Abstract
Multiple sclerosis was long considered a relatively rare entity in the Middle East, but research over the past 10 years and the publication of the Middle East North Africa Committee for Treatment and Research in Multiple Sclerosis guidelines for multiple sclerosis have allowed diagnosis and treatment to occur more efficiently. Most of the first and second-line disease-modifying therapies approved by the Food and Drug Administration and the European Medicine Agency are available in the Middle East. However, the availability of disease-modifying therapies is quite variable, with some countries having access to all multiple sclerosis disease-modifying therapies, while in others there is only one therapeutic option. Economic limitations remain a challenge for the management of multiple sclerosis, especially in countries of war. Moreover, the burden of multiple sclerosis treatment in Syrian and Palestinian refugees is likely high due to the non-availability of funds to cover the high cost of disease-modifying therapies.
Introduction
The prevalence and incidence rates of multiple sclerosis (MS) have been steadily increasing world-wide over the last few decades including the Middle East (ME).1–6 The MS treatment landscape has significantly changed during the past 10 years with the addition of several novel disease-modifying therapies (DMTs). The therapeutic armamentarium has increased from only interferon treatment in 1993 to more than 13 DMTs approved by the Food and Drug Administration 7 (FDA) and European Medicines Agency 8 (EMA) nowadays. This provides better opportunities for personalized treatment whereby patients and providers must balance considerations around efficacy and adverse events in a shared-decision process. However, due to the variety of mechanisms of actions, monitoring requirements, risk profiles together with the heterogeneity of MS and the changes in the diagnostic criteria over the years, there has been a clear need to unify and update the therapeutic paradigms across the ME. On the other hand, most of the countries in the region are in the process of establishing specialized MS centers. In this context, the Middle East North Africa Committee for Treatment and Research in Multiple Sclerosis (MENACTRIMS) has joined forces to provide updated, evidence-based guidelines 9 for the diagnosis and treatment of patients with MS.
Treatment of MS in the ME: MENACTRIMS guidelines
The MENACTRIMS treatment guidelines 9 for MS do not significantly differ from international recommendations.
The guidelines recommend starting DMTs early once the diagnosis of MS is established to prevent axonal damage and decrease the long-term accumulation of disability. Interferon-beta, glatiramer acetate, teriflunomide and dimethyl fumarate can be initiated in treatment-naïve relapsing–remitting patients. In patients with needle phobia or contraindications/adverse events related to the above DMTs, fingolimod can be used as first-line therapy. In patients with aggressive or highly active relapsing–remitting MS (RRMS), fingolimod, natalizumab, or alemtuzumab may be initiated following careful risk stratification (serum anti-JC virus antibody, prior immunosuppressant use, cardiac disease, diabetes, retinal disorders, previous autoimmune diseases, and thyroid disorders). 9
In RRMS patients with sub-optimal response to first-line therapies, treatment escalation to fingolimod, natalizumab, or alemtuzumab should be considered. The choice among them should be based on risk stratification 9 (Figure 1).

Algorithm for the management of relapsing–remitting multiple sclerosis.
In secondary progressive multiple sclerosis (SPMS) patients with evidence of superimposed relapses, interferon-beta 1b subcutaneous (SC) or interferon-beta 1a SC (high dose) is recommended. In SPMS patients without relapses, mitoxantrone may be offered after comprehensive discussion with the patient regarding its serious adverse event profile. 9 Ocrelizumab, not available yet in the ME, was recently approved for the treatment of both primary progressive MS (PPMS) and relapsing forms of MS. Off-label use of rituximab may be considered in relapsing forms of MS and PPMS.
Availability of DMTs in the ME
Most of the first and second-line DMTs (interferon beta 1a and 1b, pegylated interferon beta 1a, fingolimod, teriflunomide, dimethyl fumarate, natalizumab, alemtuzumab, and mitoxantrone) approved by the FDA and EMA for the treatment of MS are available in the ME. A noticeable exception is glatiramer acetate which is not offered in the majority of the countries of the region because it is marketed by Teva Pharmaceutical Industries, an Israeli pharmaceutical company. Ocrelizumab is only available through multinational randomized clinical trials or through special individualized request. Cladribine has recently been approved in Europe for the treatment of highly active forms of RRMS; however, it is not yet available in most countries in the ME.
The introduction of novel DMTs to the ME region is usually delayed. Drug approval can take several months to years once approved for use by the FDA and EMA, depending on the country. On the other hand, the ME is a region of developing nations, with considerable disparities in socioeconomic status. Thus, the high cost of treatments for MS is likely to represent a barrier to care for many patients in the region, with limited reimbursement for DMTs. This is the main reason behind the different levels of coverage and reimbursement for MS medications seen in different ME countries. For instance, while all current DMTs are available and reimbursed in Lebanon and Kuwait, only a few treatment options are offered in countries such as Syria or Iraq. Access to drugs is becoming increasingly problematic for MS patients in such countries where, the ministry of health provides treatment with interferons (Syria) and occasionally fingolimod or natalizumab (Iraq).
A well-known situation in Iran and many other countries around the world, is the emergence of generic DMTs or biosimilars. There are more than 18 biosimilars prescribed for MS patients in the region, most of which are manufactured in Iran. 10 In 2005, CinnoVex was the first biosimilar interferon beta 1a produced. Currently, approximately 50% of MS patients in Iran are on DMTs; 2/3 of whom are on generic DMTs. 10 The efficacy and safety of those products were assessed and established in multiple local experimental and clinical controlled studies.11–18
Treatment of MS in refugees: the Lebanese experience of the Nehme and Therese Tohme Multiple Sclerosis Center
The protracted and violent nature of conflicts in the ME has resulted in a large population of refugees facing long-term displacement, and creating an unprecedented strain on the host countries’ health systems. At the close of 2013, there were 641,915 refugees in the Kingdom of Jordan, including 585,300 Syrians and 20,300 Iraqi refugees assisted by the United Nations High Commissioner of refugees (UNHCR). 19 Similarly, Lebanon continues to host a large number of Syrian and Palestinian refugees, accounting for almost 40% of its population. As of October 2017, the UNHCR registered almost one million Syrian refugees along with almost 300,000 Palestinian refugees in Lebanon. 20 The actual number of Syrian refugees is most probably much higher.
In Lebanon, a high proportion of refugees are living within local communities (only 17% live in transit centers) and have the same access to health care as Lebanese nationals. In fact, the Ministry of Public Health (MOPH) provides primary care services through its regional centers to refugees residing in Lebanon at minimal cost. The current package includes vaccination, consultation, laboratory and diagnostic tests, acute medications, ultrasounds for pregnant women, and emergency hospitalizations. However, expensive chronic medications used for the treatment of chronic diseases such as MS remain inaccessible due to limited financial support from the UNHCR and other non-governmental organizations (NGOs) supporting displaced Syrians. In addition, there are no data on the incidence and prevalence of MS among Syrian and Palestinian refugees although the estimated burden is likely high.
Economic limitations remain a challenge for the treatment of MS in the ME, especially among displaced Syrian and Palestinian patients. Although MS should be considered a high health risk disease, there is a lack of awareness among international and regional supporting organizations given its status as a low prevalence disease, resulting in non-availability of funds to cover DMTs. Displaced Syrians have only access to interferon-based therapy for the treatment of MS through the Syrian Ministry of Public Health, but patients have to travel to get their monthly medication supply personally from Syria, which is not always feasible due to political and security issues. In addition, the DMTs provided by the Syrian government are almost exclusively Iranian generics. As for Palestinian refugees, the United Nations Relief and Works Agency for Palestine Refugees in the Near East (UNRWA) used to provide interferons for Palestinian MS patients with a minimal copayment, but due to recent restrictions on funding especially from the US government, this support has been dwindling. No other treatment options exist currently for Syrian and Palestinian refugees.
Around 142 Syrian and 37 Palestinian MS patients have been treated at the Nehme and Therese Tohme Multiple Sclerosis Center (MSC) at the American University of Beirut Medical Center (AUBMC) in Lebanon between October 2011–December 2018. One hundred and twenty-three patients (68.7%) were diagnosed with RRMS and 36 (20.1%) with progressive MS (PMS) (Table 1). Among Palestinian RRMS patients, the most commonly prescribed DMTs were interferons (n=13; 35.2%) followed by rituximab (n=8; 21.6%). Similarly, most of the Syrian RRMS patients were maintained on interferons (n=40; 28.2%) followed by rituximab (n=36; 25.3%). While two-thirds of Palestinian patients with PMS were treated with off-label immunosuppressive medications (mycophenolate mofetil or methotrexate) due to its cheap cost, 6.4% (n=9) of Syrian patients with PMS remained off-treatment either due to advanced progressive disease or due to financial reasons, 4.2% (n=6) received off-label cyclophosphamide, 3.6% (n=5) rituximab, 2.8% (n=4) mycophenolate mofetil and 2.8% (n=4) ocrelizumab through their enrollment in the CONSONANCE clinical trial (a study to evaluate ocrelizumab treatment in participants with preogressive multiple sclerosis) (Figure 2). The most commonly used DMTs among Syrian and Palestinian refugees were (a) interferons (n=55; 30.7%), (b) rituximab (n=50; 27.9%), and (c) off-label immunosuppressive medications such as mycophenolate mofetil, cyclophosphamide, azathioprine, and methotrexate (Figure 3). Only a few refugees have access to expensive novel second-line DMTs such as fingolimod or natalizumab through a private insurance plan, which means that if they fail interferon-based therapy offered by the Syrian MOPH or UNRWA, they will have no other treatment options. At our MSC, a special fund was secured to cover the cost of rituximab treatment for refugees. Rituximab at a dose of 1000 mg intravenously every six months is provided by this fund as first-line therapy for RRMS patients who cannot obtain interferons from Syria, and as second-line therapy for both Syrian and Palestinian patients who have suboptimal response to injectable DMTs or early progressive disease.
Diagnosis of Syrian and Palestinian refugees treated at the Nehme and Therese Tohme Multiple Sclerosis Center (n=179).

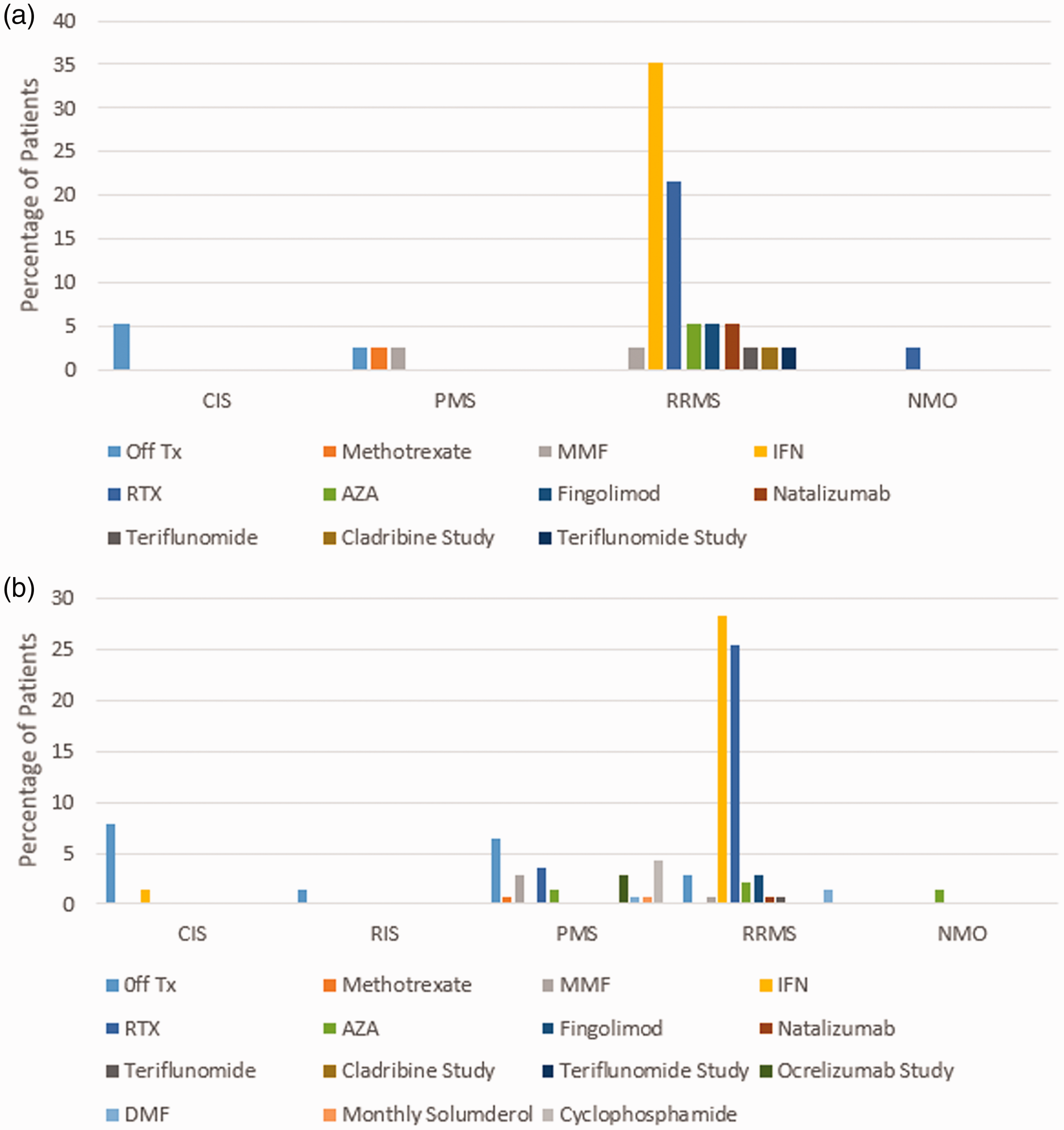
Disease-modifying therapies for Syrian and Palestinian refugees treated at the Nehme and Therese Tohme Multiple Sclerosis Center. (a) Palestinian refugees (n = 37) and (b) Syrian refugees (n = 142).

Disease-modifying therapies for all refugees (n=179) treated at the Nehme and Therese Tohme Multiple Sclerosis Center.
Rituximab is not FDA-approved for the treatment of MS but its off-label use has increased considerably in many countries all around the world since the original phase II HERMES trial (helping to evaluate rituxan in relapsing-remitting multiple sclerosis) that showed robust efficacy in RRMS based on clinical and radiological parameters. 21 This was further encouraged by the recent FDA and EMA approval of ocrelizumab as the first B-cell depleting therapy in MS. The obvious reasons for treating refugees at our MS center with rituximab are: the similar mechanisms of action of rituximab and ocrelizumab as they both deplete B cells by binding to the CD-20 surface antigen, the good long-term safety of rituximab based on experience in oncology and rheumatology, the efficacy and safety data of rituximab for MS derived from multiple observational studies mainly from Sweden, and, most importantly, the much cheaper price of rituximab compared to newer DMTs. Most publications regarding the off-label use of rituximab in MS have come from Sweden,22–25 where rituximab accounts for almost 40% of all DMTs used in MS. Salzer et al. 22 reported on 822 MS patients derived from the national Swedish MS registry and treated with rituximab for a mean period of 23.1 months. Rituximab induced a significant decrease in relapse rate in both RRMS (annualized relapse rate (ARR)=0.0440) and PMS (ARR=0.015–0.038). In addition, rituximab effectively suppressed magnetic resonance imaging (MRI) activity with only 4.6% of patients showing enhancing lesions while on treatment as opposed to 26.2% at baseline. The median Expanded Disability Status Scale (EDSS) remained unchanged in RRMS patients and showed a statistically non-significant increase in PMS patients.
We reported our own experience with rituximab in MS at the AUBMC MSC. 26 We reviewed 89 MS patients treated with rituximab for a mean duration of 22.2±24.8 months. During treatment, the ARR decreased from 1.10 at baseline to 0.01 in RRMS (p<0.0001) and from 0.25 to 0.005 in PMS patients (p=0.022). The mean EDSS remained unchanged in both RRMS and PMS patients. Between baseline and last follow-up, the percentage of patients free from any new MRI lesions increased from 18.6% to 92.6% in the RRMS group and from 43.3% to 82% in the PMS group. 26 On the other hand, from a financial perspective, the price of rituximab is around 2000$/year compared to approximately 24,000$/year for fingolimod or natalizumab. Accordingly, and due to the limited available funds, rituximab is practically the most-cost effective therapy for Syrian and Palestinian refugees who do not have access to other DMTs.
In conclusion, despite the publication of MENACTRIMS consensus recommendations, the treatment of MS varies markedly between different countries in the ME based on DMTs availability, reimbursement, and political circumstances especially in war-torn countries. Syrian and Palestinian refugees have limited access to DMTs which poses a major problem in managing their disease. Our MSC at AUBMC is providing those patients with rituximab free of charge through a special fund, both as a first-line and an escalation therapy. There is an urgent need to develop a support network to help refugees not only in Lebanon but also across the ME region in order to optimize management and secure a larger fund that will allow access to more effective MS therapies.
Footnotes
Acknowledgements
The authors wish to thank their research coordinator Tracy J Abi Hanna for her assistance in collecting the data of refugees treated at their center.
Conflicts of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
