Abstract
Objectives:
To describe incidence and mortality rates of oral and of oropharyngeal cancers in sub-Saharan Africa using available sources and to discuss shortcomings.
Methods:
Incidence and mortality rates were obtained from GLOBOCAN online (IARC 2012) and the pathology-based South African National Cancer Registry (SA-NCR). Descriptions of oral and of oropharynx cancers for the four main geographical regions were derived from the literature. For South Africa, the incidence by ethnic group of lip and intra-oral cancer over the period 1988–2012 could be calculated from SA-NCR. The age distributions for intraoral and for oropharynx cancer in both genders in South Africa were compared for the period 1996–2002.
Results:
Age standardized incidence rates and mortality rates of lip and of oral cancer in sub-Saharan Africa varied substantially, depending on the regions, the mix of carcinogenic substances consumed in each country and the quality of the cancer registry data. African islands in the Indian Ocean and the Southern and Eastern African countries report the highest rates; these are generally reported to be much lower in West African countries. Available data are now old: not beyond 2010. Up until that time, in the Republic of South Africa, there appears to have been a slight decline in oral cancer rates, except in Black females, but a rise this millennium in cancer of the oropharynx, especially in Coloureds (mixed-race). At least until 2002, the Human Immunodeficiency Virus infection (HIV) pandemic had little or no stimulating influence on the incidence rates of oral squamous carcinoma in South Africa, although Kaposi’s sarcoma has been a major burden in the past. Asian and mixed-race South African females had the highest levels of oral squamous cell carcinomas in the <45 year age group.
Conclusion:
The quality of cancer registry data in sub-Saharan Africa is variable, unreliable and out of date, suggesting gross under-ascertainment. Records of all causes of death are poor, which can result in both under- and over-attribution of cancer mortality. Improvements of the necessary resources for cancer registration and death certification deserve a high priority.
Introduction
The continent of Africa is enormous; it is the world’s second-largest and second-most populous continent. At about 30.2 million km2 including adjacent islands, it covers 6% of Earth’s total surface area and 20.4% of its total land area. It is composed of 54 independent states and, according to the United Nations, its population currently exceeds 1.23 billion souls. It is the region with the most rapidly growing population in the world, currently equivalent to 16.4% of the total world population. Mainland Africa has a current population of over 962,268,000 which is predicted to rise to nearly 1.4 billion by 2030. 1 The countries of North Africa are traditionally included with the Middle Eastern region (Middle Eastern North African Region), and cancer data for these regions are discussed in this journal by Kujan et al. 2
At least 847,000 new cancer cases (6% of the world total) and 591,000 deaths (7.2% of the world total) were estimated in the 54 countries of Africa in 2012, with about three quarters in the sub-Saharan countries: However, we believe there is significant under-reporting. While the published profiles often differ markedly between regions, the most common cancers in men were prostate (16.4% of new cancers), liver (10.7%) and Kaposi sarcoma (KS; 6.7%); in women, by far the most important are cancers of breast (27.6% of all cancers) and cervix uteri (20.4%). 3 It is important to note that head and neck cancers (except for oesophagus), and therefore oral cancer and pharyngeal cancer, do not get the attention they deserve in papers dealing with the incidence and mortality of major cancers. Even when African websites and publications dealing with major cancers record head and neck cancer (on average 10% of all cancers), the incidence of cancers of the lip, oral cavity and tongue, pharynx and larynx is often aggregated. 4 In this regard, the oral cavity plus the oropharynx was by far the most commonly reported site among head and neck squamous cell carcinomas (HNSCC) in sub-Saharan Africa. 5
This critique provides an account of the current parlous state of the quality and extent of information on cancer across Africa and a secondary data analysis on the epidemiology of oral and related cancers in sub-Saharan Africa (41 mainland countries). We then give a short review of aetiology and risk factors and recommendations for improved funding and organization of data services in the future.
Sources of data
The African Cancer Registry Network (AFCRN; available at http://afrcn.org) was formally inaugurated on 1 March 2012 to expand activities of the East African Cancer Registry Network, which had been established in 2011, enabled by a grant from the USA-based Doris Duke Charitable Foundation (available at: http://www.ddcf.org, accessed 11 June 2017). The AFCRN is a project of the Cancer Registry Programme of the International Network for Cancer Treatment and Research (INCTR) (available at: http://www.inctr.org, accessed 11 June 2017). It is supported financially through the INCTR Challenge Fund (available at: http://www.challengefund.org, accessed 11 June 2017).
Support to AFCRN is a recognition of the increasing burden of non-communicable diseases, especially cancer, in the continent, and the need for adequate surveillance as a fundamental part of any rational programme for cancer control. Since September 2012, the International Agency for Research on Cancer (IARC), in the framework of its global initiative for cancer registry development in low- and middle-income countries (available at: http://gicr.iarc.fr/; accessed 11 June 2017), has partnered with AFCRN to provide a regional hub for cancer registration in sub-Saharan Africa.
The role of this hub is in providing technical and scientific support to countries, delivering tailored training in population-based cancer registration and use of data, advocating the cause of cancer registration in the region and facilitating setting up associations and networks of cancer registries and coordinating international research projects and disseminating findings. A summary of AFCRN activities can be accessed at http://afcrn.org/programme-of-activities (accessed 10 June 2017). The 5th AFCRN Annual Review Meeting was hosted by the Kumasi Cancer Registry in Ghana from 18–20 December 2016. The website only mentions percentages of the major cancers in East Africa in 2008, and lip plus oral cavity accounts for 3.1% of all cancers in males in East Africa (available at: http://activities-in-2011/72-cancer-patterns-and-trends, accessed 11 June 2017).
Member countries each have an affiliated website with a description of location, logistics and the (intended) activities. The Kampala Cancer Registry report for 2007–2009 is recommended as a model to follow, but few countries have actually published data, and in incidence percentages only. A good survey of the dire functionality of the cancer registries in Africa in 2014 has been provided. 6
The South African National Cancer Registry (SA-NCR) was established in 1986 and has been primarily collecting histologically verified data; recognized methods of analysis and routine reporting were introduced in 1990 and the Cancer Research Unit is funded by the South African Medical Research Council. The detailed incidence data per gender and population group are published online by the National Institute for Occupational Health which is a division of the South African National Health Laboratory Service (available at: http://www.nioh.ac.za/?page=national_cancer_registry&id=41, accessed 11 June 2017) but must be interpreted with caution (see below).
Data in age standardized incidence rates (ASIRs) format from the world’s cancer registries are made available online by GLOBOCAN (available at: http://globocan.iarc.fr/Default.aspx, latest accessed 11 June 2017). GLOBOCAN is a project of the IARC, the World Health Organization (WHO) body that undertakes research on carcinogens and cancer epidemiology. It uses data reported from local, regional and national cancer registries to estimate incidence, prevalence, mortality and disability adjusted life years lost to cancer. GLOBOCAN statistics are widely quoted and used by national governments and international organizations to set health priorities. GLOBOCAN published national estimates of cancer epidemiology in 184 countries for 2002, 2008 and 2012. The source dates vary between 2006 and 2012. Incidence and mortality rates are derived from 22 sub-Saharan countries: 20 are population-based registries, there is one registry collecting data from a single hospital and five are pathology based: overall the coverage and quality is dispiriting. We have, in addition, reviewed relevant scientific publications.
Data quality
Of the 20 population-based registries, only 4 have national coverage (Botswana, the Gambia, Namibia and Swaziland) and 16 cover a region, district or a city. Twenty countries covering approximately a third of the sub-Saharan population had no cancer registries in 2012. Most of the registries are weak (with the exception of the Gambian and Ugandan national registries) and in an early stage of development and gather data from relatively limited subnational populations. Registry coverage is highly variable and ranges from approximately 2.3% of the population in Kenya to 37.3% in Congo and 100% in the Gambia. In this context, the reader should realize that the proportion of Africa’s population covered by cancer registries is only a very low 2% compared to Asia (6%), Central and South America (8%), Europe 42%, Oceania (78%) and North America (95%). 7 This very low level of coverage is somewhat disputed in a survey of cancer registries in Africa in 2014 6 by which it was estimated that the population-based cancer registries covered a total population of 90.7 million (note: South Africa alone accounts for 49.3 million) which was 10.5% of the population of sub-Saharan Africa. Nevertheless, cancer registration in this part of the continent is clearly underdeveloped – partly due to lack of funding.
The incidence data for the 20 countries (e.g. Democratic Republic of the Congo, Gabon, Tanzania, Central African Republic, Chad, Niger, etc.) which are lacking a cancer registry system were extrapolated as averages from neighbouring countries (e.g. Brazzaville, Congo, Kenya, Uganda, Nigeria, etc.) or better functioning registries in the greater region. As with other parts of the world where a region has significant ethnic and cultural differences, such data can be misleading. A classic example is mixing generations of south Asian immigrant populations with Indigenous Melanesian and Polynesian populations in the Western Pacific. 2
Of the 26 registries, 19 met less than five of the 15 cancer registration quality criteria adapted from Bray and coworkers
7,8
; only the Gambia met more than 10 criteria. The main issues are: Comparability: of the 26 registries, 16 provided no data on coding. None have published information on how they determine date when registering an incident cancer. Validity: although 81% of the registries published the proportion of cancers morphologically verified, these ranged from 15% for male patients in The Gambia National Registry to 96% in female patients in the Namibian Registry. This reflects lack of the resources for histopathology and the methods by which cancers are registered. No information on death certification is available for 15 registries. Autopsies are not performed in many countries, even if someone dies in hospital. In some African countries or regions, there is a sociopolitical resistance in recording the ethnicity of patients (like in South Africa since 1993); many registered cases lack either some or all of the vital statistics: date of birth, patient ID, cancer stage and treatment. Timeliness: Fourteen of the registries submitted data which had been collected more than 5 years previously, while four registries submitted data more than 10 years old, for example, the Rwanda data originated in 1991. Completeness: Mortality – incidence ratios are not available for any registry due to lack of data. In some registries, the mortality entries are higher than incidence data. Incidence reporting is not mandatory in most sub-Saharan countries and the data from a growing number of public and private pathology laboratories (with the exception of South Africa) and private oncology practices are not recorded; this distorts the situation considerably because many private practices do not report cancer cases. The NCR of South Africa has traditionally been a pathology-based registry; however, since April 2011, it has been made compulsory for all practitioners making a diagnosis of cancer to report each case to the NCR and non-compliance is a legal offence.
Detailed information has been provided on the 15 quality variables and the performance of the individual cancer registries 9,10 (also available at: http://globocan.iarc.fr/Pages/DataSource_and_methods.aspx, accessed 11 June 2017). This uses an alphanumeric scoring system which independently describes the availability and the methods of gauging the incidence and mortality data established at country level. The combined score is presented together with the data estimates for each country, aiming to provide a broad indication of the robustness of the estimation. Figures 1 and 3 are computed by the GLOBOCAN online system (year 2012); Tables 1 and 2 are compiled from information generated by GLOBOCAN graphs on lip plus intra-oral cancer data.

Top 20 highest incidence and mortality estimates for lip and oral cavity cancer (C00–C06) in sub-Saharan Africa (IARC/GLOBOCAN 2012). IARC: International Agency for Research on Cancer.
African regional ASIRs and mortality rates per 100,000 pa for lip and intra-oral cancer (C00–C06) in males and females (IARC/GLOBOCAN 2012).

WHO: World Health Organization; IARC: International Agency for Research on Cancer.
Countries with higher and lower ASIRs and corresponding mortality rates in males and females for lip and intra-oral cancer (C00–C06) in sub-Saharan Africa (IARC/GLOBOCAN 2012).a

IARC: International Agency for Research on Cancer.
a(x) = no own country data.
bFrequency data. The superscript indices indicate the quality of the source and estimates. Refer to: http://globocan.iarc.fr/Pages/DataSource_and_methods.aspx for legends.
There is a collection predilection for urban areas. Limited rural coverage due to disproportionate registration of cancers of those patients able or willing to seek medical attention also distorts data and understanding. 11 Many patients able to afford it go abroad for treatment so that outcomes data are lost. Mortality data published by GLOBOCAN for most countries in Africa are also somewhat inaccurate estimates: death certification is incomplete in many. 12 Medium quality cancer mortality rates are only available for two countries (Mauritius and Reunion) and of low, although improving quality in South Africa where mandatory stating of the cause of death on the death certificate has been adhered to for at least the last 3 years.
To illustrate some of the difficulties that cancer registries in Africa are faced with, we herewith refer to the SA-NCR which is functioning in a reasonably well-developed country with an emerging economy. Between 2002 and 2009, competing burdens of disease in South Africa meant that chronic diseases, and particularly cancers, were not perceived as a priority in the country. As a result, the progress and development of the SA-NCR was arrested due to a lack of funding and human resources support. This has led to a lag in reporting of cancer incidence data. In addition, there is a significant proportion of under-reporting due to lack of sustained laboratory data submission and lack of adequate medical infrastructure due to historical socio-racial inequalities. The latter comment can also be applied to the majority of African countries resulting in underestimation of data.
Data analysis and results
Geographical differences in lip and intra-oral carcinoma (C00–C06)
The captured data sketch a very heterogeneous picture, and we believe comparisons between countries are unreliable. Based on the GLOBOCAN data quality rating system, the data in Table 2 are the most reliable for La Reunion, South Africa, Zimbabwe and Malawi.
Figure 1 illustrates the top 20 ranking of African countries with the highest incidence and mortality rates for lip plus oral cavity cancer. Their combined ASIR for both genders for the whole of Africa is estimated at 2.7/100,000/per year (Table 1) which, conservatively, should translate into a minimum of 31,000 new cases per year. In general for sub-Saharan Africa, the ASIRs for males (3.5/100,000/per year) are much higher than for females (2.1/100,000/per year) with the exception of Kenya (male = 7.3 vs. female 6.4/100,000/per year, M–F ratio = 1.1:1; Table 2). The ASIRs reported for males in Western Africa (1.7/100,000/per year, M–F ratio = 1.2:1) appear considerably lower than those reported for East Central Africa (vide Kenya: 7.3/100,000/per year; Table 1), Eastern Africa (4.5/100,000/per year, M–F = 1.5:1) and Southern Africa (6.3/100,000/per year, M–F = 2.3:1; Table 1). It is also surprising that the records for the Gambia, which reportedly has a 100% cancer registry coverage, indicate a low ASIR for males of only 0.4/100,000/per year (M–F ratio = 1.33:1) in males for lip and intra-oral cancer (Table 2).
The incidence data, especially for male inhabitants of the islands in the South Western Indian Ocean, indicate much higher ASIRs (Seychelles 14.3/100,000/per year, M–F ratio = 5.7:1; La Reunion 13.7/100,000/per year, M–F ratio = 5.1:1; Madagascar 9.3/100,000/per year, M–F ratio = 2.9:1; Comoros 6.9/100,000/per year, M–F ratio = 1.5:1) as do the data for Namibia (10.2/100.00/per year, M–F ratio = 2.55:1; Figure 1 and Table 2). On the contrary, Southern African countries with large populations record 50% lower ASIRs for males (Zambia 2.3/100,000/per year, M–F ratio =1.1:1; Zimbabwe 2.3/100,000/per year, M–F ratio =1.1:1) which would not be expected for that region (Table 2). Different mixes of ethnicities, and their social/behavioural habits, would partly explain these variations. It is unexpected that the rates are so low in Zimbabwe, because this is the largest producer of tobacco in Africa. Surprisingly, Zimbabwe has recently acceded to the WHO Framework Convention on Tobacco control – perhaps with ulterior motives. 13 The WHO Tobacco Atlas (available at: http://www.tobaccoatlas.org/country-data/zimbabwe/) reports current smoking rates at 24.6% and 2.7% for adult men and women, respectively, with 4.8% of boys and 1.5% of girls smoking daily. In addition, 1% of the population uses smokeless tobacco. Referring to a WHO report from 2009 on the global tobacco epidemic, tobacco use remains uncommon in most of sub-Saharan Africa even if tobacco companies conduct(ed) aggressive marketing campaigns, particularly targeting the youth. 5
The quality of the estimated mortality rates (Figures 1 and 2; Tables 1 and 2) is questionable as recognized by the GLOBOCAN scoring system. Indications are that countries with a somewhat better health infrastructure (like South Africa, Kenya, Botswana, Zimbabwe, Nigeria, La Reunion and even Gabon) have a relatively lower mortality burden from lip and oral cancer than countries with a lesser developed health infrastructure.

Top 20 highest 5-year prevalence incidence estimates for lip and intra-oral cavity cancer (C00–C06) for the whole of Africa (IARC/GLOBOCAN 2012).
This observation is echoed by Figure 2 which was generated through the GLOBOCAN website: this ranks the top 20 African countries with the highest estimated 5-year prevalence rates or lip and oral cavity cancer. Again the data should be interpreted in a relative manner but the graph shows that generally the 5-year prevalence rates are better in countries such as South Africa (5.15/100,000/5 years), Egypt (2.57/100,000/5 years), Kenya (3.29/100,000/5years) and Tanzania (3.7/100,000/5 years) with (for African circumstances) a larger number of reasonably well or better functioning head and neck cancer treatment centres, which contributes to keeping oral cancer sufferers alive for longer (see below).
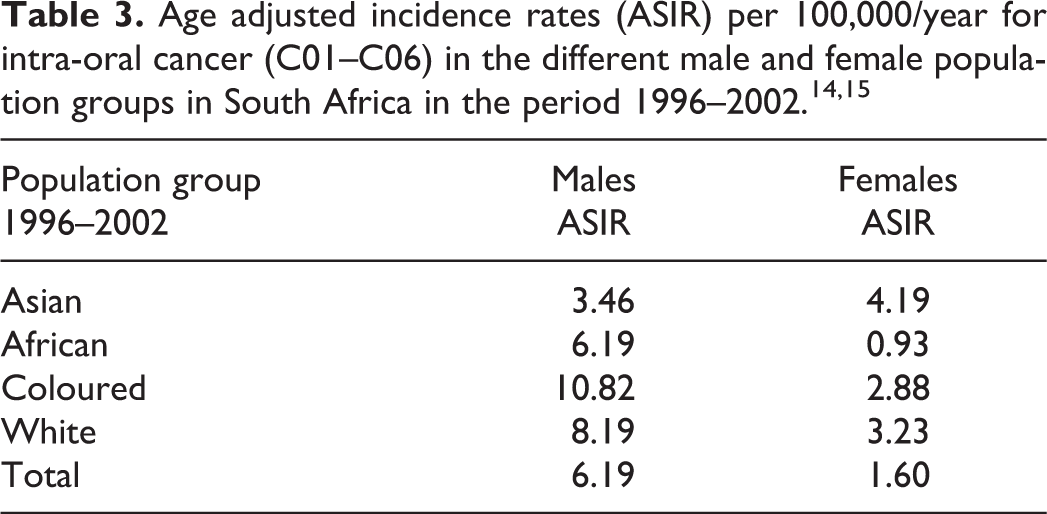
In a study by the first author and co-worker on the epidemiology of oral cancer in South Africa derived from SA-NCR data between 1996 and 2002, 14,15 the incidence of cancer of the oral cavity could be analysed separately from lip cancer. The average oral cancer ASIR for all males over the period 1996–2002 was 6.19/100,000/ per year and for all females 1.60/100,000/per year (Table 3). Significant differences between the four main population groups were noted. In males, the highest ASIR average for that period was recorded for ‘Coloured’ (mixed-race) males (10.82/100,000/per year) and the lowest for Indian (‘Asian’) males (3.46/100,000/per year). For females, Indian (‘Asian’) women had the highest ASIR (4.19/100,000/per year) while the lowest rates were among African females (0.93/100,000/per year).
The ASIR data for oral cancer calculated in this investigation are comparable with those reported by an independent South African study 16 also using the South African National Cancer Registry data but included oral cancer reports from an oral pathology department which had not submitted these data to the SA-NCR between 1997 and 2001 (see also below in the section on cancer rates of the oropharynx).
Other upper aero-digestive tract cancer incidences
A comparison of relative frequencies of SA-NCR upper aero-digestive tract (UADT) cancer data 17 revealed that the relative burden of these cancers in the 1990s (23.8%) was higher compared to those of the world (12.7%) and the United States (6.1%; Table 4). The difference in UADT rates between South Africa and the other regions is mainly due to high oesophageal cancer incidence rates, however that does not detract from the fact that 6.2% of all non-skin cancers were registered as intra-oral and lip cancer in South Africa, which was about 1.8 times the rate (3.3%) of those cases registered in the world and 2.8 times the rate in the United States (derived from SEER data). As oesophageal cancer is traditionally not regarded as a head and neck cancer, these data confirm that intra-oral and lip cancers are the most common head and neck cancers in South Africa.
Comparison of relative frequency of UADT cancer in South Africa with those in the world and the United States in the 1990s (Hille J, unpublished data 1996; sources: SA-NCR and Seer data).

UADT: upper aero-digestive tract; NOS: not otherwise specified.
Differences by anatomical sub-site
Very little information is available on detailed sub-site distribution of lip and intra-oral cancers in African countries. The comprehensive review paper by Faggons et al. 5 includes a detailed site distribution compiled on information from 28 publications. These originated from Central Africa (DRC – 1, Sudan – 4), Eastern Africa (Ethiopia – 1, Kenya – 6), Southern Africa (South Africa – 2, Zimbabwe – 1) and West Africa (Ghana – 1 and Nigeria – 13). Cumulated results include a total of 7750 cases: lip 290 (4%), oral mucosa ‘not otherwise specified’ (NOS) 368 (5%), alveolar ridge 1007 (13%), floor of mouth 325 (4%), palate 375 (5%), tongue NOS 773 (10%) and with the majority of 4441 cases recorded in a non-specified site (57%). When data from the eight publications which do not or incompletely specify the site of occurrence were subtracted, site distributions recalculate as follows (Figure 3): lip 275 (8%), oral mucosa NOS 368 (10%), alveolar ridge 1000 (28%), floor of mouth 325 (9%), palate 373 (11%), tongue NOS 766 (22%), oropharynx 171 (5%) and non-specified site 229 (7%).

Site distribution of lip (C00), intra-oral cancer (C01–C06) and oropharynx (C10) for the different regions of Africa (recalculated from Faggons et al. 5 ).
An analysis study of the recorded data of oral malignancies in Zimbabwe between 1988 and 1997 (N = 873) 18 demonstrated that, at least from 1994, the palate (average malignancy ASIR 5.53/100,000/per year) was the most common site (46.7%) between the ages of 20 and 55 because of the high incidence of oral KS (51% of all recorded malignancies) due to the Human immunodeficiency virus infection and acquired immunedeficiency syndrome (HIV/AIDS) epidemic, especially in the young who are usually not affected by squamous carcinoma (38.4% of all recorded malignancies; Figures 4 and 5). The tongue (21.4% – ASIR 3.11/100,000/per year) was more affected by squamous cell carcinoma (60.4%) than by KS (31%). The data are old and the situation will have changed dramatically since then due to the widespread availability of highly active antiretroviral therapies, but later comparable data have not been identified. Anyway the other relative site incidences of malignancy recorded that period were mouth NOS 11.2% (ASIR N/A), floor of mouth 6.6% (ASIR 1.18/100,000/per year), gum 5.2% (ASIR 0.66/100,000/per year), lips total 4% (ASIR 0.63/100,000/per year; upper lip 1.8%, lower lip 2.2%), base of tongue 2.2% (ASIR 0.41/100,000/per year) and buccal mucosa 1.3% (ASIR 0.18/100,000/per year). 18 Another study performed in 2005 on the Zimbabwean population (a total of 358 patients) indicates that the mandibular gingiva is most commonly affected (18.2%), followed closely by the tongue (17.9%) and floor of the mouth (16.2%). The maxillary gingiva was involved in 9%, and interestingly the hard palate in 8% which is unusual for squamous carcinoma. Squamous carcinoma of the buccal mucosa accounted for 9% and the soft palate only for 5%; the lips were the least frequently involved (3.9%). 19

ASIR trends of oral (C01–C06) KS and SCC for the Zimbabwean population 1988–1997. 18 KS: Kaposi sarcoma; SCC: squamous cell carcinoma.

Site specific age-group incidence rates for malignancies (including KS) at the four most commonly affected intra-oral sites (C01, C02, C04, C05) in Zimbabwe between 1988 and 1997. 18 KS: Kaposi sarcoma.
In the study on the SA-NCR data (1996–2002), 14 the tongue with a 34% incidence and mouth (29% incidence) were the sites with the highest oral malignancy rates, followed by much lower rates for tonsils, palate and gum. This can be attributed to the fact that KS was recorded separately from all other cancers by the SA-NCR. Cancer of the lips was recorded in 11% of the sample and the oropharynx had the lowest proportion (1.2%).
Past trends over time in South Africa: Differences by sex and ethnicity
The most comprehensive data for time trends for oral, lip, oropharynx and nasopharynx cancer in South Africa are derived from a detailed analysis of the South African studies on the SA-NCR data between 1988 and 2012. Figure 6 shows that, over this period, the incidence of intra-oral cancer in males shows a decline of about 30% (except in coloured males; see linear trends in Figure 7), 15 whereas the trends in lip cancer in males and intra-oral cancer in females suggest a slight decline only, except in Black females (Figure 8). 15 The decline of oral cancer rates in males could be explained by the following: (1) more aggressive public campaigns warning against the dangers of tobacco with relative steep increases in the price of tobacco products resulting in less consumption, (2) lower reporting rates of cancer data and/or a decline in receipts of reported cases at the SA-NCR in general in the last two decades and (3) an increase in mortality with a decline in the overall survival age of the South African population susceptible to cancer, due to the high HIV/AIDS rates. However, these findings are not supported by the personal observation of oral cancer incidence in the first author’s public histopathology laboratory (greater Western Cape, South Africa).

Line graph with ASIR (100,000/year) trends for lip (C00) and intra-oral cancer (C01–C06) in the males and female population groups in South Africa between 1988 and 2002.

Line graph with ASIR (100,000/per year) trends for intra-oral cancer (C01–C06) in the various male population groups in South Africa between 1996 and 2002. 15

Line graph with ASIR (100,000/per year) trends for intra-oral cancer (C01–C06) in the various female population groups in South Africa between 1996 and 2002. 15
In order to assess differences in epidemiologic trends in squamous carcinoma incidence between the oral cavity (OSCC) and the oropharynx (OPSCC), the respective incidences were separately calculated for South Africa in the period 1997–2001, which is at the beginning of the human papillomavirus (HPV) epidemic. 16 These data revealed that OSCC in all South African males and females accounted for 1.91% and 0.61% of all non-skin cancers, respectively, whereas new OPSCC in all South African males and females accounted for 0.55% and 0.13% of all non-skin cancers, respectively. Interestingly, 7.28% of all OSCC recorded in males of all age groups were found in males younger than 45 years, and 7.84% of all OSCC recorded in females of all age groups were found in females younger than 45 years, compared to 5.2% of OPSCC in males and 11.17% of OPSCC in females younger than 45 years. Indian (‘Asian’) and mixed-race (‘Coloured’) females had the highest proportion of OPSCC incidence in the younger (i.e. <45 year old) age group, that is, 10.9% and 11.45%, respectively. It was concluded that there is a greater incidence of OPSCC in younger females <45 years old when compared to younger males. 16
It has been reported in an independent study conducted by the second author of the present review and colleagues, based on data from by the South African Cancer Registry for the years 1992–2001, that the incidence rates of oral plus oropharyngeal cancer were stable overall but increased significantly among Coloured South Africans over the period (p < 0.05). 20 Cancer specifically involving the oropharynx was most common among Cape Coloureds and showed an increasing trend indicating that at least this section of the population has become affected by the worldwide HPV-driven oropharyngeal cancer epidemic.
In contrast, a significant shift over time (1996–2002) to an earlier age of onset for both OSCC and OPSCC for all males and females was not detected by Ndui 14 and Ndui and Hille 15 (Figures 9 and 10); these authors suggested that there was perhaps a mild but no substantial increase in (putatively HPV-related) tonsillar and oropharyngeal cancers in males during that particular period of the HIV/AIDS epidemic in South Africa (Figure 9). These data are, however, very out of date.

Comparison of the age distribution of combined oropharyngeal and tonsil cancer (C09 and C10) with intra-oral cancer (C01–C06) for all males in South Africa between 1996 and 2002. 15

Comparison of the age distribution of combined oropharyngeal and tonsil cancer (C09 and C10) with intra-oral cancer (C01–C06) for all females in South Africa between 1996 and 2002. 15
Forward projections
GLOBOCAN contains software to project ASIRs. The estimates for sub-Saharan Africa are based on collective data for C00–C06: that is, no separate data for lip, intra-oral and oropharynx cancers; nor for other pharynx (C09–C10, C12–C14). Some registries record cases in crude separate sites as ‘lip NOS’, ‘oral cavity NOS’, ‘tongue’ and ‘gum’ only (e.g. South Africa NCR). Many ASIRs are calculated on low recorded numbers, and as already indicated, we believe there is substantial under-reporting. The five leading causes of death in Africa are AIDS, lower respiratory tract infections (including tuberculosis), diarrhoea, malaria and stroke (available at https://africacheck.org/factsheets/factsheet-the-leading-causes-of-death-in-africa/, accessed 09 June 2017). The reduced lifespan of Africans arising from infectious diseases thus reduces the cancer burden which will increase in the future by both population growth and increasing longevity. To demonstrate the minimum projected rise in intra-oral plus lip cancer burden (C00–C06), we calculated the number of new cases per year for 2020, 2030 and 2050 in sub-Saharan Africa and three major African regions from the GLOBOCAN 2012 data and the United Nations Population Pyramid website (based on United Nations data; available at http://btlas.com or http://www.populationpyramid.net/; accessed 10 June 2017; Figure 11). It can be derived that the minimum number of new oral plus lip cancer cases burdening sub-Saharan Africa will be 29,583 in the year 2020, 37,715 in 2030 and 57,327 in 2050.

Forward projection of the minimum number of new intra-oral and lip cancer (C00–C06) cases for 2020, 2030 and 2050 for sub-Saharan Africa and three major regions. Source: GLOBOCAN 2012 and Btlas/United Nations population pyramids websites.
Aetiology and risk factors of oral cancer in Africa
Traditional risk factors
Mainly due to the diverse ethnicities, extremely variable environments and socio-economic disparities, including a general lack of access to health facilities, the information on cancer aetiology in Africa, as with the epidemiology, has been erratic. Aetiological and risk factors for oral cancer in Africa constitute a long list of abuse of alcohol and/or tobacco in various smoking and smokeless (chewing/snuff) forms 21 –27 but also mixed with other leaf and bark products including Toombak, Khat/Miraa, Areca nut and Kola nut. Toombak is a product of fermented and cured tobacco containing sodium bicarbonate placed in the labial sulcus and predominantly consumed in Sudan and southern Saudi Arabia. 28,29 It is heavily contaminated with microorganisms which likely contribute to carcinogenesis. 30
Khat is a psychoactive stimulant derived from the leaves of the Catha edulis plant, which is a flowering evergreen shrub indigenous to East Africa and parts of the Middle East. 31 Khat (or Miraa as it known in Kenya) holds significant social and cultural importance, particularly in Eastern Africa and the Arabian Peninsula, with increasing consumption in the global diaspora. Adverse systemic and oral effects of Khat consumption are well documented, although it does not appear to be a strong carcinogen. 32,33
Potentially malignant white lesions such as Khat-induced leukoplakia have been described in chewers with higher prevalence among those who simultaneously consume tobacco 34 and alcohol. 35 –37
A large part of the Indian ethnic population in Africa (Kenya, South Africa in particular) still consume areca nut, commonly embedded in betel quid containing shredded tobacco, scented products and slaked lime (Paan masala). It was reported in 1993 that the areca nut is chewed alone by 93% of Indian women in South Africa. 38 The carcinogenic effects of chronically chewing betel quid or areca nut alone have been well documented, especially in the Indian subcontinent, and areca products are now regarded as a class 1 human carcinogen by the IARC. 38,39 Another substance potentially associated with risk for oral cancer is kola nut, a stimulant used both socially and ceremonially in many forested regions of sub-Saharan Africa. 40,41
The following social risk factors have been shown to have associations: poverty, indoor air pollution, poor oral hygiene and poor general health, dental trauma, poor diet lacking antioxidants, lack of general education and public ignorance, no or little access to oral health care, paucity of oral healthcare workers, especially rural and little opportunity for opportunistic oral examination to detect early and late oral cancer. All these factors and influences on global inequalities in incidence and outcomes for oral cancer have been well addressed. 42
HIV/AIDS, HPV and oral cancer
Persistent infection with high-risk genotypes of HPVs is now identified as a major aetiological factor for posterior/base of tongue, tonsillar and other oropharyngeal non-keratinising squamous carcinomas. 43 The burden of HPV-related cancer in Africa is primarily uterine cervix, but a number of head and neck cancers, perhaps of the order of 10% according to data from South Africa, are associated with high-risk HPV types. 44 Southern Africa, the west coast of West Central Africa and Sudan have surprisingly high rates of OPSCC, indicating a possible rise in HPV-related cases. 20 HIV/AIDS rates do not yet appear to have had a stimulatory effect on OSCC and OPSCC rates, unlike in many western countries, 45 but this may be simply because the information is lacking.
HPVs and HIV synergize at molecular, cellular and clinical levels 46 and are frequently co-transmitted. This is especially true of cervical, anal and penile cancer 47 and no doubt of oropharyngeal cancer. The same is true for the markedly increased risk of the human herpesvirus cancers, KS and non-Hodgkin lymphoma in HIV/AIDS patients. 48 This has yet to be established for oral squamous cancer in Africa.
It is suggested that HIV-infected HNSCC patients in sub-Saharan Africa may be younger, with infrequent tobacco and alcohol use, have more aggressive cancers, and suffer from worse clinical outcomes. 27,49 These assumptions are made on limited data.
The study on the SA-NCR data collected between 1996 and 2002 14,15 attempted to determine whether the HIV/AIDS pandemic in South Africa could have increased the incidence and reduced the age of onset of oral squamous cancer. The slightly declining trends in ASIR of oral cancer in males of the Black, Asian and White population groups (Figure 7), and for females of the Asian and White groups as well as a minimal increase in incidence trends noted in females of the Coloured population group (Figure 8) suggest that, at least until 2002, the pandemic HIV infection rate had little or no stimulating influence on the incidence rates of oral squamous carcinoma in South Africa. The year-based age distributions between 1996 and 2002 were also calculated and illustrated for males (Figure 12) and females (Figure 13). Although in general Africans tend to suffer from oral cancer about a decade earlier than in developed countries, there were no significant changes in the shape or shifts of the age-adjusted curves for both South African males and females during the years of peak HIV infection rates. This was confirmed by a study on oral cancer at Kenyatta National Hospital in Nairobi, 50 which reported neither an increase in the frequency nor a change in the pattern of oral cancer in this population in the last decades of the 20th century, despite changes in life style and the emergence of AIDS in Kenya. One might, however, expect to see rising rates of HPV-related cancers in subsequent decades. Speculation on the increasing role of HPV in oropharyngeal cancer in Africa is abundant, particularly in settings where HIV is highly prevalent and where HIV-infected individuals may have increased acquisition and persistence of oncogenic HPV strains at multiple anatomic sites. 51,52 Yet active HPV replicative infection has not been well described in HNSCC (and oral cancer) in sub-Saharan Africa except for the 2010 study by Jalouli et al. 29 who showed that 25% of 217 oropharyngeal/oral cavity squamous cell carcinoma specimens in Sudan were positive for HPV. HPV positivity was reported by anatomic subsite, but clear anatomic trends were not described and oropharynx sites were not exactly identified. Although not statistically significant, more patients reporting toombak use had HPV-positive neoplasms (27%) than those who did not consume toombak (21%), which is contrary to observations in the western world.

Year-based age distribution for intra-oral squamous cancer (C01–C06) in all males in South Africa 1996–2002. There is no significant shift or shape alteration of the line graphs over the various years coinciding with the peak of the pandemic HIV infection. 15

Year-based age distribution for intra-oral squamous cancer (C01–C06) in all females in South Africa 1996–2002. Also, here there is no evidence of influence of the HIV pandemic on the shape of the distribution curves. 15
Quite interesting though, in a follow-up study, in which 155 oral/oropharyngeal squamous carcinomas in eight different countries were tested using polymerase chain reaction for HPV, herpes simplex virus and Epstein-Barr virus, tumour HPV positivity was the highest in Sudan, the only (sub-Saharan) Africa country included in the sampling. 53
Needs for primary prevention and early diagnosis.
A deficiency of specific knowledge by a cohort of Nigerian dental students of the early signs of oral cancer was identified and only 65% of these undergraduate students believed in regular opportunistic screening for the disease. 54 There is a huge educational need among all health professionals and the public to avoid risk factors and to be aware of early signs and symptoms.
Since squamous cell carcinoma of the oral cavity (OSCC) is the most common recorded head and neck malignancy in the African continent, 5,19,25,40,55,56 –60 a lack of awareness and access to advanced head and neck healthcare facilities results in high incidences of advanced, difficult to treat cases at first presentation. 26,61,62
However, during the HIV/AIDS pandemic in Zimbabwe, the incidence of oral KS overtook OSCC in 1994 as the most common oral malignancy 18 and might well apply elsewhere and for the foreseeable future. There is a clear need for research into the role of HPV infection in oral and oropharyngeal cancer in sub-Saharan Africa through comprehensive epidemiological and molecular studies. HPV vaccination may reduce HNSCC burden in the long term, and hopefully it will be introduced for young males as well.
Conclusions and recommendations
The aetiology and risk factors of OSCC in Africa are numerous and varied. However, relatively low tobacco and alcohol consumption rates have been reported by more than half of HNSCC patients in a number of countries implying a different set of risk factors compared to high-income countries. Social norms might prevent patients from openly reporting substance use and sexual behaviours. Registry data are variable and unreliable, with gross under reporting, suitable for estimates and relative comparisons only. The most reliable oral cancer incidence data appear to originate from South Africa and La Reunion. The Zimbabwe and Malawian data report fewer cases than expected.
Southern Africa, the west coast of West Central Africa and Sudan have surprisingly high rates of OPSCC, indicating a possible rise in HPV-related cases. 20 HIV/AIDS rates do not yet appear to have had a stimulatory effect on OSCC and OPSCC rates, unlike in many western countries, but this may be simply because the information is lacking.
Recommendations for future actions in Africa
All African governments need to be lobbied to address the grossly underfunded oral/head and neck/ear, nose and throat healthcare services and the pressing need for a coordinated approach to improving the extent and quality of epidemiological data collecting services for cancer control in the continent. The glaring deficiencies in cancer surveillance systems must be addressed; specifically the quality and validity of the population and pathology-based cancer registries by enforcement of compulsory cancer notification, laboratory confirmation and death certification, using ICD10 descriptors.
8,63
Using GLOBOCAN estimates without proper consideration of their quality will result in misallocation of resources. The true incidence and prevalence of cancer in sub-Saharan Africa remain largely unknown. Much greater emphasis needs to be placed on improving the quality of data collected by these registries, especially where funders use them in support of major global health proposals.
9
Emphasis must be put on targeted education and training in cancer data collection. Correlation studies on tobacco and alcohol consumption with oral cancer/HNSCC incidence must be carried out in countries reporting high or frequencies. Research infrastructure and capacity building must be developed by establishing Oral Cancer Research Collaborations, Linkages and Networks with emphasis on improvement of data collection. HPV vaccination should reduce HNSCC burden in the long term, and it must be introduced for young males as well. The outcomes must be carefully followed. Continued emphasis on primary prevention through public campaigns to limit tobacco and alcohol abuse, to practice sexual hygiene, to encourage healthy diets and to strive for economic advances is needed across the continent.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Global Oral Cancer Forum was totally supported by the funds received from The Henry Schein Cares Foundation.