Abstract
Aim:
Epithelial-to-mesenchymal transition (EMT) is recognized as a hallmark of cancer. The change in phenotype of epithelial cells enables them to migrate and the cancer therefore, to become more aggressive. The presence of the epidermal growth factor receptor (EGFR) has been implicated in the spread and aggressive nature of oral tumors. The aims of this present study were to investigate the role of two different growth factors, both ligands of the receptor, and their influence on EMT and cellular motility.
Methods:
A number of assays were used to investigate the response of the cells to the two growth factors (EGF and transforming growth factor (TGF-α)). These techniques included the 2-D scratch assay, a 2-D-cell scatter assay, and immunocytochemistry. Normal keratinocytes (HaCaT), oral adenoid squamous cell carcinoma (TYS), and human salivary gland tumor cells were used in this study.
Results:
The results of the scratch and scatter assays indicated that EGF and TGF-α exerted differential effects upon the cells. EGF and TGF-α stimulated the migration of cancer cells in scratch assays. Scatter assays have revealed that both EGF and TGF-α induce EMT. EGF- and TGF-α-induced scattering is EGFR dependent and localization of EMT markers (E-cadherin and vimentin) might play an important role in scattering. These responses were also found to be dependent upon cell type, indicating that the assay could also be used as a simple screen for active growth factor. The observed effects were also dose dependant (data not shown).
Conclusion and implication for translation to clinic:
We have previously reported that although EGF and TGF-α have ultimately similar effects on motility, it would appear that different mechanisms are responsible. This data extends our knowledge of the action of these growth factors to epithelial cell lines and will inform future testing of the effects of EGF and TGF-α inhibitors.
Keywords
Introduction
Head and neck cancer is the sixth most common cancer in the world and squamous cell carcinoma (HNSCC) is the predominant form. 1,2 While HNSCC patients have a 5-year survival rate below 40%, patients with metastatic disease have an extremely poor prognosis and a survival rate lower than 10%. 3 The development of distant metastases, second primary tumors, and potentially inoperable recurrences with increased resistance to radiation or chemotherapy are the main reasons for mortality. 4 Tumor cells must gain invasive ability to develop metastases. The physiological process of epithelial-to-mesenchymal transition (EMT) permits cells to change their morphology, to lose polarity, and to become invasive. 5 EMT is a complex and reversible biological process, where an epithelial tumor cell modifies its polar, adhesive phenotype to a mesenchymal phenotype characterized by enhanced cell migration and invasive potential, cytoskeletal remodeling, and resistance to apoptosis. 6 –8 In recent years, EMT has been recognized as a key factor in cancer metastasis, allowing tumor cells to leave the primary tumor microenvironment and to migrate as circulating tumor cells to distant sites. Several reports have established the link between EMT, progression, metastasis, and chemoresistance in several types of cancer. 9 –11 Invasive cells often exhibit a de-differentiated morphology, that is complemented by a loss of epithelial markers such as E-cadherin and cell–cell junctions and an increase in expression of mesenchymal markers such as vimentin, N-cadherin, Twist, and Snail. 12 –14 An association between loss of E-cadherin and gain of vimentin with increased cancer cell migration and enhanced metastatic risk in HNSCC patients was found recently. 15,16 Primary HNSCC with the EMT signatures (low E-cadherin and high vimentin expression) has a 100% metastasis rate, in comparison to a 44% metastasis rate for primary HNSCC with partial or null EMT signatures. 16
Epidermal growth factor receptor (EGFR) is a tyrosine kinase receptor which is overexpressed in over 90% HNSCC patients 17 –19 and associated with poor prognosis. 20,21 EGF and transforming growth factor-alpha (TGF-α), two natural ligands, bind to the extracellular domain of EGFR and activate the receptor and its downstream signal transduction pathways such as PI3K/Akt, ERK-1/2, and Stat3 pathways, eventually causing activation or modulation of several cellular processes. 22 Activation of EGFR has been shown to decrease cell adhesion in several solid malignancies including HNSCC leading to an increase in cell migration and invasion. 23,24 A study suggested that HNSCC cells with high endogenous EGFR exhibited increased cell motility and number of invasive cells, upon addition of EGF. 25 Studies so far have revealed that EGFR plays a crucial role in the invasion and metastasis of HNSCC. However, the mechanism of EGF- and TGF-α-induced HNSCC cell migration, in terms of EMT still need to be elucidated.
The aim of this study was to investigate the EMT phenotype and expression of the related markers in EGF- and TGF-α-induced HNSCC cells. It also aimed to elucidate the subcellular localization of the EMT markers in EGF- and TGF-α-induced migrated HNSCC cells. It was hypothesized that EGF and TGF-α motogenic activities (in terms of EMT) are mediated by the EGFR and the subcellular localization of the markers is a better indicator of EMT than the total expression of markers in HNSCC.
Materials and methods
Cell culture
The oral adenoid squamous cell carcinoma (OASCC) cell line (TYS), derived from a minor salivary gland and the human salivary gland (HSG) tumor, was a kind gift from Dr. Koji Harada, University of Tokushima, Japan. Normal adult keratinocytes (HaCaT) were a kind gift from Professor S.L. Schor (late), Dundee Dental School, UK. All cells were cultured at 37°C and 5% carbon dioxide in minimum essential medium (MEM) supplemented with 10% (v/v) heat-inactivated fetal calf serum and 200 mM glutamine.
Scratch assay
The scratch assay, a directional in vitro 2-D migration assay, was performed as described earlier. 26 A cell monolayer was serum-starved overnight and then a wound was made in the monolayer using a 100-µl pipette tip and the cells were then incubated in different concentrations of EGF (#10-1001B) or TGF-α (#10-1033C; both from Insight Biotechnology Inc., London, UK) for 48 h. Images were captured by an inverted microscope (Olympus IX70, Osaka, Japan) at the starting point, at 24 and at 48 h of the assay to monitor the cell migration causing wound closure. Cells treated with serum-free (SF) medium were used as a negative control.
Scatter assay and immunofluorescent staining
Cell scattering is a dynamic process for investigating EMT in which dispersion of compact colonies of epithelial cells is induced by certain soluble factors such as growth factors. 27 The assay was performed as described earlier. 28,29 Cells were seeded, with a density of 4 × 104 cells/ml, in 60 mm dishes and grown until well-defined colonies (maximum 20 cells in a colony) were visible. The cells were washed twice, serum starved overnight, and incubated with the stated ligands diluted in SF medium for 24 h. Scattering of the cells was observed by at least two individuals before images were taken. Images were captured using an Olympus inverted microscope at either 100× or 200× magnification.
After 24 h, the cells were fixed with cold methanol for 15 min and then washed with phosphate-buffered saline (PBS). Cells were then treated with 0.2% (v/v) Triton X-100 in PBS for 5 min, small areas of the dishes were ringed with Immunopen (DAKO, Cambridgeshire, UK) and blocked with 5% (v/v) normal goat serum (NGS; Vector Laboratories, Burlingame, CA, USA) in PBS with 0.1% (v/v) Tween 20 (PBST) for 30 min. The cells were then washed with PBS and incubated with anti-E-cadherin (1:200; #3195, Cell Signaling Technology, Danvers, MA, USA), anti-vimentin (1:50; #3932, Cell Signaling Technology), and anti-EGFR (1:500; # ab52894, Abcam, Cambridge, UK) rabbit monoclonal primary antibodies diluted in 5% (v/v) NGS in PBST and kept at 4°C for overnight. They were then washed twice with PBST, once with PBS and incubated with anti-rabbit immunoglobulin G (H+L), F(ab′)2 Fragment (Alexa Fluor® 488 Conjugate) is the name of secondary antibody from cell signaling technology for 30 min at room temperature. After washing twice with PBST and once with PBS, sections were cover slipped with aqueous mounting medium (Sigma-Aldrich, St Louis, MO, USA). Sections were then viewed with an Olympus IX70 inverted fluorescent micro-scope using 20× or 40× objective lenses. Images were collected using an Olympus SC35 digital camera (Olympus). All devices were controlled through the Metamorph v6.1 software (Molecular Devices, Sunnyvale, CA, USA) and images were then processed and analyzed by ImageJ software (NIH, Bathesda, MD, USA).
Results
EGF and TGF-α stimulated the migration of cancer cells
Different concentrations (500 pg/ml, 1 ng/ml, 10 ng/ml, and 50 ng/ml) of EGF and TGF-α were used in the scratch assay. Maximum stimulation of migration was observed at 50 ng/ml of each growth factor. A 48-h incubation of the test cells in the scratch assay revealed that 50 ng/ml of EGF did not stimulate normal keratinocytes (HaCaT) to migrate (Figure 1), whereas OASCC cells (TYS) were stimulated to migrate (Figure 2) in response to the same concentration of EGF. In contrast, oral HSG tumor cells showed partial gap closure after 48 h of EGF stimulation (Figure 3). However, TGF-α stimulated the migration of normal keratinocytes after 48 h of incubation (Figure 1) and oral adenocarcinoma after 24 h (the cells started to die after 48 h; Figure 2). EGF- and TGF-α-induced migration of oral salivary gland tumor cells was not significantly higher than that of the negative control (Figure 3). Single-cell migration was observed in TGF-α-induced normal keratinocytes, whereas other cells showed collective cell migration upon EGF and TGF-α stimulation (Figure 1). SF MEM was used as negative control and EGF and TGF-α as a stimulant in this study.

Scratch assay of normal adult keratinocytes (HaCaT). After serum starvation, a wound was made in the monolayer and assays incubated for 48 h in experimental conditions. Images were then captured every 24 h by an inverted microscope at 40× magnification. Images were then edited using ImageJ software. Serum-free (SF) medium was used as a negative control. EGF: epidermal growth factor; TGF-α: transforming growth factor alpha.

Scratch assay of oral adenoid squamous cell carcinoma (TYS). A cell monolayer was serum starved overnight and then a wound was made using a pipette tip. Wounded cells were then incubated in experimental conditions for 48 h. Images were captured every 24 h at 100× magnification and edited. Serum-free (SF) medium was used as a negative control. EGF: epidermal growth factor; TGF-α: transforming growth factor-alpha.

Scratch assay of human salivary gland (HSG) tumor cells. A cell monolayer was wounded after serum starvation and incubated for 48 h in experimental conditions. Images were captured at 100× magnification. Serum-free (SF) medium was used as a negative control. EGF: epidermal growth factor; TGF-α: transforming growth factor-alpha.
Scatter assays revealed that both EGF and TGF-α induce EMT
Cells were plated at both high (4 × 105 cells/ml) and low (4 × 104 cells/ml) density to analyze the effect of EGF and TGF-α on cell scattering. No scattering of cells was observed with cells plated at high density upon addition of EGF or TGF-α (results not shown). However, cells plated at low density formed small colonies and stimulation of cells with EGF and TGF-α for 24 h revealed a colony scatter effect. A range of concentrations (500 pg/ml, 1 ng/ml, 10 ng/ml, and 50 ng/ml) of EGF and TGF-α were used in this assay and effective scattering was observed at or over 10 ng/ml. Normal keratinocytes (Figure 4) and HSG tumor cell colonies (Figure 5) were scattered into single cells and a morphological change of epithelial cells to mesenchymal type was observed upon addition of EGF and TGF-α. This type of morphological change is called EMT or EMT. Furthermore, OASCC also showed scattering into single cells upon addition of EGF and TGF-α, but EGF-induced OASCC displayed a unique change in morphology which resembled a cytoskeletal rearrangement (Figure 6). Filopodia and lamellipodia were observed in EGF-induced scattered OASCC cells; these are the classic features of actin remodeling, another mechanism involved in cell migration.

Scatter and immunofluorescence assays of HaCaT cells. Small colonies of cells were serum starved and incubated in experimental conditions for 24 h. Cells were then fixed and stained with E-cadherin, vimentin, and EGFR antibodies. Images were captured by inverted fluorescence microscope with a CCD camera and edited using ImageJ software. Normal light images were captured at 40× magnification and fluorescent images were captured at 200× magnifications. Serum-free (SF) medium was used as a negative control. EGFR: epidermal growth factor receptor; TGF-α: transforming growth factor-alpha.
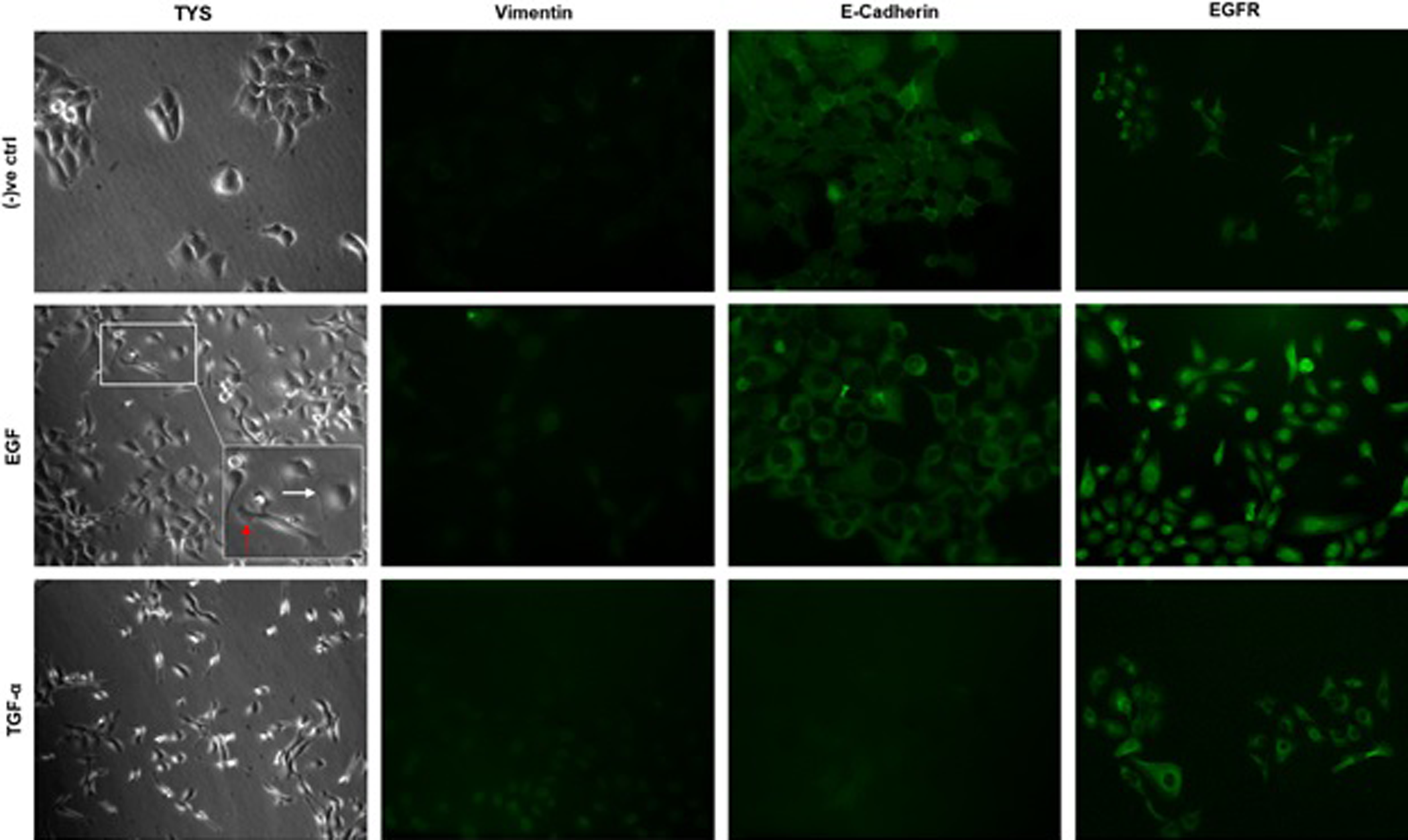
Scattering of oral adenoid squamous cell carcinoma (TYS) with immunofluorescent staining. Scattering of cells were observed within 24 h of adding growth factors. Cells were then fixed and stained with E-cadherin, vimentin, EGFR primary antibodies, and fluorescent-tagged secondary antibodies. Images were captured at 100× (normal light) and 200× (fluorescent) magnification and edited using ImageJ software. White arrow: lamellipodia; red arrow: filopodia. Serum-free (SF) medium was used as a negative control. EGFR: epidermal growth factor receptor; TGF-α: transforming growth factor-alpha.

Immunofluorescent assay of scattered and fixed salivary gland tumor cells (HSG). Small colonies of cells were scattered within 24 h of adding growth factors. Scattered cells were then fixed and stained with E-cadherin, vimentin, and EGFR antibodies. Images were captured at 40× (normal light) and 200× (fluorescent) magnification. Images were then edited using ImageJ software. Serum-free (SF) medium was used as a negative control. EGFR: epidermal growth factor receptor; TGF-α: transforming growth factor-alpha.
EGF- and TGF-α-induced scattering is EGFR dependent and localization of EMT markers might play an important role in scattering
Scattering is the visible phenotype at the end of an extensive cellular signaling cascade. 28 Hence, it was investigated whether the results of the scattering assays are reflected by EGFR-dependent signaling. After 24 h of scatter assay observation, immunofluorescent assay of the cells, after fixing, was employed to investigate the expression and localization of vimentin (a mesenchymal marker), E-cadherin (an epithelial marker), and EGFR in the cells. SF medium-treated normal keratinocytes expressed E-cadherin, vimentin, and EGFR. Vimentin was found to be highly expressed in a diffuse pattern, while weak cytoplasmic E-cadherin and strong expression of EGFR in the cell membrane were observed in SF medium-treated normal keratinocytes (Figure 4). EGF-treated scattered normal keratinocytes also expressed vimentin, E-cadherin, and EGFR (Figure 4). A strong diffuse expression of E-cadherin and EGFR was observed. Contrary to predictions, vimentin was observed on the nuclear membrane of the EGF-treated scattered normal keratinocytes (Figure 4). Moreover, TGF-α-treated scattered normal keratinocytes revealed a strong diffuse expression of vimentin and EGFR but a very weak expression of E-cadherin (Figure 4). SF medium-treated OASCC cells displayed upregulated E-cadherin expression in contrast to downregulated vimentin (Figure 5). An elevated and diffuse expression of EGFR was also observed in SF media-treated OASCC cells. EGF-treated scattered OASCC cells displayed filopodia (red arrow) and lamellipodia (white arrow), which are the characteristic features of cytoskeletal rearrangement (Figure 5). A cytoplasmic upregulation of E-cadherin, very weak expression of vimentin, and an elevated diffuse expression of EGFR were exhibited in EGF-treated scattered OASCC cells (Figure 5). However, TGF-α-treated scattered OASCC cells displayed an EMT phenotype, downregulated E-cadherin and nuclear vimentin, and strong cytoplasmic EGFR expression (Figure 5). HSG tumor cells, treated with SF medium, expressed elevated vimentin, EGFR, and E-cadherin (Figure 6). The expression of vimentin was diffuse, E-cadherin was downregulated, and EGFR was in the cell membrane (Figure 6). EGF-treated HSG tumor cells were scattered after 24 h and showed EMT phenotype. Downregulated E-cadherin and upregulated vimentin expression confirms EMT in EGF-treated salivary gland tumor cells (Figure 6). Vimentin expression was observed in the nuclear membrane and EGFR expression was in the cytoplasm and/or in the cell membrane. TGF-α-treated scattered salivary gland tumor cells also showed an EMT phenotype (Figure 6). The expression pattern of vimentin, E-cadherin, and EGFR was similar to that of EGF-treated tumor cells (Figure 6).
Discussion
In this study, wound healing (scratch) and scatter assays were included to investigate the two different modes of cell migration. The wound healing (or scratch) assay is a popular 2-D in vitro assay to visualize collective cell migration that allows polarization, force generation, and mechanism of cell–cell adhesion to be studied during the movement of confluent monolayers. 30 –32 Normal keratinocytes (HaCaT) were stimulated to migrate upon addition of TGF-α in the wound healing assay, whereas EGF was not able to stimulate normal keratinocytes to migrate. In this assay, TGF-α-induced normal keratinocytes migrated as single cells, the opposite to collective migration. A recent wound healing study suggested that keratinocytes (HaCaT) migrate as single cells via EMT during the healing process 33 . Moreover, OASCC cells were stimulated and HSG tumor cells were partially stimulated to migrate as a collective sheet by both EGF and TGF-α. In comparison, both normal keratinocytes and HSG tumor cells showed a distinctive EMT phenotype during single-cell migration, upon addition of EGF and TGF-α in the scatter assay. OASCC cells also exhibited an EMT phenotype upon addition of TGF-α, whereas EGF-stimulated scattered cells showed filopodia and lamellipodia in contrast to the EMT phenotype. EGFR was highly expressed in both SF medium-treated nonscattered and EGF- and TGF-α-induced scattered cells. But EGFR was localized in the membrane of nonscattered cells, whereas it was found either in the cytoplasm and/or in the nucleus of scattered cells. The localization pattern of EGFR in migrated cells fits with the concept of intracellular membrane trafficking during the cell signaling process. At the cellular level, receptor tyrosine kinases such as EGFR need to be localized in a specific location to function as signal receiving and transmitting devices. 34 EGFR must be exposed at the surface of the plasma membrane to receive signals, that is, to bind extracellular ligands, such as EGF and TGF-α. To transmit signals after ligand binding by EGFR, suitable signaling machinery has to be available within the intracellular compartments of the cytoplasm, in association with membrane bound organelles and in the nucleus. 35,36 Increasing evidence suggests that intracellular membrane trafficking, such as endocytic internalization (entering of the components of plasma membrane, cell surface receptors, and various soluble molecules into the cell by endocytosis 36 ), degradation, and recycling, can profoundly affect the signaling properties of EGFR. 34,37,38 To explain the fate of EGF- and TGF-α-induced EGFR activation in scattered cells requires further investigation. This study revealed that intracellular localization as well as expression of EGFR has an important effect on cell migration. To confirm the EMT phenotype of migrated cells, immunofluorescent staining of two widely used EMT markers, E-cadherin and vimentin, was carried out. EGF-treated normal keratinocytes and HSG tumor cells showed perinuclear vimentin expression in scattered cells, in comparison to cytoplasmic vimentin expression in SF medium-treated nonscattered cells. Moreover, TGF-α-treated scattered cells exhibited a varied pattern of vimentin localization such as cytoplasmic/perinuclear (normal keratinocytes), perinuclear (salivary gland tumor), and weak nuclear (OASCC). Recent studies have revealed that vimentin interacts directly with the microtubules via microtubule-associated protein, kinesin, or indirectly through intermediate filament-associated proteins, such as plectin at the perinuclear region, and mediates microtubule polarization to support cell migration. 39 –41 High expression of cytoplasmic E-cadherin and very weak vimentin expression in EGF-treated OASCC scattered cells also demonstrated another mechanism of cell migration via actin reorganization, that is, lamellipodia and filopodia. Subcellular localization of markers has already been shown to be a better indicator of EMT than only the expression of the markers. 16,42 –44 This study therefore revealed that EGF and TGF-α stimulate the migration of both normal and cancer cells, which supports the idea that these ligands could induce cell migration during both wound healing and cancer metastasis. The mode of cell migration and associated signaling cascade, however, largely depend on the cell types, assay format, incubation time, and available concentrations of the ligands. Moreover, it may not be prudent to interpret the data in terms of squamous cell carcinoma (as the cell lines used were OASCC cell line (TYS), derived from a minor salivary gland, HSG tumor, and normal adult keratinocytes (HaCaT)). A direct comparison of results from HaCaT with a squamous cell carcinoma cell line would be required to extrapolate the data directly to oral cancer. Investigating the downstream signaling pathways of EGFR responsible for EMT in oral squamous cell carcinoma would be essential in translating the research into clinical practice in future.
Conclusion
This study has demonstrated the role of EGF and TGF-α on the cell migration of HNSCC through EMT. However, there are several omissions in the published data regarding EMT which need to be addressed to gain a more comprehensive understanding of the EMT process in HNSCC. HNSCC covers several subsites and each subsite and stage may have unique features which could regulate EMT through overlapping and diverse mechanisms. A better understanding of the role of EMT in HNSCC metastases and associated signaling pathways could lead to the design and therapeutic use of EMT-targeting drugs and may ultimately help to decrease HNSCC-associated morbidity and mortality.
Footnotes
Acknowledgments
This work was conducted as part of a laboratory project for the degree of MRes in Oral Cancer at the University of Dundee. The laboratory work was performed by Shraddha Mane and Erum Hyder under the supervision of Ian Ellis, Sarah Jones, and Mohammad Islam. The authors wish to thank Dr. Margaret Florence for her dedication and technical ability.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
