Abstract
Background and Objectives
Late-onset MS (LOMS, symptom onset after age 50) remains underrepresented in clinical trials, leading to a gap in knowledge about the efficacy of disease-modifying therapies (DMTs). This study aims to evaluate treatment outcomes in relapsing LOMS.
Methods
A retrospective electronic medical record study at Northwestern University analyzed patients with LOMS presenting between 2004 and 2021. Demographic, clinical, DMT, and MRI data were extracted. Statistical analyses evaluated progression based on DMT efficacy.
Results
Overall, 63 patients (63% female, 76% white, median onset 55 years) were followed for a median of 7.6 years. Most patients (73%) were started on low/moderate efficacy DMTs (LET/MET). Increasing baseline EDSS was associated with an increased risk of reaching EDSS 6 (P < .001), but increasing age at diagnosis/treatment was not associated with increasing disability attainment (P = .527). Patients on LET/MET had no difference in progression to EDSS 6.0 compared to no DMT (P = .354) or change in Age-Related Multiple Sclerosis Severity Score (ARMSS) from the start of treatment/diagnosis to last follow-up (P = .477).
Discussion
The effect of LET/MET DMTs is less pronounced in older adults and may not significantly impact long-term disability outcomes.
Keywords
Introduction
Multiple sclerosis (MS) is an autoimmune disease characterized by inflammation and demyelination of the central nervous system.1–3 Most of the nearly 1 million persons in the United States living with MS are diagnosed between ages 20–40, although there has been growing recognition of late-onset multiple sclerosis (LOMS), diagnosed after age 50, and very late-onset multiple sclerosis (VLOMS), diagnosed after age 60.4,5 LOMS is more frequently associated with a progressive disease course, delayed diagnosis, and more rapid disability accumulation, particularly in motor function.6,7 Primary progressive MS (PPMS) is more common in older patients, but recent studies also suggest a rising incidence of late-onset relapsing MS, particularly among women.5,8,9
One study conducted in 2002 found that 3.4%–4.7% of MS cases were diagnosed after age 50, and 0.45% were diagnosed after age 60.10,11 German and Italian cohorts from 2024 show that 6% of diagnoses now occur after age 50.8,12 A Norwegian cohort found that 11.9% of MS cases were diagnosed after age 50 from 2010 onward. 13 These trends suggest a notable shift in the demographics of MS, reflecting better recognition of the disease in older adults, advancements in diagnostic criteria, increased awareness, and an aging population.2,10,14
Despite this recognition of LOMS, clinical trials have overlooked older populations. Most disease-modifying therapy (DMT) trials in relapsing MS focus on younger individuals, with participant ages averaging around 36 with a maximum age of 55, leaving a significant gap in knowledge regarding the effectiveness of DMTs in older MS patients. 15 Sustained DMT use may reduce the risk of disability accumulation in older patients, although the efficacy appears lower than in younger individuals. 16
This study aimed to address the characteristics, progression, and treatment outcomes of LOMS in order to contribute to improved care and management for this growing patient population.
Materials and methods
Study design and setting
Retrospective data was collected at Northwestern Medicine MS and Clinical Neuroimmunology Center and Northwestern Lake Forest Neurology Clinic (NM).
Participants
Patients with MS, as defined by the diagnostic criteria at the time care was provided, with initial symptom onset after age 50 (between 1990 and 2020), were included. Patients must have had an initial visit for MS at NM between January 1, 2004, and December 31, 2021, due to a complete electronic medical record (EMR). Patients were excluded if the follow-up duration was less than 12 months. Patients with PPMS (n = 36/129) were excluded from the study due to limitations in comparative data evaluation, as FDA-approved DMTs were not widely available for these patients before 2018. 17 A breakdown of participant selection is provided in Figure 1.

Participant selection.
Procedures
To reduce clinician recall or selection bias, a systematic search of the EMR was performed using the diagnosis code of multiple sclerosis (G35) and age greater than 50. Records were then manually searched to confirm MS symptom onset after age 50 and that an initial consultation and follow-up notes were available.
Standard protocol approvals, registrations, and patient consents
NM Institutional Review Board (IRB) approved the retrospective analysis of EMR-derived data (STU00217309). This study was exempt from informed participant consent.
Demographic and clinical data collection
Co-investigators extracted demographic and MS clinical data from the EMR. In cases where the date was unknown (i.e., MS diagnosis date), the month was approximated as July and/or the date as the 15th.
MS clinical history included date of MS onset and diagnosis, type (RRMS, PPMS), relapses, and DMTs, Expanded Disability Status Scale (EDSS) at diagnosis, treatment start, and last follow-up (either listed or extrapolated by MS physician (EG, GP, NG, BC), when able). DMT was selected by the clinician based on currently available treatments at the time of the clinic visit. When EDSS was not listed by the provider, it was retrospectively estimated using the Clinical Instrument to Retrospectively Capture Levels of the EDSS (CIRCLE) algorithm. 18 Only EDSS scores recorded more than 30 days after the onset of a preceding relapse were considered. Each EDSS score was independently reviewed by two MS physicians. 18 Time to EDSS 6.0 was calculated from treatment start (or from diagnosis if no treatment received) to the first visit where EDSS was 6 or higher. Annualized relapse rate (ARR) is defined as the total number of relapses divided by the total person-time at risk of relapse. Reports of MRI brain, cervical, and thoracic spine were reviewed for evidence of new T2 lesions or gadolinium-enhancing lesions.
MS disease severity scales
EDSS, the Multiple Sclerosis Severity Score (MSSS), and the Age-Related Multiple Sclerosis Severity Score (ARMSS) were used to assess disease severity and progression (Table 1).19–21
Comparison of MS disease severity scales.

DMT efficacy
Patients were in the high efficacy DMT (HET) group if they had received ocrelizumab, rituximab, natalizumab, or alemtuzumab; moderate efficacy DMT (MET) group if they received fumarates or S1P modulators; low efficacy DMT (LET) group if they received glatiramer acetate, interferons, or teriflunomide; no DMT group if they were not started on treatment.
MRI analysis
MRI brain, cervical, and thoracic spine were reviewed by an MS physician (EG, GP, NG) for evidence of new T2 lesions or gadolinium-enhancing lesions at least 6 months after treatment onset (or 6 months after diagnosis for patients without DMT).
Statistical analysis
Patient clinical data were summarized overall and by initial DMT therapy using means and standard deviations (SD) or medians and ranges or interquartile ranges (IQR) for continuous variables, and frequencies and percentages for categorical variables.
Time to EDSS 6.0 (right-censored at the last date of follow-up) for those not started on DMT and those started on LET/MET was presented using a Kaplan–Meier plot. A log-rank test was used to examine if there was a difference in survival times between groups. Cox proportional hazards model was used to evaluate this association adjusted for EDSS at the start of treatment/diagnosis and age at that EDSS measure. Hazard ratios (HR) and 95% confidence intervals (CI) were reported.
Multivariable linear regression models were used to examine change in the three MS disease severity scales from diagnosis/start of DMT to last follow-up date by initial DMT (LET/MET compared to none). Models were adjusted for known predictors of change in EDSS (time to last follow-up in years, the measure at start of treatment, age at the follow-up measurements); exploratory models also adjusted for smoking history (former/current versus never) and cardiovascular comorbidities. Beta estimates and 95% CIs were reported.
An exploratory analysis was conducted to estimate change in EDSS at 3 years follow-up (the median length of follow-up in the high efficacy DMT group) by DMT efficacy (none, low, moderate, and high efficacy DMT) using a mixed effects linear regression model adjusted for the known predictors above. Least-square means and 95% CIs of change in EDSS at 3 years follow-up for each treatment group were estimated using population average values for starting EDSS and age at follow-up of 2.4 and 64.2 years, respectively.
All statistical analyses were conducted using R statistical software (version 4.1.2). 22 The statistical significance threshold was set at P < .05 for two-sided comparisons. These analyses were exploratory, and corrections for multiple testing were not conducted.
Results
Patient characteristics
Data were collected on 63 patients at NM (90.5% LOMS, 9.5% VLOMS). The cohort was predominantly female (63.5%) and white (76.2%). Median age at symptom onset of 55.2, and median age at diagnosis of 56.2. A majority (74.6%) had at least one comorbidity, with cardiovascular disease and mental health comorbidities being the most common.
The median time from symptom onset to diagnosis was longer in the no DMT group (8 months) compared to the LET or MET groups (3 and 2 months). At diagnosis, 9.5% of patients used a mobility device, and 17.5% reported cognitive dysfunction. Characteristics are further broken down in Table 2.
Demographic and clinical characteristics at onset by DMT efficacy.

Median (IQR) for continuous variables, n (%) for categorical variables.
BMI: body mass index; DMT: disease-modifying therapy; EDSS: Expanded Disability Status Scale; CSF: cerebrospinal fluid.
a Or from the first clinic visit if no DMT.
DMT initiation. Of the 63 patients, 16% were not started on DMT, 59% were started on LET, 14% on MET, and 11% were started on HET. Of the 10 patients on no DMT, 6 declined, and 4 were not recommended to start. Of the 53 patients on treatment, 48 were started on DMT within 12 months. For the 5 patients who started after 12 months, the time frame was 18, 23, 24, 31, and 52 months, respectively.
All 32 patients seen before 2013 were started on LET (interferon = 22, glatiramer = 6) or no DMT (n = 4). The first MET used was dimethyl fumarate for a patient diagnosed in 2013. The first HET used was rituximab in 2018. After 2018, the distribution of treatments were no DMT = 2, LET = 3 (teriflunomide = 3), MET = 2 (dimethyl fumarate = 2), and HET = 7 (natalizumab = 2, rituximab = 1, ocrelizumab = 4).
Overall, 29 (46%) patients switched to different DMTs from the initial DMT. The reasons for switching from the initial DMT were as follows: 11 switched for clinical relapse, 10 switched from side effects, 5 for a new asymptomatic MRI lesion, 2 stopped DMT due to age (one at 61 and one at 74), and 1 for secondary progression. Additionally, 14 (22%) patients switched to higher efficacy DMT.
Follow-up duration. The median length of follow-up was 7.6 years. Patients were seen on average 2.3 times per year from DMT start to last follow-up. The median years of follow-up varied by DMT treatment (none: 6.2, low: 9.8, moderate: 6.8, high: 3.3). During follow-up, 22.2% of patients changed their DMT, with 27.0% of those initially on LET and 44.4% of those on MET switching to a different DMT. None of the patients on HET or in the no DMT group changed their treatment.
Clinical relapses. The majority of patients (71%) had no relapse after onset. Given the long duration of follow-up, the mean corrected ARR over the follow-up was low across all groups, with no significant differences between those on LET (mean ± SD: 0.0 ± 0.1), MET (0.1 ± 0.3), and those on no DMT (0.0 ± 0.0) (Table 3). Most patients who relapsed had 1–2 relapses; however, three patients had six relapses each (2 on LET, 1 on MET). For the one patient who experienced a relapse in the HET group, this occurred within 1 month after the first ocrelizumab infusion, which was before therapeutic efficacy.
Outcomes by initial DMT efficacy.

Mean (SD) for continuous outcome variables, n (%) for categorical variables.
These are outcomes observed over follow-up and not adjusted for varying lengths of follow-up time.
a Or > 6 months from diagnosis if None for DMT efficacy.
MRI outcomes. There was baseline MRI data for every patient. All patients had MRI surveillance after DMT start, with a median number of brain MRIs of 1.0 per year from DMT start. Radiographic progression was observed in 39.7% (n = 25) of patients with RRMS. New MRI T2 lesions were found in 30% of patients on no DMT, 43.2% on LET, 55.6% on MET, and 14.3% on HET. Among these, 19.0% (n = 12) of patients had gadolinium-enhancing lesions. No enhancing lesions were observed in patients on HET, who had the shortest follow-up duration.
Three of 10 patients on no DMT that had T2 lesion progression, but none started DMT for the following reasons: patient preference/managed with Wahl's diet (n = 1), not recommended by provider due to mild disease (n = 1), immediate first dose side effect to glatiramer (n = 1).
Clinical outcomes based on none vs low/moderate efficacy disease-modifying therapy
Patients on HET (n = 7) were excluded due to a shorter follow-up duration observed (median 3.3 years). The following analyses are based on patients who were on no DMT vs combined LET or MET (n = 56), who were followed for a median of 8.5 years (Table 3).
Time to EDSS 6.0
A total of 22% (n = 12/55) of LET/MET patients reached an EDSS score of 6 during the follow-up period (one patient had EDSS 6 at the start of DMT), with the same rate between the no-DMT group (22%) and the LET/MET group (22%). In the unadjusted Cox regression model accounting for follow-up time and time to the EDSS 6 event, there was a lower risk of EDSS 6 in the LET/MET group compared to the no DMT group, but this was not statistically significant (HR 0.48, 95% CI 0.10 to 2.28, P = .354). Adjusting for baseline EDSS and age at treatment/diagnosis, the LET/MET group showed a trend toward lower risk of EDSS 6, but this difference was also not statistically significant (aHR 0.16, 95% CI 0.02 to 1.17, P = .071). Increasing baseline EDSS was associated with an increased risk of EDSS 6 (aHR 3.04, 95% CI 1.66 to 5.58, P < .001 per unit increase), while age at the start of diagnosis/treatment was not associated with reaching EDSS 6 (aHR 0.96, 95% CI 0.84 to 1.09, P = .527).
Time to EDSS 6.0 among those with starting EDSS ≤3.5 vs ≥4.0
In this cohort, 49 (89%) individuals had an EDSS score of less than or equal to 3.5 at the start of treatment or diagnosis. Of those on LET/MET (n = 41), 17% (n = 7) progressed to EDSS 6, with a median time to progression of 5.9 years (range 3.2, 12.4). Among those who did not progress (n = 36), the median follow-up time was 8.6 years (range: 1.0, 14.3). In contrast, 1 of 8 (13%) individuals who were not on DMT progressed to EDSS 6, with a time to progression of 1.9 years. The remaining individuals without DMT (n = 7) had a median follow-up time of 5.3 years (range: 2.8, 10.4).
In the smaller group with an initial EDSS score of 4.0 or greater (n = 6), 60% (3 of 5) of individuals on LET/MET progressed to EDSS 6, with a median time to progression of 5.6 years (range 2.0, 11.9). The single patient without DMT progressed to EDSS 6 with time to progression of 3.0 years.
Comparing patient outcomes using EDSS, MSSS, and ARMSS
Box and whisker plots for distributions of changes in EDSS, MSSS, and ARMSS measured at start of treatment/diagnosis to the last follow-up by DMT are shown in Figure 2.

Difference in measure from follow-up to treatment start based on no DMT vs low/moderate efficacy DMT.
Expanded Disability Status Scale (EDSS). Patients on LET/MET had a similar baseline EDSS (mean 2.3, SD 1.0) as those on no DMT (mean 2.4, SD 1.9). Over time, EDSS increased for both groups, with a mean increase of 1.6 in the no DMT group and 1.2 in the LET/MET group. In the regression model adjusting for follow-up time, EDSS at start of treatment/diagnosis, and age at last follow-up, treatment with LET/MET had a lower average increase in EDSS compared to no DMT (β = −1.12, 95% CI −2.54 to 0.29, P = .126). Increasing follow-up time was associated with a greater increase in EDSS (β = 0.21, 95% CI 0.04 to 0.38, P = .021 per year).
Original MSSS. At the start of treatment/diagnosis, patients on LET/MET had a slightly higher mean MSSS than those on no DMT (mean (SD) of 5.4 (2.3) and 4.6 (2.8), respectively). In the model adjusting for time to last follow-up, MSSS at diagnosis/treatment, and age at last follow-up, there was, on average, a decrease in MSSS in the LET/MET group, while the no DMT group stayed around the same on the MSSS (β = −1.06, 95% CI −2.89 to 0.77, P = .261). A greater MSSS at start of treatment/diagnosis was associated with a smaller change in MSSS (β = −0.27, 95% CI −0.52 to −0.03, P = .032 per decile increase in MSSS).
ARMSS and disability progression. At the start of treatment/diagnosis, patients on LET/MET had a higher average global age-related MS severity score (ARMSS) compared to those on no DMT (mean (SD) of 2.2 (1.4) and 1.8 (1.6), respectively). In the model adjusting for time to last follow-up and ARMSS at diagnosis/treatment, there was no difference in the change in ARMSS from the start of treatment/diagnosis to last follow-up between the two groups (β = −0.58, 95% CI −2.15 to 1.00, P = .477). The length of follow-up time was not associated with ARMSS changes (β = 0.07, 95% CI −0.08 to 0.23, P = .345).
Impact of vascular comorbidities. Adjusting for smoking history and cardiovascular comorbidities did not change the EDSS, MSSS, ARMSS, or ARR outcomes.
Exploratory analysis: predicted EDSS at 3 years
Patients on MET and HET had the lowest predicted EDSS at 3 years with the least increase from baseline (indicated by the dots in Figure 3), but the differences in predicted change across groups were not statistically significant as confidence intervals overlapped.
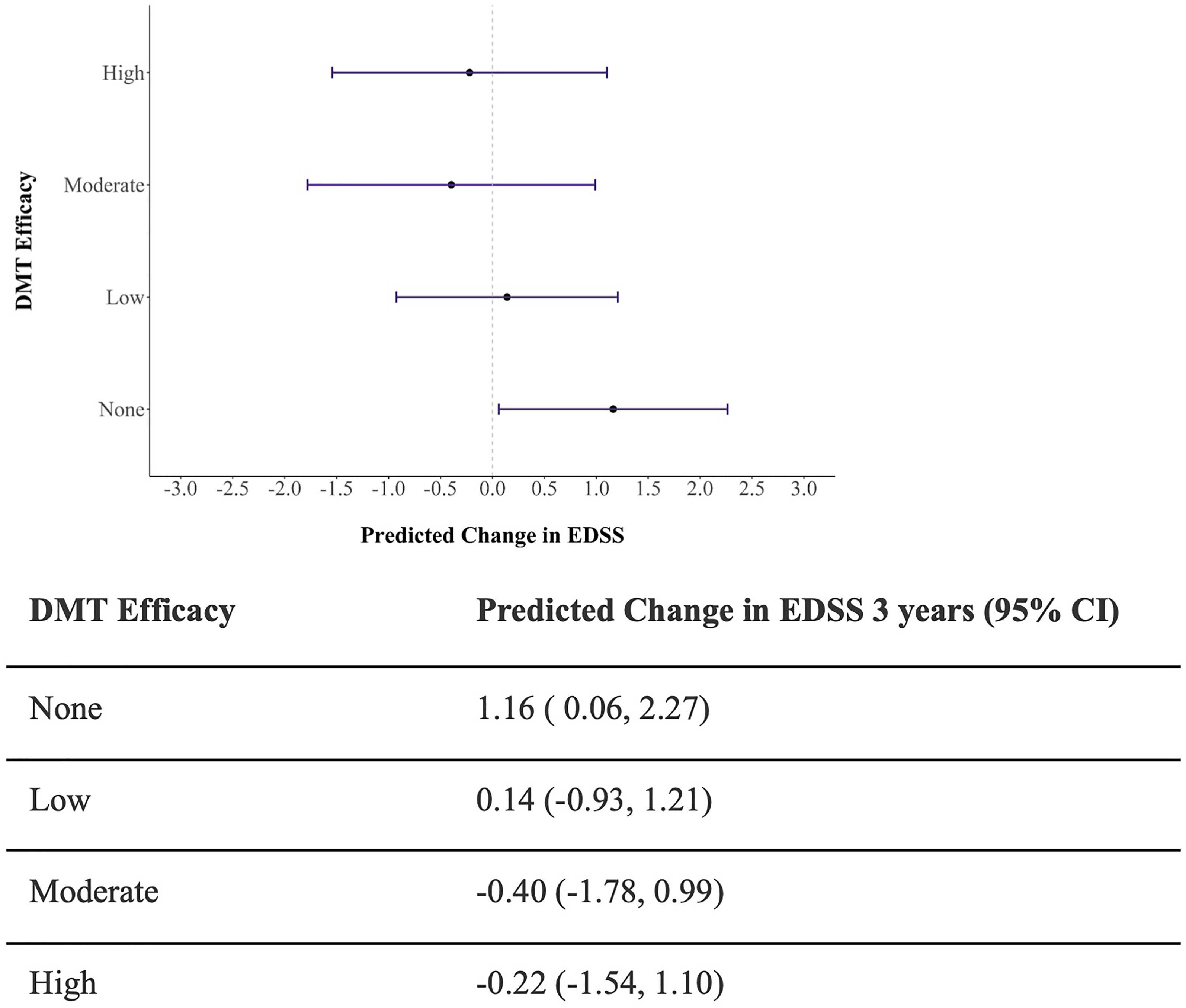
Model estimated change in EDSS at 3-year follow-up.
Discussion
Unlike other studies, our study did not show that age at diagnosis was a predictor for reaching EDSS milestones, but did show that higher baseline EDSS was associated with reaching EDSS 6.0.12,23 Importantly, our findings showed no significant difference in progression to EDSS 6.0 between patients on LET/MET and those on no DMT, aligning with the Swedish Nationwide study, which found no long-term disability benefit of DMTs in LOMS. 16 These findings differ from another study showing that DMTs can decrease the risk of disability accumulation in a dose-dependent manner, even in older MS patients. 24 Our cohort may be too small to detect this difference or perhaps the effectiveness of DMTs is lower in LOMS populations compared to younger individuals. 25
Our study also highlights the importance of using age and disease duration-adjusted measures like ARMSS to better evaluate progression and treatment impact in older MS patients. While EDSS scores increased in all patients over time, the rise was less pronounced in the DMT group compared to no DMT groups. This trend was less evident when using the age-adjusted ARMSS.
In our cohort, relapse rates were low across all groups; however, the low ARR observed here contrasts with studies suggesting a substantial relapse risk in individuals over 50. 26
Moreover, 39% of our patients continued to have new T2 lesions on MRI after diagnosis, underscoring ongoing disease activity despite treatment. The finding of higher new T2 lesions in both the LET and MET groups compared to the no DMT group may suggest that disability is not impacted by being on LET/MET; however, patients on no DMT may be more likely to have remote relapse as recent activity might prompt DMT. We cannot exclude that some new lesions might reflect comorbid vascular pathology.
The rate of HET use in our cohort was still relatively low because anti-CD20 therapies were not approved until the end of the study period. This trend mirrors the German MS registry, where less than half of LOMS patients received HET compared to over 70% of adult-onset MS patients. 16 Another reason for the infrequent use of HET may be due to concern for infections, although our study did not find an increased rate of hospitalizations in our high efficacy group. The rate of adverse events might not be any different than a younger population, but we do not have comparison data. This highlights an unmet need for longitudinal data on more aggressive treatment approaches in older patients, particularly as anti-CD20 therapies, which have shown promising efficacy and safety profiles in older adults, are being increasingly adopted.
Limitations
This study's limitations include a small sample size from a single center, varying follow-up lengths, and the retrospective design, which may have introduced bias and reduced power to detect significant differences, especially for HETs. Variability in clinical decisions, treatment availability, and interpretation of patient records, such as EDSS and relapse documentation, further constrained analyses. Finally, we were unable to include patients with PPMS in the study due to limited patient numbers and the availability of HET during the study period.
Conclusions
Our study emphasizes the challenges of managing LOMS, including progressive disability and limited DMT efficacy. High-efficacy DMTs, such as anti-CD20 therapies, may slow disability progression. Unless a patient has a history of significant recurrent infections, we are currently using high-efficacy DMT in LOMS patients, similar to the overall MS population. We will assess this as we continue to collect information on this cohort. Larger, long-term studies are needed to optimize treatments, balancing efficacy and safety in older adults. Including older populations in clinical trials is crucial to improving outcomes and guiding treatment decisions for this unique patient group.
Footnotes
Abbreviations
Authors note
Sophie Ahmad, Department of Neurology, Rush University, Chicago, IL, USA
Consent to participate
Informed consent for information published in this article was not obtained because this study was deemed exempt by the Northwestern University IRB.
Consent for publication
Not Applicable.
Data availability
Anonymized data may be shared at the request of a qualified investigator for purposes of replicating procedures and results up to 3 years after publication of this article.
Declaration of conflicting interest
S. Ahmad reports no disclosures relevant to the manuscript. E.L. Graham has received research support from F. Hoffman La-Roche Ltd and consultancy fees from Novartis, Horizon Therapeutics, Roche/Genentech, EMD Serono, TG Therapeutics. N. Lancki reports no disclosures relevant to the manuscript. N.G. Caldito reports no disclosures relevant to the manuscript. G. Perez-Giraldo has received consultancy fees from TG Therapeutics. B.A. Cohen has received personal compensation from WCG clinical services for serving as an independent adjudicator for clinical trials.
Ethical approval
This study received ethical approval from the Northwestern University IRB (approval #STU00217309) on August 3, 2022. This is an IRB-approved retrospective study, all patient information was de-identified, and patient consent was not required.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
