and target molecules pertaining to those pathways. We have previously reported perturbations in various tyrosine kinases in response to curcumin in HNSCC. 10 However, in our previous study, we did not study the role of altered kinases that could be targeted as an alternative to curcumin.
Serine/threonine kinases (STKs) play an important role in maintaining cellular homeostasis by regulating various transcription factors and cell cycle proteins. Dysregulation of STKs has been associated with various tumours where they are known to regulate tumour growth and metastasis. 11 To gain more insights into curcumin-mediated effects, in this study, we profiled STKs and their substrates regulated by curcumin. Stable isotope labeling with amino acids in cell culture (SILAC)-based phosphoproteomic analysis of an HNSCC cell line, CAL 27 treated with curcumin and/or dimethyl sulfoxide (DMSO) was carried out. Phosphoproteomic analysis of curcumin-treated CAL 27 cells resulted in the identification of 5921 phosphopeptides, among which 275 and 183 phosphopeptides were found to be hyper- and hypo-phosphorylated (2-fold), respectively, in response to curcumin treatment. Among the kinases dysregulated, calcium/calmodulin-dependent protein kinase kinase 2 (CaMKK2) was found to be hypo-phosphorylated at Ser-100 (0.3-fold) upon curcumin treatment.
CaMKK2, a non-receptor STK, is known to play a role in calcium/calmodulin-dependent kinase signalling cascade. 12 This signalling cascade plays an important role in cell proliferation, differentiation and metabolism. CaMKK2 has been reported to be elevated in gastric, prostate and hepatic cancers. 13 –15 CaMKK2 has been shown to regulate growth, migration and progression of prostate cancer cells. 13,16,17 Previous study from our group demonstrated that silencing or inhibiting CaMKK2 activity inhibited cellular proliferation, colony formation and invasive ability of gastric cancer cells. 15 In this study, we determined the role of CaMKK2 as an alternative to curcumin in HNSCC.
Materials and methods
Cell culture conditions
CAL 27 and FaDu cells were purchased from the American Type Culture Collection (ATCC, Manassas, VA). JHU-O22, JHU-O28 and FaDu cells were grown in Roswell Park Memorial Institute (RPMI) medium supplemented with 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin mixture. CAL 27 cells were grown and maintained in Dulbecco’s Modified Eagle Medium (DMEM) containing 10% FBS and 1% penicillin/streptomycin mixture. All cells were grown at 37ºC in a humidified incubator supplied with 5% CO2.
SILAC labelling and sample preparation for mass spectrometry analysis
SILAC labelling of CAL 27 cells was carried out as described in our previous study. 10 CAL 27 cells grown in regular media were treated with curcumin at a dose of 11.5 µM for 4 h and cells cultured in SILAC media were treated with DMSO. Post-treatment, the cells from both the conditions were washed with 1× phosphate buffered saline (PBS) and harvested in urea lysis buffer (20 mM (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid) [HEPES] (pH8.0), 9 M urea, 1 mM sodium orthovanadate, 2.5 mM sodium pyrophosphate and 1 mM β-glycerophosphate). The samples were then sonicated and centrifuged at 13,000 rpm at 15°C for 20 min. Then, protein concentration was determined by bicinchoninic acid (BCA) assay 18 and equal amount of protein lysate from both the conditions was mixed and subjected to reduction, alkylation and protein digestion, as described previously. 10
Basic reversed phase liquid chromatography fractionation
Peptides were fractionated by basic pH reverse phase liquid chromatography as described previously. 19 Briefly, 10 mg of lyophilized peptides mixture was resuspended in 1 ml of basic reversed phase liquid chromatography (bRPLC) solvent A (7 mM triethylammonium bicarbonate [TEABC], pH 8.5) and fractionated by bRPLC chromatography on a XBridge C18, 5 µm 250 mm × 4.6 mm column (Waters, Milford, MA) by employing an increasing gradient of bRPLC solvent B (7 mM TEABC, pH 8.5, 90% acetonitrile) on an Agilent 1100 LC system (Agilent Technologies, Waldbronn, Germany). A total of 96 fractions were collected which were then concatenated into 12 fractions and lyophilized.
Enrichment of phosphopeptides using titanium dioxide
Titanium dioxide (TiO2)-based phosphopeptide enrichment of 12 fractions obtained was carried out as described by Larsen and group. 20 Briefly, TiO2 beads were incubated with 2, 5-dihydroxybenzoic acid (DHB) solution (80% acetonitrile [ACN], 1% trifluoroacetic acid [TFA], 3% DHB) for 2–4 h at room temperature. Each fraction was resuspended in DHB and incubated with pre-treated TiO2 beads (1 mg). Phosphopeptide-bound TiO2 beads were washed three times with DHB solution and twice with 40% ACN. Peptides were eluted three times with 40 μl of 2% ammonia into 10 μl of 20% TFA.
LC-MS/MS and data analysis
LTQ-Orbitrap Velos mass spectrometer (Thermo Fischer Scientific, Bermen, Germany) interfaced with Proxeon Easy nLC system (Thermo Fischer Scientific) was used for the analysis of the bRPLC-fractionated phosphopeptides as described previously. 10
The raw data obtained were processed using Proteome Discoverer software suite version 1.4.1.14 (Thermo Fisher Scientific) and searched using Sequest and Mascot search algorithms against human RefSeq protein database 59 and trypsin as proteolytic enzyme. The search criteria included carbamidomethylation at cysteine as fixed modification, oxidation at methionine, phosphorylation at serine, threonine and tyrosine and SILAC labels 13C6-Lysine; 13C6-Arginine as variable modifications with maximum of two missed cleavage. Precursor and fragment mass tolerance were set to 20 ppm and 0.1 Da, respectively. The peptide and protein data were extracted using high peptide confidence and top one peptide rank filters. The data were also searched against a decoy database to calculate the false discovery rate (FDR). Peptide spectrum matches at 1% FDR were used for protein identifications. Precursor ion quantifier node was used for peptide quantification, and PhosphoRS node (version 3.1) was used for the calculation of phosphosite probabilities. Further phosphopeptides with phosphosite probabilities ≥75% were used for the identification of protein phosphorylation sites.
Availability of data
Data generated from mass spectrometry analysis has been submitted to and can be accessed at ProteomeXchange Consortium (http://www.proteomecentral.proteomexchange. org) via the PRIDE public data repository and can be accessed using the data identifier – PXD007256.
MTT assay
To determine the effects of curcumin on HNSCC cell viability, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay was carried out as per manufacturer instructions (ATCC 30-1010K). Briefly, JHU-O22, JHU-O28 and FaDu cells were seeded at a density of 8 × 103 and treated with varying doses of curcumin ranging from 0 to 25 µM for 48 h at 37ºC. Post-incubation, MTT reagent was added and incubated for 2–4 h until purple formazan crystals formed. The purple crystals were solubilized using 100 µl detergent solution and the absorbance was read at 570 and 650 nm.
Colony formation and invasion assays
Colony formation and invasion assays of CAL 27, JHU-O22, JHU-O28, FaDu and CAL 27 cells were carried out as described previously. 21
Western blot analysis
Western blot analysis of the samples was carried out as described previously. 21 Cells were grown to 70–80% confluence and proteins were harvested in radioimmunoprecipitation assay buffer (RIPA) lysis buffer (10 mM Tris pH 7.4, 150 mM NaCl, 5 mM EDTA, 1% Triton-X-100, 0.1% SDS containing protease and phosphatase inhibitor cocktails) and sonicated. The cell lysate of 30 µg was resolved by SDS-PAGE and transferred onto nitrocellulose membrane. The membrane was blocked with 5% non-fat dry milk or 5% bovine serum albumin in PBS-T and incubated overnight with indicated antibodies. Then secondary antibody was added and proteins on the membrane were visualized using enhanced chemiluminescence detection kit as per manufacturer’s instructions. β-actin was used as a loading control. CaMKK2 antibody was purchased from Sigma (St Louis, Missouri, USA). Total AMPK-α and p-AMPK-α (T172) and total AMPK-β1 and p-AMPK-β1 (S108) were purchased from Cell Signaling Technology (Beverly, Massachusetts, USA).
Results
Identification of signalling pathways altered in response to curcumin
In our previous study, we reported that curcumin inhibits cellular proliferation, colony formation and invasion ability of HNSCC cell line, CAL 27. Mass spectrometry–based analysis revealed dysregulation of various tyrosine kinases which may be involved in the anti-neoplastic effects of curcumin. 10 In this study, we studied the effects of curcumin on STKs using quantitative mass spectrometry–based approach. We investigated the molecular alterations of curcumin-induced signalling using SILAC-based quantitative phosphoproteomic analysis. Cells treated with DMSO (control cells) were adapted to ‘heavy’ SILAC media, whereas the cells grown in ‘regular’ media were treated with curcumin for 4 h. Post-curcumin treatment, cells were harvested, lysed and equal amounts of lysates were pooled and digested with trypsin. The phospho-Ser/Thr peptides were enriched by TiO2-based enrichment method and analysed on LTQ-Orbitrap Velos mass spectrometer. The schematic workflow of SILAC-based phosphoproteomic analysis is shown in Figure 1. The data acquired were processed and searched using the search algorithms, SEQUEST and MASCOT, against human RefSeq database. This resulted in the identification of 5921 phosphopeptides corresponding to 1878 proteins. Among these, 275 and 183 phosphopeptides corresponding to 335 and 242 proteins were found to be hyper- and hypo-phosphorylated (≥2.0-fold) upon curcumin treatment, respectively. A complete list of proteins and their phosphopeptides is available on request from the authors.
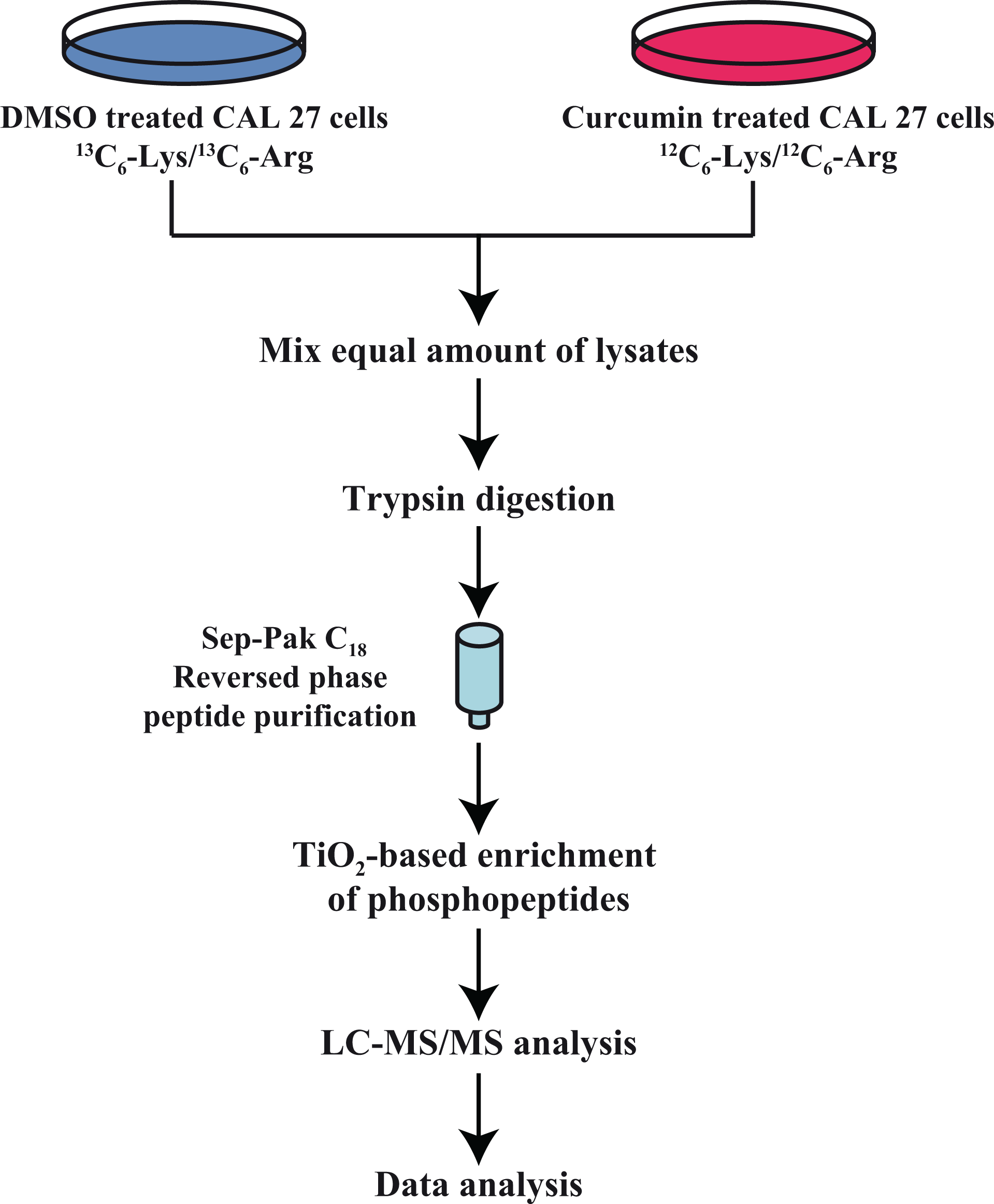
Experimental strategy followed to study curcumin-mediated anti-neoplastic effects in HNSCC cells: CAL 27 cells were treated with curcumin and DMSO for 4 h. Cells treated with DMSO served as control and were cultured in SILAC medium and CAL 27 cells treated with curcumin were cultured in regular DMEM. Post-treatment, cells were serum-starved, harvested and lysed. The lysates were then subjected to reduction, alkylation and trypsin digestion. The digested peptides were then enriched for serine/threonine phosphopeptides by TiO2-based enrichment method and subjected to LC-MS/MS analysis. HNSCC: head and neck squamous cell carcinoma.
Curcumin induces hyper- and hypo-phosphorylation of several STKs.
Mass spectrometry analysis revealed aberrations in various kinases and their downstream targets upon exposure of CAL 27 cells to curcumin. A partial list of proteins found to be hyper- and hypo-phosphorylated is given in Table 1. Kinases including protein tyrosine kinase 2 (PTK2) (S910; 3.7-fold) and STK 17a (STK17A) (S28; 2.8-fold) were found to be hyper-phosphorylated in CAL 27 treated with curcumin. Other kinases including mitogen-activated protein kinase 1 (MAPK1), mitogen-activated protein kinase 3 (MAPK3) and protein kinase D2 (PRKD2) were found to be hypo-phosphorylated at multiple sites upon curcumin treatment (Table 1). CaMKK2, an STK (S100; 0.33-fold), and its downstream targets including protein kinase, AMP-activated, beta 1 non-catalytic subunit (PRKAB1 or AMP-dependent protein kinase (AMPK)-β1) (S108-0.35), acetyl-CoA carboxylase 1 (ACC1 or ACACA) (S62, S66; 0.067; S60-0.084; S117- 0.11), p21 protein (Cdc42/Rac)-activated kinase 2 (PAK2) (S197; 0.3) and glucosamine-fructose-6-phosphate aminotransferase [isomerizing] 1 (GFPT1) (T262; 0.6) were found to be hypo-phosphorylated upon curcumin treatment (Figure 2). Our data indicate an orchestrated downregulation of CaMKK2 and its targets, suggesting that CaMKK2-mediated signalling could be one of the pathways through which curcumin mediates its anti-neoplastic effects. In this study, we determined whether targeting CaMKK2 and its downstream targets could serve as an alternative to curcumin in HNSCC patients. Representative MS/MS spectra of CaMKK2 and AMPK-β1 are given in Supplementary Figure 1.
Partial list of proteins that are hyper- or hypo-phosphorylated in response to curcumin in CAL 27 cells.

CAMKK2: calcium/calmodulin dependent protein kinase kinase 2.
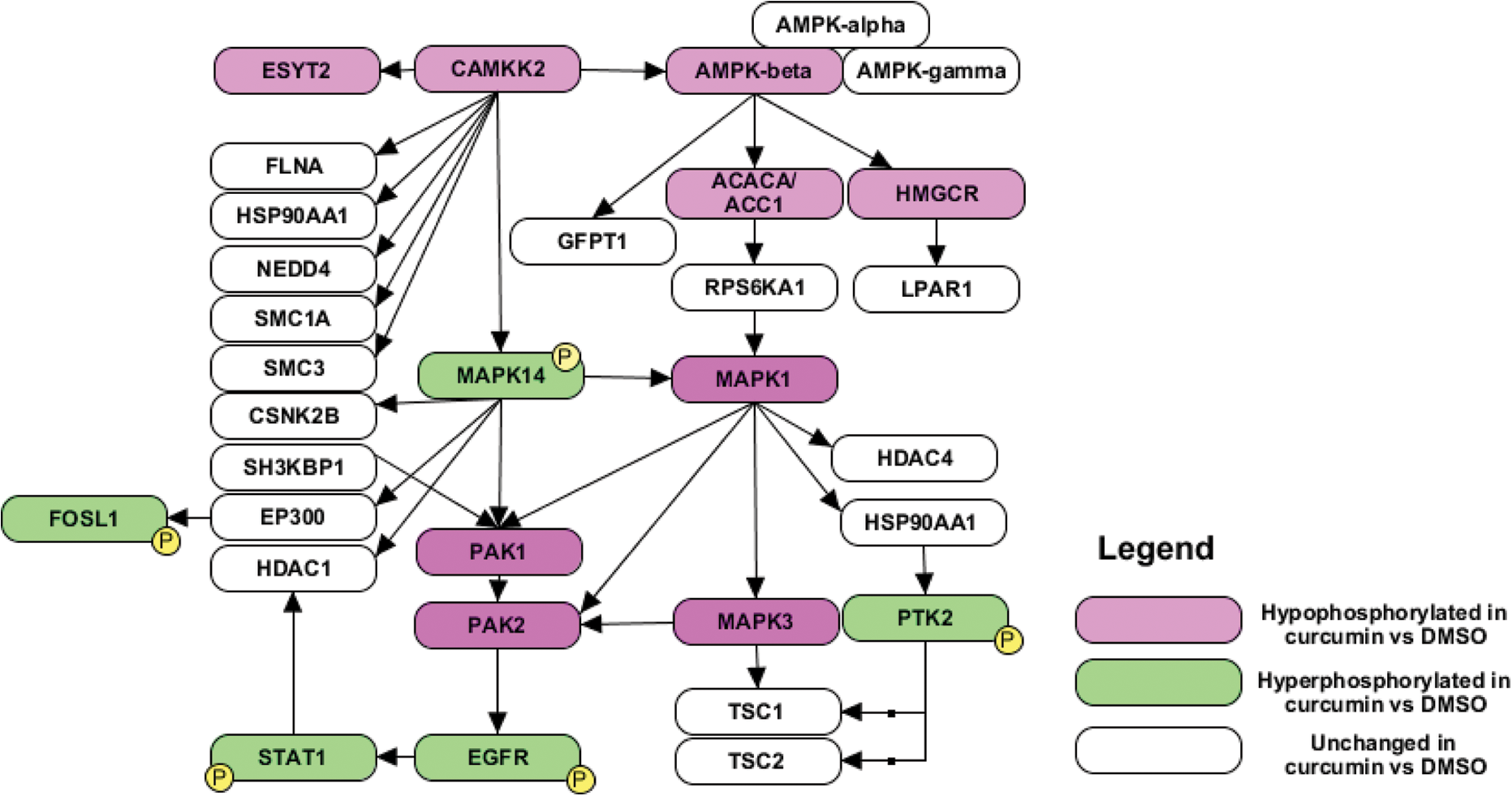
A schematic diagram showing CaMKK2 and its interactors identified in the study. CAMKK2: calcium/calmodulin dependent protein kinase kinase 2.
Curcumin inhibits invasion and colony forming ability of HNSCC cells
In our previous study, we showed that curcumin significantly inhibited colony forming and invasive ability of CAL 27 cells. Here, we studied the effects of curcumin on other HNSCC cells lines. We observed that treatment with curcumin led to decreased invasive and colony forming ability of HNSCC cell lines FaDu, JHU-O22 and JHU-O28 (Figure 3(a) to (d)). In addition, curcumin was found to inhibit the proliferation of these cells (data not shown). Taken together, these results suggest that curcumin inhibits cellular proliferation, colony formation and invasive ability of HNSCC cells.

Effects of curcumin on invasion and colony formation abilities of HNSCC cells. (a) Invasion assays were carried out in a transwell system using Matrigel-coated filters and the number of cells that migrated to the lower chamber was counted. Cells that migrated were visualized following methylene blue staining in FaDu, JHU-O22 and JHU-O28 cell lines in the presence or absence of curcumin. The images were captured at 10× magnification. (b) A graphical representation of the invasive ability of FaDu, JHU-O22 and JHU-O28 cells upon curcumin treatment, *
Inhibition or silencing of CaMKK2 reduces colony formation and invasion ability of HNSCC cells
We observed hypo-phosphorylation of CaMKK2 in CAL 27 cells upon curcumin treatment. We and others have shown the role of CaMKK2 as a therapeutic target in cancer; however, its role in HNSCC is poorly understood. We studied the effects of both silencing and inhibiting CaMKK2 in a panel of HNSCC cells. small interfering RNA (siRNA)-mediated silencing and inhibiting CaMKK2 using its inhibitor STO-609 led to a significant decrease in the colony formation ability of CAL 27 and other HNSCC cells (Figure 4(a) to (d)). In addition, both siRNA-mediated silencing and inhibition of CaMKK2 with STO-609, resulted in a significant decrease in the invasive property of all HNSCC cells (Figure 5(a) to (d)). Taken together, our results indicate that CaMKK2 may play an essential role in HNSCC metastasis and mimics the effects of curcumin in controlling the oncogenic potential of HNSCC cells.

Effects of CaMKK2 silencing/inhibition on colony formation ability of HNSCC cells: colony forming assay following ((a) and (b)) siRNA-mediated silencing of CAMKK2 or control siRNA (scrambled siRNA). A graphical representation of the colony forming ability of HNSCC cells upon CAMKK2 silencing, *

Effects of CAMKK2 silencing/inhibition on invasion ability of HNSCC cells. (a) and (b) Invasion assay was carried out using CAL 27, FaDu, JHU-O22 and JHU-O28 cells. Cells were transfected with either control (scrambled) or CAMKK2 siRNA and invaded cells were photographed. A graphical representation of the invasive ability of HNSCC cells upon CAMKK2 silencing, *
Delineation of signalling pathways in CaMKK2-mediated effects
Having observed that CaMKK2 and its downstream targets, AMPK-β1 and acetyl-CoA carboxylase 1 (ACC1), hypo-phosphorylated upon curcumin treatment and that inhibition of CaMKK2 corroborates the phenotypic effects of curcumin, we sought to study the signalling mechanisms of CaMKK2. Western blot analysis confirmed hypo-phosphorylation of AMPK-β1 (S108) upon treatment with curcumin. However, the total levels of AMPK-β1 and CaMKK2 remained unchanged (Figure 6(a)). Since AMPK-α exists in complex with AMPK-β and Ca2+/CaM, and phosphorylation of AMPK-α at Thr-172 is essential for its activation, 22,23 we determined the effects of curcumin on AMPK-α. Curcumin treatment resulted in hypo-phosphorylation of AMPK-α at T172 (Figure 6(a)).
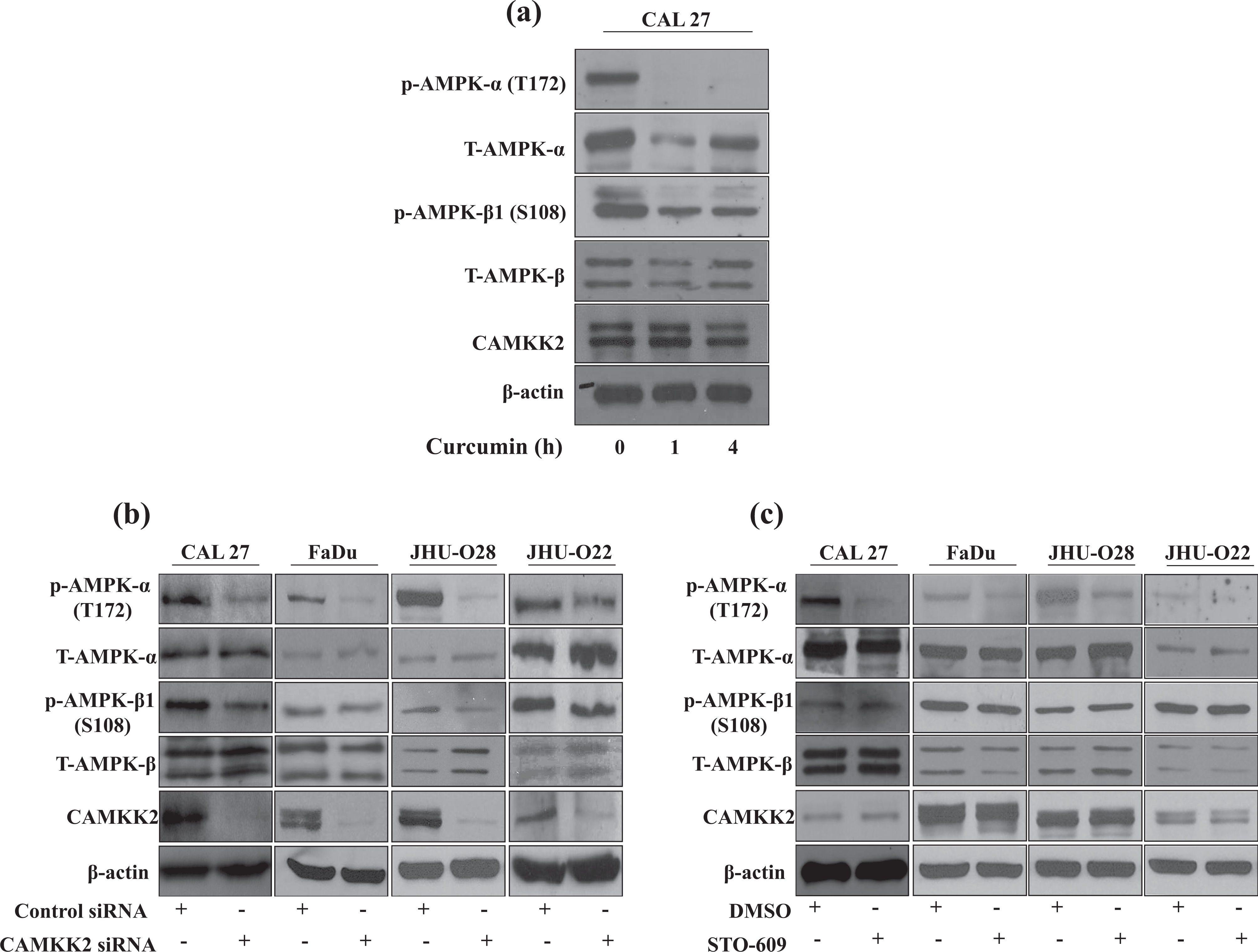
(a) CAL 27 cells were treated with curcumin for the indicated time points. Western blot was performed using phospho-AMPK-α and AMPK-β1, total AMPK-α and AMPK-β1, and total CaMKK2 antibodies. β-actin was used as a loading control. CAL 27, FaDu, JHU-O22 and JHU-O28 cells were treated with (b) CaMKK2 siRNA or (c) STO-609. Western blot was performed for the indicated proteins. β-actin was used as a loading control. CAMKK2: calcium/calmodulin dependent protein kinase kinase 2; AMPK: AMP-dependent protein kinase.
We next studied the effects of CaMKK2 silencing and inhibition on the signalling cascade using a panel of HNSCC cells, in addition to CAL 27. siRNA-mediated silencing of CaMKK2 led to a decreased phosphorylation of AMPK-α. However, the phosphorylation of AMPK-β1 remained unchanged upon silencing CaMKK2 (Figure 6(b)). In agreement with the siRNA results, inhibition of CaMKK2 using its inhibitor STO-609 also showed a decreased phosphorylation of AMPK-α but not AMPK-β1 (Figure 6(c)). Taken together, these results suggest that CaMKK2 corroborates the signalling pathway of curcumin and mediates its effects through AMPK-α.
Discussion
HNSCC is one of the most common cancers in India and sixth most common cancer worldwide. Despite improvements in diagnostic and therapeutic modalities, the clinical outcome/prognosis remains poor with a 5-year survival rate being less than 50%. Therefore, new therapeutic targets are needed for the early diagnosis of disease and to improve patient survival. Multiple groups have evaluated the role of curcumin as an anti-cancer agent. However, the use of curcumin is limited due to its poor solubility and bioavailability. In this study, we evaluated the signalling pathways through which curcumin mediates its effects and identified proteins that could be targeted as an alternative to curcumin by mass spectrometry–based proteomic analysis. The current study demonstrates the anti-cancer effects of curcumin on HNSCC cells and signalling pathways through which it mediates its effects. This is the first high-throughput study to profile STKs and their downstream targets that are altered in response to curcumin.
Curcumin is shown to affect the phosphorylation of MAPK1/3 and also reported to regulate the cleavage and activation of p21 (RAC1)-activated kinase 2 (PAK2). 10,24 –26 In addition, curcumin has also been reported to affect the autophosphorylation of epidermal growth factor receptor (EGFR) where it has also been shown to decrease phosphorylation of EGFR. 27,28 Our group has studied dysregulation in tyrosine kinases in the same cell line, Cal 27, in response to curcumin. 10 Comparison between our previous study and this study revealed dysregulation of 66 proteins to be common between the two data sets. Various kinases including EGFR, EPH receptor A2 (EPHA2), PTK2 and EPH receptor B4 (EPHB4) were found to be dysregulated in both data sets, thus showing concordance between data published previously. In addition, we identified several STKs including CDC-like kinase 1 (CLK1), STK10, CaMKK2 and STK38 whose role are to be studied in curcumin-mediated effects. Although studies have reported the dysregulation of PTK2, its downstream targets were not reported. In our previous study, using phosphoproteomic-based mass spectrometry approach, we mapped the downstream targets of PTK2 pathway. 10 In the current study, in addition to PTK2 pathway, we have shown that curcumin can also mediate its effects through CaMKK2 and its downstream targets. Thus, the two studies together implicate that curcumin-mediated anti-neoplastic effects may be due to perturbations of PTK2 and CaMKK2-mediated signalling pathways.
It is established that calcium (Ca2+) binds to intercellular receptor, calmodulin (CaM), and regulate various cellular processes and pathways. 29 –31 Binding of Ca2+ to CaM activates various calcium-binding proteins including CaMKK2. Ca2+/CaM binding relieves autoinhibition of CaMKK2 and results in the activation of kinase. CaMKK2 is an STK involved in the regulation of various processes including glucose homeostasis, inflammation, adiposity and cancer. Several substrates are reported for CaMKK2 which include CaMKI, CaMKIV and AMPK. 32 –34 CaMKI and CaMKIV have been shown to be activated independent of Ca2+/CaM signalling; however, activation of AMPK is dependent on Ca2+/CaM signalling cascade. 35,36 AMPK is an enzyme that plays a role in the regulation of energy homeostasis, cytoskeletal organization and cell cycle. 22 CaMKK2 has been shown to phosphorylate and activate protein kinase AMP-activated catalytic subunit alpha 1 (PRKAA1; AMPK-α) and these proteins exist as a complex with Ca2+/CaM and AMPK-β. 35,37,38
We evaluated the role of CaMKK2 as an alternative to curcumin (due to poor solubility and bioavailability) that could be targeted in HNSCC. Mass spectrometry analysis revealed hypo-phosphorylation of CaMKK2. Further, inhibition/siRNA-mediated silencing of CaMKK2 resulted in a significant reduction in colony formation and invasion ability of HNSCC cells. In addition to CaMKK2, mass spectrometry analysis also revealed the hypo-phosphorylation of downstream targets of CaMKK2, AMPK-β1 and ACC1 upon curcumin treatment of CAL 27 cells. Phosphorylation status of CaMKK2 upon curcumin treatment could not be evaluated by Western blotting due to unavailability of site-specific phospho-CaMKK2 antibody. However, Western blot analysis showed that the total levels of CaMKK2 remained unchanged upon curcumin treatment. AMPK-α exists in complex with AMPK-β and CaMKK2 and mediate signalling. Western blot analysis showed decreased phosphorylation of AMPK-α at T172 upon treatment with curcumin and on silencing or inhibition of CaMKK2, indicating that CaMKK2 may mediate its effects through AMPK-α. Taken together, these results indicate that curcumin mediates its effects through CaMKK2 and AMPK-α. Our current study does not rule out the role of other molecules and/or genomic factors towards curcumin-mediated signalling. The findings of this study substantiate the crucial role of CaMKK2 in HNSCC and that it can serve as a potential therapeutic target in HNSCC. A detailed understanding of the function of CaMKK2 in HNSCC is needed, which requires evaluation in preclinical mouse models and is beyond the scope of this article. Our work provides a scaffold for future studies to systematically investigate the role of CaMKK2 in HNSCC, especially as an alternative to curcumin due to curcumin’s poor solubility and bioavailability.
Footnotes
Acknowledgements
We thank the Department of Biotechnology, Government of India, for research support to the Institute of Bioinformatics, Bangalore. Gajanan J. Sathe and Pavithra Rajagopalan are recipients of Senior Research Fellowship from the Council of Scientific and Industrial Research (CSIR), New Delhi, India. Harsha Gowda is a Wellcome Trust/DBT India Alliance Intermediate Career Fellow. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. Remya Raja is a recipient of Startup research scheme from DST. We thank Dr. S. K. Shankar and Dr. Anita Mahadevan (NIMHANS), for providing access to the microscopy imaging facility.
Author contribution
Vishalakshi Nanjappa and Gajanan J. Sathe both have contributed equally to this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
