Abstract
Background
Monoclonal antibodies that target the CD20 antigen and cause B-cell depletion have proven to be a highly effective treatment strategy in relapsing-remitting MS (RRMS). Ofatumumab, the first fully human anti-CD20 monoclonal antibody, was approved for RRMS treatment in 2021 following successful phase III clinical trials. Real-world patient populations are inherently heterogeneous and offer further insights into the impact of therapies on disease activity, adverse events and immunological parameters.
Objective
To evaluate the safety and effectiveness of ofatumumab in a clinic-based MS population.
Methods
Adult RRMS patients attending a clinic in Sydney, Australia, treated with ofatumumab were included. A retrospective review of medical records was undertaken.
Results
A total of 170 patients were enrolled. Mean age was 46.2 years (±11.0). Median duration of treatment with ofatumumab was 18.1 months (11.0–26.0). 96.5% of patients met three-parameter criteria for no evidence of disease activity (NEDA-3) during treatment. No relapses occurred. Eighteen patients had progression independent of relapse activity (PIRA) in the 12 months before ofatumumab, 12 of whom stabilised. Serious infections occurred in 2.9%, and 6.4% stopped treatment due to adverse events. Immunoglobulin levels decreased on treatment, though this was only significant for IgM (
Conclusion
This study demonstrates high effectiveness of ofatumumab in an older RRMS population with longer disease duration than pivotal trials. A positive effect on PIRA was observed in 66.7% of those experiencing it in the year prior. Discontinuation due to adverse events was rare.
Introduction
MS is the most common autoimmune disease of the central nervous system. The frequency of the disease is increasing, with the global prevalence estimated at 2.8 million people worldwide. 1 It is one of the commonest neurological causes of disability in young people and a source of significant burden on healthcare systems. 2
MS was historically considered a T-cell-mediated disease 3 but demonstration of intrathecal antibody synthesis, along with B-cell-containing follicles in parenchymal plaques and leptomeninges, prompted investigation of B-cell depletion as a therapeutic strategy. 4 Monoclonal antibodies that target the CD20 antigen and cause B-cell depletion have since proven to be a highly effective treatment strategy in relapsing-remitting MS (RRMS). 5 Since the approval of ocrelizumab in 2018 and ofatumumab in 2021, B-cell-depleting therapies have significantly reshaped the MS therapeutic landscape in Australia and have become the most frequently initiated class of disease-modifying therapy (DMT) in the country. 6
Ofatumumab, the first fully human anti-CD20 monoclonal antibody, is administered as a monthly subcutaneous injection. In the phase III ASCLEPIOS I and II trials, it demonstrated superior efficacy to teriflunomide in reducing relapse rates and disability progression in RRMS, with a favourable safety profile. 7
Amid growing evidence favouring the early use of high-efficacy DMTs, 8 ofatumumab has become the most frequently initiated treatment for MS in Australia over the past 2 years. 9 Real-world evidence (RWE) is critical for evaluating the long-term safety and effectiveness of therapies across heterogeneous patient populations. Post-marketing safety data from the U.S. Food and Drug Administration's adverse event reporting system align with clinical trial findings, demonstrating a low rate of serious infections on treatment. 10 Furthermore, high efficacy has been reported in several real-world cohorts, particularly among treatment-naïve individuals and those switching from lower-efficacy therapies.11,12
This study provides novel RWE in a highly treatment-experienced RRMS cohort, the majority of whom had prior exposure to high-efficacy therapies (HET), including alemtuzumab, natalizumab and ocrelizumab. It contributes to the understanding of ofatumumab's potential impact on smouldering disease mechanisms, through observations on progression independent of relapse activity (PIRA) – an area of ongoing investigation. 13 It also characterises the immunological profile (lymphocyte subsets and immunoglobulins) of a B-cell-depleted cohort with a significant history of prior DMT exposure. This is clinically relevant, as anti-CD20 therapy reduces immunoglobulin levels, with treatment history and duration potentially influencing hypogammaglobulinaemia risk. 14
Methods
This is a retrospective cohort, observational study of RRMS patients treated with ofatumumab between March 2021 and December 2023. Ethics approval was obtained from the Northern Sydney Local Health District Human Research Ethics Committee. Given the retrospective cohort design and the use of routinely collected clinical data within a real-world MS clinic setting, individual informed consent was not required.
Study population
Patients were identified from a clinic database at a tertiary MS referral centre. Inclusion criteria were (i) age over 18 years inclusive; (ii) continuous treatment with ofatumumab for a minimum of 6 months; (iii) prescribing of ofatumumab is compliant with the Australian Pharmaceutical Benefits Scheme (PBS) eligibility criteria 15 ; (iv) a diagnosis of RRMS according to the 2017 McDonald criteria. 16
In the Australian setting, ofatumumab is subsidised under the PBS for relapsing forms of MS only. Although patients with sustained progression independent of relapse activity (sPIRA) in this study may eventually transition to secondary progressive MS (SPMS), the criteria for SPMS – defined here as continuous disease progression over a period of at least 12 months without any relapse activity 17 – were not met by any patients by the study end date.
Study design
A retrospective review of electronic medical records, MRI, and pathology results was conducted for the 12 months prior to, and during, ofatumumab treatment. Data extracted included patient demographics, disease duration (from symptom onset), and prior DMT exposure. Patients were assessed every 6 months by their treating neurologist, with documentation of adverse events and scoring on the Expanded Disability Status Scale (EDSS). Brain and spinal MRI scans were performed at 6-month intervals and included T1, T2-FLAIR, and post-contrast T1-weighted sequences. All imaging was performed on 1.5 or 3 Tesla Phillips MRI scanners and reported by neuroradiologists.
The primary outcome was No Evidence of Disease Activity (NEDA-3), assessed at 6-month intervals, including retrospectively for the 12 months preceding ofatumumab initiation in treatment-experienced patients. NEDA-3 was defined as: (i) absence of clinical relapses; (ii) no EDSS progression; (iii) no new or enhancing lesions on MRI.
Patients who did not meet NEDA-3 criteria were further classified as having relapse-associated worsening (RAW) or PIRA. RAW was defined as a confirmed disability accumulation event following a stable or improving neurological state for at least 30 days, accompanied by new or worsening neurological symptoms and objective findings and/or new T2-FLAIR or contrast-enhancing lesions on MRI. PIRA was defined as a sustained EDSS increase over 90 days of ≥1.0 point if the baseline EDSS was <5.5, or ≥0.5 if the baseline EDSS was ≥5.5, in the absence of any relapse activity.
Patients underwent blood sampling before and six-monthly during treatment with ofatumumab to assess lymphocyte subsets and immunoglobulin levels. Immunoglobulins, total lymphocyte count, CD4+ and CD8+ counts were recorded chronologically for each patient. Only those with blood results at the time of or before drug commencement and after a minimum of 3 months of treatment were included.
Adverse events were obtained from medical records. Mild injection-related reactions were not included, as these are inconsistently recorded. Serious infections were defined as those requiring intravenous antibiotics, hospital admission or resulting in permanent organ injury. Infections not meeting these criteria are referred to as minor infections. Patients were classed as having recurrent infections if two or more were reported per 12 months.
Statistical analysis
Statistical analysis was undertaken using SPSS, version 28.0. The continuous outcomes are summarised with a mean and standard deviation (SD) when normally distributed and with a median and interquartile range (IQR) otherwise. Paired samples t-test was used to determine significant changes in pathology results over time. Continuous variables, including demographics, were compared between pPIRA and non-pPIRA groups using independent sample
Results
Demographics and clinical characteristics
A total of 170 patients were identified. The cohort was 74.7% female, with a mean age of 46.2 years (±11.0). Patients over the age of 55 comprised 27.1% of the sample (Figure 1). The median duration of ofatumumab treatment was 18.1 months (IQR 11.0–26.0). Median EDSS was 1.0 (IQR 0.0–1.5), and median MSSS was 0.64 (IQR 0.3–1.8). The mean disease duration, defined from symptom onset, was 12.0 years (±9.2). Most patients (89%) were DMT-experienced, with ocrelizumab being the most common prior DMT (30%), followed by natalizumab (25.3%) (Figure 2). A minority (11%) were DMT-naïve, with a lower median EDSS of 0 (IQR 0–1).

Age distribution of patients (%).

DMT prior to ofatumumab.
The leading indication for switching to ofatumumab was disease progression in the preceding 12 months, accounting for 25.9% of cases (Figure 3). Among these, 69.0% had prior RAW (pRAW) and 31.0% had prior PIRA (pPIRA). ‘Other drug side effects’ were cited in 17.1% of cases, most associated with fingolimod (30.7%), ocrelizumab (19.2%), and teriflunomide (15.4%). Natalizumab was discontinued in 15.9% of patients due to JC virus seroconversion. The main indications for switching from ocrelizumab to ofatumumab were recurrent infections (29.4%) and ‘wearing off’ effect (29.4%). The latter was characterised by increase in MS symptoms such as fatigue and brain fog in the weeks before the next ocrelizumab infusion.

Indication for switching to ofatumumab.
Efficacy
No patients experienced a relapse on ofatumumab, and 96.5% of patients met NEDA-3 criteria.
Eighteen patients (10.6%) had pPIRA, all of whom were DMT-experienced (Table 1). Of these, 12 (66.6%) showed no further progression on ofatumumab after initiating ofatumumab (median treatment duration of 18 months (IQR 7–30). Two patients had EDSS improvement, while six had sustained PIRA (sPIRA) and accounted for the 3.5% of patients who did not achieve NEDA-3 on ofatumumab (Figure 4). Of these six, three had pPIRA on ocrelizumab, two on natalizumab and one on dimethyl fumarate.
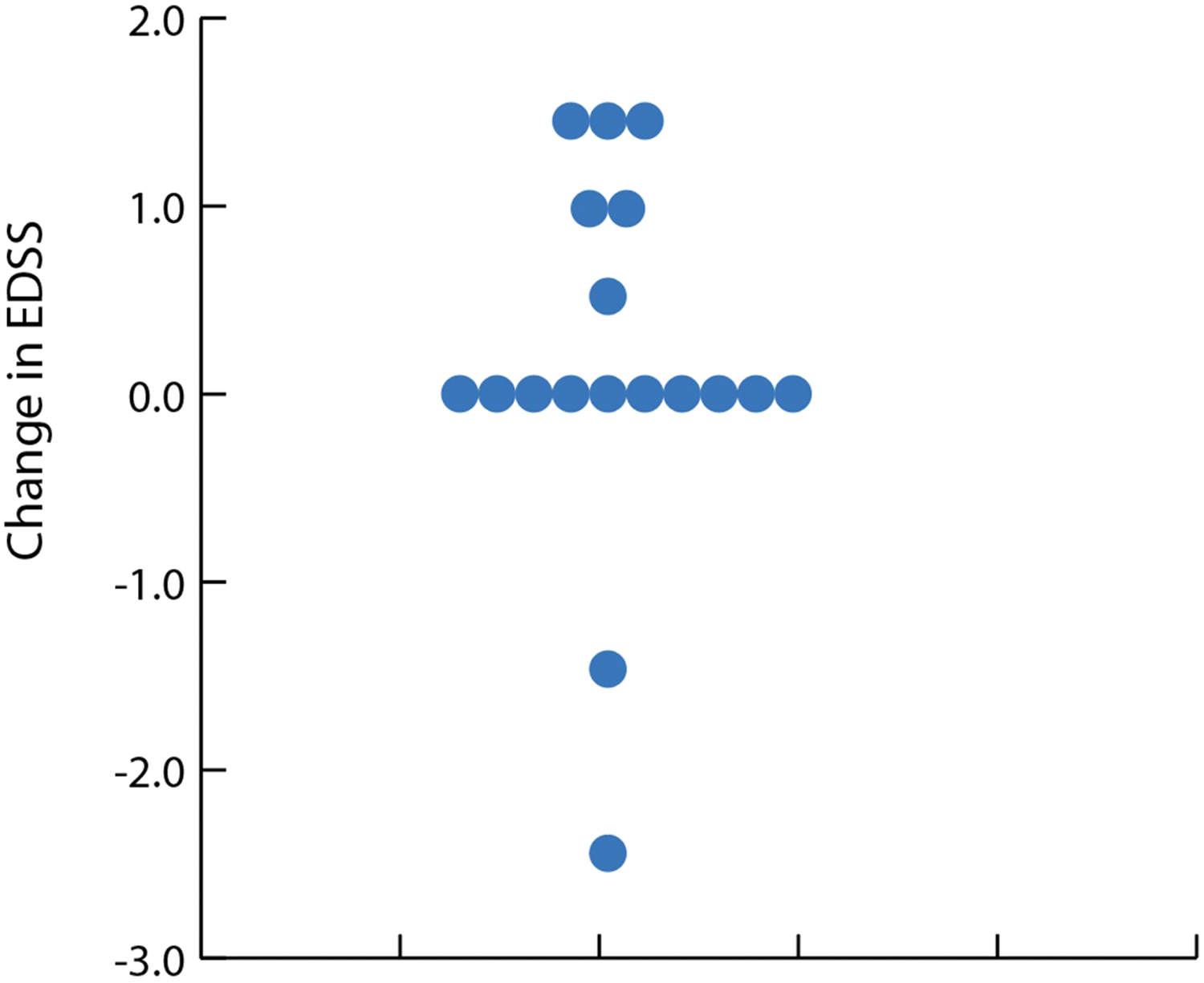
Dot plot of change in EDSS after a median duration of 19.5 months (12–30) treatment with ofatumumab in pPIRA patients.
Characteristics of patients with pPIRA, pRAW and sPIRA.

pPIRA: prior progression independent of relapse activity; pRAW: prior relapse-associated worsening; sPIRA: sustained progression independent of relapse activity.
The pPIRA subgroup had distinct clinical features: their mean age was 60.2 ± 9.3 years – 14 years higher than the overall cohort (
Disease activity in ≤55 years compared to ≥55 years.

NEDA-3: no evidence of disease activity; pPIRA: prior prior progression independent of relapse activity; sPIRA: sustained progression independent of relapse activity.
Safety
6.5% (11) discontinued ofatumumab due to adverse events, including disease progression (Table 3). Three patients progressing on ofatumumab stopped treatment in preparation for autologous haematological stem cell transplant (AHSCT). Five patients stopped due to serious or recurrent infections. Three patients developed new-onset seronegative spondyloarthritis requiring treatment cessation.
Indications for stopping treatment.

25.3% (43) reported infections on treatment (Figure 5). Minor respiratory tract infections, including COVID, were most frequently reported. Only 2.9% experienced serious infections, which were community-acquired pneumonia and urinary sepsis. Serious infections were more common in those over 55 years (6.52% vs 1.6%). 58.7% of this age group had no recorded adverse events, similar to the under 55 years’ group at 60.5%.

Adverse events of ofatumumab.
Immunological parameters
62 patients met criteria for assessment of immunoglobulins. The remainder had incomplete or missing pathology results. A significant reduction in IgM levels occurred (
Ocrelizumab to ofatumumab group
The main indications for switching from ocrelizumab to ofatumumab were recurrent infections (29.4%) and a ‘wearing off’ effect (29.4%). The latter was characterised by a significant increase in MS symptoms such as fatigue and brain fog in the weeks before each ocrelizumab infusion. Seven people (13.7%) had pPIRA on ocrelizumab, and none had pRAW. Three pPIRA patients stabilised prior to switching to ofatumumab and only switched due to preferred route of administration. Three of the pPIRA patients had sustained progression and one stabilised on ofatumumab. Of those switching due to infections, only 27% continued experiencing recurrent infections on ofatumumab.
Discussion
In this real-world retrospective analysis, ofatumumab is highly effective in treating RRMS, with 97% of patients demonstrating NEDA-3 after a median of 18 months. Ofatumumab is well tolerated in practice, with only 7% of patients discontinuing treatment due to adverse events. Compared to the pivotal clinical trial, this study population is older (46.2 years) with a longer disease duration (12.0 years). 7 Importantly, 27% of the cohort are over the age of 55 years, a group that were excluded from ASCLEPIOS I and II but has considerable relevance clinically, and in whom the balance between disease control and immune senescence is pertinent. Older patients also exhibited a high frequency of NEDA-3 (98%), and the percentage reporting no adverse events was similar to those under 55 years.
Compared to clinical trials, where 40% of patients were DMT naïve,
7
only 12% of this study population started ofatumumab
Nonetheless, controlling MS is more difficult with longer disease duration, 18 and so we sought to identify patients who showed disability worsening despite HET. Of DMT-experienced patients, 44 (30%) patients started ofatumumab due to disease activity in the 12 months before switching, 41% of whom had PIRA (pPIRA). By including PIRA patients, this real-world study facilitated the investigation of ofatumumab upon ‘non-acute’ inflammatory mechanisms and by inference smouldering inflammation, where the benefit of B-cell depletion is less established. 13
Only six patients had confirmed disability accumulation on ofatumumab, all of whom had pPIRA. No one demonstrated relapse activity and ofatumumab reduced PIRA by 66.7% in patients who experienced it in the 12 months prior to switching. Our findings support existing research that PIRA, rather than RAW, is the primary driver of disability progression in RRMS patients on high efficacy HET. 19 We found that PIRA patients were older than RAW patients by 9 years, had a longer disease duration and a higher level of baseline disability. Similar phenomena were reported by Portaccio et al., 20 albeit only 6% of that cohort were on high efficacy DMTs.
Although anti-CD20 therapies have been shown in some studies to have limited effect on chronic active lesions (CALs), 13 the clinical findings from this analysis indicate PIRA, and therefore some form of indolent inflammatory activity, is partly treatable by B-lymphocyte depletion. In a retrospective study determining the utility of serum neurofilament light (sNfl) in the OPERA I and II studies, Ocrelizumab was shown to suppress sNfl levels compared to Interferon-Beta 1b. 21 In patients on interferon-beta 1b without acute activity, elevation in sNfl correlated with increasing brain atrophy, disability progression and chronic active lesions (CALs), whereas among Ocrelizumab-treated patients if sNfl was elevated, this was associated only with increasing brain atrophy and T1 lesion volume. 21 Taken together, these findings raise the possibility that the inflammatory component of progression (for example, CALs might respond partly to treatment, but that degeneration (increasing T1 hypointensity and some contribution to brain atrophy) does not. As volumetric MRI analysis was not performed here, we cannot confirm that the reduction in PIRA on ofatumumab is attributable to an effect on CALs, and this is an important future direction.
It is interesting that three of the four patients who switched from ocrelizumab to ofatumumab due to PIRA continued to progress, and we postulate that their disability increase is predominantly related to neurodegenerative mechanisms. The one patient who stabilised following the switch may have benefited from cumulative duration of B-cell treatment and efficacy of one over the other cannot be inferred from this. Existing real-world evidence comparing efficacy of ocrelizumab to ofatumumab reported comparable control of disease activity over 12 months. 22
Immunological parameters and infection
We observed that IgG was not significantly reduced in patients on ofatumumab, excepting the study period was relatively short and a minority of patients were included in analysis of immunological parameters. Long-term monitoring is appropriate. The rate of infections reported by patients was moderately high at 25%. Serious infections were rare (3%) but more frequent in those over 55 years of age (6.5%) and clinicians should be aware of this difference. There was a significant decrease in IgM, but this did not correlate with increased infection rates. This is consistent with findings from the ASCLEPSIOS trials and supports the understanding that reduction in IgG, total lymphocyte count, CD4 and CD8 cells are more pertinent to infection risk. 23 Ofatumumab did not have a significant deleterious effect on total lymphocyte count or T-cell counts.
Other adverse events
Overall, ofatumumab was well tolerated across both age groups in this study. The occurrence of secondary autoimmune phenomena, albeit rare, is of note and was characterised here by onset or exacerbations of psoriasis and seronegative spondyloarthritis in six patients. This has also been observed with rituximab and ocrelizumab. 24
Study limitations
This study has several limitations. The real-world data approach is prone to several biases. 25 Clinical data, such as EDSS and adverse events relied on medical records. Although the EDSS is a standardised tool, there may be variability between raters. MRI scans did not include volumetric analysis of lesion size or cerebral volume, and as such, we cannot comment on the impact of slowly expanding lesions or brain atrophy, which are important prognostic indicators in MS. Documentation and patient reporting bias cannot be excluded entirely, although the data was derived from one clinic with the oversight of an experienced group of clinicians. Therefore, it is likely that most significant adverse events were identified. Further, the data presented here is granular, and this may provide measures beyond population-based analyses. As this study was performed within a tertiary MS referral centre, the cohort may not represent the full spectrum of RRMS patients, and generalisation should be cautioned.
Conclusion
This real-world analysis reinforces the high efficacy and tolerability of ofatumumab in a broader RRMS population, including older patients with longer disease duration. Our findings highlight the relevance of PIRA as a driver of disability and suggest that ofatumumab reduces this process. Future efficacy studies should include analysis of CALs, brain volume and measurement of global markers of neural injury, particularly serum neurofilament light-chain and glial fibrillary acidic protein.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a research grant from Novartis Pharmaceuticals.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: John Parratt received honoraria for speaking engagements from Novartis Pharmaceuticals. Turlough Montague received travel support to attend ECTRIMS 2024 from Novartis Pharmaceuticals. The other authors have no conflicts of interest to declare.
Data availability statement
The data that has been generated and analysed during the current study is not publicly available due to institutional data-sharing policies and confidentiality considerations. However, these datasets are available from the corresponding author upon reasonable request and with appropriate permissions.
