Abstract
Introduction
Severe dengue and sepsis account for tens of millions of cases and deaths annually in low-resource settings, where spot-check vital sign monitoring misses early physiological deterioration. Affordable wearable sensors that stream continuous data and feed machine learning algorithms promise earlier detection and improved outcomes.
Methods
We conducted a structured narrative literature review of human studies on PubMed, Embase, Scopus, and Google Scholar that evaluated wearable vital-sign devices for predicting severe dengue or sepsis in resource-limited areas. Two reviewers independently screened, extracted, and synthesized data on study design, devices, vital streams, predictive performance, and implementation barriers.
Results
Seven eligible studies (2019–2024) from Vietnam, Rwanda, Bangladesh, and South Korea tested photoplethysmography or single-lead electrocardiography wearables in intensive care units and emergency departments of hospitals. The algorithms achieved an area under the receiver operating characteristic curve of 0.83–0.86 for forecasting dengue shock up to 2 hours and sepsis deterioration up to 5–48 hours before conventional recognition. The precision reached 0.79 for the three-level dengue severity classification. Continuous monitoring reduces the time to intervention and matches or exceeds the accuracy of manual charting.
Conclusion
Early evidence shows that low-cost wearable sensors can deliver clinically meaningful lead times for severe dengue and sepsis in developing countries’ hospitals. However, generalizability, artifact suppression, power autonomy, and economic sustainability remain unproven. Multicentre pragmatic trials, edge-optimized algorithms, and full cost-effectiveness analyses are needed before routine adoption.
Introduction
Dengue and sepsis, while distinct in their epidemiology and pathophysiology, impose overlapping and intensifying burdens on health systems worldwide. Dengue is a mosquito-borne viral disease endemic to tropical regions, often causing explosive outbreaks. In contrast, sepsis is a life-threatening syndrome triggered by dysregulated host responses to severe infections from various pathogens, typically presenting as sporadic cases rather than outbreaks.1,2 Despite these differences, both conditions converge at the point of acute clinical deterioration, share overlapping physiological signatures (such as fever, hypotension, and shock), and disproportionately affect resource-limited low- and middle-income countries (LMICs). This shared clinical endpoint of rapid progression to critical illness provides a rationale for considering them together in evaluating wearable sensor-based early warning systems.
The World Health Organization (WHO) estimates that over 6.5 million cases of dengue and 7300 deaths were reported in 2023 alone, while in 2024, more than 14 million cases were reported with over 10,000 deaths, marking the highest number of global dengue cases ever recorded to date, thereby surpassing previous annual records.3–5 In parallel, sepsis continues to exact a staggering toll, accounting for an estimated 48.9 million cases and 11 million deaths each year, representing roughly one-fifth of all global mortality, with nearly 20 million cases occurring in children under 5 years of age. 6 Importantly, sepsis and dengue shock syndrome (DSS) exhibit several similar clinical features, which create a diagnostic dilemma, particularly in dengue-endemic regions where both conditions coexist. 7 This underscores the need for rapid physiological monitoring to aid frontline clinicians in differentiating between early manifestations of dengue and sepsis. Evidence from an Indian case study highlights how wearable vital-sign sensors can support earlier recognition in these challenging scenarios. 8
Recent advances in wearable vital-sign sensors offer a promising countermeasure: a low-cost wrist-mounted photoplethysmography (PPG) device that deployed predicted severe dengue deterioration 2 hours in advance with an area under the receiver operating characteristic curve (AUROC) of 0.83 and recall of 0.95, supporting real-time ward triage in a resource-constrained hospital. 9 A gradient-boosted model using only six continuously streamed vitals from consumer-grade wearables forecasted sepsis onset up to 48 hours earlier with AUROCs of 0.84–0.83 across external test sets. 10 Another study showed machine-learning–driven alerting systems reduced sepsis mortality by 44% (RR 0.56, 95% CI 0.39–0.80) compared with rule-based care. 11 Additional clinical deployment reports describe continuous ward monitoring via ViSi Mobile and similar platforms that shift observation from 4-hour spot-checks to minute-level streams, shortening antibiotic-to-door times and lowering sepsis deaths across 12 hospitals. 12
Despite these advantages, critical gaps remain. Most sensor algorithms are validated in tertiary centers rather than primary care or community settings typical of dengue-endemic LMICs9,13; head-to-head comparisons of signal quality loss during high humidity, extreme ambient heat, or pediatric shock are scarce14,15; interoperability with minimal electronic infrastructures and cost-effectiveness over the device life cycle are understudied.16,17 Furthermore, there is limited evidence on how real-time data streams integrate with frontline clinical workflows to trigger fluid resuscitation or vector-control responses in LMIC outbreaks.18,19 Bridging these gaps is important because variables captured in our review tables such as heart-rate variability, pulse transit time, peripheral perfusion index, SpO₂, skin temperature, NEWS2/mSOFA scores, device cost, and battery autonomy, map directly onto outcomes that drive both scientific inquiry (algorithm generalizability), policy (procurement and reimbursement), and bedside practice (earlier intravenous fluid or platelet transfusion), 20 especially where clinician-to-patient ratios are low and laboratory turnaround is slow. Thus, this study aims to synthesize and critically appraise human studies of wearable vital-sign sensors for the early detection/prediction of severe dengue and sepsis in resource-limited settings, with a particular focus on predictive performance and implementation barriers in LMIC hospitals.
Methods
We conducted a structured narrative literature review to map the clinical evidence, challenges, and public health relevance of wearable vital sign sensors for the early detection of severe dengue and sepsis in low-resource settings. Although not a full systematic review, the methodology was strengthened by aligning with the elements of the PRISMA reporting framework to enhance transparency and reproducibility. PubMed, Embase, Scopus, and Google Scholar were searched for relevant studies. The search strategy used a combination of controlled vocabulary (e.g. Medical Subject Headings [MeSH]) and free-text keywords. Search strings combined exposure terms such as “wearable,” “biosensor,” “continuous monitoring,” AND and OR “photoplethysmography” with device-specific terms like “ECG patch,” paired with condition terms including “dengue,” “dengue shock,” “sepsis,” and “septic shock,” using Boolean operators AND and OR. Studies published after 2015 were prioritized. Eligible sources were peer-reviewed journal articles in English that enrolled humans and reported diagnostic, prognostic, or implementation outcomes of wearable vital sign sensors. Inclusion was restricted to studies conducted in low-resource or LMIC-relevant contexts, which were determined based on the study's geographic setting, explicit reference to resource-constrained environments, or descriptions of infrastructure limitations. We excluded editorials, commentaries, conference abstracts without full data, and studies that did not focus on dengue, sepsis, or LMIC-relevant contexts from the review. Additional exclusion criteria included studies limited to simulation or bench testing without clinical deployment and those evaluating non-wearable devices (e.g. handheld monitors or fixed bedside equipment). Two reviewers (MMA and ZKO) independently screened the titles, abstracts, and full texts, with disagreements resolved through discussion with the third reviewer (OJO). Data from the included studies were extracted into standardized evidence tables capturing the characteristics of the included studies, devices, clinical settings, monitored vital signs, target conditions, predictive performance, usability, and key limitations of wearable approaches. The findings were narratively synthesized into domains covering study characteristics across the included investigations, wearable sensor platforms, care settings, physiological streams, model precision, clinical lead time, and implementation challenges.
Results
Study characteristics across included investigations
Seven eligible studies were published between 2019 and 2024 and represented Vietnam, Rwanda, Bangladesh, and South Korea, with Vietnam contributing half of the total.9,13,18,21–24 Prospective observational designs predominated (six of seven, 85%), while one study used a prospective cohort framework, underscoring a descriptive emphasis in early stage validation.9,13,18,21–24 Dengue-focused work accounted for three Vietnamese studies (Table 1), whereas sepsis motivated the remaining five.9,18
Characteristics of included studies.

Wearable sensor platforms, care settings, and physiological streams
All deployments occurred in hospital environments, including five intensive care units and two high-acuity emergency departments, reflecting a preference for closely monitored clinical contexts during early implementation13,18 (Table 2). Photoplethysmography wrist or fingertip devices were used exclusively in dengue investigations, whereas chest or forehead electrocardiography (ECG) patches formed the backbone of sepsis monitoring; a pattern more likely reflecting the specific expertise and established practices of Vietnamese dengue studies rather than intrinsic disease-specific physiology.21,23 Heart rate was captured in every dataset, respiratory rate in five studies, peripheral oxygen saturation or compensatory reserve indices in three studies, and skin temperature in two studies, producing multimodal vital-sign streams of varying breadth. 22 Across diseases, each team pursued an early warning paradigm: dengue studies aimed to stratify severity or forecast shock up to 2 hours in advance, whereas sepsis investigations sought to detect advanced disease, short-term mortality several hours before conventional assessments.9,24
Devices, clinical settings, monitored vital signs, and target conditions.

CRI: compensatory reserve index; PPG: photoplethysmography; HR: heart rate; RR: respiratory rate; ECG: electrocardiography.
Model accuracy, clinical lead time, and implementation challenges
The discrimination metrics were uniformly strong. A convolutional neural network applied to SmartCare wristband signals classified three dengue severity levels with a precision of 0.79
21
(Table 3). A multimodal transformer predicted hemodynamic collapse 2 hours ahead of time with an AUROC of 0.83.
9
A compensatory reserve index threshold below 0.40 signaled recurrent dengue shock within 12 hours, achieving a sensitivity of 0.66 and specificity of 0.86.
22
Sepsis models showed comparable accuracy: ridge regression analysis of VitalPatch data in a pediatric intensive care unit reached an AUROC of 0.86 and delivered alerts a median of 2 hours earlier than clinical recognition.
18
Gradient-boosted ECG features detected septic shock 5–9 hours sooner than intermittent observations and slightly outperformed manual charting (AUROC 0.86 vs. 0.84).
24
Agreement studies corroborated sensor validity, with heart rate correlation of

Operational barriers are reported across wearable-sensor studies.
Predictive performance, usability, and key limitations of wearable approaches.
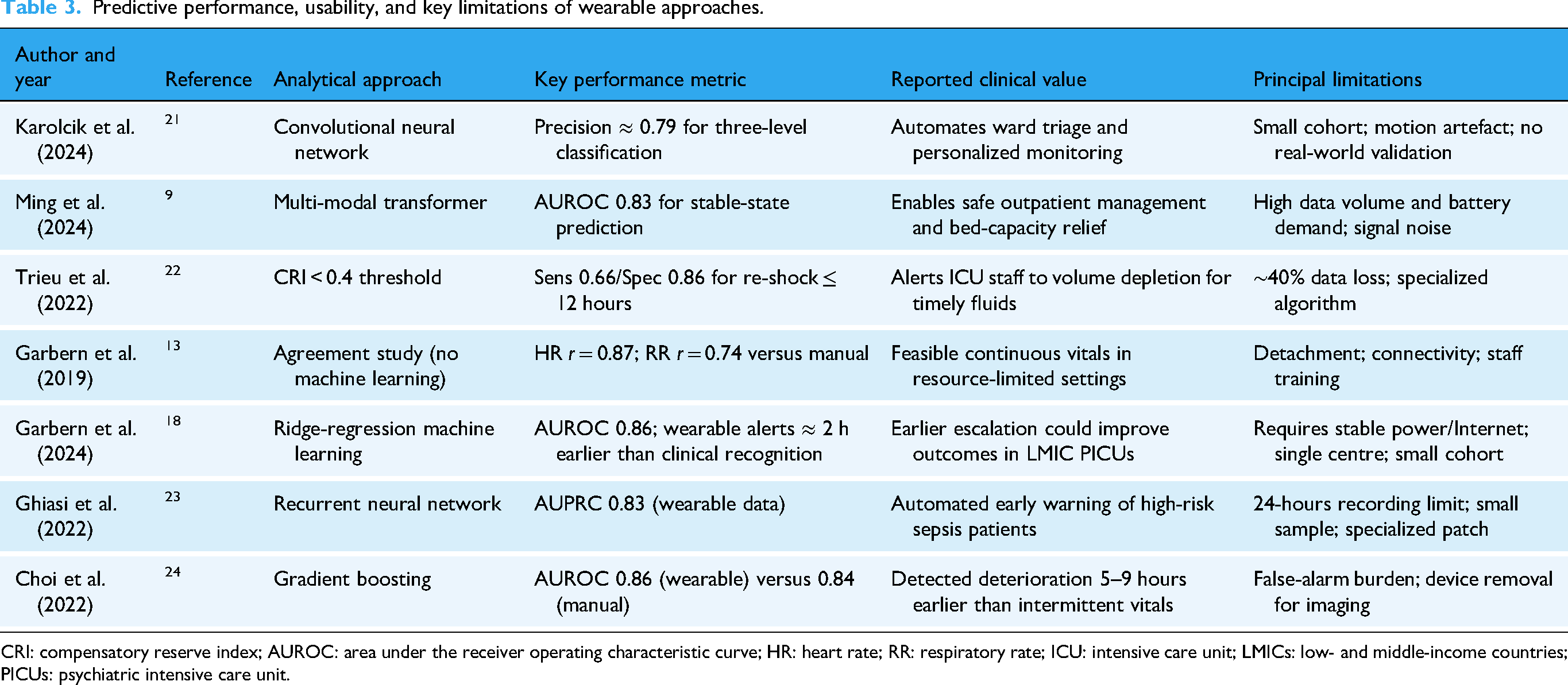
CRI: compensatory reserve index; AUROC: area under the receiver operating characteristic curve; HR: heart rate; RR: respiratory rate; ICU: intensive care unit; LMICs: low- and middle-income countries; PICUs: psychiatric intensive care unit.
Discussion
Our synthesis shows that across diverse low-resource hospitals, the reviewed wearables consistently provided clinically useful lead times and strong discrimination for incipient shock or advanced sepsis, enabling earlier escalation of care than conventional four-hourly spot-checks. The physiology behind this performance is plausible: dengue plasma leakage and early sepsis vasodilation first alter pulse-pressure amplitude, heart-rate variability, peripheral perfusion index, and skin-temperature gradients, signals that photoplethysmography and single-lead ECG patches capture at sub-minute resolution, while transformer and gradient-boosting algorithms exploit the resulting high-frequency dynamics to detect decompensation well before absolute thresholds are breached.25,26 In addition, one feasibility pilot study demonstrated the practicality of coupling a medical-grade wrist sensor to an artificial intelligence–enhanced NEWS2 engine for bedside monitoring, which identified sepsis deterioration hours earlier than standard nursing rounds. 27
These findings align with a propensity-matched analysis of 7971 medical ward admissions, where continuous wireless monitoring cut the combined odds of ICU transfer or in-hospital death by nearly threefold, underscoring how uninterrupted vital-stream analytics translate into real-world outcome benefits. 28 Moreover, cost and sustainability concerns are mitigated by emerging sub-US$150 prototypes whose PPG-based blood pressure trend and SpO₂ estimates met clinical accuracy thresholds in volunteer trials, suggesting that device affordability no longer precludes adoption in lower-income settings. 29 However, the literature remains skewed toward tertiary ICUs; a study found that the validation of wearable data flows into frontline decision-making and interoperability with sparse electronic infrastructures are critical gaps for broad implementation. 30
The main limitations of the studies center on study design, signal fidelity, power management, and economic uncertainty. Most were single-centered investigations with small cohorts (fewer than 200 participants). Half of the included studies originated from a single country (Vietnam), which raises the possibility of selection bias and constrains external validity. Continuous PPG and ECG streams are highly vulnerable to motion artifacts.31,32 Laboratory tests that used multichannel wavelength filtering still lost accuracy during ambulation, and clinical studies reported discarding up to 43% of raw epochs because of poor signal quality. Power constraints further limited continuity. 33 The low-cost wrist system that predicted dengue shock required recharging after a median of 18 hours, creating unavoidable surveillance gaps. Algorithmic generalizability is also uncertain because models are usually trained and tested on data from the same site, 34 and only one large propensity-matched ward study has linked continuous monitoring to lower intensive care transfers and mortality, leaving causality provisional. Finally, none of the trials presented full cost-effectiveness analyses that accounted for device amortization, connectivity fees, consumables, or staff training; therefore, sustainability in low-resource budgets remains unproven.
To move the field forward, multicenter pragmatic trials that include district hospitals and community fever clinics should externally validate algorithms, document signal dropout rates, battery endurance and lifecycle costs, and report clinical and economic end points (Figure 2). 35 Developing edge-optimized models that run on on-device microcontrollers, together with robust motion-suppression pipelines and energy-harvesting or ultra-low-power chipsets, would extend operating time and reduce dependence on cloud bandwidth. 36 Parallel health-economic evaluations should incorporate procurement, maintenance, and training costs so that ministries of health can negotiate pooled tenders or local assembly schemes. 37 Interoperability and data-governance standards ought to align with the WHO Regional Digital Health Action Plan 2023–2030, which explicitly calls for evidence-based scaling of remote patient monitoring and secure stewardship of health data. 38 At the service level, hospitals can begin by embedding validated wearable dashboards into existing early warning systems, training nurses in sensor placement and escalation protocols, and establishing local maintenance pathways. Policymakers should update national sepsis and dengue guidelines to include wearable-enabled monitoring and create financing mechanisms that subsidize devices for frontline facilities. Together, these actions help convert continuous wearable surveillance from a promising research prototype into a reliable, sustainable safety net across LMICs’ health systems.

Strategies for wearable surveillance integration.
Conclusion
Across the included studies, continuous photoplethysmography and ECG wearables consistently detected impending dengue shock or sepsis deterioration hours ahead of standard four-hourly observations, demonstrating AUROCs above 0.80 and translating to faster escalation of care. These gains plausibly reflect the devices’ ability to capture rapid fluctuations in pulse amplitude, heart rate variability, perfusion index, and skin temperature gradients that preceded over hemodynamic collapse. Nonetheless, evidence is still largely confined to small, single-center cohorts in tertiary hospitals; high motion-related signal loss, limited battery life, and uncertain lifecycle costs threaten scalability; and few investigations evaluate integration with frontline workflows or health-economic impact. To move from promising prototypes to sustainable clinical tools, future research must undertake multicenter trials in district hospitals and community fever clinics, rigorously benchmark artifact-suppression pipelines and on-device inference against resource constraints, and embed cost-effectiveness analyses that include procurement, maintenance, and training.
Footnotes
Acknowledgements
ORCID iDs
Ethical approval
Not required for this study.
Consent to participate
Not applicable.
Consent to publication
Not applicable.
Author contributions
MMA conceptualized and designed the study. ZKO and SMM performed the literature review and data curation. MMA and OJO drafted the manuscript. SSM and FB revised the manuscript. DELP III supervised the study. All authors have read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Not applicable; no new datasets were generated or analyzed.
Clinical trial registration
Not applicable.
