Abstract
Background
The UK Risk Sharing Scheme (RSS) provided information on the effect of first-line multiple sclerosis (MS) disease-modifying treatments on long-term disability.
Objective
The aim is to provide results specific to glatiramer acetate (GA; Copaxone®) from the final 10-year analysis of the RSS.
Methods
A Markov model was used to assess clinical effectiveness measured as Expanded Disability Status Scale (EDSS) progression and utility loss. Untreated patients from the British Columbia MS cohort (1980–1995) were used as a ‘virtual comparator’ group. A separate Markov model assessed cost-effectiveness, based on a 50-year time horizon (with a 50% treatment waning effect imposed at 10 years) and using NHS list price (£513.95 per 28 days). Results were expressed in quality-adjusted life years (QALYs).
Results
In total, 755 patients with relapsing–remitting MS (RRMS) received GA, with a mean follow-up of 7.1 (standard deviation 1.3) years. EDSS progression was reduced by 23% (progression ratio 76.7, 95% confidence interval [CI] 69.0–84.3) and utility loss by 39% (progression ratio 61.0, 95% CI 52.7–69.3) compared with no treatment. There was no persistent waning in GA treatment effect over time (EDSS: p = 0.093; utilities: p = 0.119). The cost per QALY was £17,841.
Conclusion
GA had a beneficial effect on long-term disability and was a cost-effective treatment for RRMS.
Keywords
Introduction
Multiple sclerosis (MS) is a neurological disease that imposes a considerable burden on patients, healthcare systems and society.1–4 At diagnosis, relapsing forms of MS are most prevalent, 5 with relapsing–remitting MS (RRMS) accounting for around 80% of MS cases. 6 RRMS is characterised by periodic, transient relapses interspersed with periods of recovery and an accumulation of disability associated with incomplete recovery from relapses. 7 Disease-modifying treatments (DMTs) for MS, such as glatiramer acetate (GA) and the beta interferons, have been available since the late 1990s, and are able to reduce the rate of relapses in RRMS. 8 GA, under the brand name Copaxone®, has accumulated more than 2 million patient-years of experience in people with relapsing forms of MS over the past two decades. 9 GA is classified as a non-biological complex drug (NBCD), with a composition that is dependent on a closely controlled manufacturing process.10–12 The exact mechanism of action for GA is not fully understood, but it is thought to modify immune processes responsible for the pathogenesis of MS.13,14
Copaxone® and the beta interferons (Avonex®, Betaferon®, Rebif®) were assessed by the National Institute for Clinical Excellence (NICE; now known as National Institute for Health and Care Excellence) in 2002, 15 the first DMTs for MS to be appraised by NICE. In response to the limitations highlighted during this appraisal regarding the extrapolation of long-term trends from short-term clinical studies, the Risk Sharing Scheme (RSS) was established. 16 The RSS provided cost-effective provision of these DMTs to the National Health Service (NHS) (through discounted costs for some products including Copaxone®) while collecting long-term data on their clinical and cost-effectiveness through a 10-year observational study. 16 The RSS included mechanisms for price changes based on interim analyses to ensure that the scheme DMTs continued to be supplied at a cost-effective price. 16 Copaxone®, in line with the other drugs within the RSS, was assigned an individual cost-effectiveness target for reduction of Expanded Disability Status Scale (EDSS) worsening predicated on data from its registration studies. Based on its performance in exceeding its target, Copaxone® was granted an increase to list price after the scheduled analyses at year 6 demonstrated its cost-effectiveness.17,18
The aggregate results for all four DMTs in the RSS at 2 years, 19 6 years, 20 and, recently, the final 10-year analysis have been published. 21 For the RSS DMTs in aggregate, the 10-year analysis, using the Markov model, reported a 14% reduction in EDSS progression (ratio 86%) and 31% reduction in utility loss (ratio 69%) in patients with RRMS. The treatment effect corresponded to an absolute reduction of 0.25 EDSS units compared with that predicted by natural history. It was also found that while the treatment effect of the DMTs was maintained over 10 years, effectiveness appeared to decrease with time. 21 The results for GA alone in the RSS are presented in this paper, focussing on the clinical and cost-effectiveness of this DMT over 10 years of real-world usage.
Methods
RSS methodology
Full details on the methodology of the RSS have been published previously.20,22 In brief, eligibility for entry into the RSS was governed by the 2001 Association of British Neurologists (ABN) criteria which, for RRMS, were: aged 18 years or older; two clinically significant relapses in the previous 2 years; and an EDSS score ≤5.5. 16 Patients were recruited at 72 sites between January 2002 and July 2005. Treatment allocation for eligible patients was through patient and physician preference at the time of prescribing. Patients had annual reviews, with assessment of EDSS carried out irrespective of changes to treatment (except when in relapse).
Comparator cohort
The RSS included no untreated/placebo patients and instead relied on a ‘virtual comparator’ group based on data from untreated patients in the British Columbia MS (BCMS) database (1980–1995) who met the ABN 2001 eligibility criteria. 22 Comparison of baseline characteristics between RSS and BCMS patients was assessed by t-test and Chi-square test, as appropriate. Transition probabilities for EDSS progression were derived from the BCMS data and used to drive a continuous Markov model that modelled the expected progression of disability without DMT treatment in the RSS cohort. The model adjusted for differences in baseline characteristics between the BCMS cohort and the RSS population. The model used EDSS figures rounded down to the nearest integer (for half point scores), age of onset as a covariate (<28 years or ≥28 years), and had a time horizon of 10 years. 22
Assessment of clinical effectiveness
Disease progression
The primary clinical effectiveness outcome of the RSS was disease progression measured as an accumulation of disability. This was measured in two ways: through progression measured on the EDSS and through utilities to provide a measure of quality of life (derived from EDSS scores based on surveys conducted by the MS Trust). 20 These outcomes were expressed in two different ways: as a progression ratio and as an implied hazard ratio (HR). The progression ratio was the ratio (expressed as a percentage) of the observed progression in the RSS cohort to expected progression off treatment, as calculated by the Markov model from the ‘virtual comparator’ cohort. Implied HRs were calculated as the treatment HR that was required for the modelled data to match the observed outcome data.
Other outcomes
The absolute reduction in EDSS progression on therapy versus that predicted without therapy was calculated, as was the cumulative benefit of GA in EDSS years over the 10-year study. Median time to sustained EDSS 6 was estimated by Weibull analysis, controlling for gender and baseline EDSS. Waning of treatment effect over time was evaluated for both EDSS progression and utility loss by linear regression and also by t-test on year-2 versus year-10 implied HRs.
Sensitivity analyses
A range of sensitivity analyses were conducted to check the robustness of the Markov model in terms of the GA data, similar to those conducted on the aggregate data. 21 These included: eliminating late starters (more than 90 days between baseline assessment and therapy initiation); including patients with switch to another RSS DMT only (primary analysis excluded all patients with a therapy switch); including patients with any therapy switch; missing values for year 10 used year-9 data carried forward; and using year 1 as the baseline.
Assessment of cost-effectiveness
Cost-effectiveness was modelled using a separate Markov model developed for the original NICE appraisal.23,24 The final model was adapted to use the BCMS transition probabilities, 22 costs were inflated to 2015/16 prices, 25 and the analyses run over a 50-year time horizon. The implied HR for utility was used as the clinical effectiveness input, with a 50% treatment waning effect applied at 10 years. 20 The list price of Copaxone® was used for all analyses (£513.95 per 28 days/£6,701 per annum). 26 Results were expressed using quality-adjusted life years (QALYs).
Sensitivity analyses
Sensitivity analyses included disability progression being varied to match the 95% confidence intervals (CI) of the implied HR; the time horizon being reduced to 40, 30 and 20 years; use of UK MS Survey health state costs (the preference of NICE in recent multiple sclerosis appraisals), 27 rather than costs derived by the School of Health and Related Research at the University of Sheffield (ScHARR) as part of the original NICE appraisal, which were used in the base case; 24 and all other inputs being varied by ±10%.
Analyses and modelling were conducted using Excel 2010, including Visual Basic for Applications (VBA), and SPSS 15.0 for Windows.
Standard protocol approvals, registrations, and patient consents
Ethical approval for the RSS was given by the South East Medical Research Ethics Committee (MREC 2/01/78) and all patients gave written consent. 22 BCMS patients gave consent to be enrolled in the BCMS database and the University of British Columbia's Clinical Research Ethics Board approved the study. 22
Data availability statement
The BCMS database is held at the host institution, and analysis and access to the data are limited to on-site access. 22 The RSS operated under the oversight of a Steering Group representing all the parties to the Scheme (the four UK health departments, the four companies whose products were made available through the Scheme, the Association of British Neurologists, the UK MS Specialist Nurses Association, the Royal College of Nurses, and the MS Trust). The Steering Group was administered by the MS Trust.
Results
Patient disposition and baseline characteristics
In total, 979 patients with RRMS were treated with GA in the RSS, with data available from 755 patients (77.1%) at the 10-year analysis. Two patients with secondary progressive MS (SPMS) received treatment with GA in the RSS, but their results were excluded from analysis. Patients who received GA were older at disease onset (median 30 vs. 28 years, respectively; p = 0.0158) and had a higher EDSS at baseline (median 3 vs. 2; p = 0.0019) compared with untreated patients in the BCMS cohort (Table 1).
Baseline population characteristics.

BCMS: British Columbia Multiple Sclerosis; EDSS: Expanded Disability Status Scale; GA: glatiramer acetate; IQR: interquartile range; SD: standard deviation.
EDSS scores are half-integers.
Clinical effectiveness
GA was associated with a 23.3% reduction in EDSS progression (ratio 76.7; 95% CI 69.0–84.3; p < 0.001) and a 39.0% reduction in utility (quality of life) loss (ratio 61.0; 95% CI 52.7–69.3; p < 0.001) compared with no treatment (Table 2). The implied HR was 83.5% (95% CI 78.3–88.8) for EDSS progression and 75.0% (95% CI 69.8–80.3) for reduction in utility loss. The results corresponded to an absolute reduction of 0.36 EDSS units versus that predicted with no therapy (1.2 actual vs. 1.5 predicted). The cumulative benefit of GA over the 10-year study (and the basis for any cost-effectiveness calculation over this period) was 2.4 EDSS years (95% CI 1.7–3.2). The median length of time for the GA cohort to reach an EDSS score of 6.0 was calculated to be 12.9 years.
Clinical effectiveness of glatiramer acetate using 10-year data from the RSS.
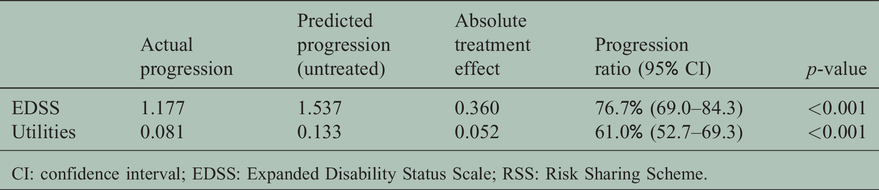
CI: confidence interval; EDSS: Expanded Disability Status Scale; RSS: Risk Sharing Scheme.
There was no evidence of a substantial waning of treatment effect for GA over time (EDSS: p = 0.093; utility: p = 0.119; Figure 1). EDSS progression and utility progression (loss) over time for GA versus that predicted without treatment are shown in Figures 2 and 3, respectively. To further evaluate waning, direct comparison between implied HRs for GA at year-2 and year-10 was conducted and showed no significant difference in terms of utilities (69.3% vs. 75.0%, respectively; p = 0.142), but a significant difference for EDSS (74.6% vs. 83.5%; p = 0.0213).

Implied hazard ratios over 10 years for glatiramer acetate.
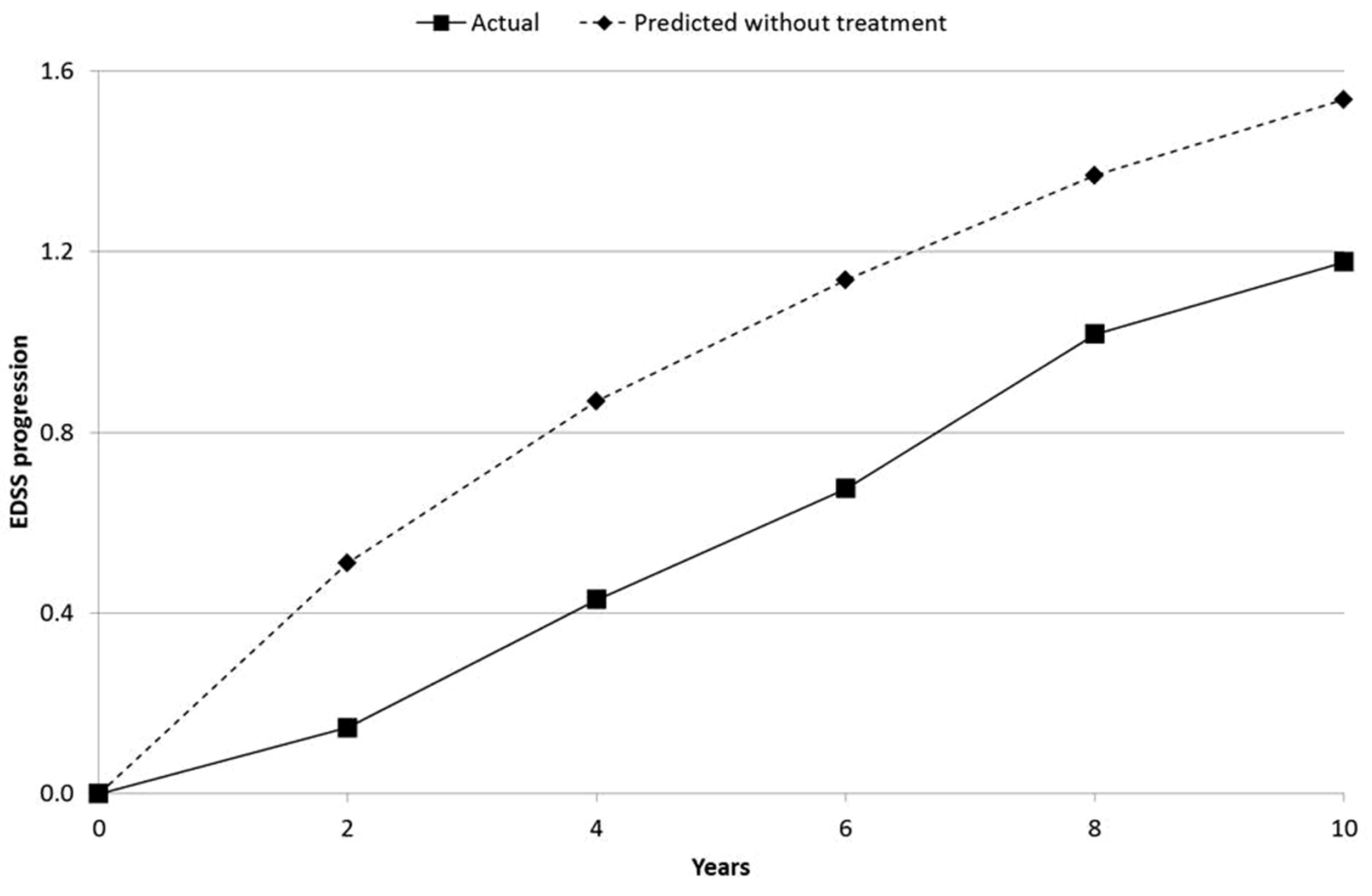
EDSS progression over time for glatiramer acetate versus that predicted without treatment.

Utility progression (loss) over time for glatiramer acetate versus that predicted without treatment.
Sensitivity analyses
The sensitivity analyses had a relatively limited impact on the progression ratios, particularly for elimination of late starters and imputation of missing year-10 values, with all results being within 8% of the primary analysis for EDSS progression and 6% for utilities (Table 3). The inclusion of patients who switched treatments (either to another RSS DMT or to any DMT) shifted the results towards a lower effectiveness (EDSS: +3.5–5.8%; utilities: +3.0–5.4%), but remained within the CIs of the primary analysis. In contrast, the use of year-1 data as treatment baseline led to increased effectiveness of GA (EDSS: 69.0% vs. primary analysis 76.7% [–7.7% improvement]; utilities: 55.2% vs. 61.0% [–5.8% improvement]); results, again, remain within the primary analysis CIs.
Sensitivity analyses on clinical effectiveness results for glatiramer acetate.

*Annual follow-up date was more than 90 days after the baseline assessment date and therapy initiation. #Year 9 assessments carried forward where year 10 data were missing. †Year 1 data used as the baseline (i.e. Year 0 was disregarded). DMT: disease-modifying treatment; EDSS: Expanded Disability Status Scale; RSS: Risk Sharing Scheme.
Cost-effectiveness
The cost per QALY for GA was £17,841 at Copaxone® list price during the RSS study.
Sensitivity analyses
One-way (univariate) sensitivity analysis (assessing the impact of varying one variable in the model at a time) revealed that the implied HR for utility input had a substantial impact on cost-effectiveness (Supplementary Figure 1; Supplementary Table 1). The cost per QALY ranged from £9,633 to £30,429 at the lower and upper bounds of the 95% CI of the implied HR. Updating the health state costs to those from the UK MS Survey increased the cost per QALY to £33,308. Other inputs had a relatively limited impact on the cost-effectiveness of GA, with the cost per QALY remaining below £30,000 when the time horizon was reduced to 20 years.
Discussion
The RSS was established to provide long-term disability progression data for GA and the beta interferons, while supplying these DMTs to the UK NHS at a cost-effective price. 16 For patients with RRMS, the final 10-year analysis supported the DMTs in aggregate having a positive effect on long-term disability, with the Markov model producing an EDSS progression ratio of 86% and a utility progression ratio of 69%. 21 Including all patients (RRMS and SPMS), the cumulative benefit of the RSS DMTs was 1.3 EDSS years and there was a median of 12.5 years to reach EDSS 6. While the benefit of the DMTs was maintained over 10 years, there was evidence of a waning in treatment effect over time. 21 Our study has now reported the corresponding results for GA alone.
The final, 10-year results from the RSS suggest long-term effectiveness of GA and its ability to slow disability progression in patients with RRMS. GA was associated with a 23% reduction in EDSS progression and a 39% reduction in loss of utility compared with no treatment. The clinically relevant outcome of time to EDSS 6 was estimated to be a median of 12.9 years on GA therapy. Importantly, there was no evidence of a substantial waning in treatment effect for GA within the 10-year timescale of the study; there was no significant trend in treatment effect over time (EDSS: p = 0.093; utility: p = 0.119), and the implied HRs (i.e. treatment effect) at 2 and 10 years were not significantly different for utility loss (p = 0.142), albeit different for EDSS progression (p = 0.0213). The sensitivity analyses undertaken reinforced the validity of the results, with none of the analyses having a meaningful impact on the progression ratios. Other studies have also reported the longer-term effectiveness of GA at reducing relapses and delaying disability.28,29 At the 20-year follow-up of the US Glatiramer Acetate Trial, the cumulative annualised relapse rate over the entire study period in patients on GA treatment was 0.2, with 63% of patients with an EDSS score of <4 at baseline remaining below EDSS 4. 30
GA was calculated to be a cost-effective treatment for RRMS in the RSS (at Copaxone® list price), with a cost per QALY below the stringent threshold of £20,000 (base case: £17,841). In almost all modelled scenarios, GA remained highly cost-effective, with the main exceptions being when using the upper 95% CI for the implied HR for utility and when using an alternative set of health state costs (UK MS Survey), where, in both cases, the cost per QALY increased to approximately £30,000. Nursing/infrastructure contributions to MS services provided by Teva (and the other companies) were also considered as part of the RSS methodology for assessing cost-effectiveness of the DMTs; taking these contributions into consideration would reduce the cost per QALY below the current base case of £17,841. It is worth noting that the imposed waning assumption used in the modelling (50% step reduction at 10 years) should be questioned and may be considered disproportionate for GA, with the observed reduction in the implied HR for utility from year 2 to year 10 being only 18.5%; again, supporting the cost-effectiveness of this treatment.
The strengths and weaknesses of the RSS have been discussed in a number of publications.19–24 Key weaknesses include the facts that there was no comparator group, treatment allocation was non-random (by patient and physician preference), and the availability of utility data relating to EDSS scores and associated health state costs was somewhat limited. The BCMS data come from an earlier period than the RSS data and, as such, might reflect a more severe MS population, as evidence from the placebo arms of clinical trials have indicated a steady improvement in the natural history of MS since the 1980s.31,32 This may represent a confounding factor in relation to the treatment effect seen in the RSS. However, it should also be recognised that the apparent improvement in the natural history of MS might be an artefact of evolving trial inclusion criteria and conduct, as population-based studies, such as the BCMS, have not shown evidence for milder prognosis in more recent cohorts. 33 Furthermore, during the course of the RSS the number of MS specialist nurses employed rose from 80 to over 240 across England and Wales. 34 Although their initial key task in the RSS was to support the administration of injectable DMTs, patients’ treatment adherence and monitoring of (adverse) events, the role of the MS nurse evolved significantly such that a direct impact of improved nursing care on disability and utility outcomes cannot be excluded. 35 While this element of the MS RSS has been neglected in previous publications on the subject,20–22 it is evidently of generic relevance, that is, not specific to the outcomes for GA reported here.
The RSS was not designed to be a comparative study and caution should be exercised when drawing any conclusions about the relative effectiveness of the DMTs. Although there was undoubtedly a degree of bias in selection of treatment, the results from the RSS reflect the real-world effectiveness of GA within NHS practice. The aggregate RSS results were also modelled using a multilevel model, which provided insights into the strengths and weaknesses of the Markov model. 21 Of note, it was found that the Markov model was particularly strong at modelling utility loss. 21 This insight indicates that the implied HR for utility was arguably the optimal clinical effectiveness input for analysing cost-effectiveness in the Markov model. Perhaps more importantly, it also strengthens the conclusions that can be drawn, as the utility results for GA showed a stronger treatment effect and less evidence of waning than did those for EDSS.
The RSS is a unique study that provides long-term clinical and cost-effectiveness data on GA in a large cohort of patients followed for 10 years. The RSS demonstrates that, in a real-world setting, GA is an effective treatment that is able to slow disease progression in RRMS, with no evidence of a substantial waning in treatment effect over time. GA was also shown to be a cost-effective treatment at Copaxone® list price. The RSS provides prospective, long-term disability data, albeit with some limitations, which are lacking for most of the other DMTs, whose long-term benefits are inferred by their demonstration of short-term anti-inflammatory effects.
Conflicts of interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: Gavin Giovannoni has received compensation for serving as a consultant or speaker for, or has received research support from AbbVie, Atara Bio, Actelion, Bayer Schering Healthcare, Biogen, Canbex, Eisai, Elan, Five Prime Therapeutics, Genentech, GlaxoSmithKline, Ironwood Pharmaceuticals, Merck Serono, Novartis, Pfizer, Roche, Sanofi-Genzyme, Synthon BV, Teva Pharmaceutical Industries, UCB and Vertex Pharmaceuticals.
Peter Brex has received honoraria from Teva, Genzyme, Biogen, Merck Serono and Roche for attending advisory boards or speaking at meetings. He has attended meetings as the guest of Biogen, Merck Serono and Genzyme. His department has received a grant from Biogen to support service developments.
Dalbir Dhiraj is an employee of Teva UK Limited, the manufacturer of Copaxone®.
John Fullarton, Matthew Freddi and Barry Rodgers-Gray are employees of Strategen Limited which has received consultancy and research fees from Teva.
Klaus Schmierer has been a PI on trials sponsored by Medday, Novartis, Roche and Teva, and a member of the steering committee of MAGNIFY-MS (Merck). He has received honoraria and meeting support from Biogen, Lipomed, Merck Serono, Sanofi-Genzyme, Novartis, Roche and Teva.
Supplemental Material
MSO893103 Supplemental Material1 - Supplemental material for Glatiramer acetate as a clinically and cost-effective treatment of relapsing multiple sclerosis over 10 years of use within the National Health Service: Final results from the UK Risk Sharing Scheme
Supplemental material, MSO893103 Supplemental Material1 for Glatiramer acetate as a clinically and cost-effective treatment of relapsing multiple sclerosis over 10 years of use within the National Health Service: Final results from the UK Risk Sharing Scheme by G Giovannoni, PA Brex D Dhiraj J Fullarton, M Freddi, B Rodgers-Gray and K Schmierer in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Supplemental Material
MSO893103 Supplemental Material2 - Supplemental material for Glatiramer acetate as a clinically and cost-effective treatment of relapsing multiple sclerosis over 10 years of use within the National Health Service: Final results from the UK Risk Sharing Scheme
Supplemental material, MSO893103 Supplemental Material2 for Glatiramer acetate as a clinically and cost-effective treatment of relapsing multiple sclerosis over 10 years of use within the National Health Service: Final results from the UK Risk Sharing Scheme by G Giovannoni, PA Brex D Dhiraj J Fullarton, M Freddi, B Rodgers-Gray and K Schmierer in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Source of funding for RSS: Health Departments of England, Wales, Scotland and Northern Ireland, Teva Pharmaceuticals Industries, Biogen Idec, Merck Serono, Bayer Schering Pharmaceuticals, UK National Institute of Health Research’s Health Technology Assessment Programme. Strategen Limited provided analytical and editorial support for this article, which was funded by Teva UK Limited.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
