Abstract
The Middle East North Africa (MENA) region falls in the low-to-moderate multiple sclerosis (MS) prevalence zone, with prevalence rates slightly lower than Southern Europe but much higher than sub-Saharan Africa. However, there is clearly a trend toward increased MS prevalence over the last few decades, consistent with the globally rising prevalence of the disease. We analyzed all data collected from the MENACTRIMS (Middle East North Africa Committee for Treatment and Research in Multiple Sclerosis) registry by the end of December 2018. A total of 6885 patients were eligible for the study. Overall, the clinical phenotype of MS in the MENA region was not different from the Western phenotype except for earlier age at onset and a more aggressive clinical course leading to earlier disability.
Introduction
Multiple sclerosis (MS) is an immune-mediated demyelinating disorder affecting predominantly patients aged 20–40 years. The incidence and prevalence rates of MS continue to rise worldwide, with the highest rates originating from North America and Europe. 1 Most countries from the MENA (Middle East North Africa) region fall in the low-to-moderate MS prevalence zone, with prevalence rates slightly lower than Southern Europe but much higher than sub-Saharan Africa.
Epidemiology
We reviewed the most relevant epidemiologic studies published in the MENA region, to determine the prevalence of MS in this part of the world (Table 1). Most epidemiologic studies performed before the year 2000 showed low prevalence rates of MS ranging between 3 and 20/100,000, and came mostly from Saudi Arabia, Iraq, Libya, Kuwait, Tunisia and Jordan.2–8 Prevalence started rising during the first decade of the 21st century, with studies from Kuwait, Saudi Arabia, Palestine and Jordan showing rates in the range of 30–38/100,000.9–12 The only exception was Oman, that had a persistently low prevalence rate reaching 4/100,000 in 2005, possibly due to different genetic factors compared with neighboring Arab countries. 13 More recent data from Kuwait, Qatar, Bahrain and United Arab Emirates revealed a further increase, with prevalence rates currently ranging between 55 and 85/100,000.14–18 A single study from Egypt 19 reported in 2013 a low prevalence of 13.7/100,000 in a small city located south of Egypt near the Sudanese border, possibly reflecting the lower prevalence rates seen in African countries. Interestingly, Iran has always shown the highest disease prevalence in the region, increasing from 51.9 in 2010 to 148.1/100,000 in 2017, which might be due to genetic factors related to the different ethnic origins of the Iranian population.20,21 It is of note that the methodology to calculate prevalence in the above-mentioned studies varied widely, from national registry-based to hospital or community-based studies, while some of the older studies used the Kurtzke approximation. This approximation estimates MS prevalence by comparing the number of MS and motor neuron disease patients seen during a defined period of time based on the assumption that the prevalence rate of motor neuron disease is fixed at 5/100,000.
Prevalence of multiple sclerosis in the Middle East North Africa region.

*Kurtzke Approximation
There is clearly a trend toward increased MS prevalence in the Middle East over the last few decades, consistent with the globally rising prevalence (Figure 1). The Multiple Sclerosis International Federation Atlas recorded a 10% increase in global prevalence of MS from 30 to 33/100,000 between 2008 and 2013. The rapid rise of prevalence rates in the MENA region might be partly due to earlier diagnosis, especially with the advent of magnetic resonance imaging in the 1980s and the new McDonald diagnostic criteria. 22 On the other hand, prolonged survival resulting from medical advances and new efficacious MS therapies 23 might have increased prevalence simply through more patients with MS reaching older ages. Marrie et al. have clearly shown that the rise in MS prevalence in the province of Manitoba-Canada between 1984 and 2004 was associated with a shift in the MS population age distribution curve toward older ages. 24 However, other factors might be contributing to a direct “true” increase in MS incidence in the Middle East, such as increased urbanization and migration from rural areas to large cities. Urbanization is associated with less sun exposure, higher body weight, improved hygiene, increased stress and smoking, all of which are potential risk factors for developing MS. 25 A single study from Kuwait 13 showed that this increase in MS incidence is mostly in females, similar to what has been demonstrated in other regions of the world. 26

MS Prevalence rates in the MENA region by decade according to published studies (Each bar represents a single study).
Clinical characteristics
The clinical characteristics of MS in the MENA region have been described in isolated studies from single countries, most of which included relatively small number of patients. The Middle East North Africa Committee for Treatment and Research in Multiple Sclerosis (MENACTRIMS) created in 2016 the MENACTRIMS registry as a regional substudy of MSBase, with the aim of recruiting patients with MS from different countries in the MENA region. MSBase is an international observational MS cohort study, registered with the World Health Organization International Clinical Trials Registry Platform, 27 and uses the Imed© software system, 28 a clinical database where different demographic, clinical and laboratory variables are collected. A total of 11 centers joined the MENACTRIMS registry, contributing 6885 patients as of December 2018.
We performed an analysis of all data collected up to the end of year 2018. The Statistical Package for Social Sciences (SPSS), version 24.0 was used for data cleaning, management and analyses. Descriptive statistics were summarized by presenting the number and percentage for categorical variables, whereas continuous ones were presented by mean and standard deviation (±SD). In the bivariate analysis, the association between relapsing onset and progressive onset MS phenotypes and other categorical variables was assessed using the Chi-square test, whereas Student’s t-test was used for the association between age and phenotype. The time to reach an Expanded Disability Status Scale (EDSS) score of 6.0 was analyzed using Kaplan–Meier survival analysis. A p-value <0.05 was used to indicate statistical significance.
Mean age at onset was 27.8 (±9.3) and at diagnosis 31.6 (±10.1) years, with an approximate 3.5 years delay in diagnosis (Table 2). Almost 60% of patients started below 30 and 17% below 20 years of age. The proportion of females in our registry was 72.3%, and a positive family history of MS was reported in 17.8% of patients. Presenting symptoms were spinal in 34.2%, optic in 24.5%, brainstem/cerebellar in 22.3% and supratentorial in 20.0%. Onset was relapsing in 96.1% and progressive in 3.2%. A total of 161 cases of neuromyelitis optica spectrum disorder (NMOSD) were identified in the registry, accounting for 4.7% of all patients and an NMOSD/MS ratio of around 1:40.
Demographic and clinical characteristics.

Comparing primary progressive MS (PPMS) and relapsing–remitting MS (RRMS) (Table 3), patients with PPMS were older (35.1±10.1 vs. 27.7±9.1 years, p-value <0.0001), and more likely to present with spinal symptoms (49.3% vs. 33.7%, P-value <0.0001) rather than optic neuritis (11.4% vs. 26.9%, P-value<0.0001).
Comparison of clinical characteristics of patients with relapsing and progressive onset MS.
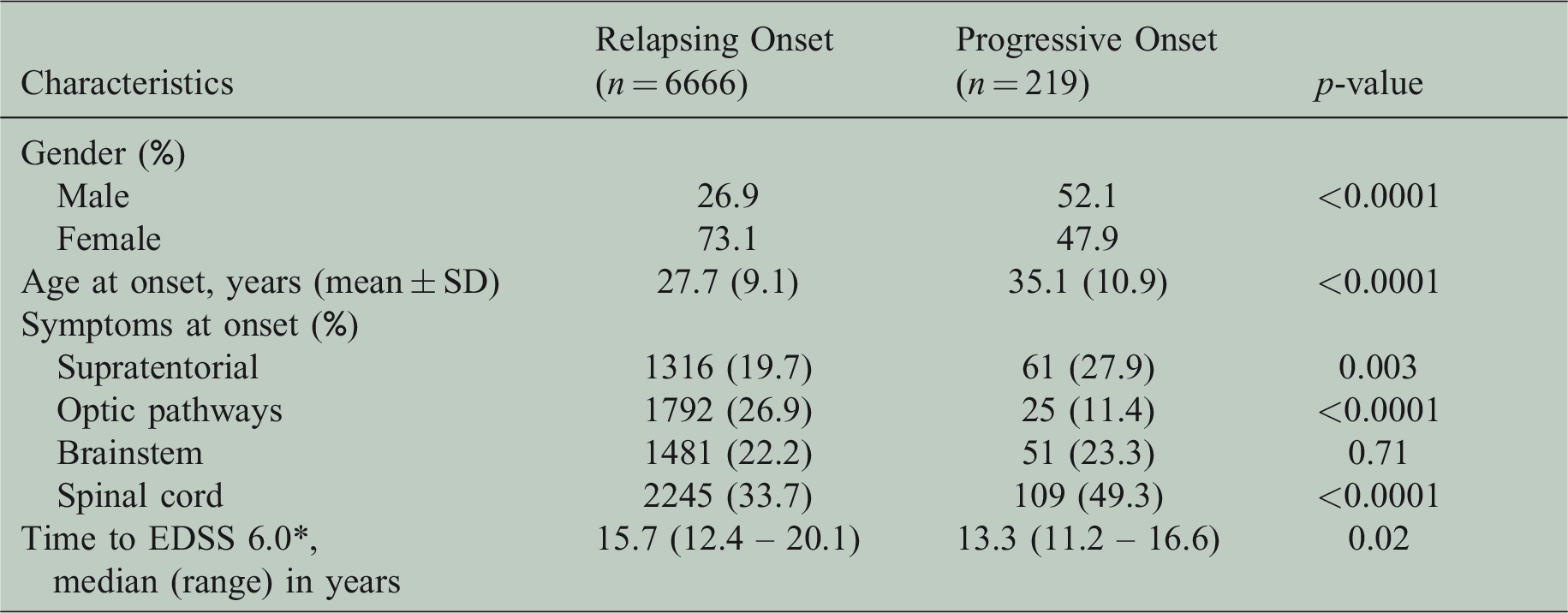
In order to assess the long-term course of the disease, we selected patients with at least 10 years of follow-up since onset of symptoms (Table 3). A total of 741 patients were eligible for the analysis. Irreversible disability was defined as persistence of an EDSS score for two consecutive visits at least 6 months apart. Median time to reach an EDSS score of 6 was 15.7 (12.4–20.1) and 13.3 (11.2–16.6) years, respectively, for the RRMS and PPMS groups.
Compared with recent epidemiological studies from the West, our patients had a younger mean age at onset: 27.8 years vs. 30.5 years in Italy, 32.5 years in France and 30.0 years in the UK.29–31 A total of 10.6% had onset of symptoms below the age of 18 as compared with 5.5% in the French cohort. The proportion of females, however, was similar: 72.3% vs. 71.1% and 67% in the French and Italian registries, respectively. Patients with PPMS constituted 3.2% of the total cohort, reflecting a lower proportion compared with modern Western populations: 10.7% in France, 12.9% in the UK, 12.4% in British Columbia and 6% in Italy, but similar to Japan (3%).29–31 However, the possibility of under-diagnosis of PPMS in our registry could not be ruled out, as the latter’s diagnosis is usually more difficult in clinical practice. The NMOSD/MS ratio in our cohort was close to what has been reported in the West. 33
Our patients with RRMS reached an EDSS score of 6.0 after a median time of 15.7 years, which is much shorter than what has been described in Western MS populations. Median time to EDSS 6.0 in RRMS patients was 21 (19–24) and 27.3 (16.3–39.7) years in two French studies,34,35 and 30.3 (28.6–32.0) years in British Columbia. 32 Similar conclusions were reached by two French studies comparing disability progression between Caucasian French, French of North African origin and Tunisian RRMS patients. French patients of North African origin reached an EDSS score of 6.0 almost 10 years earlier compared with French patients of Caucasian origin (17.0 vs. 27.3 years). 35 Similarly, French patients of Caucasian origin reached an EDSS score of 6.0 five years later than Tunisian patients (21 vs. 16 years). Our data are indicative of a more aggressive and disabling MS course in the MENA region compared with the West.
Disease-modifying therapies (DMTs) are available to different degrees in the MENA region countries. Gulf countries and Lebanon have access to nearly all DMTs with adequate reimbursement by governmental agencies. In the remaining countries, new DMTs are either not registered or not fully reimbursed by governments. Most MENA countries, however, have access at least to first-line injectables except for glatiramer acetate due to its Israeli origin. First-line injectables still account for almost half of the DMTs used in our region (Table 4). This is followed in descending frequency by fingolimod, natalizumab, rituximab and dimethyl fumarate. It is of note that locally manufactured generics, mostly Iranian-produced, are used in the MENA region. However, as shown in Table 4, they account for less than 4% of the total DMTs used. In total, 76.0% of all patients in the registry were on DMTs.
Disease-modifying therapies at last follow-up.

Our study has certain limitations, common to all registry-based studies. The data were collected retrospectively, which might have introduced a bias especially in determining the course of the disease and its phenotype. In addition, and due to the nature of the data collected, there were no unified definitions of clinical phenotypes, disability progression and other clinical parameters. Finally, by choosing patients with a disease duration of at least 10 years, we might have introduced a selection bias toward patients with more aggressive disease, as the latter might be more prone to maintain long-term follow-up.
In conclusion, the prevalence of MS has been steadily rising in the MENA region over the past few decades. The clinical phenotype of MS in the MENA region is similar to what has been described in the West except for an earlier age at onset and a more aggressive clinical course leading to earlier disability.
