Abstract
In this review, we aim to discuss the rising role of artificial intelligence (AI) across all phases of stroke care from prehospital triage, acute management, rehabilitation, to postdischarge care, and secondary prevention. AI has shown a remarkable potential in improving stroke patients’ care through prehospital triage systems, refining risk stratification and personalizing rehabilitation protocols. The use of automated notification systems in prehospital triage has reduced door-to-neurointerventional times by approximately 40 min, speeding thrombectomy access and providing stroke patients time critical treatment that hugely affects their outcomes. In the hyperacute setting, deep learning helps to provide a more accurate estimation of lesion(s) on brain scans than standard conventional quantitative markers, even offering a faster processing time. In rehabilitation, the use of kinematic data in real time led to increasing utilization of robotics and virtual reality (VR) to aid motor recovery poststroke, and machine learning to map out complex nonlinear recovery paths for speech and cognition. However, the evidence linking AI integration to measurable improvements in patients outcomes is yet to be fully evaluated. Moving forward, these models need to be validated through multicenter prospective trials and standardized benchmarks. There is also a need to focus on developing systems that are designed for the user, ensuring they support rather than replace clinical expertise. AI's ultimate value will depend on rigorous validation, integration into clinical workflows, and sustained collaboration between developers and clinicians.
Introduction
Artificial intelligence (AI) refers to the development of machines capable of simulating human intelligence. 1 Unlike classical statistical methods which are limited in their ability to predict outcomes from unstructured datasets, AI has enormous ability to identify patterns from big datasets.2,3 Consequently, AI technologies, particularly machine learning (ML) and large language models (LLMs), have attracted attention, with potential applications encompassing multiple domains of clinical medicine, including stroke. 4 The use of AI in healthcare is growing at 40% per year, and it has been estimated that AI use can cut up to 150 billion USD in healthcare costs by 2026. 5 Recognizing the rising role of AI and its ability to analyze huge and complex clinical and imaging data, the FDA has approved more than 20 AI/ML-enabled technologies for indications specifically related to stroke diagnosis and rehabilitation in recent years. 6
ML can be grouped into supervised and unsupervised learning. Supervised learning, which is more commonly used, includes linear regression, logistic regression (LR), random forest (RF), support vector machines (SVM), and neural networks. The principle of such algorithms is to train models using known input and output data to predict and classify new data. Another promising technology is deep learning (DL) which mimics the structure and function of neural networks in the human brain. This technology is capable of automatically learning and extracting features through multilayer neural networks. Owing to its multilayer structure, DL has significant advantages in identifying nonlinear patterns, making it particularly suitable for stroke imaging tasks. 7
Stroke is a time sensitive medical emergency where clinical outcomes are directly linked to the accuracy and speed of detection and prompt treatment decisions. Brain imaging remains the gold standard for diagnosing stroke, distinguishing ischemic from hemorrhagic stroke and detecting large vessel occlusion (LVO) and ischemic tissue during acute ischemic stroke (AIS). However, modern management of stroke is multifaceted, requiring the processing and synthesis of a large amount of multimodal data ranging from clinical symptoms to complex neuroimaging (CT/MRI) and perfusion maps in a short amount of time. This high-pressure data rich environment creates an ideal landscape for AI integration. Through leveraging DL capabilities to process complex imaging datasets faster than human capacity, AI is transforming stroke care from a reactive model to a precision-based discipline.
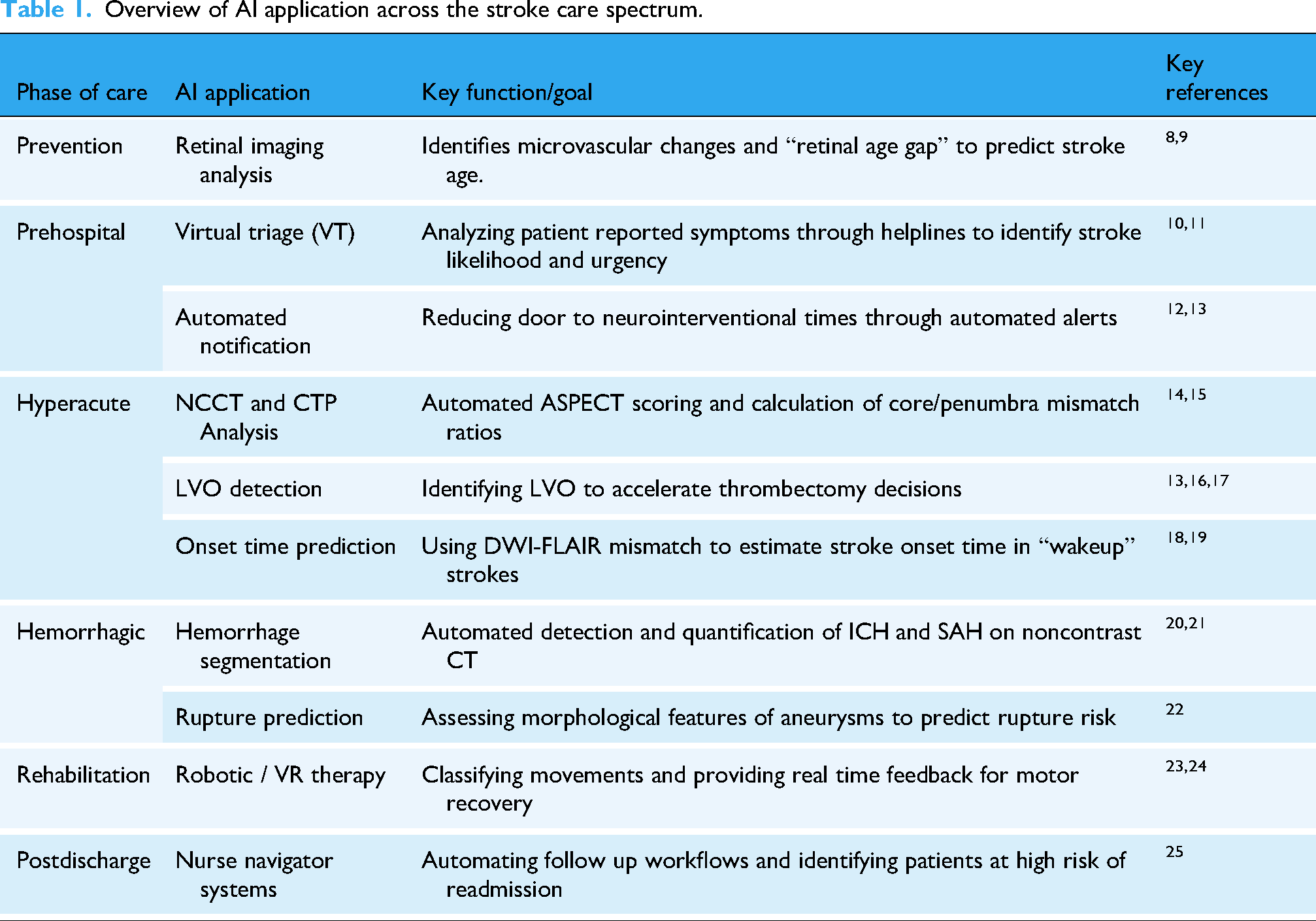
This review aims to summarize the present body of literature regarding the rising role of AI in the management of stroke patients, its promise, and limitations. We will cover AI applications across stroke prevention, acute management, rehabilitation, and outcome prediction while addressing the hurdles these technologies face in clinical practice (Table 1).
Overview of AI application across the stroke care spectrum.
Method
We searched PubMed and Google Scholar for AI-related literature published in peer-reviewed journals specializing in Neurology, Radiology, and Medical Informatics during the period from 2014 to 2024.
First, we used the following using a combination of Medical Subject Headings (MeSH) terms and free-text keywords for our literature search: [(“Artificial Intelligence”[MeSH] OR “Machine Learning”[MeSH] OR “Deep Learning”[MeSH]) AND (“Stroke”[MeSH] OR “Ischemic Stroke” OR “Neuroimaging” OR “Hemorrhagic Stroke” OR “Stroke Rehabilitation”)]. Secondly, additional studies were included through “backward snowballing” by manually screening the reference lists of key papers. We limited our search to papers published in English language. Finally, a “forward snowballing” search was conducted using Google Scholar's “Cited by” feature for landmark papers in the field.
Both original and review papers were included, we did not include editorials, letters to the editor or studies not published in peer-reviewed journals. Studies were included based on their contribution to the narrative themes of the review, focusing on clinical implementation and diagnostic accuracy and/or directly assessing the clinical performance of an AI-enabled technology for stroke diagnosis or management in humans. The decision to include papers was conducted by two authors and any conflict was solved by consulting the senior author.
No quality assessment was conducted as this is not a systematic review. The selection process, including identification, screening, and final inclusion, is summarized in Figure 1.
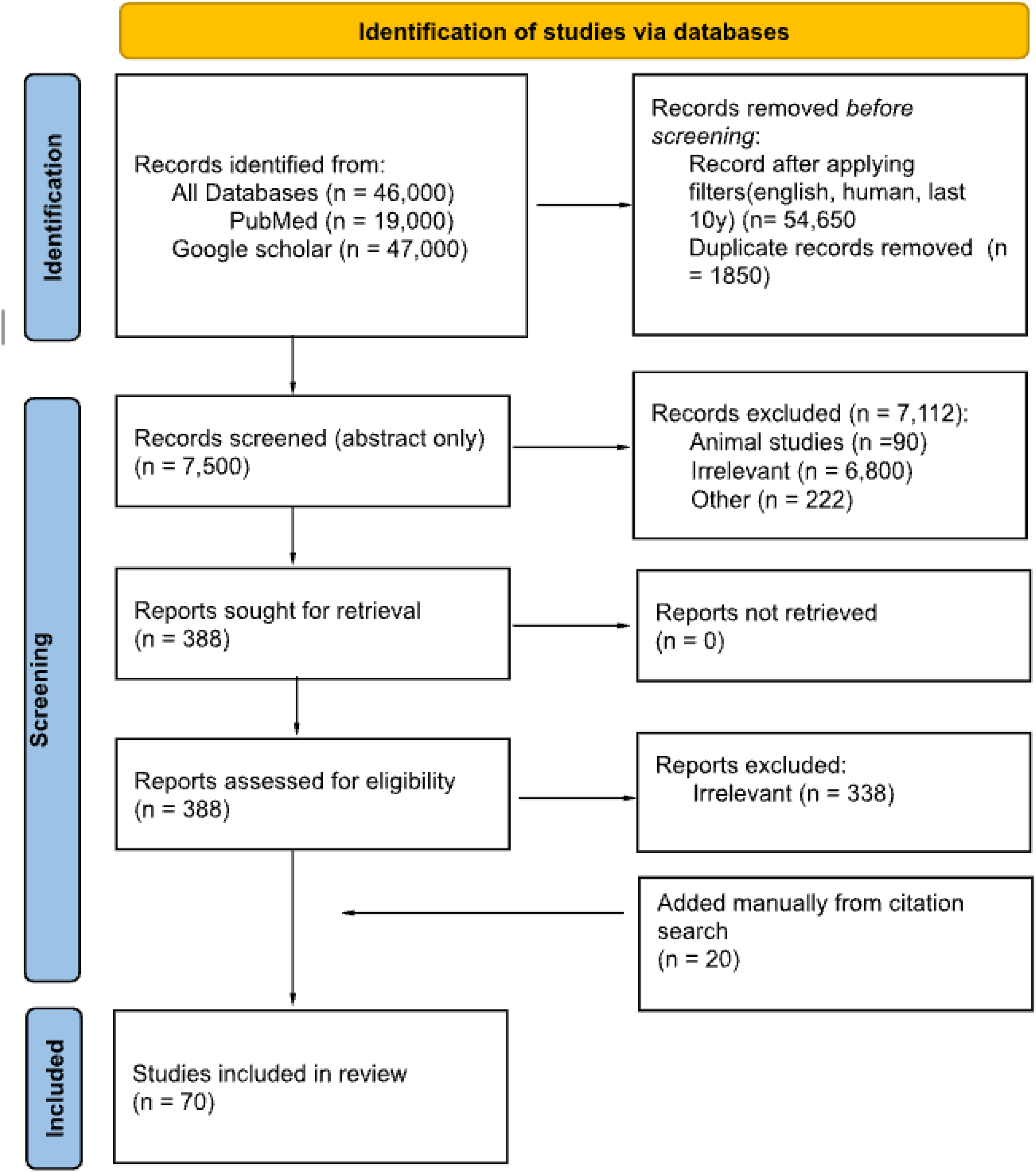
The process of studies identification.
AI in stroke prevention
Primary prevention
AI may have a role in identifying patients at high risk for future stroke to tailor and intensify preventive strategies. Changes in the retina and its microcirculation may precede overt cerebrovascular disease, and DL models and retinal imaging can be used to assess an “age gap” marker—the difference between actual and predicted age from retinal images—to forecast 10-year stroke risk with an AUROC of 0.676. 8 In another study, a model trained on 1.6 million unlabeled retinal images using self-supervised learning predicted 3-year ischemic stroke incidence with an AUROC of 0.754 internally, and an AUROC of 0.594 when externally validated. 9
Role of AI in the prehospital setting
Recognizing stroke
Early recognition of stroke symptoms and activation of Emergency Medical Services (EMS) contribute to reduced prehospital delays and more timely, higher-quality care. 26 Improved ability to predict the likelihood of a stroke is vital for effective triage systems in prehospital settings and to determine the appropriate level of care and the most suitable medical facility for the patient. 27 In a study evaluating machine-learning assisted stroke recognition for medical helpline calls, ML framework substantially improved stroke recognition in comparison to solely relying on human call takers suggesting that it should be used as a supportive tool for call-takers in the decision-making process. 10
Virtual triage (VT) AI systems can enhance early detection and ensure timely medical intervention for stroke patients. 28 For example, the Infermedica Symptomate VT engine employs ML algorithms and natural language processing to analyze patient-characteristics and reported symptoms during initial emergency contacts like phone calls or telehealth consultations. By simulating the reasoning of a human clinician through dynamic, evidence-driven interviews, these AI-driven systems use Bayesian probabilities to rapidly assess symptom severity, identifying critical conditions like stroke with high sensitivity and specificity. After analyzing all patient data, the VT computes the probabilities of likely conditions and advises on probable causes, illness severity, and recommended levels of medical care—from self-care to emergency services. Crucially, VT AI can engage patients who might underestimate the seriousness of their symptoms. 11 Studies involving thousands of VT user interviews have shown that a significant percentage of individuals requiring emergency care initially had no intent to consult a physician. In one study, 35% of patient-users changed their healthcare-seeking actions based on VT evaluations and recommendations. These findings suggest that integrating VT AI into prehospital care pathways can significantly alter patient behavior by accelerating care engagement and emergency care referral, particularly for severe conditions where misunderstanding or indecision may cause dangerous delays.
Stroke triage
A recent study analyzed data from more than 14,000 acute stroke consultations seen by tele-specialist neurologists in the emergency departments across 166 facilities in 17 states reported that the door-to-neurointerventionalist notification time was 39.5 min less in hospitals which used a disease detection and care coordination AI-powered model compared to those without it. This resulted in a statistically significant improvement in time-to-clot recognition and retraction. 12 In another study, the use of automated LVO detection and secure communication at primary stroke centers reduced door-in-door-out time by a median of 106 min and increased the proportion of patients receiving endovascular therapy after transfer. 11
Risk assessment
Various risk assessment models, such as HAT, SEDAN, SPAN-100, THRIVE, MSS, and SIT, have been employed in clinical settings to assess the risk of hemorrhagic transformation (HT) after thrombolysis. While such models provide valuable insights, they often depend heavily on predefined features and may exhibit variability across different healthcare systems due to differences in data collection practices and patient population. Recent studies exploring ML ability in risk assessment of HT found improved accuracy in comparison to these traditional risk assessment models. For example, an ML model developed only based on clinical information reported an AUC of 0.94 compared to the best performing model, SIT, which scored 0.65. 29
A recent systematic review presents compelling evidence that ML models—especially ones integrating clinical and imaging data—can outperform traditional risk scores such as the HAT and SITS-SICH 30 in predicting HT by leveraging nuanced interactions between variables such as NIHSS, infarct heterogeneity on CT radiomics, and platelet counts to achieve median AUCs of 0.91 across studies, compared to 0.65 for conventional scores. 30
While many of the currently available AI technologies are still experimental and lack integration into clinical workflows, the preceding findings clearly illustrate that the integration of AI and ML in the prehospital/ triage/risk assessment setting can be a vital and transformative step in acute stroke care.
Role of AI during the hyperacute phase of stroke
AI in acute ischemic stroke
CT head analysis in AIS
DL has been extensively applied to segmentation of medical imaging. New advances in accurate segmentation of small targets and its scalable network architecture and simplifying key aspects from pre- to postprocessing with minimal human involvement have led to improved architecture for automatic medical image segmentation including early noncontrast CT (NCCT) changes in hyperacute and acute phase of AIS. In a study comparing DL model to two human readers, the model performed well compared to human experts and in some instances identified lesion parts that some experts missed. 31
The use of conventional neural network-radiomics (CNN-R) model, an approach where a multitude of imaging features are extracted from a region of interest and subsequently reduced to convey diagnostic or prognostic information, trained upon chronometric lesion age (time-from-stroke-onset-to-scan) can be employed to estimate the biological lesion age from NCCT. In one study, the CNN-R used for estimating acute ischemic lesion age from NCCT was approximately twice as accurate as the NWU (NET water uptake), a quantitative imaging marker of the fractional water content of ischemic tissue compared to a contralateral homologous region. Despite being only trained on time-based data, the CNN-R was able to outperform not only NWU but also the actual chronometric age in predicting the biological lesion age. 32 For example, the CNN-R showed a better correlation with core penumbra mismatch ratios and lesion growth prediction. These findings suggest that CNN-R could perhaps better reflect tissue viability when physicians are faced with important treatment decisions, particularly in cases such as stroke where onset time is unknown.
Detection of large vessel occlusion (LVO)
AI models trained for the detection of intracranial LVO have been developed with the aim of assisting in rapidly triaging the stroke patient, especially in limited resource centers that lack neuroradiologists or trained stroke specialists.
A fully automated AI software developed to detect LVO's was able to achieve a sensitivity of 86% and a specificity of 97%. Tested in a reader assessment study, the model was able to achieve an improved sensitivity of LVO detection with AI assistance compared to reading without AI assistance, 92% and 88%, respectively. 16 These results highlight the added value of such systems; Not only do they perform well as standalone diagnostic tools, but they also serve as highly-functioning assistance tools to augment the capabilities of less experienced early career physicians. This can have important utility to standardize the quality of care in settings where vascular expertise is lacking or not available 24/7.
LVO-automated detection using convolution neural networks was compared to board-certified neuroradiologists’ CTA interpretation in two different analyses. Both the primary and secondary analysis included a large set of data and a variety of clinical environments. 13 In the primary analysis, the CNN model evaluated ICA or MCA-M1 occlusions. It illustrated a sensitivity of 78.2%, specificity of 97%, and the kappa coefficient for agreement with neuroradiologists’ interpretation was 0.67. In the secondary analysis, the CNN model evaluated ICA, MCA-M1, or proximal MCA-M2 branch occlusion; sensitivity was 60.6%, specificity 98.2%, and kappa's coefficient 0.65. 13 The sharp decline in sensitivity when the model is faced with a more distal M2 occlusion presents a common challenge for current AI models, where their effectiveness or reliability decreases when faced with smaller or more distal targets. Such limitations need to be addressed in future research, in particular as thrombectomy criteria continue to expand to include select M2 occlusions.
The use of DL-based detection can improve LVO detection. In one study, DL-based algorithm demonstrated a sensitivity of 70.5% and specificity of 98.9% for detecting all occlusions independent of scanner type, and showed significantly better performance on photon-counting computed tomography CTA images for detecting all occlusions, with a sensitivity of 84.0% and specificity of 99%, compared to CTA images from conventional CT scanners, which had a sensitivity of 65.1% and specificity of 98.8%. 17 These results illustrate an important point often overlooked in software evaluations, which is that algorithmic performance is fundamentally linked to the quality of the input data from the imaging hardware. The full potential of AI in stroke imaging goes hand in hand with parallel advancements in scanner technology.
Another study utilized DL through a 3D self-configuring medical object detection system to detect LVO's. The model was fed maximum intensity projection CTA images. It was able to achieve an accuracy exceeding 98% on an independent external test data. The model was able to also demonstrate a strong agreement in assigning collateral scores of CTA images, their performance was comparable to or even at instances surpassing individual radiologists’ consensus as reference standard. 33 Suggesting that more complex modern network architectures like the 3D self-configuring model may be able to overcome previous challenges with older algorithms.
CT perfusion (CTP) analysis
The ability of AI-driven algorithm for CTP analysis was tested in real-world setting. 14 The model successfully postprocessed all CTP studies (100% success rate) rapidly (median time of 8 min), and when compared to syngo.via, a conventional imaging software, it reported a core infarct volume, penumbra and mismatch ratio that correlated strongly with syngo.via. For assessment of eligibility for thrombectomy, it aligned in 87.5% of cases (core<70 mL, penumbra >15 mL, mismatch ratio>1.8) with discrepancies only near the threshold values.
AI-assisted automated analysis of CTP scans might also be used as a screening tool to detect large and medium vessel occlusions (LVO/MeVO) in resource-limited settings lacking timely neuroradiological interpretation. In a retrospective study which compared automated CTP to CT angiography in 795 patients with AIS, automated CTP demonstrated high sensitivity (95.55%) for identifying occlusions, with Tmax >6 s having the highest accuracy (AUC = 0.905). However, specificity (81.73%) and positive predictive value (63.54%) were lower, and 25/562 occlusions (4.44%) were missed by CTP, mostly related to challenges in detecting MeVOs (e.g., distal M3 occlusions) due to technical challenges such as motion artifacts or cardiac conditions (e.g., atrial fibrillation) affecting the perfusion maps. 15
AI role in stroke of unknown onset time
DWI-FLAIR mismatch is used to estimate the time from stroke onset to imaging, where DWI-positive, FLAIR-negative lesions imply onset-time ≤4.5 h. 34 AI has utilized DWI-FLAIR mismatch to facilitate the process utilizing three ML models: LR, SVM, and RF, each with unique strengths, where LR estimates the probability of a binary outcome, through probabilistic predictions instead of fixed classifications; SVM is useful for high-dimensional data and effectively reduce the impact of outliers; and RF is characterized by its fast training and ability to rapidly select important features. A recent study found no significant performance differences between the models with sensitivity rates (75.8% for LR; 72.7% for SVM; and 75.8% for RF) versus 48.5% for expert readers, and specificity rates (82.6% for LR; 82.6% for SVM; and 82.6% for RF) versus 91.3% for expert readers. 18 The higher sensitivity and lower specificity of the ML models compared to human readers suggest that the use of these models may help to improve decision-making regarding thrombolysis.
Another study developed ML classifiers that use extracted features obtained from acquired MRI images to determine stroke onset time, or a DL approach to extract hidden representations to enhance classification performance, or a combination of both. All models achieved an AUC of at least 0.6 when trained on either one of them. The combination of both, improved the classification performance across most of the models achieving an AUC of 0.765, with a sensitivity of 0.788 and a negative predictive value of 0.609; and outperformed the conventional method (DWI-FLAIR mismatch) for determining stroke onset time. 35 In addition, ML models that work on DWI and perfusion-weighted imaging “DP fusion” have shown that DP fusion outperformed traditional DWI- or PWI-based approaches. 36
The use of a DL model to predict DWI-FLAIR mismatch from NCCT images has been shown to improve human performance/accuracy through improving less-experienced physician in detecting the DWI-FLAIR mismatch from NCCT from 0.53 to 0.61. 19
Integration of AI in stroke networks and regional stroke centers
Beyond triage, the integration of AI-powered perfusion tools like RAPID ANGIO enables flat-panel CT perfusion (FD-CTP) imaging directly in the angio suite, bypassing the usual CT workflows. In a small multicenter study of 20 AIS patients with LVO who underwent thrombectomy, qualitative and quantitative perfusion results from FD-CTP with AI processing correlated well with traditional CTP (AUC 0.84 for infarct core) while also minimizing time-to-decision during thrombectomy triage. This integration in the network can facilitate direct-to-angio protocols, which streamline transfers in hub-and-spoke networks and helps in reducing delays for patients with LVO. 37
AI in hemorrhagic stroke
Identification of intracranial (ICH) and subarachnoid hemorrhage (SAH)
Innovative DL models like the Hybrid 2D/3D UNet promise to potentially advance ICH and SAH diagnosis by automating hemorrhage identification and quantification on NCCT scans. By combining 2D and 3D CNNs, which interconnects intraslicing details with spatial context, this framework addresses challenges such as irregular lesion morphology and overlapping hemorrhage subtypes. In a retrospective study involving 1355 aneurysmal SAH (aSAH) cases, the model was able to achieve high accuracy in detecting all ICH subtypes (Dice scores 0.993–0.999) and segmentation performance (0.550–0.897), outperforming current algorithms. 20 This platform also reduced manual processing time from 5 to 10 min to under 40 s per scan. However, in another study, using a large multimodal model (GPT-4) to analyze cerebral hemorrhages on noncontrast CT scans, the model was able to achieve an overall hemorrhage identification completeness of 72.6%, with notable variability across subtypes. 21 For SAH, GPT-4 had a complete identification (measure of the thoroughness or the success of the model's ability to identify all the relevant elements) of 71.5% but demonstrated a high misidentification rate (50.5%), reflecting challenges in the model's ability to distinguish SAH from overlapping features or artifacts. Performance was stronger for epidural (89.0%) and parenchymal hemorrhages (86.9%), while chronic subdural hemorrhages posed significant difficulties (37.3% completeness), likely due to their subtle CT density and structural changes. These studies demonstrate both the limitations of existing AI models in hemorrhage identification, and the potential promise of efficiency to augment emergency triage and treatment planning. Clearly, more studies are needed.
Detection of aneurysms and prediction of aneurysmal rupture
The role of AI in the diagnosis and prognosis of unruptured cerebral aneurysms is rapidly evolving. A commercial AI algorithm established a high sensitivity (100% for aneurysms >4 mm) in detecting incidental intracranial aneurysms during routine MRI screenings. 38 Although, its high alert rate (17.8%) and low positive predictive value (11.5%–43.8%) created a substantial burden in follow-up imaging. It identified 59 suspicious findings in 907 MRIs, including 13 intradural aneurysms missed by initial radiology reports, underscoring its potential to reduce underdiagnosis. Notably, 84.7% of these findings were newly detected, highlighting AI's role as a second reader in high-volume clinical settings. In a Chinese multicenter validation study, a DL model for intracranial aneurysm detection achieved a patient-level sensitivity of 95.7% and specificity of 79.7% in internal validation and was able to outperform clinicians in external validation with a sensitivity of 94.3% versus 65.8% reported by clinicians, particularly for small aneurysms (<3 mm), although sensitivity dropped to 67.5% for <3 mm cases. 39 As a second reader, the model was able to improve clinicians’ diagnostic accuracy both on a per-patient basis by increasing the AUC from 0.795 to 0.878, and on a per-aneurysm basis from 0.765 to 0.865. It was also able to reduce the reading times from 87.5 to 82.7 s. In prospective validation, the negative predictive value was 99.8%, reducing missed diagnosis by 23.5% among radiologists. The model was also able to flag high risk morphological features such as irregular shape and size predictive of rupture potential.
Lastly, a systematic review and meta-analysis involving 18,670 participants examined the efficacy of ML algorithms in predicting cerebral aneurysm rupture risk. The pooled sensitivity and specificity were 0.83 with a high diagnostic odds ratio of 23.69 and an AUC of 0.90, 22 indicating that the use of ML algorithms to predict the risk of rupture in cerebral aneurysm can be effective with good levels of accuracy.
Prediction of long-term functional outcome
Modified Rankin Scale (mRS) is the most commonly used measure of poststroke functional outcome. 40 Outcome prediction can be important to counsel patients and their families and in decision-making regarding high-risk treatment options and aggressiveness of care. Studies using AI have focused on predicting unfavorable outcomes, however, predicting the exact mRS scores has been challenging. 41 The models’ performance varied depending on the true mRS scores, with more accurate predictions for patients with mRS scores ranging from 0 to 4, however with severe scores the model tended to underestimate the true scores. 41 The poor ability of AI models to predict the exact mRS scores in these models is likely due to the fact that prediction of long-term outcomes should incorporate other postacute phase factors such as rehabilitation and medical complications, which were not widely used in AI models in the literature (Table 2).
Summary of ML and DL models applied in ischemic stroke for outcome prediction.
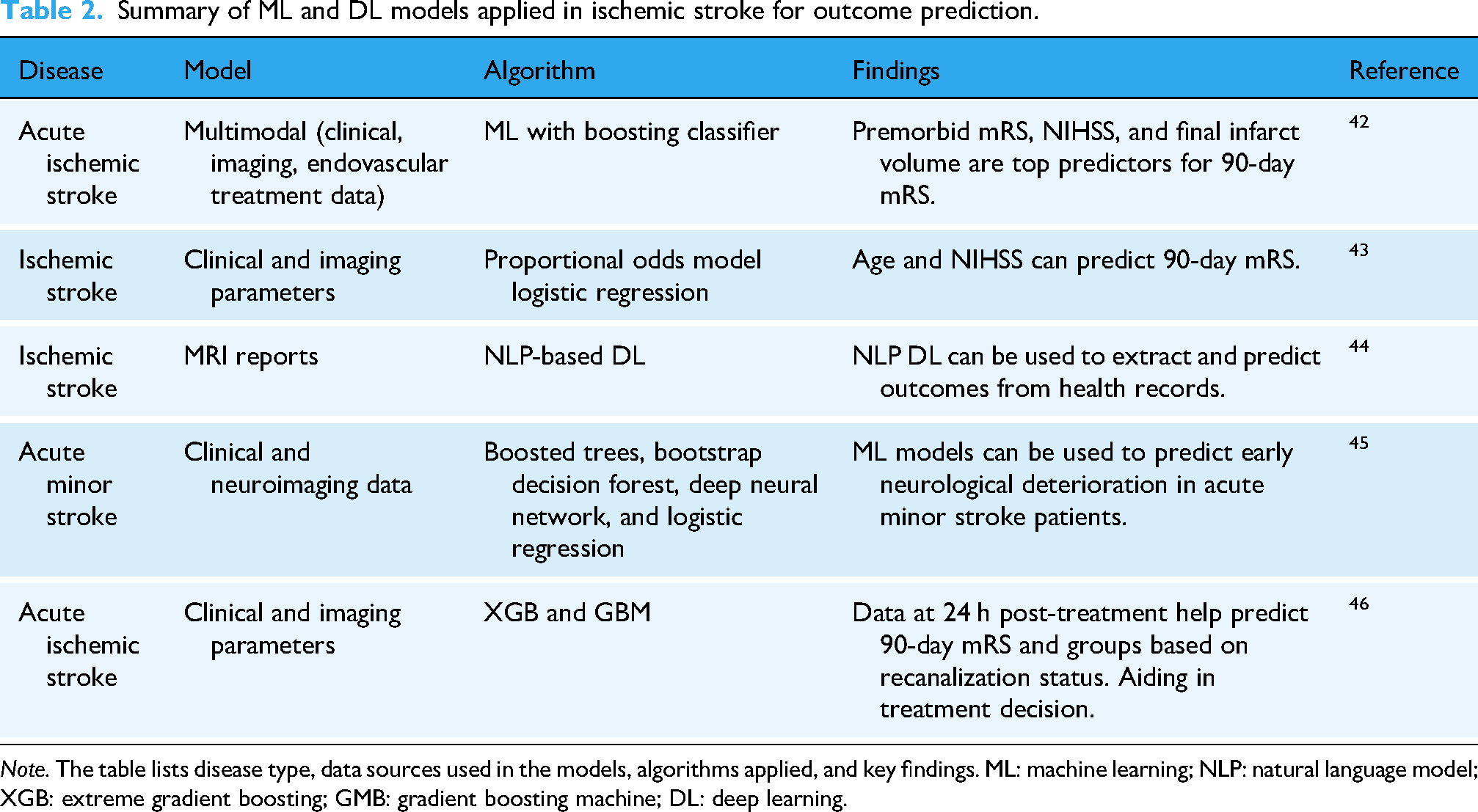
Note. The table lists disease type, data sources used in the models, algorithms applied, and key findings. ML: machine learning; NLP: natural language model; XGB: extreme gradient boosting; GMB: gradient boosting machine; DL: deep learning.
ML and LR models were shown to have comparable predictive accuracy for 90-day mRS scores in acute ischemic stroke patients, 47 however, a study comparing multiple different ML models showed that the XGBoost model was the best in predicting mortality among patients, with an accuracy of 94.5%, when compared with a traditional approach as LR. 48 However, it is important to note that predicting mortality presents a unique challenge for AI models, as a large proportion of stroke patients die from poststroke complications or withdrawal of treatments that are no longer perceived to be beneficial or necessary for a patient's wellbeing.
Different models have incorporated various clinical and imaging factors into their models, such as NIHSS score, ASPECTS score, presence of hyperdense MCA sign, degree of vascular occlusion, premorbid mRS, and length of stay.49,50 Compared to AI models using clinical data for outcome prediction, models that incorporate images seem to have a more accurate prediction. 42 One study demonstrated a higher proportion of mRS score predictions within ±1 category when using an imaging model compared to only a clinical model, with 79% correctly predicted scores. 41 The effect of imaging data is more prominent in predicting the outcome after endovascular treatment, as predictive accuracy for image-incorporating models was found to be 0.69 in comparison to 0.66. 51 In one study, final infarct volume (<19.5 cm3), NIHSS score ≤7 after 24 h, and premorbid mRS score ≤2 emerged as important predictors of favorable clinical outcome after 3 months, 42 combining the final infarct volume and location improved prediction accuracy, emphasizing the importance of spatial data.
Prediction of hematoma expansion
Hematoma expansion (HE) is a consistent predictor of early neurological deterioration and mortality after ICH. Early identification of patients at risk for HE can have important treatment and triage implications. Several imaging markers and clinical scales/scores have been developed to predict HE, but their predictive abilities are modest at best. 52 AI may play an important role in this regard. A study employing CNN to predict HE based on CT scan and clinical data reported an accuracy of 0.90. 53 Moreover, the CNN model performed better when compared to other ML models such as SVM and decision trees, which are dependent only on clinical data.53,54 Because it is critical not to miss patients at high-risk of HE, it has been suggested that the better the sensitivity the better the model would be. 54 Others argue that it is equally important to balance AUC and sensitivity when developing HE models. 55 AI models for HE prediction still require further refinement and simplicity. Although DL models have higher predictive ability, their complexity may limit their clinical application, especially during the hyperacute and acute phase. Moreover, increasing the number of variables makes the predictive model less effective in clinical settings because it is challenging to ensure that all patients have the required data.56–58
Prediction of stroke recurrence
Stroke recurrence depends on the number of modifiable and some nonmodifiable risk factors. However, it is challenging to identify the factors for each individual or establish a specific scale due to the variability and limitations of available data between patients. ML can integrate radiomics and clinical features to improve prediction models. A personalized, AI- and statistically-based stroke recurrence prediction using three classifiers (RF, AdaBoost (ADA), and extreme gradient boosting (XGB)) incorporated several predictive factors (time from previous stroke, Barthel Index, atrial fibrillation, dyslipidemia, age, diabetes, sex, glycemia, body mass index, high blood pressure, cholesterol, tobacco dependence, and alcohol abuse) found that RF and ADA classifiers outperformed the Cox model in predicting early, late, and long-term recurrence risks, with statistical significance while the XGB classifier did not show a significant improvement. 59 In another study, 4 ML models (LR, Support Vector Classification (SVC), LightGBM, and RF using clinical data and MRI radiomics) were evaluated for their ability to predict ischemic stroke recurrence in the first year. The light GBM achieved the best performance when combining radiomics and clinical features, with 0.85 sensitivity, 0.805 specificity, and AUC = 0.789. Performance was lower when using clinical data alone (AUC = 0.735) or radiomics data alone (AUC = 0.647). 60
AI in poststroke rehabilitation
AI-powered rehabilitation can benefit a wide range of stroke patients. For patients with motor impairments, AI-driven robotic exoskeletons and virtual reality (VR)-based therapy enhance limb function by classifying movements as correct or incorrect and predicting their accuracy. 23 In cognitive and speech rehabilitation, models such as SVM, RF, and DNN achieved high predictive accuracy, however these models have not been tested widely or used in real clinical settings. 61 Gait rehabilitation using AI has been widely explored to guide personalized rehabilitation, but choosing the suitable gait measures and making an accurate prediction is still challenging. 61 Current evidence supports a structured approach to AI implementation in rehabilitation based on the patient's specific functional deficit. Robotic exoskeletons and VR systems are most effective for motor recovery as they use real time kinematic data to correct limb trajectory. 23 Moreover, ML classifiers, for example, support vector machines demonstrate higher utility in prognostication, particularly for predicting if there are nonlinear recovery trajectories in speech and cognition. 61 Lastly, wearable sensors address the limitation of intermittent clinic visits by providing continuous, objective metrics of gait and mobility in the home environment. 62
Also, a home care system to enhance rehabilitation and improve motor function in chronic stroke patients through wearable devices combined with AI to assist in long-term recovery is being studied. 61 Table 3 lists different applications, which apply ML and DL techniques to assess patient progress.
Summary of studies applying AI in poststroke rehabilitation.

Note. This table outlines the rehabilitation type, study focus, AI classifiers used, and key findings. SVM: support vector machine; RF: random forest; ANN: artificial neural network; CNN: convolutional neural network; k-NN: k-nearest neighbor; DNN: deep neural network; RNN: recurrent neural network; DT: decision tree; LR: logistic regression; ML: machine learning; DL: deep learning.
Personalized recovery plans are enhanced using AI technology in poststroke care. Through using wearable devices and AI-driven analytics, patients can be continuously monitored for motor and cognitive functions, enabling early detection of complications and stroke recurrence risks. The collected data is transmitted directly to healthcare providers, for evaluation and more accurate assessments of each patient's progress. By analyzing these data patterns, AI systems create customized rehabilitation programs that fits to everyone's abilities, cognitive function, and recovery trajectory. 67
Role of AI in postdischarge care
AI nurse navigator workflows
Fragmented workflow where responsibility is split between various healthcare providers often results in inconsistent patient follow-up and delivery of optimal postdischarge care. Nurse navigator implementation in stroke patients’ care is important to address such gaps to facilitate postdischarge continuity-of-care, assure compliance and adherence with prescribed medications, and reduce 30-day unplanned readmission risk. 68 AI-enabled solutions and systems could improve follow-up adherence by automating a standardized workflow, closing communication gaps, and freeing nurse navigators to focus on high-risk cases. 25 In practice, these workflows use algorithms to screen Electronic Medical Records for things such as risk factors, polypharmacy or prior appointment nonattendance. For example, an AI system detecting a missed pharmacy refill 48 h postdischarge can automatically trigger a text message to the patient assessing adherence. A negative or ambiguous response immediately flags the case for the nurse navigator; this allows nurse navigators to prioritize high risk patients and offer intervention over routine follow up calls. Also, an AI model using natural language processing could be used for specific tasks while leaving tasks that are best handled by humans to the nurse navigators. This does not only reduce workload on already overworked nursing system but also ensures that no certain patient population is lost/overlooked in the follow-up, while also bringing attention to cases that are at higher risk of missing a follow-up to nurse navigators. These models have been successfully implemented in radiology to communicate actionable incidental findings. 68 Stroke systems would benefit immensely from similar AI applications to enable stroke nurse navigators to dedicate time to patient education, address disparities, and give a personalized outreach to every unique stroke patient, which is key in preventing stroke recurrence.
Challenges, limitations, and future directions for AI in stroke
This review highlights the wide range of potential AI uses in stroke patients, from triage to rehabilitation and postdischarge care. However, the adoption of AI into real-world clinical settings still faces many challenges.
Regulations and guidelines regarding the use of AI in clinical practice are still limited. Only recently, did the Food and Drug Administration issue a draft guidance on using AI in medical products aiming to ensure safety and effectiveness while fostering innovation. 69 Although various AI models have shown great accuracy, it is still questionable whether such accuracy will be achieved when prospectively studied across different settings and a wide range of clinical pictures. 70 The complexity of AI models still limits their clinical application, especially during the hyperacute and acute phase. Moreover, the large number of variables makes the predictive model less effective in clinical settings because it is challenging to ensure that all patients have the required data elements built into the model.
Another issue is the “black box”, we know the model made a decision but do not know why. To rely on AI systems in real-world clinical settings, we must have some transparency about why the model made such a decision or prediction. It has been suggested that implementing explainability methods 71 may help to address these concerns. Explainability is the model's ability to explain why it made a decision. Such a feature is crucial for safety and accountability, especially in emergencies and for regulatory purposes.
Moreover, there are ethical challenges and considerations to AI implantation in real world stroke management, related to institutional workforces and need for multivalidation studies to ensure that the model is trained on data from multiple races, genders, and backgrounds. Data privacy remains an issue as many patients may refuse to share their data. It is impossible to have an ideal model, and an acceptable level of bias may be necessary.
Conclusion
The integration of AI into stroke care offers a method to standardize high-quality care across diverse clinical settings. Through the literature reviewed, we can clearly see that these technologies effectively streamline time-sensitive decisions in the prehospital phase, improve the scope of risk stratification, and allow for the customization of protocols.
However, the translation of these technologies from research to the clinical world is hindered by critical limitations. Recent studies have consistently reported improved diagnostic speed and accuracy but sound evidence linking AI integration to measurable improvement in patient outcomes remains sparse. It is even less clear whether AI can directly translate to improved clinical outcomes.
Moving forward, these models will require to address such challenges through multicenter prospective trials, standardized benchmarking, and the development of explainable, user-centric systems that complement and assist—rather than replace—clinical expertise. It is of high importance to emphasize that AI cannot be a stand-alone solution and that its success depends on human skill. AI's future is promising but its ultimate value will depend on rigorous validation, seamless integration into clinical workflows, and sustained collaboration between developers, clinicians, and policymakers to align innovation with patient-centered care priorities.
Footnotes
Acknowledgment
Not applicable.
Ethical approval
Not applicable.
Contributorship
Mohammed Awamleh: conceptualization, idea formation, literature review and extraction, and writing—original draft and editing. Mohammad Hamad: literature review and extraction and writing—original draft. Abdullah Jariri: literature review and extraction and writing—original draft. Magdy Selim: supervision, mentorship, critical editing, and contribution to manuscript revision.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
